1. Introduction
The use of engineered nanomaterials (ENMs) to clean-up polluted media, including groundwater and wastewater, has attracted more and more attention in the last decade [
1]. This approach offers the possibility to take advantage of the high reactivity and high surface area of nanomaterials, opening the way towards more effective and economically sustainable remediation processes.
Nevertheless, the use of ENMs generates concerns associated with the potential risk for humans and environment, as the (eco-)toxicological impact of these solutions is often underestimated [
2].
Recently, we proposed a systematic approach for possibly overcoming this issue, which consists of the use of sustainable and bio-based nanomaterials as building blocks for the design of nano-structured and nano-porous sorbents, capable of taking advantage of the intrinsic nano-dimension of the network, while overpassing the risk of ENM release and migration [
3].
In accordance with this strategy, we first identified polysaccharides, and cellulose in particular, as ideal sources, often derived from discharged biomass, for the extraction of nano-sized particles to be further processed [
4].
Cellulose represents the most abundant biodegradable and renewable polymer source in the biosphere, with an annual production estimated as over 7.5 × 10
10 tons. This almost inexhaustible sustainable polymer possesses unique chemical, physical, and mechanical properties, which have suggested its use for the production of a wide range of materials [
5].
Moreover, it is possible to cleave cellulose hierarchical structure in order to obtain nanocellulose (NC) in the form of nanocrystals (cellulose nanocrystals (CNC)) and nanofibers (cellulose nanofibers (CNF)). NC has been widely proposed as building block for the design of a wide range of sorbent materials to be used in wastewater treatment [
6,
7,
8].
Among the several mechanical and chemical approaches for NC extraction, in recent years we focused on the one we considered the most effective and economically convenient, consisting of the selective oxidation of C6 alcoholic groups of the cellulose glucopyranose units to the corresponding carboxylic acids [
9,
10]. This transformation is mediated by 2,2,6,6-tetramethylpiperidine 1-oxyl free radical (TEMPO)/NaClO/NaBr system [
9,
10], and allows to promote nanodefibrillation at basic pH, thanks to the electrostatic repulsion among negatively charged TEMPO-oxidized CNF (TOCNF), due to the deprotonation of carboxylic moieties. The obtained nanofibers present a diameter in a range of 5–100 nm and a length of several microns.
Following the strategy previously described, in 2015 we reported a new class of nanostructured sorbent cellulose nanosponges (CNS), obtained by thermally promoting the cross-linking between TOCNF and branched polyethyleneimine 25 kDa (bPEI), a polymer bearing a high amount of primary, secondary, and tertiary amino groups, thanks to which it is able to interact with a wide range of ions and molecules [
11].
In recent years, we modified the CNS formulation by adding an optimized amount of citric acid (CA) as an additional source of carboxylic groups, in order both to strength the mechanical properties of the final material by increasing cross-linking nodes [
12], and to better fix bPEI into the network, so that it was also possible to reduce the amount of the same polymer and to support an eco-safe and sustainable design [
13,
14]. Moreover, we also demonstrated the versatility of the system, with the possibility of grafting bPEI with suitable moieties before undergoing cross-linking, in order to give the material additional functional properties, such as the sensing of fluoride ions [
15,
16].
Due to the chelating action of amino-groups, CNS have shown superb performances as heavy metals sorbents from both fresh- [
11] and seawater [
13,
17]. On the contrary, the investigation into the behavior of this sorbent material with organic pollutants was limited to few examples, namely phenols, and simply based on an acid–base interaction.
However, by considering the morphology of the sponge, characterized by a high micro- and nano-porosity, as evidenced in previous works [
12,
18] and better discussed later on, we envisioned the possibility of exploiting the potential of CNS, and to better clarify the possible mechanisms of interaction between the sorbent and the selected organic pollutants.
Anionic organic dyes were chosen as model molecules, due to the continuous request for new and more sustainable technologies for treating a multiplicity of colored industrial wastewater, whose discharge in superficial water bodies can significantly impair freshwater use.
Nowadays, over 100,000 dyes are commercially available, with 7 × 10
5 tons/year of dyestuff being produced [
19]. Synthetic dyes are widely used in several industrial fields, including food, rubber, paper, cosmetic, pharmaceutical, automotive, and textile production chains. Inevitably, a certain fraction of dyes ends up in the washing water during the process and it has to be removed before release in the environment, due to the potential toxicity and negative effects on the aquatic ecosystem.
Due to their complex and variable chemical structure, many dyes are difficult to be removed by following a standard photo- and/or oxidative degradation approach, and sorption is often the process selected for wastewater treatment, with activated carbon mainly used for this purpose. In fact, sorption onto activated carbon is a well-established process, effective at removing a wide range of molecules, including color-responsible molecules from textile wastewater. Moreover, recently it has been reported that the efficiency of this process can be improved by driving the growth of nanoparticles into activated carbon pores, so that it is possible to combine sorption and degradation of the organic dye pollutant [
20]. More generally, sorption represents a simply managed process, with no important drawbacks and a relative low cost, compared to other processes, such as ozonation.
In 2016, Zhu and coworkers first reported the grafting of cellulose with hyperbranched polyethyleneimine for the selective sorption of a wide range of anionic and cationic organic dyes [
21]. The material was obtained by NaIO
4-mediated oxidation to afford dialdehyde cellulose, which in turn was reacted with bPEI in ethanol. While the approach is quite interesting, the synthetic procedure required controlled and anhydrous conditions and the use of flammable solvents, which could limit the scalability of the synthesis. One year later, Wang and coworkers reported the use of TEMPO-oxidized cellulose membranes modified with linear PEI to remove both anionic (xylenol orange (XO)) and cationic (methylene blue (MB)) dyes from wastewater [
22]. However, in this procedure the cross-linking is obtained by means of glutaraldehyde, which is known to be toxic. In both cases a good sorption performance was observed, and it was ascribed to an electrostatic interaction between the cationic polymer linked to cellulose and the dyes, with a sorption efficiency depending on the charge present on the surface. This aspect somehow limits the operating conditions of the process, as it would require wastewater pH adjustment before (and then, after) sorption treatment, with a consequent increase in the economic impact.
Herein, we report an investigation on the sorption efficiency of CNS towards four commercially available and highly used organic dyes (Naphthol Blue Black (NBB), Orange II Sodium Salt (OSS), Brilliant Blue R (BB), Cibacron Brilliant Yellow (CBY)). All the selected dyes presented a similar structure but differed in terms of molecular weight and the number of sulphate functional groups. The results in terms of sorption efficacy and regeneration efficiency were correlated with both the porosity of the sorbent sponge and the chemical and dimensional differences of the dyes, much more than on the charge present on the material. In fact, and quite surprisingly, CNS performed even better at a slightly basic pH, that is, one above the value of point of zero charge. Moreover, a comparison with an activated carbon could highlight the potentialities and limits of the proposed solution.
2. Materials and Methods
All the reagents were purchased from Sigma-Aldrich (Milano, Italy). Cotton linters was provided by Bartoli Spa paper mill (Capannori, Lucca, Italy). Deionized water was produced with a Millipore Elix® Deionizer with Progard® S2 ion exchange resins. UV spectra and data were recorded on a V-600 Series UV-vis spectrophotometer from JASCO (Cremella (LC), Italy). Other equipment used in the procedures include a Branson Sonifier 250 equipped with a 6.5 mm probe tip, a Heldolph multi-reax shaker (Schwabach, Germany) and a SP Scientific BenchTop Pro Lyophilizer (Perugia, Italy).
2.1. TOCNF Synthesis and Titration
Cellulose was oxidized to a degree of 1.546 mmol
COOH/g
TOCNF according to a procedure previously reported in the literature [
9,
10]. Briefly, cotton linters (190 g) were dispersed in deionized water produced with a Millipore Elix
® Deionizer with Progard
® S2 ion exchange resins (Milan, Italy) (total volume 5.7 L), in the presence of TEMPO (2.15 g, 13.8 mmol) and KBr (15.42 g, 129 mmol) and a solution of NaClO (12.5%
w/
w aqueous solution, 437 mL) was slowly added under vigorous stirring. During oxidation, pH was maintained in the range of 10.5-11 by dripping 4 N NaOH water solution. The solution was maintained stirred for 16 h. Oxidized cellulose nanofibers were aggregated by using concentrated HCl and then washed with deionized water on a Büchner funnel to reach a neutral pH (see
Supplementary Materials for further information).
To estimate the concentration of carboxyl groups on the cellulose structure after oxidation, a titration was performed with 0.1 N NaOH water solution, using phenolphthalein as a colorimetric indicator. The first step was the titration of NaOH 0.1 N. The detailed procedure and the equation used to calculate the concentration of the carboxyl groups are reported in
Supplementary Materials.
2.2. Synthesis of CNS
CNS were synthesized according to the procedure previously reported [
13]. First, 3.5 g of TOCNF were suspended in deionized water, adding a stoichiometric amount of granular NaOH. The suspension was ultrasonicated with a Branson Sonifier 250 equipped with a 6.5 mm probe tip to further promote the separation of the nanofibers, obtaining a homogeneous solution, which was then acidified with 12 M HCl, filtered on a Büchner funnel under vacuum, and washed with deionized water until neutrality. Then, two aqueous solutions of 25 kDa bPEI (3.5 g of bPEI in 10 mL) and anhydrous citric acid (CA) (0.896 g in 10 mL) were slowly added to the TOCNF solution, while continuously stirring until obtaining a white and homogeneous hydrogel, which was placed in well-plates, quickly frozen at −35 °C, freeze-dried for 48 h using a SP Scientific BenchTop Pro Lyophilizer (at –52 °C temperature and 140 μbar pressure) and then thermally treated in the laboratory oven at a maximum temperature of 102 °C for 16 h. At the end of the process, CNS was grinded with a mortar and then washed with water to remove the excess bPEI (for further information, see
Supplementary Materials). The particle size distribution of the grinded CNS powder was measured in the Laboratory Chemical Analysis (LAC) of Politecnico di Milano by means of a Malvern Mastersizer 3000 Particle Size Analyzer (Malvern, UK) with Fraunhofer modeling, which considers opaque non-spherical particles. The CNS powder was suspended under stirring in 500 mL of water to reach an obscuration level in the range of 8–12%.
2.3. Point of Zero Charge (PZC) Calculation
The pH of the point of zero charge (pH
PZC), namely the pH above which the total surface of the sorbent material is negatively charged, was measured by the pH drift method [
23]. For this purpose, 20 mL of a 0.01 M NaCl solution was placed in a Falcon vial and N
2 was bubbled through the solution to steady the pH by preventing the dissolution of CO
2 in the solution. The pH was then adjusted to selected initial values between 2 and 12, by dripping HCl 0.01 N or NaOH 0.01 N, and the sorbent (0.06 g) was added to the solution. The final pH, reached after 4 h, was measured and plotted against the initial pH. The pH at which the curve crosses the bisector pH(final) = pH(initial) is the pH
PZC of the given sorbent. The considered sorbents were CNS and the activated carbon SAE SUPER, provided by Norit (Italy) (see
Supplementary Materials for general characteristics).
2.4. Preliminary Sorption Tests
The tests were carried out by dipping 12 mg of CNS powder in 15 mL of aqueous solutions of the selected dye for 24 h, under static conditions and at room temperature in a Falcon vial. The selected dyes are reported in
Figure 1. The concentration of the buffer dye solution was selected considering the extinction coefficient of each dye (20 mg/L for OSS and NBB, 100 mg/L for BB and CBY) and the type of buffer was chosen according to the solubility of the dye in the buffer solution at room temperature (OSS: 116 g/L, NBB: 30 g/L, BB: 70 g/L, CBY: 50 g/L). Tests at pH 5.5 were carried out in piperazine and citrate buffer. Tests at pH 7.6 were conducted in deionized water using the normal buffering power of CNS. Each sample has been reproduced in triplicate. After 24 h, one collection was taken from each sample and analyzed by UV analysis. UV-Vis spectra (
Figure S1), characteristic λ
max (
Table S1), extinction coefficients (
Table S2) and calibration lines (
Figures S2–S5) for all the four dyes are reported in
Supplementary Materials.
2.5. Isotherms and Kinetics
Isotherm and kinetic sorption tests were carried out under dynamic conditions (using the shaker at 450 rpm) at room temperature (25 °C). Isotherm tests were carried out by maintaining a constant sorbent quantity and solution volume throughout the data gathering (24 h), while changing the solution concentration. Eight different concentrations were tested for each dye and three trials were carried out for each concentration. The sponge-to-solution ratio used was 12 mg of CNS/15 mL of mono-contaminated dye solution (0.8 mg/mL). The selected concentrations for each dye are described in
Table S3 in
Supplementary Materials. The absorbance of the solutions was measured at time zero and after 24 h.
As for kinetic tests, the initial concentrations for each dye are reported in
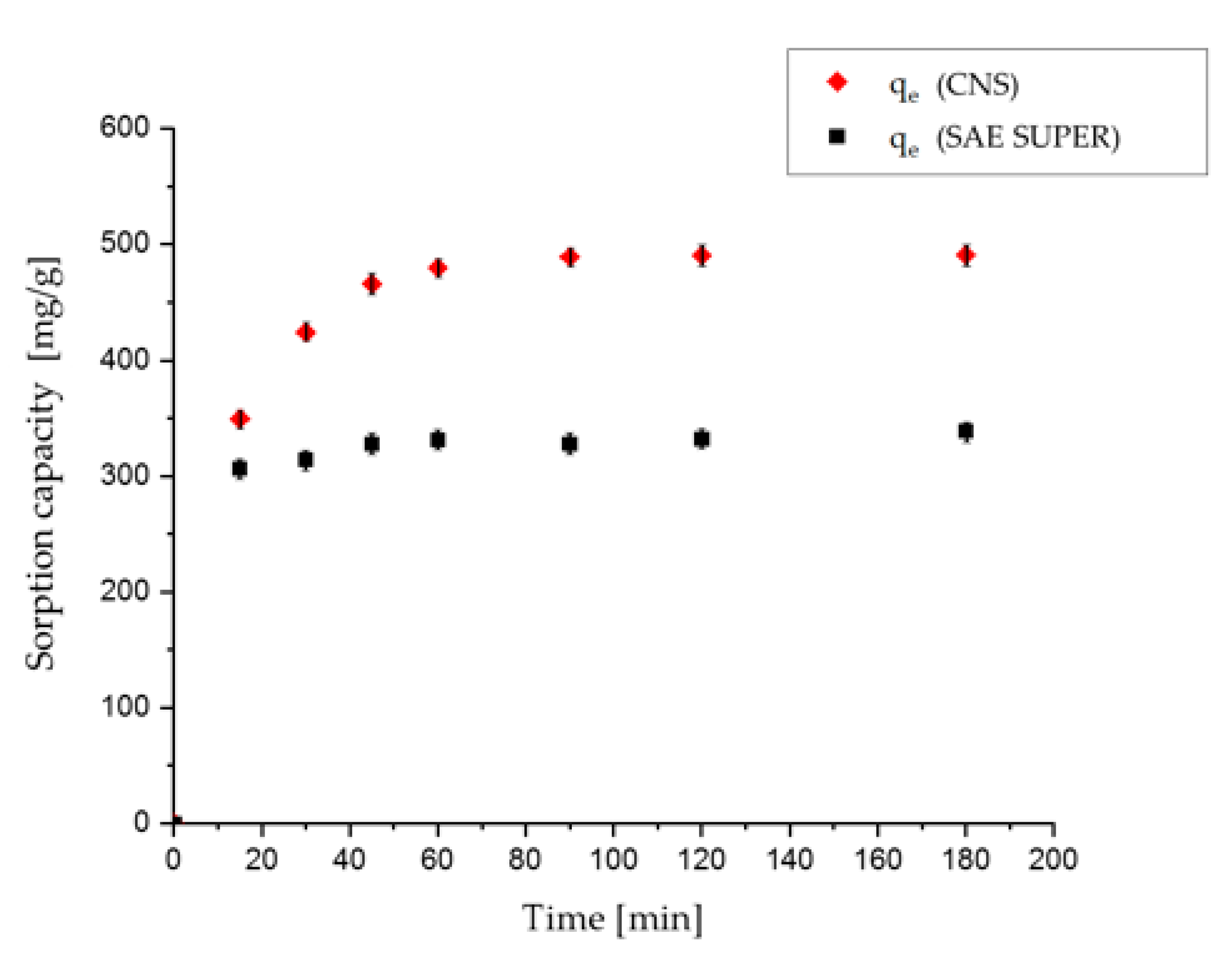
Table 1. The trials were performed under dynamic conditions and at room temperature, as described above. Samples were shaken and analyzed after 15, 30, 45, 60 and 90 min, and 2, 3, 4, 6, 8 and 24 h. A volume of 25 mL of solution was used for each test, to allow withdrawals from the solution 11 times, keeping the total diminution of the volume below 10%. The quantities of sorbent for each kinetic test were 20 mg for the lowest concentration and 40 mg for the highest concentration.
2.6. Desorption and Reusability Tests
Tests were conducted to evaluate the possibility of reusing the sponges. At first, colored CNS was produced by leaving white CNS powder in contact with a solution of the selected dye in static conditions (5 g/L concentration, 30 mL, 200 mg of CNS) for 24 h, then filtering it on a Büchner funnel and washing it with deionized water.
The desorption test was conducted with HCl 0.1 N and NaOH 0.1 N. A total of 20 mg of colored CNS was soaked in 20 mL of each solution under static conditions at room temperature for 24 h. After the first test, which decreed the efficiency for only the NaOH solution, three different molar concentrations of NaOH were compared—0.5, 0.1 and 0.05 N—following the same discoloration procedure. Other types of alkaline solutions were tested using the previously reported conditions: the test was repeated with triethylamine (TEA), NH3 30% aqueous solution and KOH 0.1 N aqueous solution.
The reusability test consisted of a sorption test conducted on the decolored sponge. The test was carried out only on the OSS discolored CNS and following the same procedure as for previous sorption tests: 12 mg of sorbent in 15 mL of 20 mg/L solution of OSS dye for 24 h under static conditions at room temperature. Five sorption–desorption cycles were carried out. For this purpose, 100 mg of OSS colored CNS were soaked in 110 mL of NaOH 0.05 N. The resulting sponge was then vacuum-filtered with the aid of a Büchner funnel and washed with deionized water until neutrality. Once dried, the sponge was weighted for the next phase of sorption with a constant sponge-to-solution ratio of 0.8 mg/mL. Each sample was carried out in triplicate. After 24 h of static sorption at room temperature, absorbance was analyzed. The sponge was then gravity-filtered and air-dried before the new desorption phase, carried out with NaOH 0.05 M solution.
2.7. Comparison between CNS and Activated Carbons
A comparison test was carried out by evaluating the sorption capacity of the activated carbon SAE SUPER. The experimental setup was the same as the previous tests, using a sorbent-to-solution ratio of 0.8 mg/mL. Two initial concentrations were tested for each dye (C
0 low and C
0 high, reported in
Table 1). This test was carried out in dynamic conditions and equilibrium was reached after 24 h in the multi reax shaker at 450 rpm. Filtration via syringe filter was required for this test due to the fine particulate dispersion of activated carbons in the solution.
Kinetic tests were carried out using SAE SUPER activated carbon in OSS solutions at high and low concentrations. For the high-concentration trial, 240 mg of SAE SUPER activated carbon were dispersed in 150 mL of 800 mg/L OSS solution. Three trials were prepared and agitated by magnetic stirring. A volume of 1 mL was withdrawn for each measurement, filtered through a cotton filter and opportunely diluted. Withdrawals were performed after 15, 30, 45, 60 and 90 min, 2, 3 and 24 h. The total amount of withdrawn solution after eight samplings was still lower than 10% of the total volume of the solution.
4. Conclusions
In this work, we reported the use of a nanostructured-cellulose-based sorbent material for water decontamination from anionic organic dyes. The material was prepared starting from biomass sources, by combining TEMPO-oxidized cellulose nanofibers, branched polyethyleneimine, and citric acid, and following a simple thermal protocol. The sorption performance was tested on four commercial dyes (OSS, BB, NBB, and CBY), differing for both molecular dimension and the number of sulfonate groups present onto the molecular structure (one for OSS, two for BB and NBB, three for CBY). The sorbent was effective also at a slightly basic pH, even if under these conditions the nanostructured sponge is negatively charged. This result suggested that the sorbent–solute interaction should not be simply ascribed to electrostatic attraction between opposite charges, but other intermolecular interactions could occur between the sulfonate groups of the dyes and the amino groups present on the nano-sponge. The role of bPEI in the network was crucial, as cellulose alone was not able to reproduce significant sorption.
Isotherm and kinetic investigation revealed a molecular-size dependence of sorption performance, as the smallest OSS is much more trapped on the material, probably because of the possibility of it being more diffused in the nano-porous network. Nevertheless, these studies also showed that the strength of sorbent–solute binding was higher when two or more sulfonate groups were present on the dye. This evidence was also confirmed by conducting regeneration and reusability tests, as once again OSS was much more easily removed from the nano-sponge under alkaline conditions, so that the sorbent system could be reused several times, by maintaining its sorption efficiency.
The dye-removal efficiency of the material herein described was compared to that of the commercially available activated carbon SAE SUPER. While with the nano-sponge the sorption kinetic was slightly slower, probably due to the nano-porous structure with respect to a microporous structure of the activated carbon, the sorption capacity was higher for all dyes except BB. In any case, the advantages of the use of this cellulose-based material can be found in its easy handling, reusability, eco-safety, and sustainability, as it can be produced from wasted biomass, following the virtuous route of the circular economy.