Fabrication and Characterization of Ceftizoxime-Loaded Pectin Nanocarriers
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals and Strains
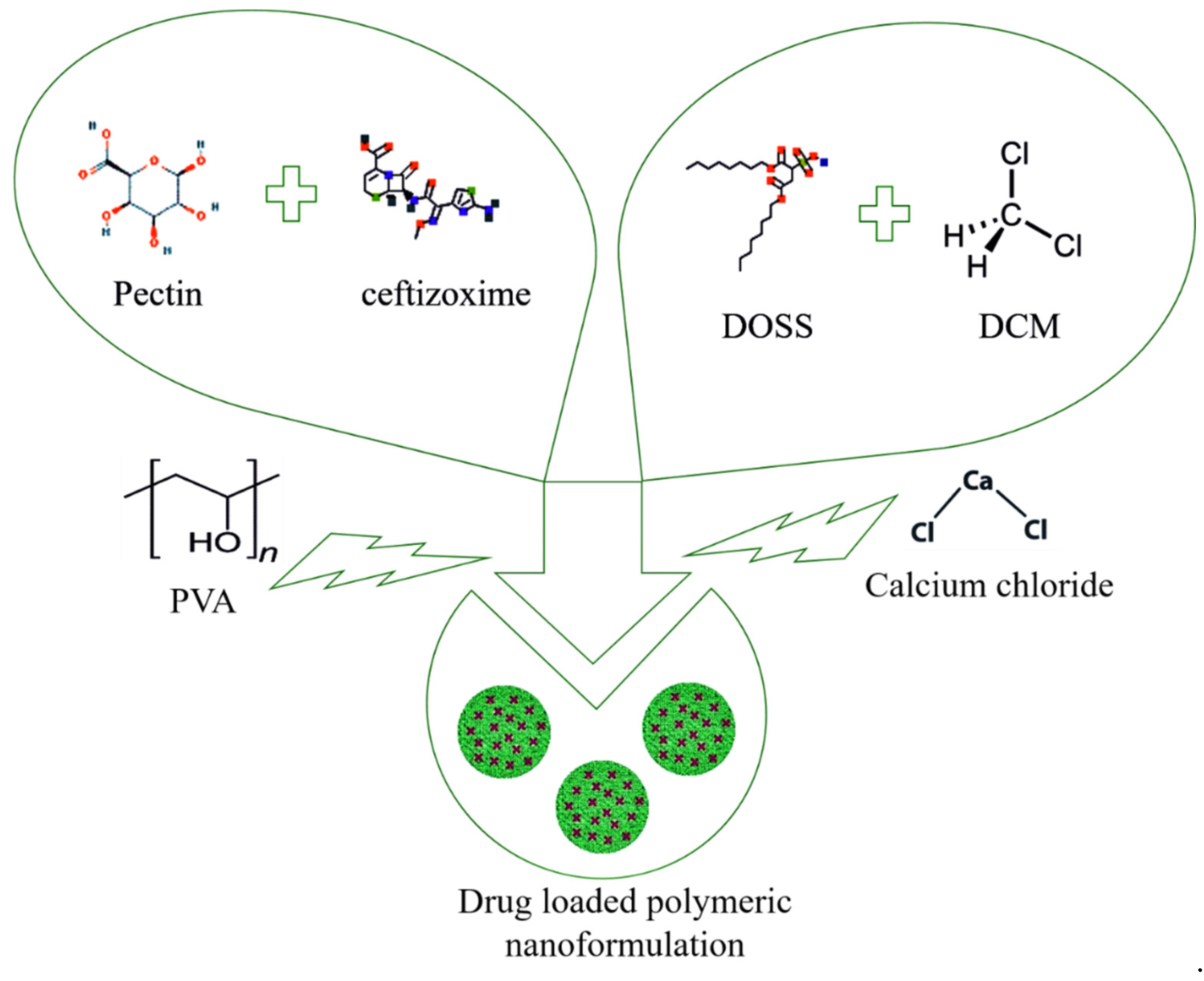
2.2. Nanoformulation Synthesis
2.3. Optimization of Nanoformulation
2.4. Characterization
2.5. Drug Loading and Efficiency of Encapsulation
2.6. In Vitro Drug Release
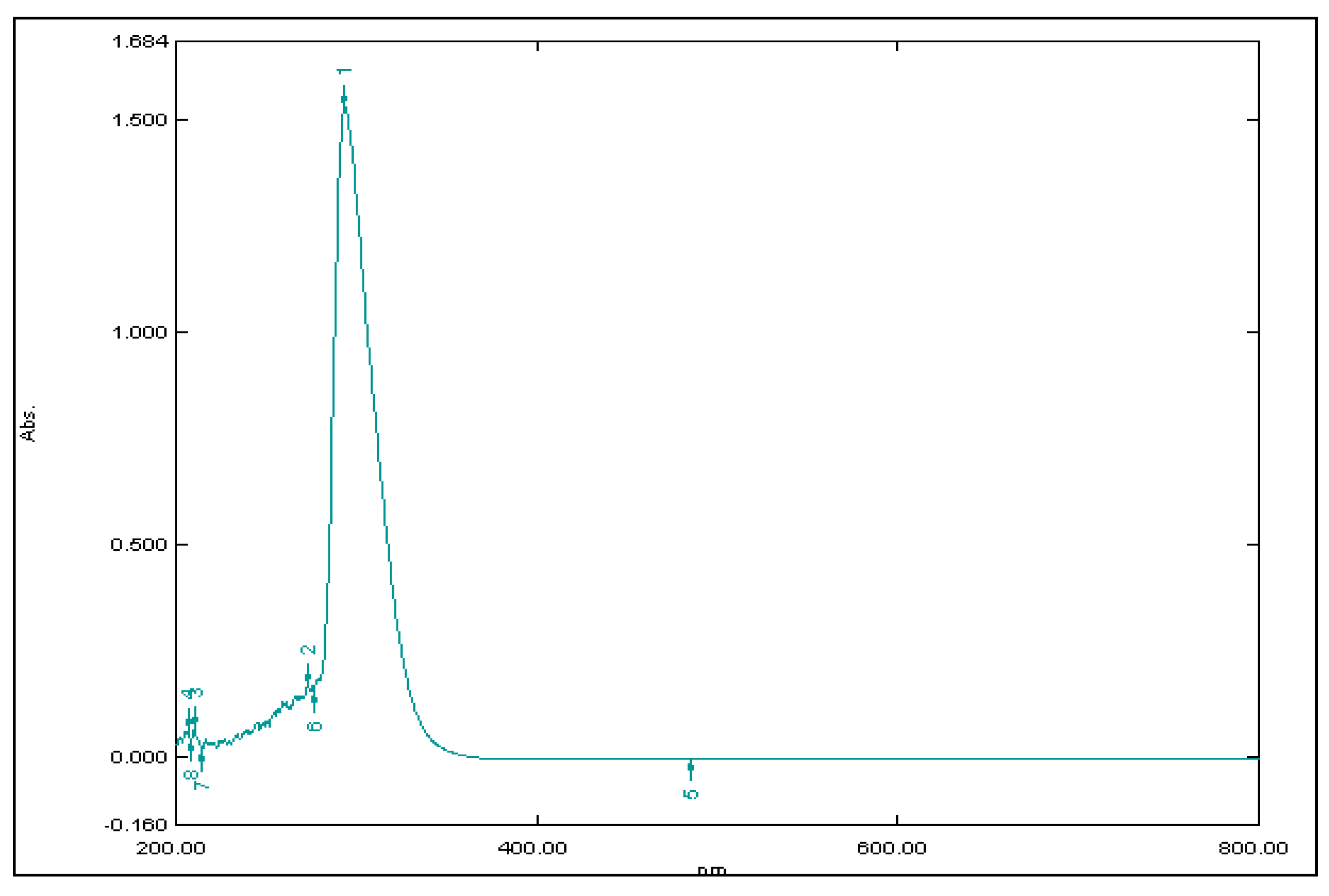
2.7. Standard Calibration Curve
2.8. Optimization of Nanoformulation
2.9. Cytotoxicity Study
2.10. Antibacterial Activity
3. Results and Discussion
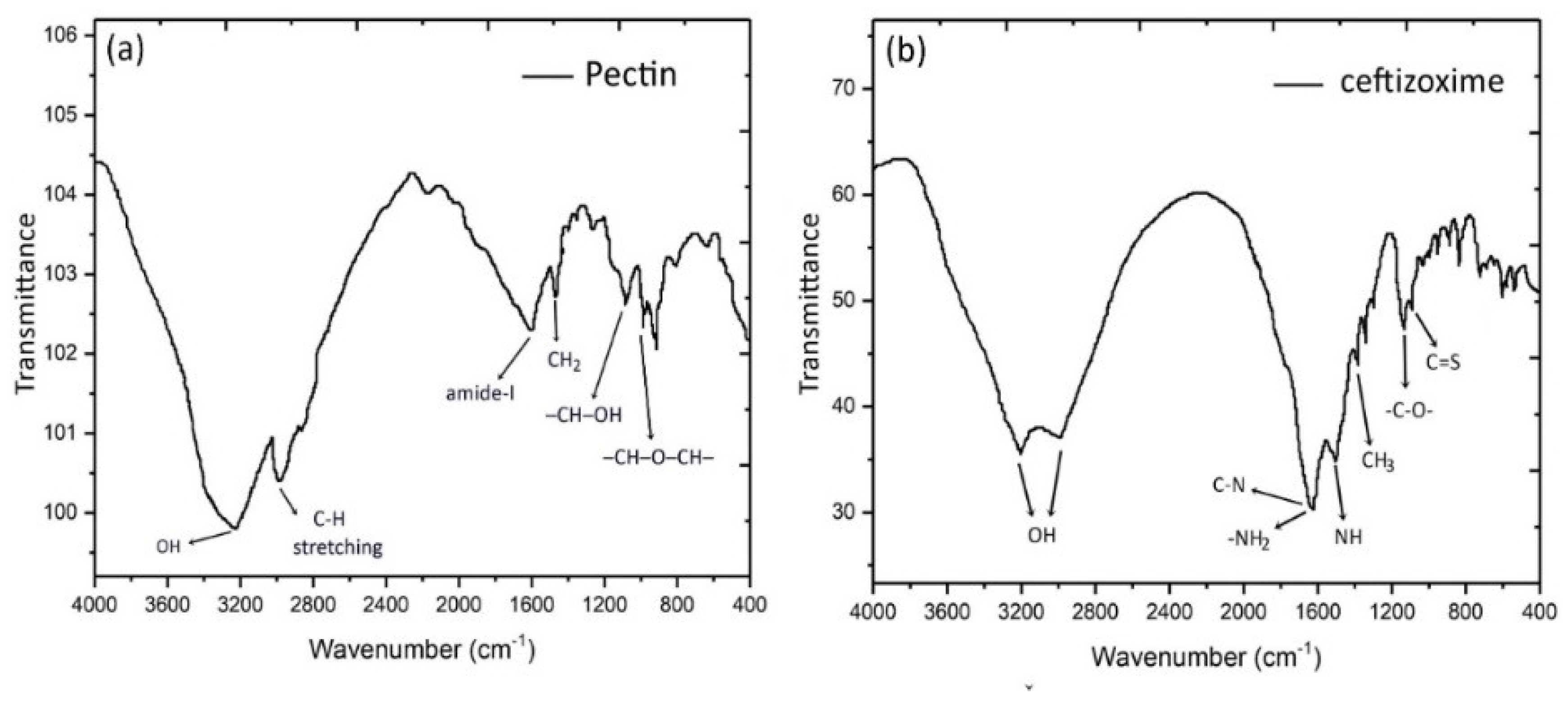
3.1. Physiochemical Analysis
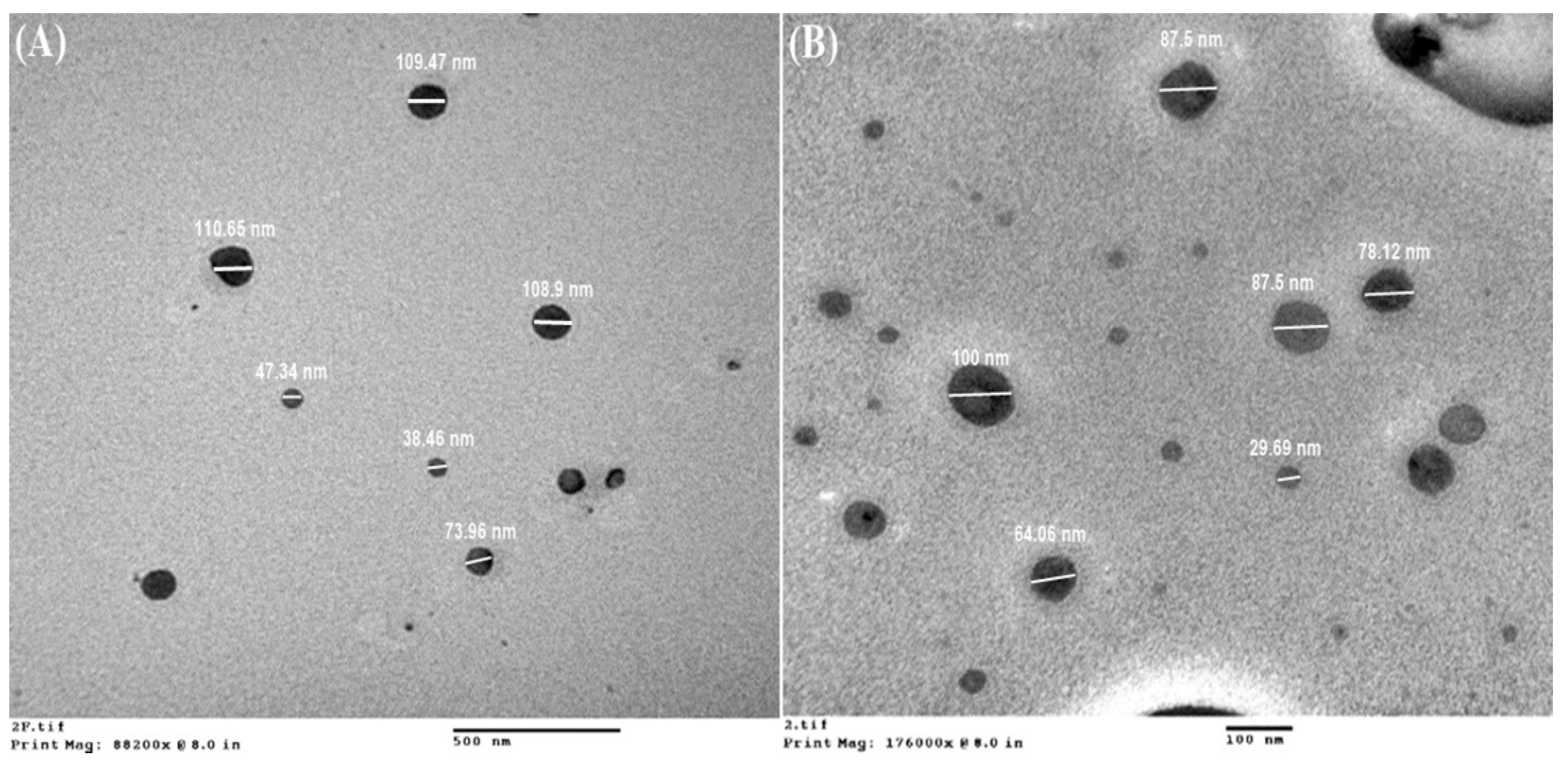
3.2. Morphological Analysis
3.3. Optimization of Nanoformulation
3.4. Drug Loading and Encapsulation Efficiency
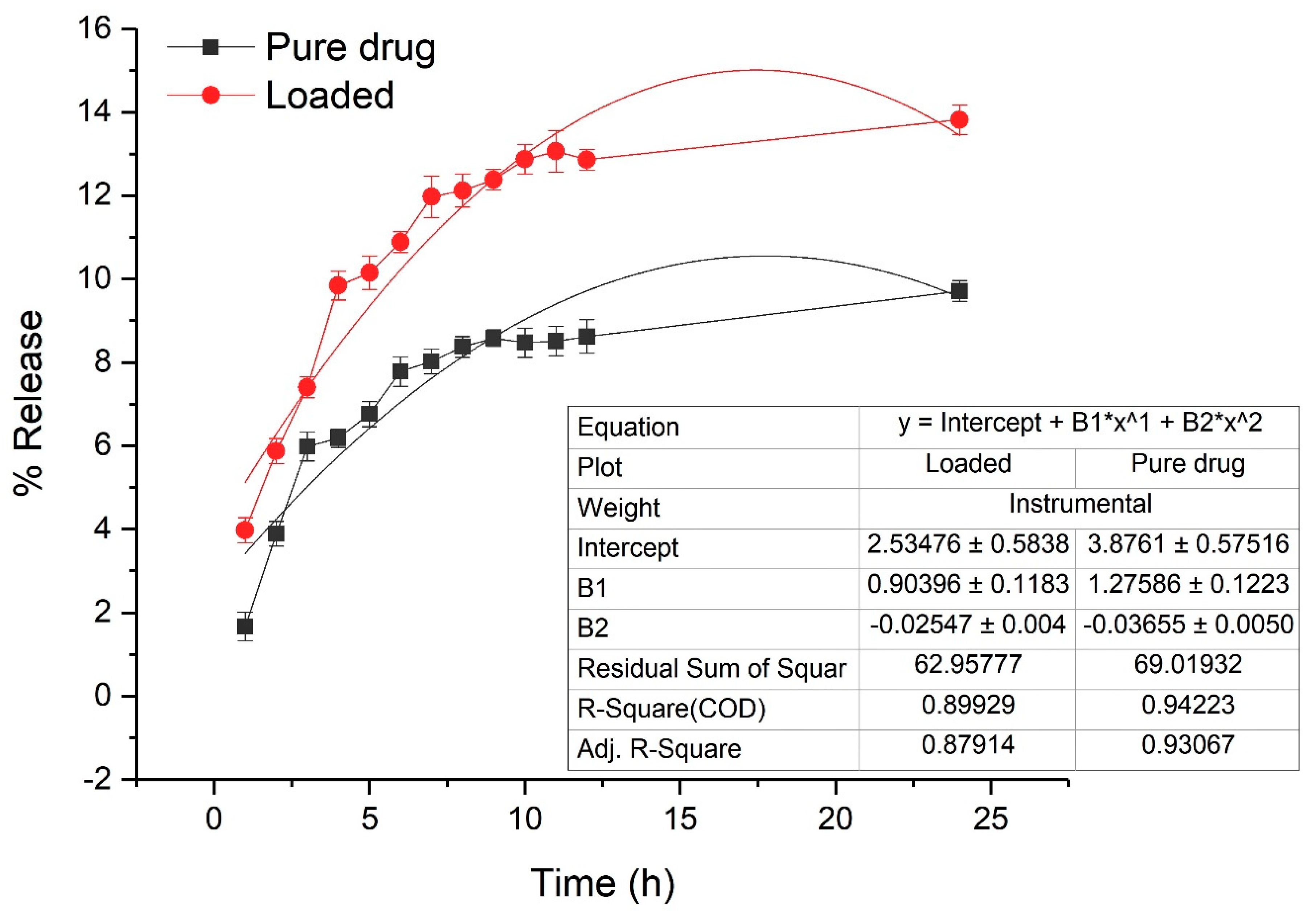
3.5. In Vitro Drug Release
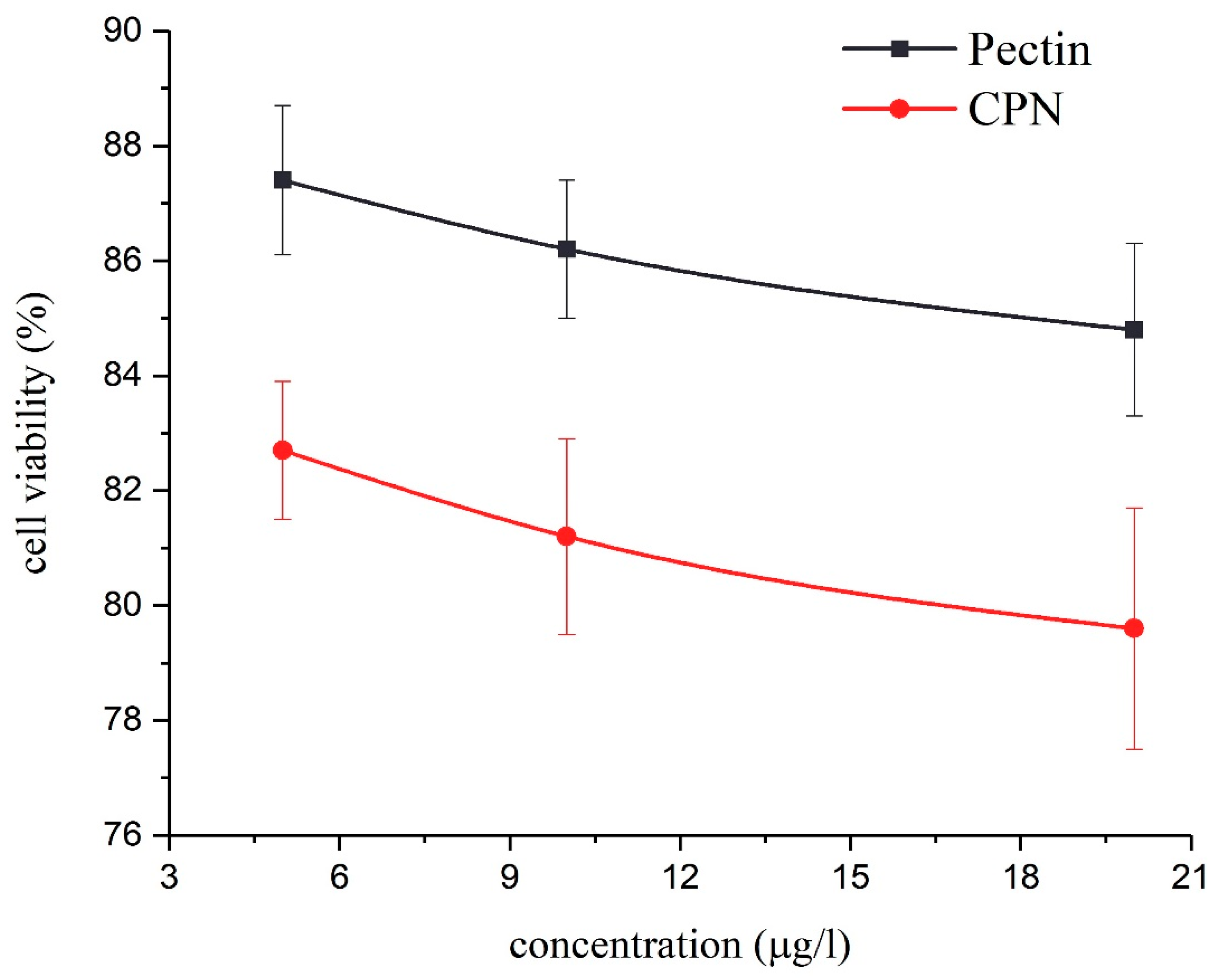
3.6. In Vitro Cytotoxicity
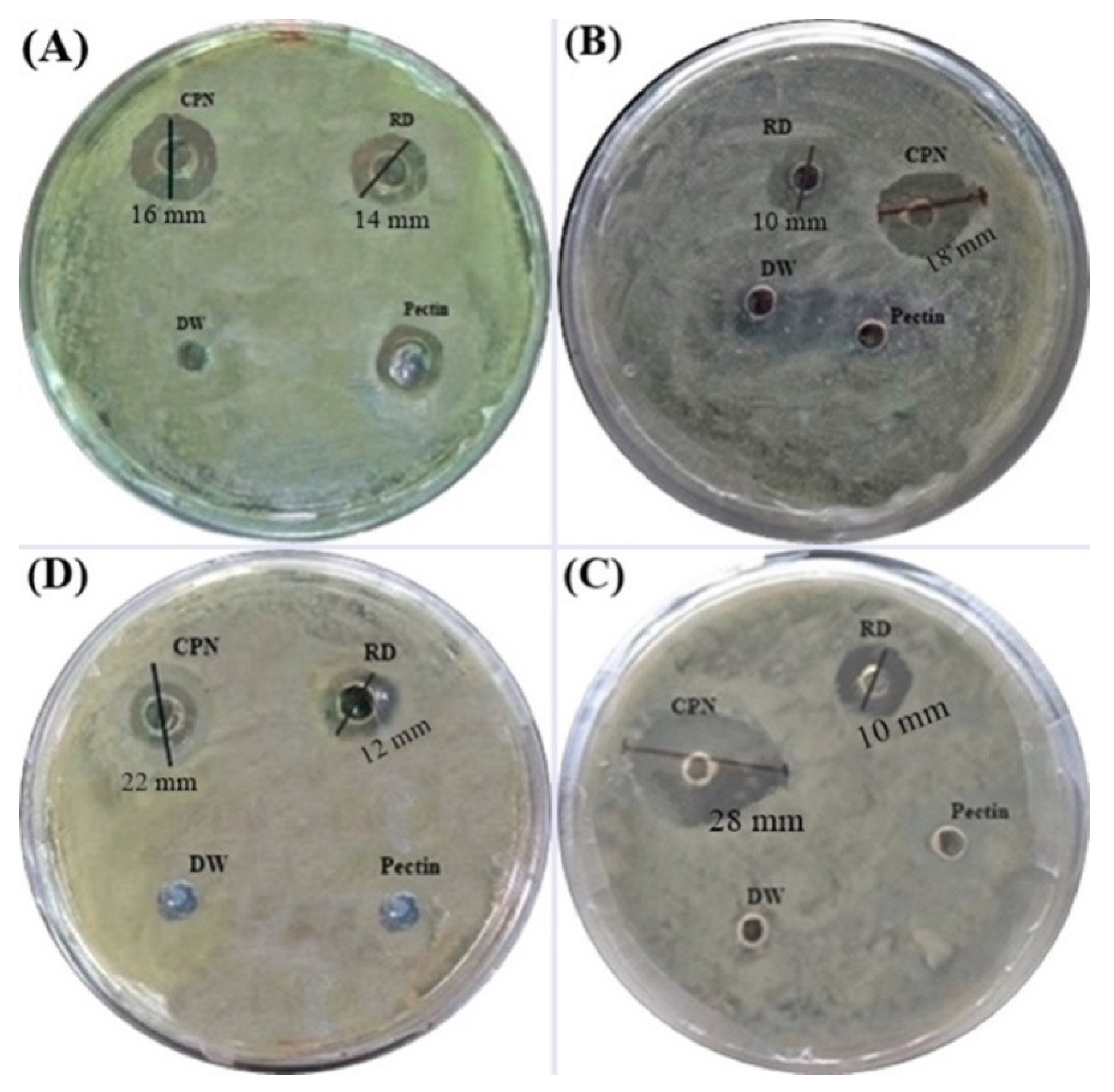
3.7. Antibacterial Activity
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Homayun, B.; Lin, X.; Choi, H.J. Challenges and recent progress in oral drug delivery systems for biopharmaceuticals. Pharmaceutics 2019, 11, 129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsui, H.; Shimokawa, O.; Kaneko, T.; Nagano, Y.; Rai, K.; Hyodo, I. The pathophysiology of non-steroidal anti-inflammatory drug (NSAID)-induced mucosal injuries in stomach and small intestine. J. Clin. Biochem. Nutr. 2011, 48, 107–111. [Google Scholar] [CrossRef] [Green Version]
- Wen, H.; Jung, H.; Li, X. Drug Delivery Approaches in Addressing Clinical Pharmacology-Related Issues: Opportunities and Challenges. AAPS J. 2015, 17, 1327–1340. [Google Scholar] [CrossRef] [PubMed]
- Park, K. Drug Delivery Research: The Invention Cycle. Mol. Pharm. 2016, 13, 2143–2147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jain, D.; Raturi, R.; Jain, V.; Bansal, P.; Singh, R. Recent technologies in pulsatile drug delivery systems. Biomatter 2011, 1, 57–65. [Google Scholar] [CrossRef] [Green Version]
- Hadizadeh, M.; Toraji, A. Amoxicillin-Loaded Polymeric Nanoparticles of Less than 100 nm: Design, Preparation and Antimicrobial Activity Against Methicillin-Resistant Staphylococcus aureus. Iran. J. Sci. Technol. Trans. A Sci. 2019, 43, 379–386. [Google Scholar] [CrossRef]
- Motiei, M.; Kashanian, S.; Azandaryani, A.H. Effect of Fabrication Parameters on the Physiochemical Properties of Amphiphilic Chitosan Nanoparticles. Iran. J. Sci. Technol. Trans. A Sci. 2018, 42, 1873–1879. [Google Scholar] [CrossRef]
- Singh, P.K.; Jairath, G.; Ahlawat, S.S. Nanotechnology: A future tool to improve quality and safety in meat industry. J. Food Sci. Technol. 2016, 53, 1739–1749. [Google Scholar] [CrossRef] [Green Version]
- Martău, A.G.; Mihai, M.; Vodnar, C.D. The Use of Chitosan, Alginate, and Pectin in the Biomedical and Food Sector-Biocompatibility, Bioadhesiveness, and Biodegradability. Polymers 2019, 11, 1837. [Google Scholar] [CrossRef] [Green Version]
- Minzanova, S.T.; Mironov, V.F.; Arkhipova, D.M.; Khabibullina, A.V.; Mironova, L.G.; Zakirova, Y.M.; Milyukov, V.A. Biological Activity and Pharmacological Application of Pectic Polysaccharides: A Review. Polymers 2018, 10, 1407. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sriamornsak, P. Application of pectin in oral drug delivery. Expert Opin. Drug Deliv. 2011, 8, 1009–1023. [Google Scholar] [CrossRef]
- Srivastava, P.; Malviya, R. Sources of pectin, extraction and its applications in pharmaceutical industry—An overview. Indian J. Nat. Prod. Resour. 2011, 2, 10–18. [Google Scholar]
- Thakur, B.R.; Singh, R.K.; Handa, A.K.; Rao, M.A. Chemistry and uses of pectin—A review. Crit. Rev. Food Sci. Nutr. 1997, 37, 47–73. [Google Scholar] [CrossRef] [PubMed]
- Lara-Espinoza, C.; Carvajal-Millán, E.; Balandrán-Quintana, R.; López-Franco, Y.; Rascón-Chu, A. Pectin and Pectin-Based Composite Materials: Beyond Food Texture. Molecules 2018, 23, 942. [Google Scholar] [CrossRef] [Green Version]
- Munarin, F.; Tanzi, M.C.; Petrini, P. Advances in biomedical applications of pectin gels. Int. J. Biol. Macromol. 2012, 51, 681–689. [Google Scholar] [CrossRef]
- Li, C.; Wang, J.; Wang, Y.; Gao, H.; Wei, G.; Huang, Y.; Yu, H.; Gan, Y.; Wang, Y.; Mei, L.; et al. Recent progress in drug delivery. Acta Pharm. Sin. B 2019, 9, 1145–1162. [Google Scholar] [CrossRef]
- Mabrouk, M.; Rajendran, R.; Soliman, E.I.; Ashour, M.M.; Beherei, H.H.; Tohamy, M.K.; Thomas, S.; Kalarikkal, N.; Arthanareeswaran, G.; Das, B.D. Nanoparticle- and Nanoporous-Membrane-Mediated Delivery of Therapeutics. Pharmaceutics 2019, 11, 294. [Google Scholar] [CrossRef] [Green Version]
- Canaparo, R.; Foglietta, F.; Giuntini, F.; Della Pepa, C.; Dosio, F.; Serpe, L. Recent Developments in Antibacterial Therapy: Focus on Stimuli-Responsive Drug-Delivery Systems and Therapeutic Nanoparticles. Molecules 2019, 24, 1991. [Google Scholar] [CrossRef] [Green Version]
- Flaherty, J.F.; Barriere, S.L. Third-generation cephalosporins: A critical evaluation. Am. J. Hosp. Pharm. 1984, 41, 1652–1663. [Google Scholar]
- Liu, Y.; Breukink, E. The Membrane Steps of Bacterial Cell Wall Synthesis as Antibiotic Targets. Antibiotics 2016, 5, 28. [Google Scholar] [CrossRef] [PubMed]
- Richards, D.M.; Heel, R.C. Ceftizoxime. Drugs 1985, 29, 281–329. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.; Lillard, J.W. Nanoparticle-based targeted drug delivery. Exp. Mol. Pathol. 2009, 86, 215–223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Feng, Q.; Tong, R. Anticancer nanoparticulate polymer-drug conjugate. Bioeng. Transl. Med. 2016, 1, 277–296. [Google Scholar] [CrossRef] [PubMed]
- Beck-Broichsitter, M.; Rytting, E.; Lebhardt, T.; Wang, X.; Kissel, T. Preparation of nanoparticles by solvent displacement for drug delivery: A shift in the “ouzo region” upon drug loading. Eur. J. Pharm. Sci. 2010, 41, 244–253. [Google Scholar] [CrossRef]
- Parkash, C.; Singh, S.; Datusalia, A.; Deep, A. Development and characterization of nanoparticles of glibenclamide by solvent displacement method. Acta Pol. Pharm. 2010, 67, 283–290. [Google Scholar]
- Geetha, N.; Harini, K.; Joseph, M.; Sangeetha, R.; Venkatachalam, P. A Comparison of Microwave Assisted Medicinal Plant Extractions for Detection of Their Phytocompounds Through Qualitative Phytochemical and FTIR Analyses. Iran. J. Sci. Technol. Trans. A Sci. 2019, 43, 397–407. [Google Scholar] [CrossRef]
- Sun, S.-B.; Liu, P.; Shao, F.-M.; Miao, Q.-L. Formulation and evaluation of PLGA nanoparticles loaded capecitabine for prostate cancer. Int. J. Clin. Exp. Med. 2015, 8, 19670–19681. [Google Scholar]
- Jain, A.K.; Thareja, S. In vitro and in vivo characterization of pharmaceutical nanocarriers used for drug delivery. Artif. Cells, Nanomed. Biotechnol. 2019, 47, 524–539. [Google Scholar] [CrossRef] [Green Version]
- Erchiqui, F. Analysis and Evaluation of Power Formulations for Wood and Hardboard Using Radio Frequency and Microwave Energy. Dry. Technol. 2014, 32, 946–959. [Google Scholar] [CrossRef]
- Eatemadi, A.; Darabi, M.; Afraidooni, L.; Zarghami, N.; Daraee, H.; Eskandari, L.; Mellatyar, H.; Akbarzadeh, A. Comparison, synthesis and evaluation of anticancer drug-loaded polymeric nanoparticles on breast cancer cell lines. Artif. Cells Nanomed. Biotechnol. 2016, 44, 1008–1017. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Saini, M.; Kumar, V.; Dehiya, B.S.; Sindhu, A.; Fouad, H.; Ahmad, N.; Mahmood, A.; Hashem, M. Polyethylene Glycol (PEG) Modified Porous Ca5(PO4)2SiO4 Bioceramics: Structural, Morphologic and Bioactivity Analysis. Coatings 2020, 10, 538. [Google Scholar] [CrossRef]
- Kumar, P.; Saini, M.; Dehiya, B.S.; Umar, A.; Sindhu, A.; Mohammed, H.; Al-Hadeethi, Y.; Guo, Z. Fabrication and in-vitro biocompatibility of freeze-dried CTS-nHA and CTS-nBG scaffolds for bone regeneration applications. Int. J. Biol. Macromol. 2020, 149, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Dehiya, B.S.; Sindhu, A. Synthesis and characterization of nHA-PEG and nBG-PEG scaffolds for hard tissue engineering applications. Ceram. Int. 2019, 45, 8370–8379. [Google Scholar] [CrossRef]
- Kumar, P.; Dehiya, B.S.; Sindhu, A. Ibuprofen-Loaded CTS/nHA/nBG Scaffolds for the Applications of Hard Tissue Engineering. Iran. Biomed. J. 2019, 23, 190–199. [Google Scholar] [CrossRef] [Green Version]
- Kumar, P.; Kumar, V.; Kumar, R.; Kumar, R.; Pruncu, C.I. Fabrication and characterization of ZrO2 incorporated SiO2–CaO–P2O5 bioactive glass scaffolds. J. Mech. Behav. Biomed. Mater. 2020, 109, 103854. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [Green Version]
- Mishra, R.; Datt, M.; Pal, K.; Banthia, A. Preparation and characterization of amidated pectin based hydrogels for drug delivery system. J. Mater. Sci. Mater. Med. 2008, 19, 2275–2280. [Google Scholar] [CrossRef]
- Liu, Y.; Sun, Y.; Guanglong, D.; Geng, Q.; Zhu, J.; Guo, M.; Duan, Y.; Wang, B. Synthesis, Characterization, and Application of Microbe-Triggered Controlled-Release Kasugamycin–Pectin Conjugate. J. Agric. Food Chem. 2015, 63, 4263–4268. [Google Scholar] [CrossRef]
- Moreno, A.; Salgado, H. Development and Validation of the Quantitative Analysis of Ceftazidime in Powder for Injection by Infrared Spectroscopy#. Phys. Chem. 2012, 2, 6–11. [Google Scholar]





| Sr. No. | Pectin (g) (X1) | Di–Octyl Sodium Sulfosuccinate (g) (X2) | Particle Size (nm) (Y1) | Encapsulation Efficiency (%) (Y2) |
|---|---|---|---|---|
| 1 | 0.55 | 1.00 | 207.6 | 67.7 |
| 2 | 0.55 | 5.50 | 174.5 | 69.44 |
| 3 | 0.55 | 10.00 | 169.2 | 62.63 |
| 4 | 0.10 | 1.00 | 152.8 | 63.02 |
| 5 | 1.00 | 5.50 | 181.7 | 69.03 |
| 6 | 1.00 | 10.00 | 143.5 | 65.3 |
| 7 | 0.10 | 10.00 | 189.6 | 57.7 |
| 8 | 1.00 | 1.00 | 285.4 | 66.91 |
| 9 | 0.10 | 5.50 | 146.3 | 62.44 |
| S. No. | Concentration (μg/mL) | Absorbance | Standard Deviation (SD) |
|---|---|---|---|
| 1. | 1.25 | 0.09 | ±0.002 |
| 2. | 2.5 | 0.22 | ±0.035 |
| 3. | 5 | 0.48 | ±0.09 |
| 4. | 10 | 0.95 | ±0.14 |
| 5. | 20 | 1.79 | ±0.33 |
| Sr. No. | Name of Bacteria | Gram Staining Results | Accession Numbers |
|---|---|---|---|
| 1. | Bacillus cereus | Gram positive | NCDC240 |
| 2. | Bacillus polymyxa | Gram-positive | NCDC 068 |
| 3. | Enterobacter aerogenes | Gram-negative | NCDC 106 |
| 4. | Pseudomonas aeruginosa | Gram-negative | NCDC 105 |
| Wavenumbers (cm−1) | Functional Groups |
|---|---|
| 3300–3000 | OH |
| 2952 | C−H |
| 1715 | R−C−O |
| 1610 | NH2, CH stretching |
| 1598 | amide-I |
| 1490 | NH stretching |
| 1470 | −CH2 scissoring |
| 1420 | C−N=O |
| 1390 | CH3 |
| 1180 | −C−O− |
| 1098 | C=S |
| 1080 | −CH−OH bending vibration |
| 1017 | −CH−O−CH– |
| Model | Lack of Fit | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Response Factor | Prob. >F | R2 | F Value | Pred. R2 | Adeq. Prec. | C.V. | Adjust. R2 | Std. Dev. | Prob. >F | F Value |
| Y1 | 0.0045 | 0.9878 | 48.41 | 0.8817 | 21.322 | 4.30 | 0.9674 | 7.88 | 0.527 | 9.17 |
| Y2 | 0.0029 | 0.9909 | 65.06 | 0.9266 | 23.517 | 0.96 | 0.9756 | 0.63 | 0.0180 | 20.30 |
| Bacterial Strain | CPN | RD |
|---|---|---|
| Bacillus cereus | 16 ± 1.5 mm | 14 ± 0.3 mm |
| Bacillus polymyxa | 18 ±1.8 mm | 10 ± 0.2 mm |
| Enterobacter aerogenes | 28 ± 2.3 mm | 12 ± 0.2 mm |
| Pseudomonas aeruginosa | 22 ± 1.9 mm | 12 ± 0.1 mm |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, P.; Kumar, V.; Kumar, R.; Pruncu, C.I. Fabrication and Characterization of Ceftizoxime-Loaded Pectin Nanocarriers. Nanomaterials 2020, 10, 1452. https://doi.org/10.3390/nano10081452
Kumar P, Kumar V, Kumar R, Pruncu CI. Fabrication and Characterization of Ceftizoxime-Loaded Pectin Nanocarriers. Nanomaterials. 2020; 10(8):1452. https://doi.org/10.3390/nano10081452
Chicago/Turabian StyleKumar, Pawan, Vinod Kumar, Ravinder Kumar, and Catalin Iulian Pruncu. 2020. "Fabrication and Characterization of Ceftizoxime-Loaded Pectin Nanocarriers" Nanomaterials 10, no. 8: 1452. https://doi.org/10.3390/nano10081452
APA StyleKumar, P., Kumar, V., Kumar, R., & Pruncu, C. I. (2020). Fabrication and Characterization of Ceftizoxime-Loaded Pectin Nanocarriers. Nanomaterials, 10(8), 1452. https://doi.org/10.3390/nano10081452







