1. Introduction
Sea cucumber (
Apostichopus japonicus) is one of the most important marine culture species for its nutritional, medicinal and healthcare values in China [
1]. In the past decades, wild populations of
A. japonicus have declined seriously due to the overfishing and environmental destruction [
2]. Aquaculture of
A. japonicus has been attempted and gradually expanded in the Northern China, especially after overcoming the breeding and nursery technologies in the 1990s. Now, the annual output exceeds 180 thousand tons, which greatly soothes the reliance on the worldwide wild sea cucumber resources [
3]. The indoor nursery of
A. japonicus larvae and juveniles is usually challenged by a high water temperature during summer, when the incidence of disease outbreak and evisceration rate of
A. japonicus is usually increased [
4]. To deal with high temperature, sea cucumbers entered a hypometabolic state, which is called as aestivation [
5]. During aestivation, sea cucumbers showed intestinal degeneration and cessation of feed intake. Therefore, the nutritional enrichment before and after aestivation is important for the sea cucumbers to build up their strength.
Selenium (Se), as an essential trace element, is involved in regulating growth performance, nonspecific immune response and antioxidant capacity of almost all organisms [
6,
7,
8,
9,
10,
11,
12]. There are two existing forms of Se, among which organic Se is widely used as a feed additive due to its advantages of lower toxicity [
13,
14]. Selenium methionine (Se-met), bio-fermenting Selenium (Se-bio) and selenium yeast (Se-yeast) are the main forms of organic Se which have been previously used as a feed additive for
A. japonicus [
2,
15,
16,
17]. It was found that dietary Se-met at an addition level of 0.4–0.6 mg/kg promoted the growth and immune parameters of juvenile
A. japonicus [
15]. Compared to Se-met, Se-bio showed better effects on accumulation efficiency and immune capacity of
A. japonicus [
17]. A recent study found that Se-yeast at an addition level of 0.5–1.0 mg/kg was beneficial for the promotion of growth and immune response of early juvenile (appropriately, 10,000 individuals/kg)
A. japonicus [
2]. However, to the best of our knowledge, the effects of Se-yeast addition on the growth, intestinal health, immune status and body composition of juvenile (appropriately 400 individuals/kg)
A. japonicus, before and after aestivation, is unknown.
Thus, the effects of Se-yeast addition on the growth, intestinal health, immune status and body composition of juvenile A. japonicus were investigated before and after aestivation. The aim of this study was to ascertain the optimal addition level of Se-yeast for juvenile A. japonicus to better cope with high-temperature water.
2. Materials and Methods
2.1. Experimental Diets
The Se-yeast was obtained from Angel Yeast Co., Ltd. (Yichang, China). It was previously found that the optimal Se-yeast addition level was 0.5–1.0 mg/kg for the survival and growth performance in juvenile
A. japonicus [
2]. Thus, the actual Se-yeast addition level in this study was 0, 0.5, 1.0, 1.5 and 2.0 mg/kg, respectively.
Table 1 shows the formulation and proximate analysis of experimental feeds. The fine ingredients of each feed were successively weighed and then mixed evenly in a plastic bag by hand. After that, about 30% water was added before the pellets were extruded through a pellet-making machine, which were then dried at 60 °C. Finally, the pellets were crushed and sieved into proper sizes.
2.2. Feeding Experiment Program
Juvenile
A. japonicus were provided by Dalian Xinyulong Marine Biological Seed Technology Co., Ltd. (Dalian, China), which were then acclimated for two weeks to adapt to the experimental procedures. Then,
A. japonicus without obvious symptom of disease were randomly distributed into 15 tanks (500 L). Each tank was cultured with 60 individuals with an initial body weight of 2.96 ± 0.04 g. Each experimental diet was randomly assigned to three tanks of animals twice daily at 7:00 and 16:00. The whole experiment was divided into three stages. At the first stage, the water temperature was 19.0–22.0 °C, which lasted for 45 days. At the second stage, the water temperature was 23–25 °C, which was the aestivation stage and lasted for 60 days. At the third stage, the experimental water temperature was 19–22 °C, which lasted for 30 days. The detailed experiment program can be referred in
Table 2. About 30% seawater was exchanged daily from each tank during the experiment, with water salinity, pH and dissolved oxygen maintained at 30 ± 1 ‰, 8.0 ± 0.1 and above 8.0 mg/L, respectively.
2.3. Sampling Procedures
Before sampling, the feeding activities were stopped for 48 h at each stage. The body weight and the number of animals in each tank were recorded. Then, 15 individuals were chosen out of each tank, which were dissected for the coelomic fluid and digestive tract. The coelomic fluid was centrifugated (3500 rpm, 4 °C, 10 min) to obtain the supernatant, which was later analyzed for the immune parameters. The length and weight of intestine of A. japonicus were measured individually, pooled into sterile tubes, and then stored at −80 °C for analyzing digestive enzyme activities.
2.4. Analyzation of Digestive Enzymes and Immune Enzymes
The procedures of supernatant preparation have been previously described by Ning et al. [
2]. Briefly, the digestive tract from each tank was homogenated with cold 0.9% NaCl. Then, the homogenate was centrifuged (4 °C, 2500 rpm, 10 min) to obtain the supernatant. Commercial kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China) were used for the determination of the activities of digestive enzymes (protease, amylase, cellulase and lipase) and the total nitric oxide synthase (T-NOS), alkaline phosphatase (AKP), superoxide dismutase (SOD) and catalase (CAT).
2.5. Analyzation of Body Compositon
The proximate body composition (crude protein, crude lipid, carbohydrates and moisture) was analyzed according to the methods described by AOAC [
18]. The moisture was determined by drying the samples to constant weight at 105 °C. Crude protein and lipid were determined by using the Kjeldahl method and Soxhlet method, respectively. The detailed procedures of Se determination have been recorded in Ning et al. [
2]. Briefly, an appropriate amount of dry sample was cold digested overnight by adding 10 mL of the mixed acid (perchloric acid: nitric acid, 1:9), and it was then heat digested by adding 2 mL nitric acid and 5 mL HCl successively. After that, the solution was transferred and mixed with 2.5 mL K
3 Fe(CN)
6. At last, Se contents were assayed based on hydride atomic fluorescence spectrometry methods [
19].
2.6. Calculations and Statistical Analysis
Survival rate (SR, %) = Nf/Ni × 100
Weight growth rate (WGR, %) = (FBW − IBW)/IBW × 100
Relative digestive tract length (RDL, %) = DL/BL × 100
Relative digestive tract weight (RDW, %) = DW/FBW × 100
Visceral somatic index (VSI, %) = VW/FBW × 100
Evisceration rate (ER, %) = Ne/Ni × 100
where Ni, Nf, IBW and FBW are the initial number, final number, initial body weight and final body weight of A. japonicus from each tank, respectively; DL and BL are the digestive tract length and whole body length of A. japonicus from each tank, respectively; DW, VW and BW are the digestive tract weight, viscera weight and body weight of A. japonicus from each tank, respectively; and Ne was the number of A. japonicus with evisceration of each tank.
The experimental data were analyzed by using software SPSS 22.0 (Redmond, WA, USA). One-way analysis of variance (ANOVA) was used to detect whether there is a significance among dietary groups (p < 0.05). Then, Duncan multiple comparisons was chosen to compare the differences in means between dietary groups. All data are presented in the form of means ± standard error (SE).
4. Discussion
When sea water temperature rises above 22 °C, A. japonicus can enter the state of aestivation. During aestivation, A. japonicus shows a complete cessation of feeding due to the degeneration of the digestive tract. When water temperature is below about 18 °C, A. japonicus is more active and gradually returns to normal food intake.
In this experiment, the WGR of experimental animals before aestivation increased significantly with the increase in Se-yeast addition. A recent study by our research team found that the WGR of early juvenile
A. japonicus with the initial body weight of about 0.1 g was increased by 0.5–1.0 mg/kg Se-yeast but was inhibited by an overdose (2 mg/kg) addition of Se-yeast [
2]. The discrepancy could be due to the different initial body weight of experimental animals, with larger individuals less sensitive to the equal dosage of Se-yeast. After aestivation, Se-yeast showed inhibitory effects on
A. japonicus as the addition level was equal to or higher than 1.5 mg/kg. It was commonly accepted that Se showed highly toxic effects on animals when the addition level exceeded the optimal dosage [
20,
21]. Indeed, the detrimental effects of Se have been reported on tilapia (
Oreochromis niloticus) if the addition level of Se was higher than 2 mg/kg [
22]. The digestive enzyme activities of grass carp (
Ctenopharyngodon idella) reached the maximum values by Se addition at the addition level of 0.6 mg/kg [
23]. However, juvenile pacu (
Piaractus mesopotamicus) showed no significant differences in the activities of digestive enzymes when the addition of Se-yeast ranged from 0.72 to 2.51 mg/kg [
24]. The inconsistence could be related to the differences in animal species, initial body weight and feed formulation. The results of the present study showed that the activities of digestive enzymes of
A. japonicus were promoted by Se-yeast at a moderate or relatively higher addition level (1.0–2.0 mg/kg) before aestivation, while the ideal activities of digestive enzymes were achieved by Se-yeast at the addition level of no more than 1.0 mg/kg. Thus, the optimal addition level of Se-yeast was estimated to be no more than 2.0 mg/kg before aestivation, and 1.0 mg/kg after aestivation for juvenile
A. japonicus from the perspective of digestive enzyme activities. It could be the previous accumulation of Se in the body wall before aestivation that accounted for the relatively lower requirement of Se after aestivation.
In this study, Se-yeast at moderate or relatively higher addition level (1.0–2.0 mg/kg) significantly increased the immune status of sea cucumbers before aestivation. This was consistent with the findings of Durigon et al. [
8], who found that dietary Se (Na
2 SeO
3) at an addition level of 0.86 mg/kg enhanced the antioxidant capacity of juvenile tilapia (
Oreochromis niloticus). However, the optimal addition level of Se-yeast was lower than the findings of Chen et al. [
25] and Zeng et al. [
19], who found that the highest immune and antioxidant capacity were achieved by 3–12 mg/kg Se-yeast and 5 mg/kg Se-met in the diets. It was previously found that the optimal Se-yeast addition level was 0.5–1.0 mg/kg for the survival and growth performance in juvenile
A. japonicus [
2]. Thus, the highest addition level of Se-yeast was only set at 2.0 mg/kg in this study. Thus, further studies are needed to investigate the effects of Se-yeast at the addition of more than 2.0 mg/kg on the immune and antioxidant status of
A. japonicus. Furthermore, the ER of was significantly inhibited by relatively higher addition level of Se-yeast (1.5–2.0 mg/kg) before aestivation. This indicated that the good immune status was beneficial for the intestinal health.
The body wall is the main edible part of
A. japonicus, with abundant nutrients and active metabolites [
26,
27]. In this study, Se-yeast addition did not significantly affect the crude protein before or after aestivation of
A. japonicus. During aestivation, the protein content of
A. japonicus showed an increasing tendency as the addition of Se-yeast increased in the diets. This could be due to the effects of Se on the hindered degradation of protein in the body wall of
A. japonicus. In this study, the contents of crude lipid in the body wall of
A. japonicus were markedly increased by the moderate addition level (1.0 mg/kg) of Se-yeast. Zhao et al. [
28] found that the lipid deposition in the pigs (
Susscrofa domestica) were promoted by an addition level of 3 mg/kg Se in the diets. In this study, the increasing addition of Se-yeast increased the contents of carbohydrates in the body wall of
A. japonicus during or after aestivation. It is possible that the addition of Se-yeast increased the contents of polysaccharide, which is one of the most important nutrients of
A. japonicus. However, the contents of polysaccharide were not assayed in this study. Thus, further research is needed to clarify this confusion in the future. Se addition has been verified to improve the nutritional value of edible part in several aquatic animals including
A. japonicus [
2], meagre (
Argyrosomus regius) [
12], common carp (
Cyprinus carpio) [
14] and grouper (
Epinephelus malabaricus) [
29]. In accordance,
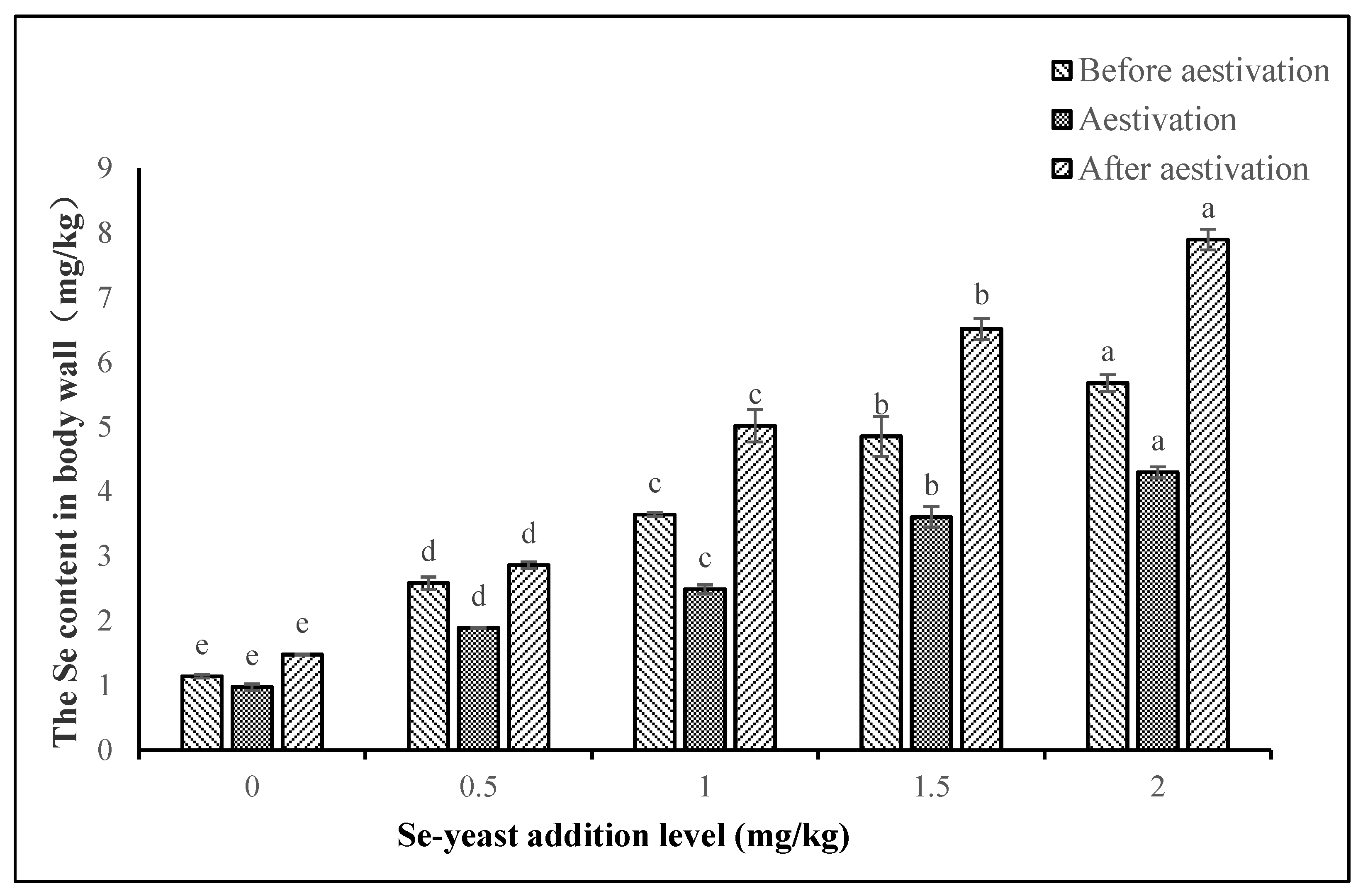
A. japonicus of this study showed an obviously retention of Se in the body wall as the addition level of Se-yeast increased. This further verified that application of Se-yeast was an effective strategy to enrich
A. japonicus with Se elements. During evisceration, the Se content decreased to some extent in the body wall of
A. japonicus. This indicated that some amount of Se was used for dealing with oxidative stress induced by high temperature.