Interrelations of Synthesis Method, Polyethylene Glycol Coating, Physico-Chemical Characteristics, and Antimicrobial Activity of Silver Nanoparticles
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation
2.1.1. Chemical Synthesis of AgNPs
2.1.2. Aloe vera Extract Preparation
2.1.3. Biological Synthesis of AgNPs Using Aloe vera Extract
2.1.4. Surface Coating of AgNPs with Polyethylene Glycol (PEG)
2.2. Characterisations of AgNPs
2.2.1. UV-Visible Spectrophotometry Analysis
2.2.2. High Resolution—Transmission Electron Microscopy (HR-TEM) Analysis
2.2.3. Fourier-Transform Infrared Spectroscopy (FTIR) Analysis
2.2.4. Dynamic Light Scattering (DLS) and Zeta Potential (ZP) Measurements
2.2.5. AgNPs’ Antimicrobial Activity Evaluation
2.3. Statistical Analysis
3. Results
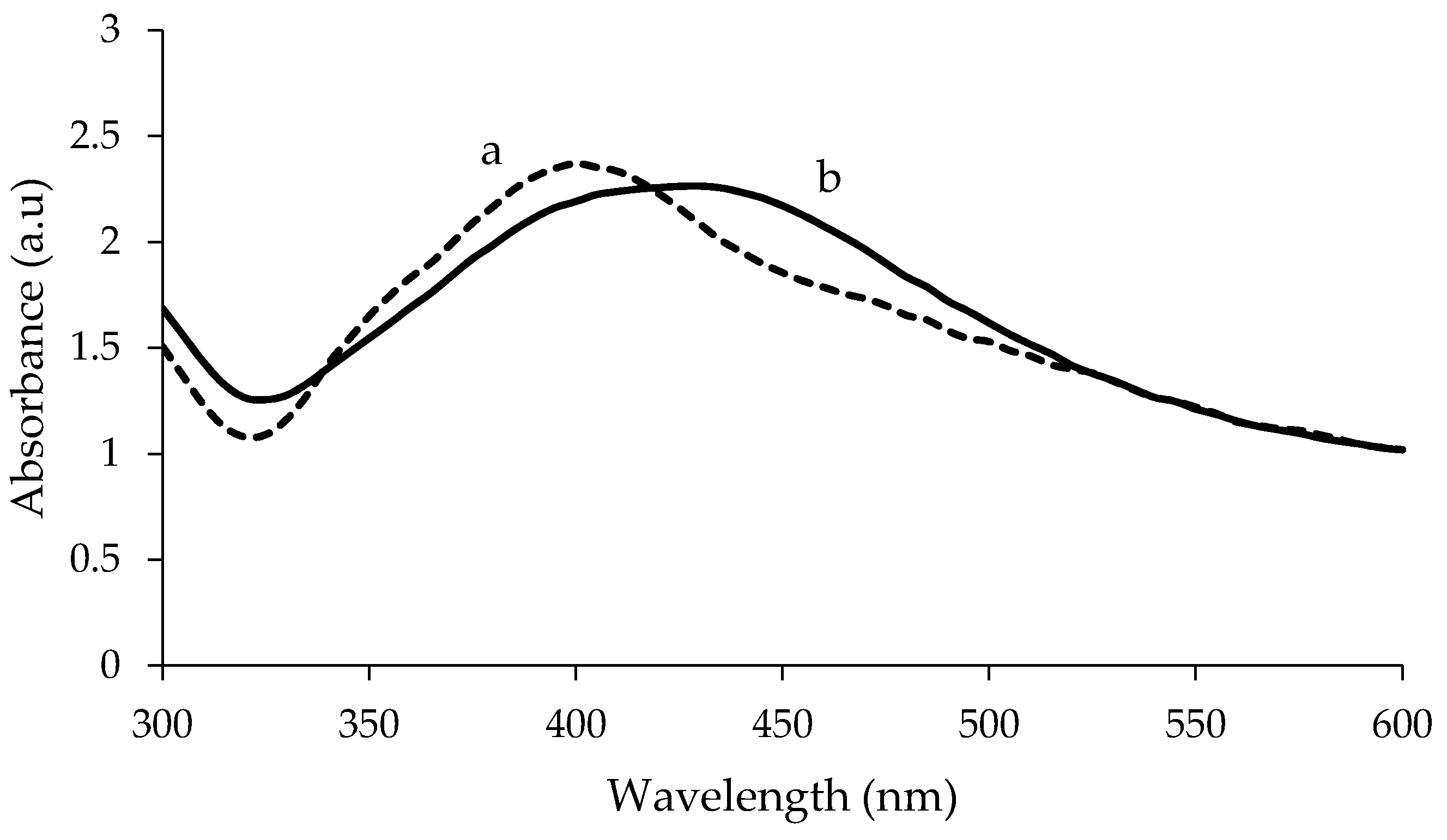
3.1. UV-Visible Absorption Spectrophotometry Analysis
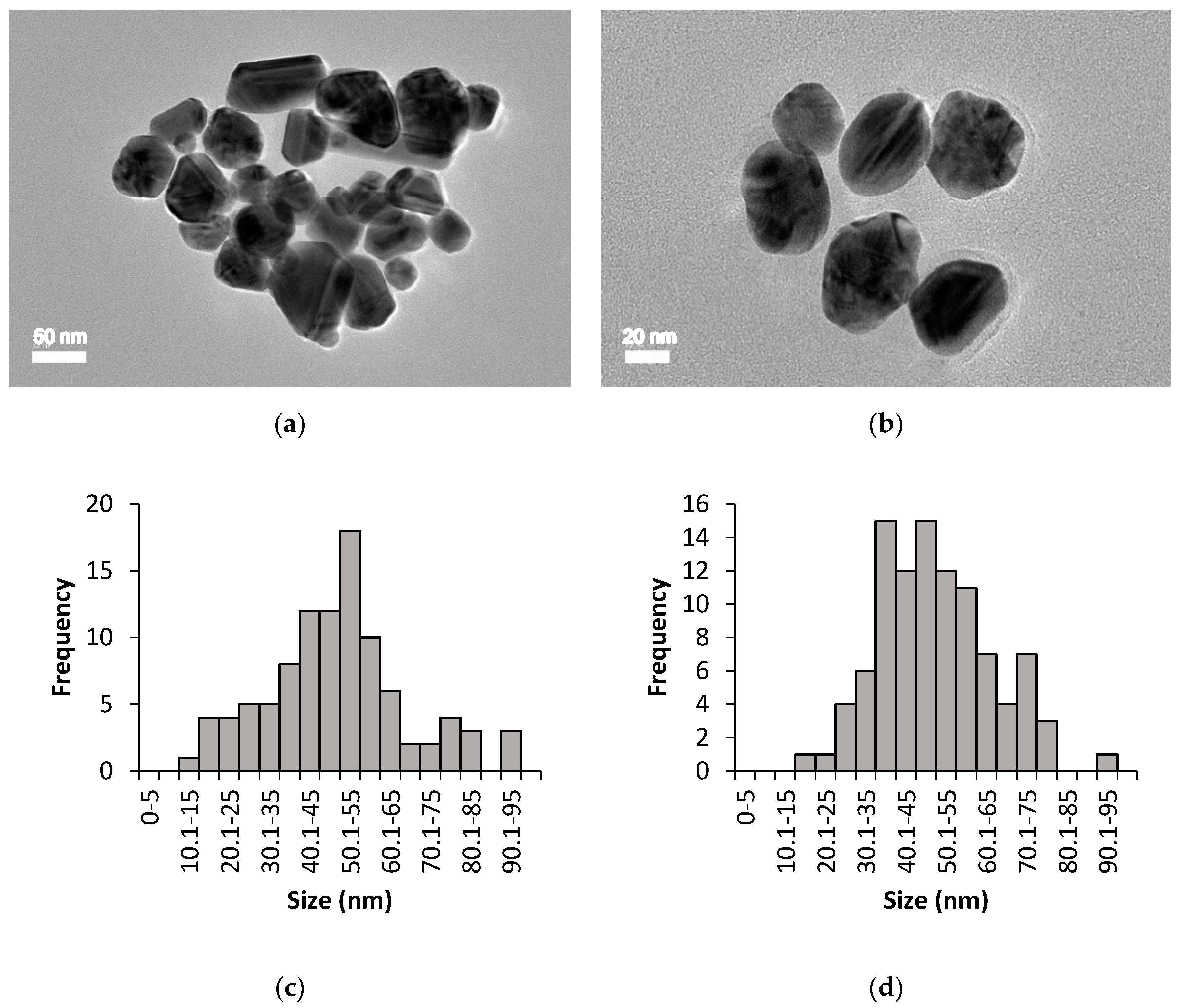
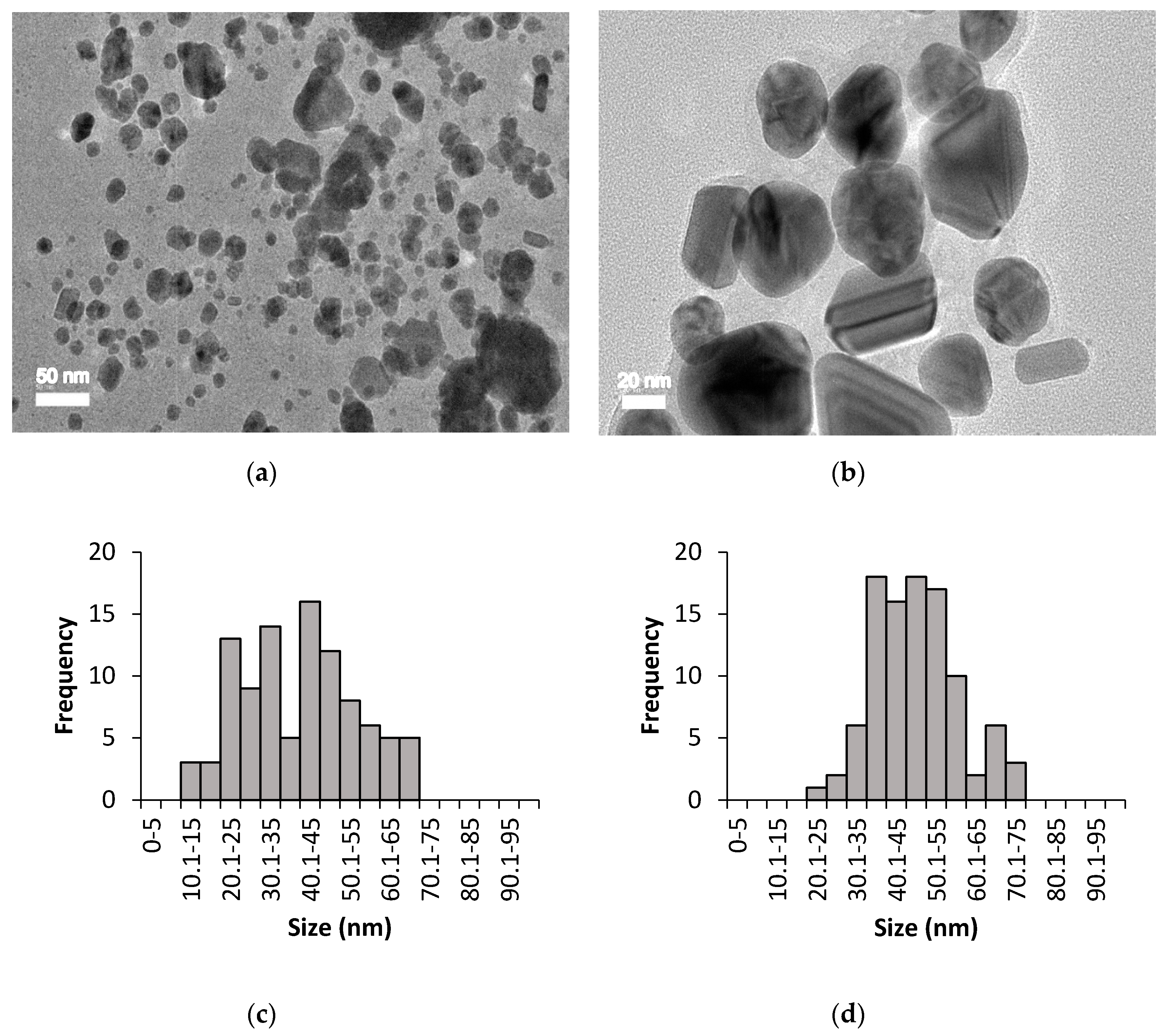
3.2. High Resolution Transmission Electron Microscopy (HR-TEM) Analysis
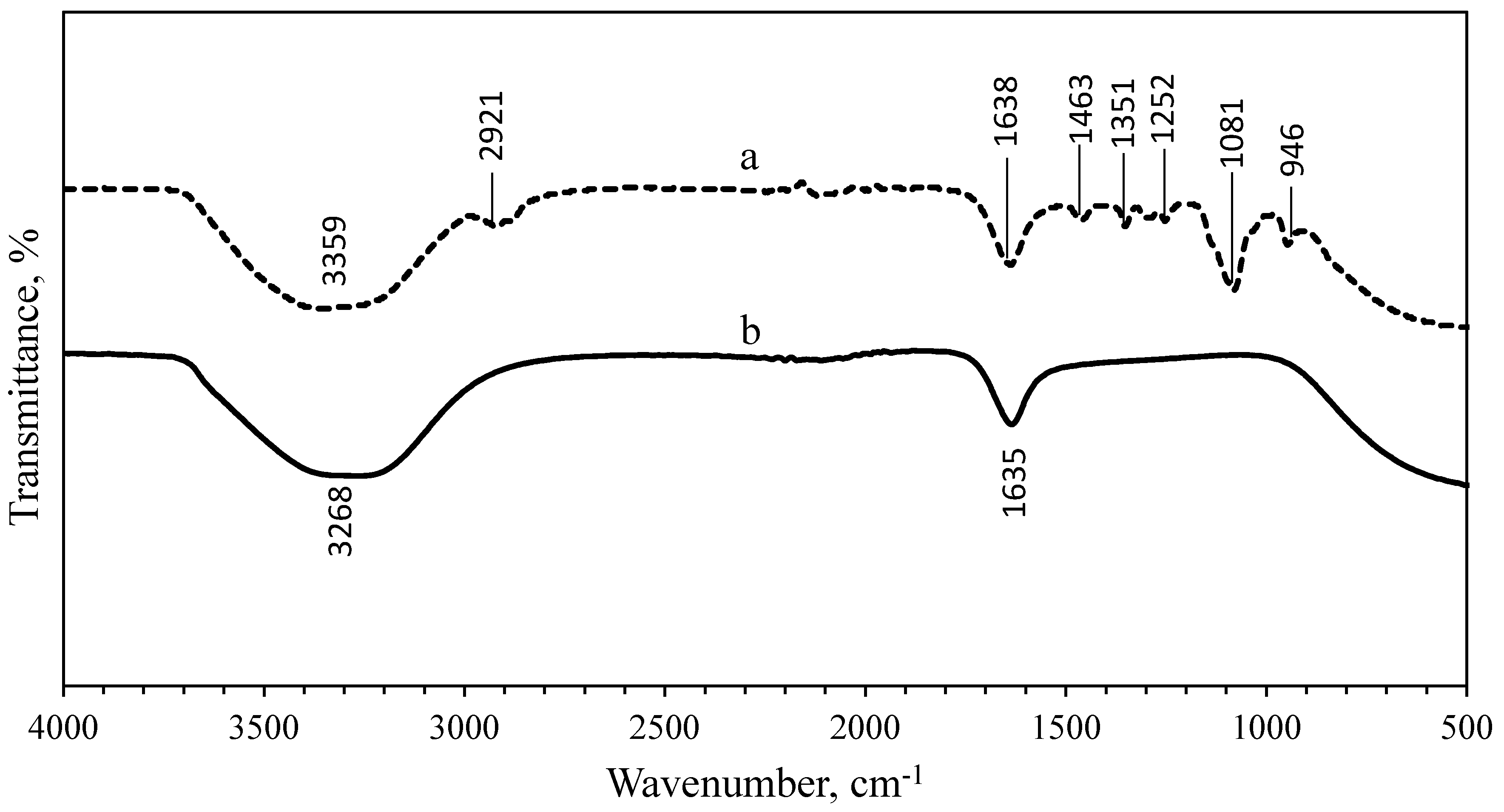
3.3. Fourier-Transform Infrared Spectroscopy (FTIR) Analysis
3.4. Dynamic Light Scattering (DLS) and Zeta Potential (ZP) Measurements
3.5. Antimicrobial Tests of AgNPs
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Ng, L.Y.; Mohammad, A.W.; Leo, C.P.; Hilal, N. Polymeric membranes incorporated with metal/metal oxide nanoparticles: A comprehensive review. Desalination 2013, 308, 15–33. [Google Scholar] [CrossRef]
- Hosokawa, M.; Nogi, K.; Naito, M.; Yokoyama, T. Structural control of nanoparticles. In Nanoparticle Technology Handbook, 2nd ed.; Elsevier: Amsterdam, The Netherlands, 2012; pp. 51–112. [Google Scholar]
- Ardani, H.K.; Imawan, C.; Handayani, W.; Djuhana, D.; Harmoko, A.; Fauzia, V. Enhancement of the stability of silver nanoparticles synthesized using aqueous extract of Diospyros discolor Willd. leaves using polyvinyl alcohol. Conf. Ser. Mater. Sci. Eng. 2017, 188, 012056. [Google Scholar] [CrossRef]
- Ju-Nam, Y.; Lead, J.R. Manufactured nanoparticles: An overview of their chemistry, interactions and potential environmental implications. Sci. Total Environ. 2008, 400, 396–414. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.; Ahmad, M.; Swami, B.L.; Ikram, S. A review on plants extract mediated synthesis of silver nanoparticles for antimicrobial applications: A green expertise. J. Adv. Res. 2016, 7, 17–28. [Google Scholar] [CrossRef]
- Singh, P.; Kim, Y.J.; Singh, H.; Wang, C.; Hwang, K.H.; Farh, M.E.A.; Yang, D.C. Biosynthesis, characterization, and antimicrobial applications of silver nanoparticles. Int. J. Nanomed. 2015, 10, 2567. [Google Scholar]
- Sondi, I.; Goia, D.V.; Matijević, E. Preparation of highly concentrated stable dispersions of uniform silver nanoparticles. J. Colloid Interface Sci. 2003, 260, 75–81. [Google Scholar] [CrossRef]
- Pal, A.; Shah, S.; Devi, S. Microwave-assisted synthesis of silver nanoparticles using ethanol as a reducing agent. Mater. Chem. Phys. 2009, 114, 530–532. [Google Scholar] [CrossRef]
- Muzamil, M.; Khalid, N.; Aziz, M.D.; Abbas, S.A. Synthesis of silver nanoparticles by silver salt reduction and its characterization. Conf. Ser. Mater. Sci. Eng. 2014, 60, 012034. [Google Scholar] [CrossRef]
- Ajitha, B.; Reddy, Y.A.K.; Reddy, P.S. Biogenic nano-scale silver particles by Tephrosia purpurea leaf extract and their inborn antimicrobial activity. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2014, 121, 164–172. [Google Scholar] [CrossRef]
- Awwad, A.M.; Salem, N.M.; Abdeen, A.O. Green synthesis of silver nanoparticles using carob leaf extract and its antibacterial activity. Int. J. Ind. Chem. 2013, 4, 29. [Google Scholar] [CrossRef]
- Kulkarni, A.P.; Srivastava, A.A.; Harpale, P.M.; Zunjarrao, R.S. Plant mediated synthesis of silver nanoparticles-tapping the unexploited sources. J. Nat. Prod. Plant Resour. 2011, 1, 100–107. [Google Scholar]
- Vázquez, B.; Avila, G.; Segura, D.; Escalante, B. Anti-inflammatory activity of extracts from Aloe vera gel. J. Ethnopharmacol. 1996, 55, 69–75. [Google Scholar] [CrossRef]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. Res. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Chandran, S.P.; Chaudhary, M.; Pasricha, R.; Ahmad, A.; Sastry, M. Synthesis of gold nanotriangles and silver nanoparticles using Aloe vera plant extract. Biotechnol. Prog. 2006, 22, 577–583. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yang, D.; Kong, Y.; Wang, X.; Pandoli, O.; Gao, G. Synergetic antibacterial effects of silver nanoparticles @ Aloe vera prepared via a green method. Nano Biomed. Eng. 2010, 2, 252–257. [Google Scholar] [CrossRef]
- Kvítek, L.; Panáček, A.; Soukupova, J.; Kolář, M.; Večeřová, R.; Prucek, R.; Zbořil, R. Effect of surfactants and polymers on stability and antibacterial activity of silver nanoparticles (NPs). J. Phys. Chem. C 2008, 112, 5825–5834. [Google Scholar] [CrossRef]
- Sarkar, A.; Kapoor, S.; Mukherjee, T. Preparation, characterization, and surface modification of silver nanoparticles in formamide. J. Phys. Chem. B 2005, 109, 7698–7704. [Google Scholar] [CrossRef]
- Srikar, S.K.; Giri, D.D.; Pal, D.B.; Mishra, P.K.; Upadhyay, S.N. Green synthesis of silver nanoparticles: A review. Green Sustain. Chem. 2016, 6, 34–56. [Google Scholar] [CrossRef]
- Guarrotxena, N.; Braun, G. Ag-nanoparticle fractionation by low melting point agarose gel electrophoresis. J. Nanopart. Res. 2012, 14, 1199. [Google Scholar] [CrossRef]
- Nabiyouni, G.; Barati, A.; Saadat, M. Surface adsorption of polyethylene glycol and polyvinyl alcohol with variable molecular weights on zinc oxide nanoparticles. Iran. J. Chem. Eng. 2011, 8, 20–30. [Google Scholar]
- Kathiraven, T.; Sundaramanickam, A.; Shanmugam, N.; Balasubramanian, T. Green synthesis of silver nanoparticles using marine algae Caulerpa racemosa and their antibacterial activity against some human pathogens. Appl. Nanosci. 2015, 5, 499–504. [Google Scholar] [CrossRef]
- Marsalek, R. Particle size and zeta potential of zinc oxide. APCBEE Procedia 2014, 9, 13–17. [Google Scholar] [CrossRef]
- Mohd Yusof, H.; Rahman, A.; Mohamad, R.; Zaidan, U.H. Microbial Mediated Synthesis of Silver Nanoparticles by Lactobacillus Plantarum TA4 and its Antibacterial and Antioxidant Activity. Appl. Sci. 2020, 10, 6973. [Google Scholar] [CrossRef]
- Hasan, S. A review on NPs: Their synthesis and types. Res. J. Recent Sci. ISSN 2015, 2277, 2502. [Google Scholar]
- Shameli, K.; Bin Ahmad, M.; Jazayeri, S.D.; Sedaghat, S.; Shabanzadeh, P.; Jahangirian, H.; Abdollahi, Y. Synthesis and characterization of polyethylene glycol mediated silver nanoparticles by the green method. Int. J. Mol. Sci. 2012, 13, 6639–6650. [Google Scholar] [CrossRef]
- Sun, C.; Sze, R.; Zhang, M. Folic acid-PEG conjugated superparamagnetic nanoparticles for targeted cellular uptake and detection by MRI. J. Biomed. Mater. Res. Part A Off. J. Soc. Biomater. Jpn. Soc. Biomater. Aust. Soc. Biomater. Korean Soc. Biomater. 2006, 78, 550–557. [Google Scholar] [CrossRef]
- Rolim, W.R.; Pelegrino, M.T.; de Araújo Lima, B.; Ferraz, L.S.; Costa, F.N.; Bernardes, J.S.; Seabra, A.B. Green tea extract mediated biogenic synthesis of silver nanoparticles: Characterization, cytotoxicity evaluation and antibacterial activity. Appl. Surf. Sci. 2019, 463, 66–74. [Google Scholar] [CrossRef]
- Mishra, S.; Shimpi, N.G.; Sen, T. The effect of PEG encapsulated silver nanoparticles on the thermal and electrical property of sonochemically synthesized polyaniline/silver nanocomposite. J. Polym. Res. 2013, 20, 49. [Google Scholar] [CrossRef]
- Englebienne, P.; Hoonacker, A.V.; Verhas, M. Surface plasmon resonance: Principles, methods and applications in biomedical sciences. J. Spectrosc. 2003, 17, 255–273. [Google Scholar] [CrossRef]
- Desai, R.; Mankad, V.; Gupta, S.K.; Jha, P.K. Size distribution of silver nanoparticles: UV-visible spectroscopic assessment. Nanosci. Nanotechnol. Lett. 2012, 4, 30–34. [Google Scholar] [CrossRef]
- El Badawy, A.M.; Scheckel, K.G.; Suidan, M.; Tolaymat, T. The impact of stabilization mechanism on the aggregation kinetics of silver nanoparticles. Sci. Total Environ. 2012, 429, 325–331. [Google Scholar] [CrossRef] [PubMed]
- Perrault, S.D.; Chan, W.C. Synthesis and surface modification of highly monodispersed, spherical gold nanoparticles of 50−200 nm. J. Am. Chem. Soc. 2009, 131, 17042–17043. [Google Scholar] [CrossRef] [PubMed]
- Singh, B.P.; Menchavez, R.; Takai, C.; Fuji, M.; Takahashi, M. Stability of dispersions of colloidal alumina particles in aqueous suspensions. J. Colloid Interface Sci. 2005, 291, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Panáček, A.; Kolář, M.; Večeřová, R.; Prucek, R.; Soukupova, J.; Kryštof, V.; Kvítek, L. Antifungal activity of silver nanoparticles against Candida spp. Biomaterials 2009, 30, 6333–6340. [Google Scholar] [CrossRef]
- Rai, M.; Yadav, A.; Gade, A. Silver nanoparticles as a new generation of antimicrobials. Biotechnol. Adv. 2009, 27, 76–83. [Google Scholar] [CrossRef]
- Otsuka, H.; Nagasaki, Y.; Kataoka, K. PEG-coated nanoparticles for biological and pharmaceutical applications. Adv. Drug Deliv. Rev. 2003, 55, 403–419. [Google Scholar] [CrossRef]
- Prema, P. Chemical mediated synthesis of silver nanoparticles and its potential antibacterial application. Prog. Mol. Environ. Bioeng. Anal. Modeling Technol. Appl. 2011, 6, 151–166. [Google Scholar]
- Hamouda, R.A.; Yousuf, W.E.; Abdeen, E.E.; Mohamed, A. Biological and Chemical Synthesis of Silver Nanoparticles: Characterization, MIC and Antibacterial Activity against Pathogenic Bacteria. J. Chem. Pharm. Res. 2019, 11, 1–12. [Google Scholar]

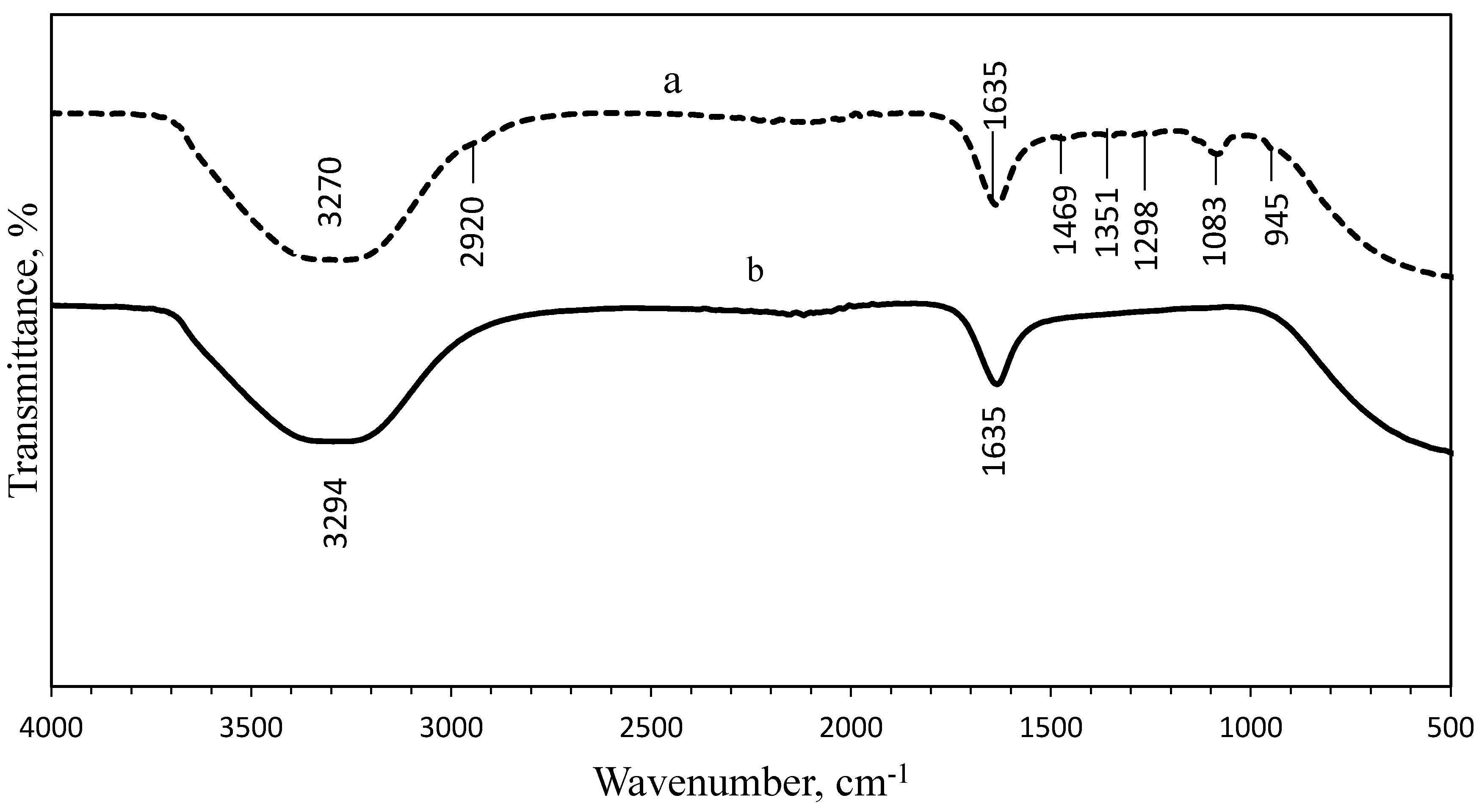

| Wavenumber (cm−1) | Vibrational Assignment | Functional Group | |||
|---|---|---|---|---|---|
| Chemical Synthesis | Biological Synthesis | ||||
| Uncoated AgNPs | PEG-Coated AgNPs | Uncoated AgNPs | PEG-Coated AgNPs | ||
| 3268 | 3359 | 3294 | 3270 | O–H stretching | Hydroxyl |
| - | 2921 | - | 2920 | C–H stretching | Carboxylic acid |
| 1635 | 1638 | 1635 | 1635 | C=O stretching, N–H stretching | Amide Ⅰ |
| - | 1463 | - | 1469 | C–H bending | Aromatic compounds |
| - | 1351 | - | 1351 | O–C–H | Alcohols |
| - | 1252 | - | 1298 | C–C stretch | Ketones |
| - | 1081 | - | 1083 | C–C=O, C–O–P | Hydroxyl from saccharides |
| - | 946 | - | 945 | C–H bend | Alkenes |
| Parameter | Chemical Synthesis | Biological Synthesis | ||
|---|---|---|---|---|
| AgNPs | PEG-Coated AgNPs | AgNPs | PEG-Coated AgNPs | |
| Polydispersity index (PdI) | 0.514 | 0.476 | 0.571 | 0.534 |
| Zeta potential (mV) | −32.3 | −36.4 | 29.8 | −30.6 |
| Samples | Zone of Inhibition (mm) for 100 µL | ||||
|---|---|---|---|---|---|
| Gram Positive Bacteria | Gram Negative Bacteria | ||||
| Staphylococcus aureus | Staphylococcus epidermidis | Escherichia coli | Salmonella sp. | ||
| Ampicillin (control) | 29.75 ± 1.28 | 41.50 ± 1.41 | 17.50 ± 2.98 | 69.2 ± 12.30 | |
| Chemically Synthesised AgNPs | Uncoated | 0.00 | 0.00 | 0.00 | 0.00 |
| PEG-Coated | 0.00 | 0.00 | 0.00 | 0.00 | |
| Biologically Synthesised AgNPs | Uncoated | 23.00 ± 1.41 | 20.00 ± 0.00 | 13.00 ± 1.41 | 26.00 ± 0.00 |
| PEG-coated | 16.00 ± 0.00 | 14.00 ± 0.00 | 12.00 ± 0.00 | 18.00 ± 0.00 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohamad Kasim, A.S.; Ariff, A.B.; Mohamad, R.; Wong, F.W.F. Interrelations of Synthesis Method, Polyethylene Glycol Coating, Physico-Chemical Characteristics, and Antimicrobial Activity of Silver Nanoparticles. Nanomaterials 2020, 10, 2475. https://doi.org/10.3390/nano10122475
Mohamad Kasim AS, Ariff AB, Mohamad R, Wong FWF. Interrelations of Synthesis Method, Polyethylene Glycol Coating, Physico-Chemical Characteristics, and Antimicrobial Activity of Silver Nanoparticles. Nanomaterials. 2020; 10(12):2475. https://doi.org/10.3390/nano10122475
Chicago/Turabian StyleMohamad Kasim, Amirah Shafilla, Arbakariya Bin Ariff, Rosfarizan Mohamad, and Fadzlie Wong Faizal Wong. 2020. "Interrelations of Synthesis Method, Polyethylene Glycol Coating, Physico-Chemical Characteristics, and Antimicrobial Activity of Silver Nanoparticles" Nanomaterials 10, no. 12: 2475. https://doi.org/10.3390/nano10122475
APA StyleMohamad Kasim, A. S., Ariff, A. B., Mohamad, R., & Wong, F. W. F. (2020). Interrelations of Synthesis Method, Polyethylene Glycol Coating, Physico-Chemical Characteristics, and Antimicrobial Activity of Silver Nanoparticles. Nanomaterials, 10(12), 2475. https://doi.org/10.3390/nano10122475





