Improved Hardness and Thermal Stability of Nanocrystalline Nickel Electrodeposited with the Addition of Cysteine
Abstract
:1. Introduction
- As demonstrated earlier, incorporating sulfur can reduce the grain size, and increase the hardness [7,8,33]. Sulfur may also yield a moderate strengthening during annealing at low temperatures (between 400 K and 600 K) [9,24]. Cysteine contains sulfur and can easily decompose into sulfur-containing components [34]. Since the sulfur atom in the cysteine molecule is relatively weakly bound as compared to the most common additive saccharin, cysteine can be a good candidate as sulfur-donor bath additive.
- Additives with –SH functional groups (i.e., thiols) tend to adsorb on metal surfaces with the sulfur atom as the anchoring entity of the adsorbed molecule. Thiols can even form monolayers that block the direct interaction between the metal and the solution. This phenomenon has a rich literature [35,36,37,38,39], especially for noble metals like Au and Ag. Although this “organic shield” does not completely block the electron transfer between the metal and the reactive solute species, the adsorbed layer inhibits the growth of the already existing grains of the deposit and promotes the formation of new nucleation sites [7]. It has been well evidenced that the metal deposition can take place through the defect sites of adsorbed thiol layers [35,36,37,38,39]. Additionally, the interaction of the polar/ionic groups of the cysteine molecule (i.e., the amino and the carboxylate groups) can stabilize the solution side of the adsorbed layer by pinning the adsorbed molecules with zwitterion formation.
2. Materials and Methods
2.1. Electrodeposition of Nickel Samples
2.2. Heat Treatment
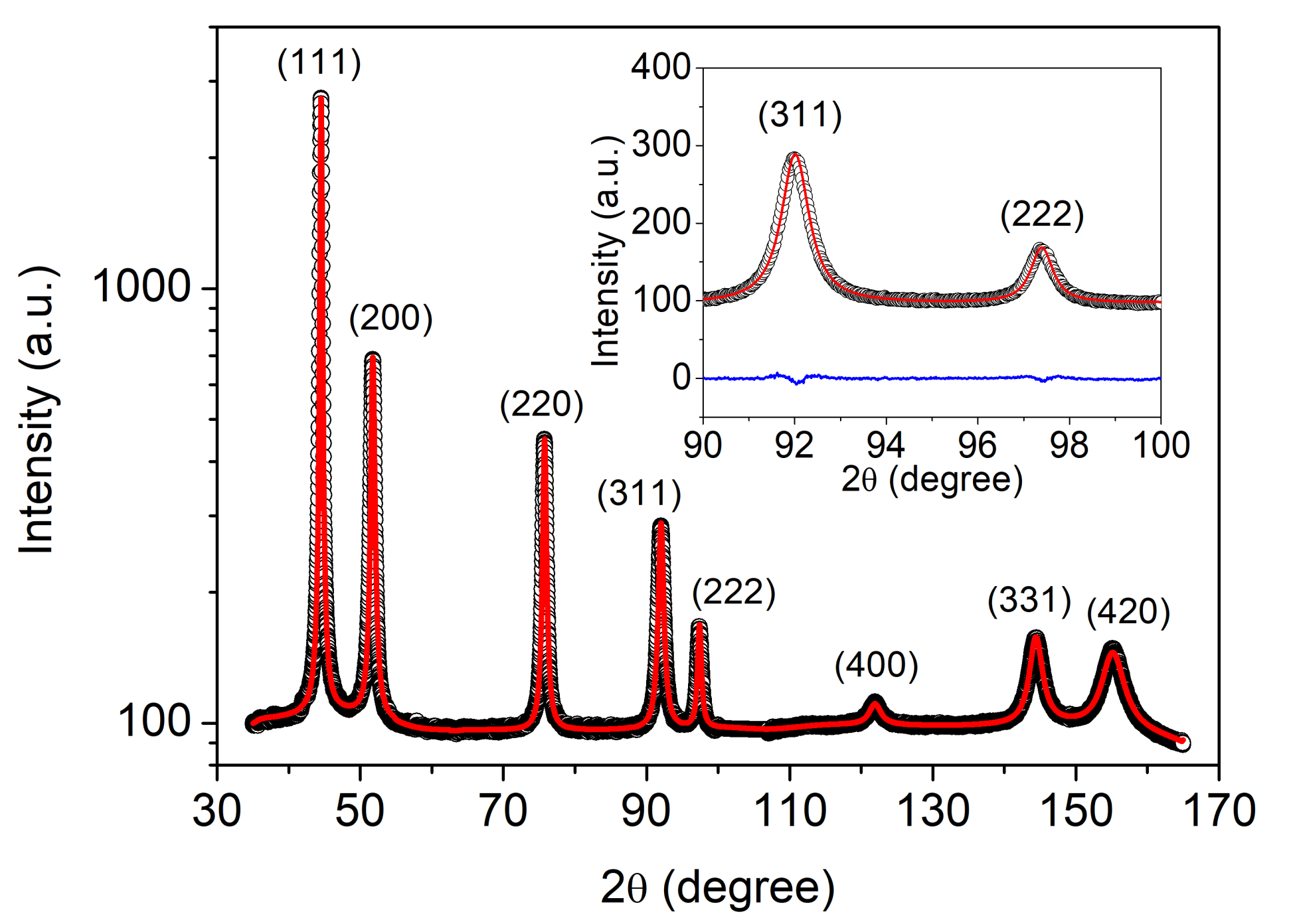
2.3. X-ray Diffraction
2.4. Transmission Electron Microscopy
2.5. Scanning Electron Microscopy and Energy-Dispersive X-ray Spectroscopy
2.6. Hardness Tests
3. Results
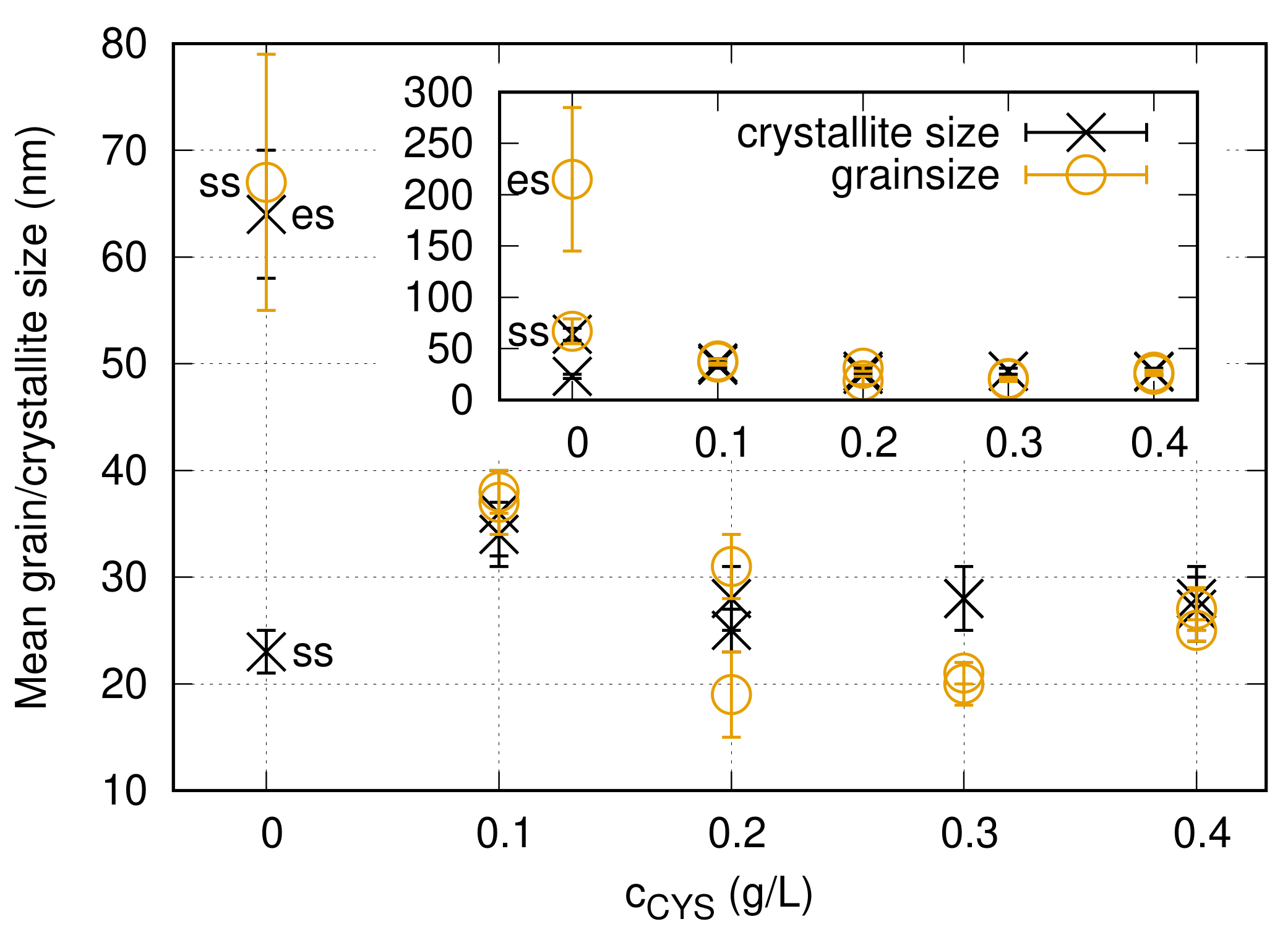
3.1. The Effect of Cysteine Addition on the Chemical Composition and the Microstructure
3.2. Hardness Evolution with Increasing Cysteine Content
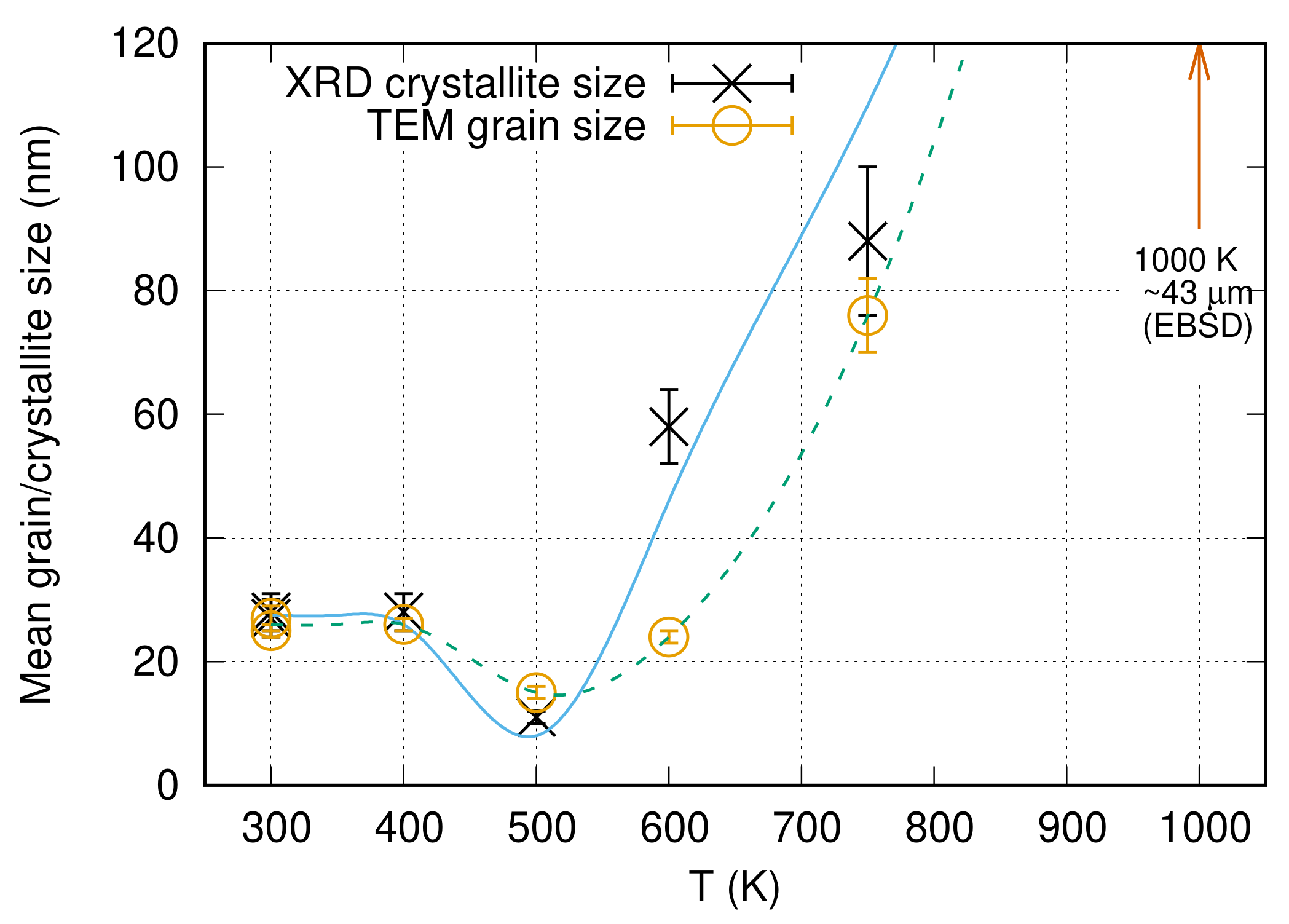
3.3. Thermal Stability of the Microstructure in the Ni Film Processed with the Cysteine Content of 0.4 g/L
3.4. Change of the Hardness During Annealing of the Ni Deposited With Cysteine Additive
4. Discussion
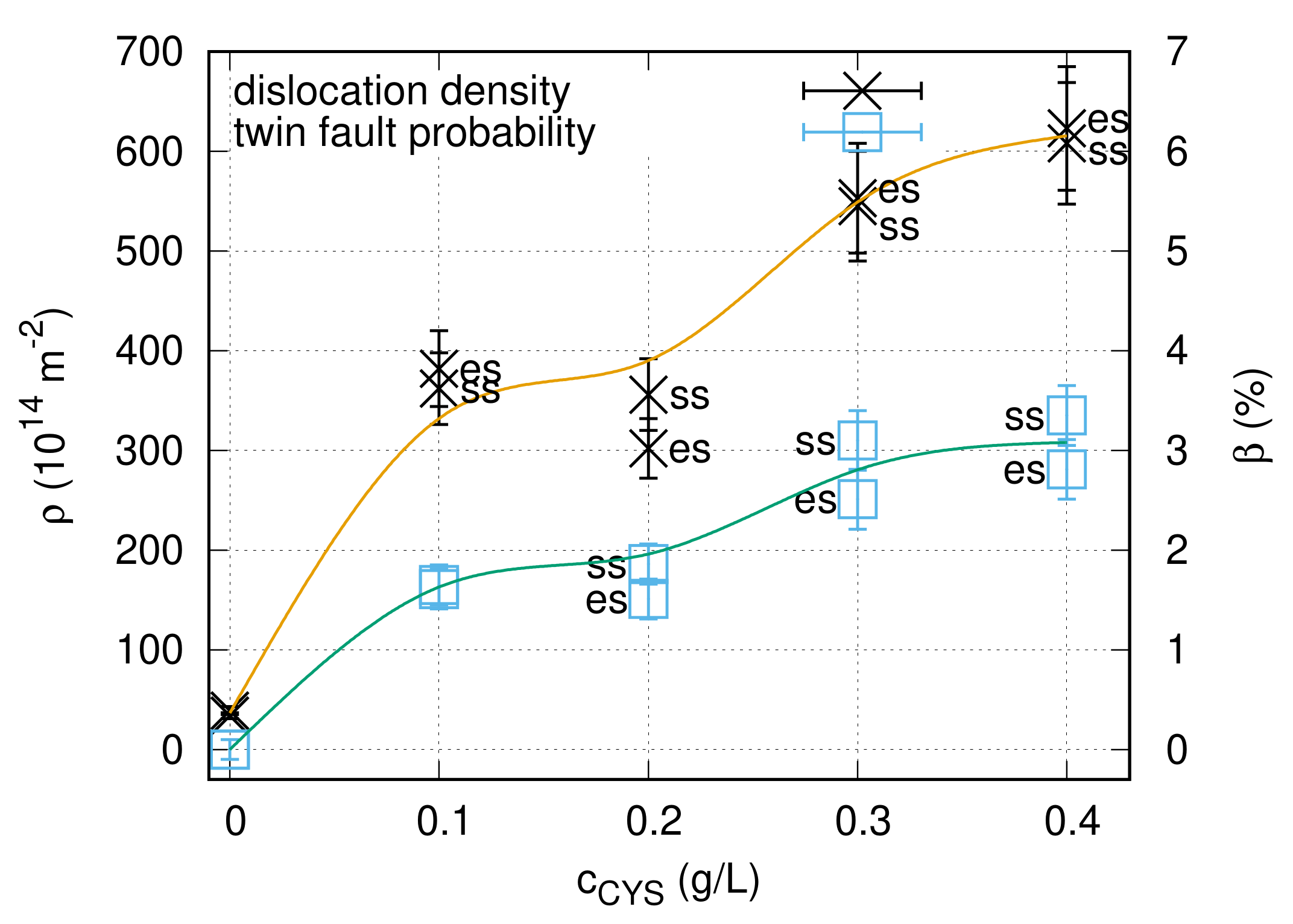
4.1. Effect of Cysteine Addition on the Texture, Grain Size and Defect Density
4.2. Influence of Concentration of Cysteine on the Hardness
4.3. Effect of Cysteine on the Thermal Stability
5. Conclusions
- The optimal parameters for electrodeposition of nickel from a cysteine-containing bath differ from the ones used for deposition from a conventional nickel-sulfate based bath. A nearly neutral pH with the value of 6.1, a high current density of –25 mA/cm and an increased concentration of boric acid (30 g/L) in the bath are recommended for obtaining nice deposits with cysteine.
- With increasing the cysteine content up to 0.3 g/L, the density of lattice defects (dislocations and twin faults) increased, while the grain size decreased in the Ni films. In addition, the (220) texture in the additive-free Ni layer changed to (200) texture. When the cysteine content was enhanced from 0.3 to 0.4 g/L, further significant variation in the microstructure was not observed.
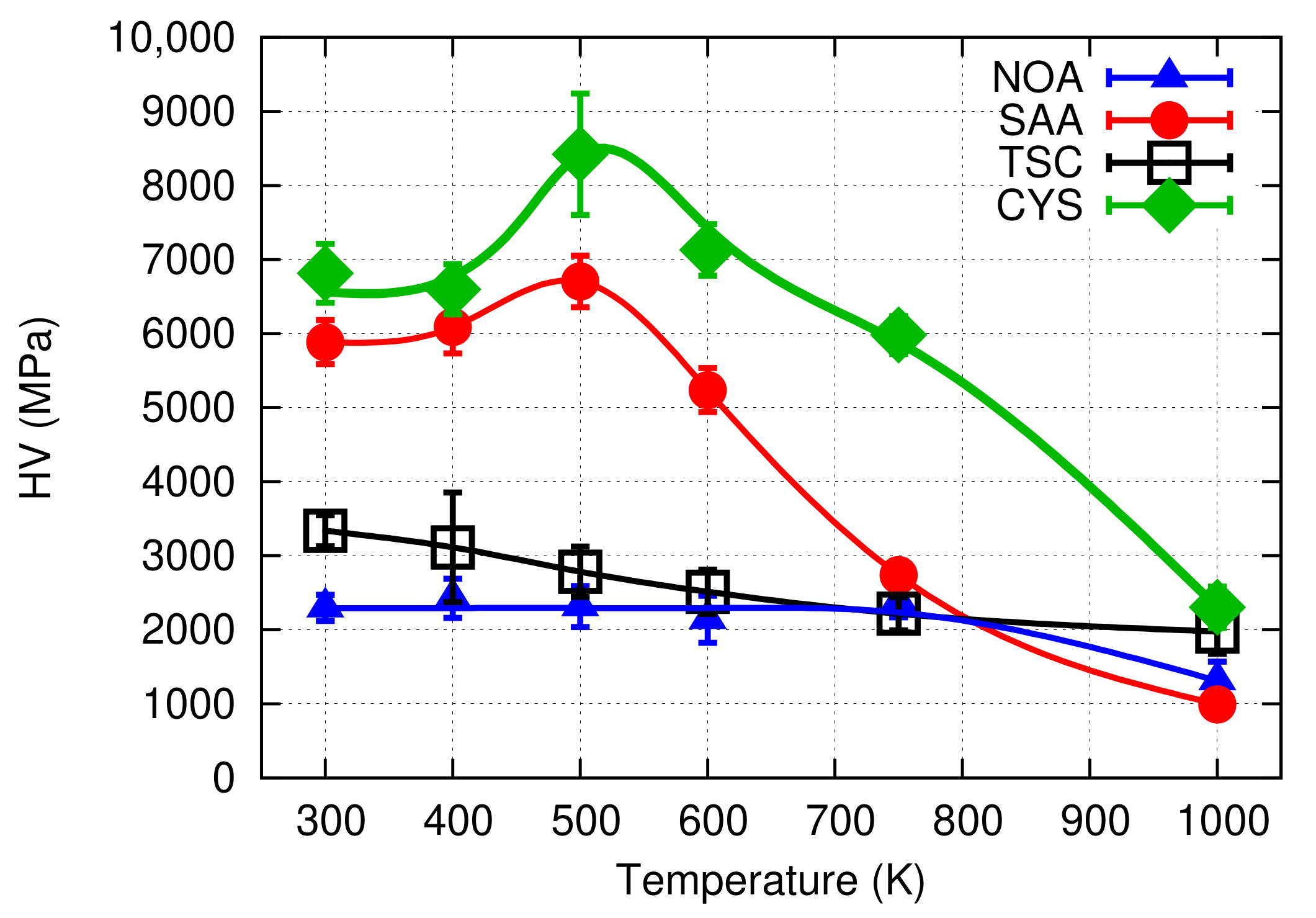
- The Ni film obtained with the addition of 0.4 g/L cysteine exhibited a higher hardness (∼6800 MPa) than the values reported for other additives in the literature. The thermal stability of the nanostructured Ni film fabricated with cysteine was exceptional; namely, the hardness of the Ni layer deposited from cysteine-containing bath remained as high as ∼6000 MPa, even after heating up to 750 K. The relatively large hardness was retained upon annealing even at 1000 K, while the (111) texture became stronger.
- When the Ni film processed with cysteine was heated up to 500 K, the grain size decreased from ∼27 to ∼15 nm and the hardness increased to ∼8400 MPa, which is an exceptionally high value among Ni electrodeposits. This very high hardness might be attributed rather to the structural changes in the grain boundaries and the strengthening of the (111) texture than to the reduction of the grain size.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kumar, K.S.; Suresh, S.; Chisholm, M.F.; Horton, J.A.; Wang, P. Deformation of electrodeposited nanocrystalline nickel. Acta Mater. 2003, 51, 387–405. [Google Scholar] [CrossRef]
- Torre, F.D.; Swygenhoven, H.V.; Victoria, M. Nanocrystalline electrodeposited Ni: Microstructure and tensile properties. Acta Mater. 2002, 50, 3957–3970. [Google Scholar] [CrossRef]
- Budrovic, Z.; Swygenhoven, H.V.; Derlet, P.M.; Petegem, S.V.; Schmitt, B. Plastic Deformation with Reversible Peak Broadening in Nanocrystalline Nickel. Science 2004, 304, 273–276. [Google Scholar] [CrossRef] [PubMed]
- Schwaiger, R.; Moser, B.; Dao, M.; Chollacoop, N.; Suresh, S. Some critical experiments on the strain-rate sensitivity of nanocrystalline nickel. Acta Mater. 2003, 51, 5159–5172. [Google Scholar] [CrossRef]
- Meyers, M.A.; Mishra, A.; Benson, D.J. Mechanical properties of nanocrystalline materials. Prog. Mater. Sci. 2006, 51, 427–556. [Google Scholar] [CrossRef]
- Kumar, K.S.; Swygenhoven, H.V.; Suresh, S. Mechanical behavior of nanocrystalline metals and alloys. Acta Mater. 2003, 51, 5743–5774. [Google Scholar] [CrossRef]
- Kolonits, T.; Jenei, P.; Tóth, B.G.; Czigány, Z.; Gubicza, J.; Péter, L.; Bakonyi, I. Characterization of Defect Structure in Electrodeposited Nanocrystalline Ni Films. J. Electrochem. Soc. 2016, 163, D107–D114. [Google Scholar] [CrossRef] [Green Version]
- Kolonits, T.; Jenei, P.; Péter, L.; Bakonyi, I.; Czigány, Z.; Gubicza, J. Effect of bath additives on the microstructure, lattice defect density and hardness of electrodeposited nanocrystalline Ni films. Surf. Coat. Technol. 2018, 349, 611–621. [Google Scholar] [CrossRef]
- Kolonits, T.; Czigány, Z.; Péter, L.; Bakonyi, I.; Gubicza, J. Influence of Bath Additives on the Thermal Stability of the Nanostructure and Hardness of Ni Films Processed by Electrodeposition. Coatings 2019, 9, 644. [Google Scholar] [CrossRef] [Green Version]
- Gertsman, V.Y.; Birringer, R. On the room-temperature grain growth in nanocrystalline copper. Scr. Metall. Mater. 1994, 30, 577–581. [Google Scholar] [CrossRef]
- Klement, U.; Erb, U.; El-Sherik, A.M.; Aust, K.T. Thermal stability of nanocrystalline Ni. Mater. Sci. Eng. A 1995, 203, 177–186. [Google Scholar] [CrossRef]
- Dake, J.M.; Krill, C.E. Sudden loss of thermal stability in Fe-based nanocrystalline alloys. Scr. Mater. 2012, 66, 390–393. [Google Scholar] [CrossRef]
- Ames, M.; Markmann, J.; Karos, R.; Michels, A.; Tschope, A.; Birringer, R. Unraveling the nature of room temperature grain growth in nanocrystalline materials. Acta Mater. 2008, 56, 4255–4266. [Google Scholar] [CrossRef]
- Boylan, K.; Ostrander, D.; Erb, U.; Palumbo, G.; Aust, K.T. An in situ TEM study of the thermal stability of nanocrystalline NiP. Scr. Metall. Mater. 1991, 25, 2711–2716. [Google Scholar] [CrossRef]
- Chookajorn, T.; Murdoch, H.A.; Schuh, C.A. Design of stable nanocrystalline alloys. Science 2012, 337, 951–954. [Google Scholar] [CrossRef] [Green Version]
- Darling, K.A.; VanLeeuwen, B.K.; Semones, J.E.; Koch, C.C.; Scattergood, R.O.; Kecskes, L.J.; Mathaudhu, S.N. Stabilized nanocrystalline iron-based alloys: Guiding efforts in alloy selection. Mater. Sci. Eng. A 2011, 528, 4365–4371. [Google Scholar] [CrossRef]
- Choi, P.; da Silva, M.; Klement, U.; Al-Kassab, T.; Kirchheim, R. Thermal stability of electrodeposited nanocrystalline Co-1.1at.%P. Acta Mater. 2005, 53, 4473–4481. [Google Scholar] [CrossRef]
- Chookajorn, T.; Schuh, C.A. Nanoscale segregation behavior and high-temperature stability for nanocrystalline W–20 at.% Ti. Acta Mater. 2014, 73, 128–138. [Google Scholar] [CrossRef] [Green Version]
- Darling, K.A.; VanLeeuween, B.K.; Koch, C.C.; Scattergood, R.O. Thermal stability of nanocrystalline Fe–Zr alloys. Mater. Sci. Eng. A 2010, 527, 3572–3580. [Google Scholar] [CrossRef]
- Burzyńska, L.; Rudnik, E.; Koza, J.; Błaż, L.; Szymański, W. Electrodeposition and heat treatment of nickel/silicon carbide composites. Surf. Coat. Technol. 2008, 202, 2545–2556. [Google Scholar] [CrossRef]
- Broszeit, E. Mechanical, thermal and tribological properties of electro- and chemodeposited composite coatings. Thin Solid Films 1982, 95, 133–142. [Google Scholar] [CrossRef]
- Gyftou, P.; Stroumbouli, M.; Pavlatou, E.A.; Spyrellis, N. Electrodeposition of Ni/SiC Composites by Pulse Electrolysis. Trans. IMF 2002, 80, 88–91. [Google Scholar] [CrossRef]
- Wang, S.C.; Wei, W.C.J. Characterization of electroplated Ni/SiC and Ni/Al2O3 composite coatings bearing nanoparticles. J. Mater. Res. 2003, 18, 1566–1574. [Google Scholar] [CrossRef]
- Wang, Y.M.; Cheng, S.; Wei, Q.M.; Ma, E.; Nieh, T.G.; Hamza, A. Effects of annealing and impurities on tensile properties of electrodeposited nanocrystalline Ni. Scr. Mater. 2004, 51, 1023–1028. [Google Scholar] [CrossRef]
- Heuer, J.K.; Okamoto, P.R.; Lam, N.Q.; Stubbins, J.F. Relationship between segregation-induced intergranular fracture and melting in the nickel–sulfur system. Appl. Phys. Lett. 2000, 76, 3403–3405. [Google Scholar] [CrossRef]
- Hibbard, G.D.; McCrea, J.L.; Palumbo, G.; Aust, K.T.; Erb, U. An initial analysis of mechanisms leading to late stage abnormal grain growth in nanocrystalline Ni. Scr. Mater. 2002, 47, 83–87. [Google Scholar] [CrossRef]
- Xiao, C.; Mirshams, R.A.; Whang, S.H.; Yin, W.M. Tensile behavior and fracture in nickel and carbon doped nanocrystalline nickel. Mater. Sci. Eng. A 2001, 301, 35–43. [Google Scholar] [CrossRef]
- Lücke, K.; Detert, K. A quantitative theory of grain-boundary motion and recrystallization in metals in the presence of impurities. Acta Metall. 1957, 5, 628–637. [Google Scholar] [CrossRef]
- Michels, A.; Krill, C.E.; Ehrhardt, H.; Birringer, R.; Wu, D.T. Modeling the influence of grain-size-dependent solute drag on the kinetics of grain growth in nanocrystalline materials. Acta Mater. 1999, 47, 2143–2152. [Google Scholar] [CrossRef]
- Hillert, M. Inhibition of grain growth by second-phase particles. Acta Metall. 1998, 36, 3177–3181. [Google Scholar] [CrossRef]
- Gottstein, G.; Shvindlerman, L.S. Grain Boundary Migration in Metals: Thermodynamics, Kinetics, Applications, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2010. [Google Scholar]
- Hondros, E.D.; Seah, M.P.; Hofmann, S.; Lejcek, P. Interfacial and surface microchemistry. In Physical Metallurgy Fourth Edn; Elsevier: North-Holland, The Netherlands, 1996; pp. 1202–1289. [Google Scholar]
- Bicelli, L.; Bozzini, B.; Mele, C.; D’Urzo, L. A Review of Nanostructural Aspects of Metal Electrodeposition. Int. J. Electrochem. Sci. 2008, 3, 356–408. [Google Scholar]
- Kouncheva, M.; Raichevski, G.; Vitkova, S.; Prazak, M. The effect of sulphur and carbon inclusions on the corrosion resistance of electrodeposited Ni-Fe alloy coatings. Surf. Coat. Technol. 1987, 31, 137–142. [Google Scholar] [CrossRef]
- Hagenström, H.; Schneeweiss, M.; Kolb, D. Modification of a Au (111) electrode with ethanethiol. 2. Copper electrodeposition. Langmuir 1999, 15, 7802–7809. [Google Scholar] [CrossRef]
- Petri, M.; Kolb, D.M.; Memmert, U.; Meyer, H. Adsorption of mercaptopropionic acid onto Au(1 1 1): Part II. Effect on copper electrodeposition. Electrochim. Acta 2003, 49, 183–189. [Google Scholar] [CrossRef]
- Langerock, S.; Ménard, H.; Rowntree, P.; Heerman, L. Electrocrystallization of Rhodium clusters on thiolate-covered polycrystalline gold. Langmuir 2005, 21, 5124–5133. [Google Scholar] [CrossRef]
- Pattanaik, G.; Shao, W.; Swami, N.; Zangari, G. Electrolytic Gold Deposition on Dodecanethiol-Modified Gold Films. Langmuir 2009, 25, 5031–5038. [Google Scholar] [CrossRef]
- Nisanci, F.B.; Demir, U. Size-Controlled Electrochemical Growth of PbS Nanostructures into Electrochemically Patterned Self-Assembled Monolayers. Langmuir 2012, 28, 8571–8578. [Google Scholar] [CrossRef]
- Ross, S.A.; Burrows, C.J. Nickel Complexes of Cysteine- and Cystine-Containing Peptides: Spontaneous Formation of Disulfide-Bridged Dimers at Neutral pH. Inorg. Chem. 1998, 37, 5358–5363. [Google Scholar] [CrossRef]
- Cox, J.A.; Gray, T.J. Controlled-potential electrolysis of bulk solutions at a modified electrode: Application to oxidations of cysteine, cystine, methionine, and thiocyanate. Anal. Chem. 1990, 62, 2742–2744. [Google Scholar] [CrossRef]
- Chen, D.; Giroud, F.; Minteer, S.D. Nickel Cysteine Complexes as Anodic Electrocatalysts for Fuel Cells. J. Electrochem. Soc. 2014, 161, F933–F939. [Google Scholar] [CrossRef] [Green Version]
- Matos, J.; Pereira, L.; Agostinho, S.; Barcia, O.; Cordeiro, G.; D’Elia, E. Effect of cysteine on the anodic dissolution of copper in sulfuric acid medium. J. Electroanal. Chem. 2004, 570, 91–94. [Google Scholar] [CrossRef]
- Silva, F.; Do Lago, D.; D’Elia, E.; Senna, L. Electrodeposition of Cu–Zn alloy coatings from citrate baths containing benzotriazole and cysteine as additives. J. Appl. Electrochem. 2010, 40, 2013–2022. [Google Scholar] [CrossRef]
- El-Deab, M.S.; Arihara, K.; Ohsaka, T. Fabrication of Au(111)-Like Polycrystalline Gold Electrodes and Their Applications to Oxygen Reduction. J. Electrochem. Soc. 2004, 151, E213–E218. [Google Scholar] [CrossRef]
- Dolati, A.; Imanieh, I.; Salehi, F.; Farahani, M. The effect of cysteine on electrodeposition of gold nanoparticle. Mater. Sci. Eng. B 2011, 176, 1307–1312. [Google Scholar] [CrossRef]
- El-Deab, M.S.; Sotomura, T.; Ohsaka, T. Size and Crystallographic Orientation Controls of Gold Nanoparticles Electrodeposited on GC Electrodes. J. Electrochem. Soc. 2005, 152, C1–C6. [Google Scholar] [CrossRef]
- Lin, T.H.; Lin, C.W.; Liu, H.H.; Sheu, J.T.; Hung, W.H. Potential-controlled electrodeposition of gold dendrites in the presence of cysteine. Chem. Commun. 2011, 47, 2044–2046. [Google Scholar] [CrossRef]
- Ebadi, M.; Basirun, W.J.; Sim, Y.L.; Mahmoudian, M.R. Investigation of Electro-Kinetic Behavior of Cysteine on Electrodeposition of Ni Through the AC and DC Techniques. Metall. Mater. Trans. A 2013, 44, 5096–5105. [Google Scholar] [CrossRef]
- Tóth, B.G.; Péter, L.; Révész, A.; Pádár, J.; Bakonyi, I. Temperature dependence of the electrical resistivity and the anisotropic magnetoresistance (AMR) of electrodeposited Ni-Co alloys. Eur. Phys. J. B 2010, 75, 167–177. [Google Scholar] [CrossRef] [Green Version]
- Weihnacht, V.; Péter, L.; Tóth, J.; Pádár, J.; Kerner, Z.; Schneider, C.M.; Bakonyi, I. Giant Magnetoresistance in Co-Cu/Cu Multilayers Prepared by Various Electrodeposition Control Modes. J. Electrochem. Soc. 2003, 150, C507. [Google Scholar] [CrossRef]
- Ribárik, G.; Gubicza, J.; Ungár, T. Correlation between strength and microstructure of ball-milled Al–Mg alloys determined by X-ray diffraction. Mater. Sci. Eng. A 2004, 387, 343–347. [Google Scholar] [CrossRef]
- Balogh, L.; Ribárik, G.; Ungár, T. Stacking faults and twin boundaries in fcc crystals determined by x-ray diffraction profile analysis. J. Appl. Phys. 2006, 100, 1–10. [Google Scholar] [CrossRef]
- Barna, A. Topographic kinetics and practice of low angle ion beam thinning. Mater. Res. Soc. Symp. Proc. 1992, 254, 3–22. [Google Scholar] [CrossRef]
- Pearson, K.X. On the criterion that a given system of deviations from the probable in the case of a correlated system of variables is such that it can be reasonably supposed to have arisen from random sampling. Lond. Edinb. Dublin Philos. Mag. J. Sci. Ser. 5 1900, 50, 157–175. [Google Scholar] [CrossRef] [Green Version]
- Arihara, K.; Ariga, T.; Takashima, N.; Arihara, K.; Okajima, T.; Kitamura, F.; Tokuda, K.; Ohsaka, T. Multiple voltammetric waves for reductive desorption of cysteine and 4-mercaptobenzoic acid monolayers self-assembled on gold substrates. Phys. Chem. Chem. Phys. 2003, 5, 3758–3761. [Google Scholar] [CrossRef]
- Solomon, E.I.; Gorelsky, S.I.; Dey, A. Metal–thiolate bonds in bioinorganic chemistry. J. Comput. Chem. 2006, 27, 1415–1428. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Li, X.; Li, H.; Zhang, W. Quantifying thiol–gold interactions towards the efficient strength control. Nat. Commun. 2014, 5, 4348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pakiari, A.; Jamshidi, Z. Nature and strength of M-S Bonds (M = Au, Ag, and Cu) in binary alloy gold clusters. J. Phys. Chem. A 2010, 114, 9212–9221. [Google Scholar] [CrossRef] [PubMed]
- Neurock, M.; van Santen, R.A. Theory of carbon-sulfur bond activation by small metal sulfide particles. J. Am. Chem. Soc. 1994, 116, 4427–4439. [Google Scholar] [CrossRef] [Green Version]
- Ulman, A. Formation and Structure of Self-Assembled Monolayers. Chem. Rev. 1996, 96, 1533–1554. [Google Scholar] [CrossRef]
- Reddy, A. Preferred orientations in nickel electro-deposits: I. The mechanism of development of textures in nickel electro-deposits. J. Electroanal. Chem. (1959) 1963, 6, 141–152. [Google Scholar] [CrossRef]
- Li, D.Y.; Szpunar, J.A. Textural evolution in electrodeposits under the influence of adsorbed foreign species: Part I Textural evolution in iron electrodeposits affected by hydrogen co-deposition. J. Mater. Sci. 1997, 32, 141–152. [Google Scholar] [CrossRef]
- Kristinsdóttir, L.; Skúlason, E. A systematic DFT study of hydrogen diffusion on transition metal surfaces. Surf. Sci. 2012, 606, 1400–1404. [Google Scholar] [CrossRef]
- Ferrin, P.; Kandoi, S.; Nilekar, A.U.; Mavrikakis, M. Hydrogen adsorption, absorption and diffusion on and in transition metal surfaces: A DFT study. Surf. Sci. 2012, 606, 679–689. [Google Scholar] [CrossRef]
- Yang, H.; Whitten, J.L. Adsorption of SH and OH and coadsorption of S, O and H on Ni(111). Surf. Sci. 1997, 370, 136–154. [Google Scholar] [CrossRef]
- Gubicza, J. Defect Structure and Properties of Nanomaterials; Woodhead Publishing: Sawston, UK, 2017. [Google Scholar]
- Clausen, B.; Lorentzen, T.; Leffers, T. Self-consistent modelling of the plastic deformation of f.c.c. polycrystals and its implications for diffraction measurements of internal stresses. Acta Mater. 1998, 46, 3087–3098. [Google Scholar] [CrossRef]
- Feng, L.; Ren, Y.Y.; Zhang, Y.H.; Wang, S.; Li, L. Direct Correlations among the Grain Size, Texture, and Indentation Behavior of Nanocrystalline Nickel Coatings. Metals 2019, 9, 188. [Google Scholar] [CrossRef] [Green Version]
- Hu, J.; Shi, Y.N.; Sauvage, X.; Sha, G.; Lu, K. Grain boundary stability governs hardening and softening in extremely fine nanograined metals. Science 2017, 355, 1292–1296. [Google Scholar] [CrossRef]
- Zhang, X.; Fujita, T.; Pan, D.; Yu, J.; Sakurai, T.; Chen, M. Influences of grain size and grain boundary segregation on mechanical behavior of nanocrystalline Ni. Mater. Sci. Eng. A 2010, 527, 2297–2304. [Google Scholar] [CrossRef]
- Chang, L.; Kao, P.; Chen, C.H. Strengthening mechanisms in electrodeposited Ni–P alloys with nanocrystalline grains. Scr. Mater. 2007, 56, 713–716. [Google Scholar] [CrossRef]
- Zhang, N.; Jin, S.; Sha, G.; Yu, J.; Cai, X.; Du, C.; Shen, T. Segregation induced hardening in annealed nanocrystalline Ni-Fe alloy. Mater. Sci. Eng. A 2018, 735, 354–360. [Google Scholar] [CrossRef]
- Ebrahimi, F.; Li, H. The effect of annealing on deformation and fracture of a nanocrystalline fcc metal. J. Mater. Sci. 2007, 42, 1444–1454. [Google Scholar] [CrossRef]
- Li, H.; Jiang, F.; Ni, S.; Li, L.; Sha, G.; Liao, X.; Ringer, S.P.; Choo, H.; Liaw, P.K.; Misra, A. Mechanical behaviors of as-deposited and annealed nanostructured Ni–Fe alloys. Scr. Mater. 2011, 65, 1–4. [Google Scholar] [CrossRef]
- Haj-Taieb, M.; Haseeb, A.S.M.A.; Caulfield, J.; Bade, K.; Aktaa, J.; Hemker, K.J. Thermal stability of electrodeposited LIGA Ni-W alloys for high temperature MEMS applications. Microsyst. Technol. 2008, 14, 1531–1536. [Google Scholar] [CrossRef]
- Rupert, T.J.; Trelewicz, J.R.; Schuh, C.A. Grain boundary relaxation strengthening of nanocrystalline Ni–W alloys. J. Mater. Res. 2012, 27, 1285–1294. [Google Scholar] [CrossRef]
- Gubicza, J. Annealing-Induced Hardening in Ultrafine-Grained and Nanocrystalline Materials. Adv. Eng. Mater. 2020, 22, 1900507. [Google Scholar] [CrossRef] [Green Version]
- Vo, N.; Schäfer, J.; Averback, R.; Albe, K.; Ashkenazy, Y.; Bellon, P. Reaching theoretical strengths in nanocrystalline Cu by grain boundary doping. Scr. Mater. 2011, 65, 660–663. [Google Scholar] [CrossRef]
- Weissmüller, J. Alloy effects in nanostructures. Nanostruct. Mater. 1993, 3, 261–272. [Google Scholar] [CrossRef]
- Donald, A.; Brown, L. Grain boundary faceting in Cu-Bi alloys. Acta Metallurgica 1979, 27, 59–66. [Google Scholar] [CrossRef]
- Nieh, T.; Wang, J. Hall–Petch relationship in nanocrystalline Ni and Be–B alloys. Intermetallics 2005, 13, 377–385. [Google Scholar] [CrossRef]









| Component | Concentration (g/L) | Role |
|---|---|---|
| NiSO6H2O | 155 | Ni source |
| H3BO3 | 30 | pH-buffer |
| NaSO4·10H2O | 96 | Supporting electrolyte |
| H2NSO3H | 10 | wetting agent |
| Cysteine HSCH2CH(NH2)COOHS | 0; 0.1; 0.2; 0.3; 0.4 | additive |
| pH Current density | 6.1 −25 mA/cm2 |
| Concentration (g/L) | Side | <x> (nm) | () | (%) | d (nm) | HV (MPa) |
|---|---|---|---|---|---|---|
| 0 | es | 64 ± 6 | 34 ± 4 | 0 ± 0.1 | 215 ± 70 | 2800 ± 350 |
| 0 | ss | 23 ± 3 | 39 ± 5 | 0 ± 0.1 | 67 ± 12 | 3700 ± 180 |
| 0.1 | es | 34 ± 3 | 380 ± 40 | 1.6 ± 0.2 | 37 ± 3 | 5100 ± 270 |
| 0.1 | ss | 36 ± 4 | 360 ± 40 | 1.6 ± 0.2 | 38 ± 2 | 5000 ± 280 |
| 0.2 | es | 25 ± 3 | 300 ± 30 | 1.5 ± 0.2 | 19 ± 4 | 6700 ± 490 |
| 0.2 | ss | 28 ± 3 | 360 ± 40 | 1.9 ± 0.2 | 31 ± 3 | 5900 ± 340 |
| 0.3 | es | 28 ± 3 | 550 ± 60 | 2.5 ± 0.3 | 21 ± 3 | 5800 ± 170 |
| 0.3 | ss | 28 ± 3 | 550 ± 60 | 3.1 ± 0.3 | 20 ± 3 | 6100 ± 340 |
| 0.4 | es | 27 ± 3 | 620 ± 70 | 2.8 ± 0.3 | 27 ± 3 | 6800 ± 400 |
| 0.4 | ss | 28 ± 3 | 610 ± 60 | 3.3 ± 0.3 | 25 ± 3 | 6500 ± 160 |
| Sample | Boric Acid (g/L) | pH | Curr. Dens. (mA/cm) | <x> (nm) | () | (%) | d (nm) | HV (MPa) | Texture |
|---|---|---|---|---|---|---|---|---|---|
| NOA-es | 15 | 3.25 | −6.25 | 42 ± 5 | 12 ± 1 | 0 ± 0.1 | 127 ± 45 | 1877 ± 211 | (220) |
| NOA-ss | 15 | 3.25 | −6.25 | 27 ± 3 | 14 ± 2 | 0 ± 0.1 | 91 ± 14 | 2837 ± 344 | NA |
| CYS00-es | 30 | 6.1 | −25 | 64 ± 6 | 34 ± 4 | 0 ± 0.1 | 215 ± 70 | 2800 ± 350 | (220) |
| CYS00-ss | 30 | 6.1 | −25 | 23 ± 3 | 39 ± 5 | 0 ± 0.1 | 67 ± 12 | 3700 ± 180 | NA |
| Temperature (K) | <x> (nm) | () | (%) | d (nm) | HV (MPa) |
|---|---|---|---|---|---|
| 300 | 27 ± 3 | 620 ± 70 | 2.8 ± 0.3 | 27 ± 3 | 6800 ± 400 |
| 400 | 28 ± 3 | 720 ± 70 | 3.1 ± 0.3 | 26 ± 2 | 6600 ± 340 |
| 500 | 11 ± 2 | 570 ± 60 | 2.2 ± 0.2 | 15 ± 2 | 8400 ± 820 |
| 600 | 58 ± 6 | 340 ± 40 | 1.5 ± 0.2 | 24 ± 2 | 7100 ± 350 |
| 750 | 88 ± 14 | 91 ± 10 | 0 ± 0.1 | 76 ± 6 | 6000 ± 260 |
| 1000 | NA | NA | NA | 43,000 ±6600 | 2300 ± 280 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kolonits, T.; Czigány, Z.; Péter, L.; Bakonyi, I.; Gubicza, J. Improved Hardness and Thermal Stability of Nanocrystalline Nickel Electrodeposited with the Addition of Cysteine. Nanomaterials 2020, 10, 2254. https://doi.org/10.3390/nano10112254
Kolonits T, Czigány Z, Péter L, Bakonyi I, Gubicza J. Improved Hardness and Thermal Stability of Nanocrystalline Nickel Electrodeposited with the Addition of Cysteine. Nanomaterials. 2020; 10(11):2254. https://doi.org/10.3390/nano10112254
Chicago/Turabian StyleKolonits, Tamás, Zsolt Czigány, László Péter, Imre Bakonyi, and Jenő Gubicza. 2020. "Improved Hardness and Thermal Stability of Nanocrystalline Nickel Electrodeposited with the Addition of Cysteine" Nanomaterials 10, no. 11: 2254. https://doi.org/10.3390/nano10112254
APA StyleKolonits, T., Czigány, Z., Péter, L., Bakonyi, I., & Gubicza, J. (2020). Improved Hardness and Thermal Stability of Nanocrystalline Nickel Electrodeposited with the Addition of Cysteine. Nanomaterials, 10(11), 2254. https://doi.org/10.3390/nano10112254








