Trastuzumab: More than a Guide in HER2-Positive Cancer Nanomedicine
Abstract
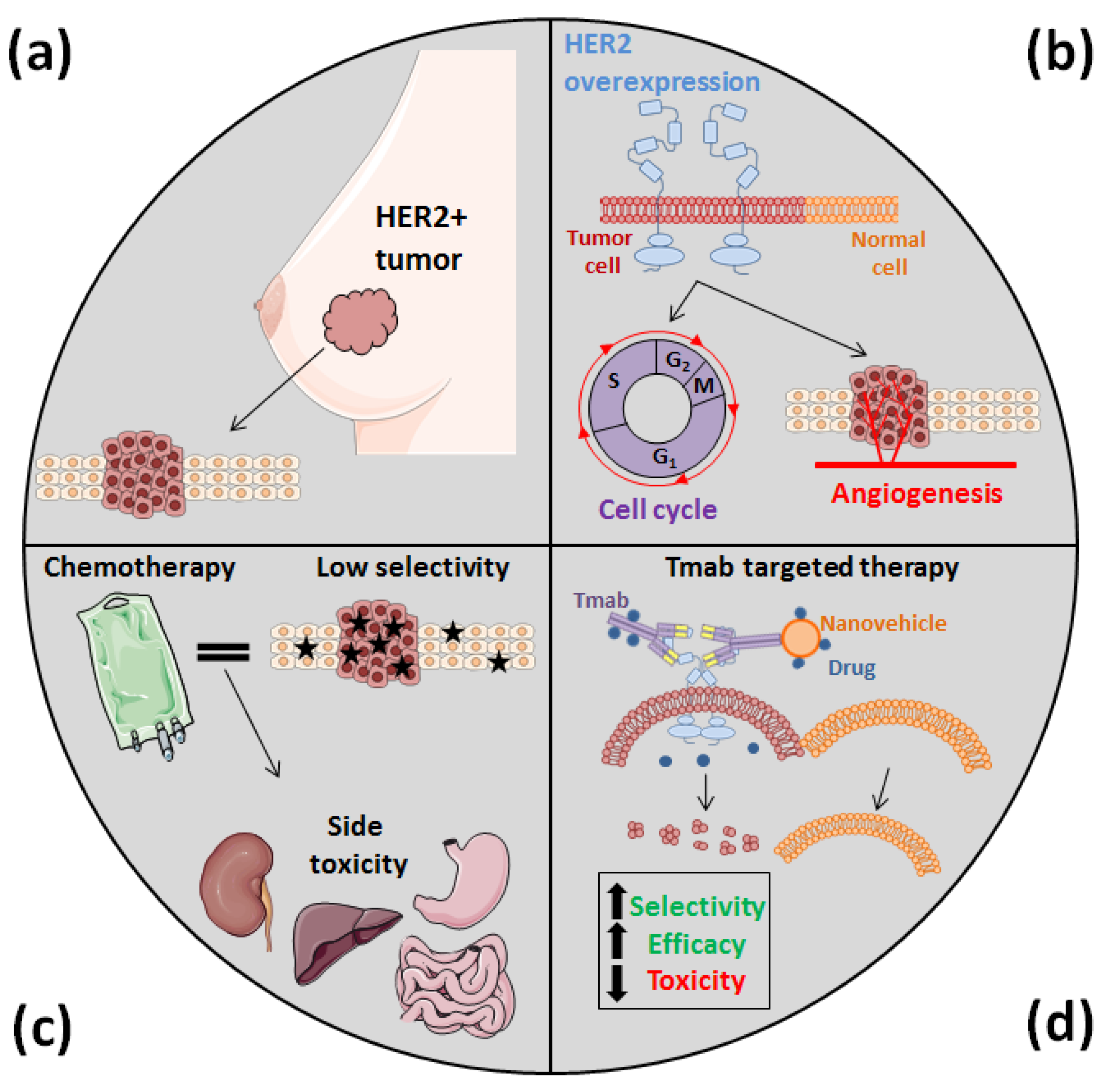
:1. Introduction
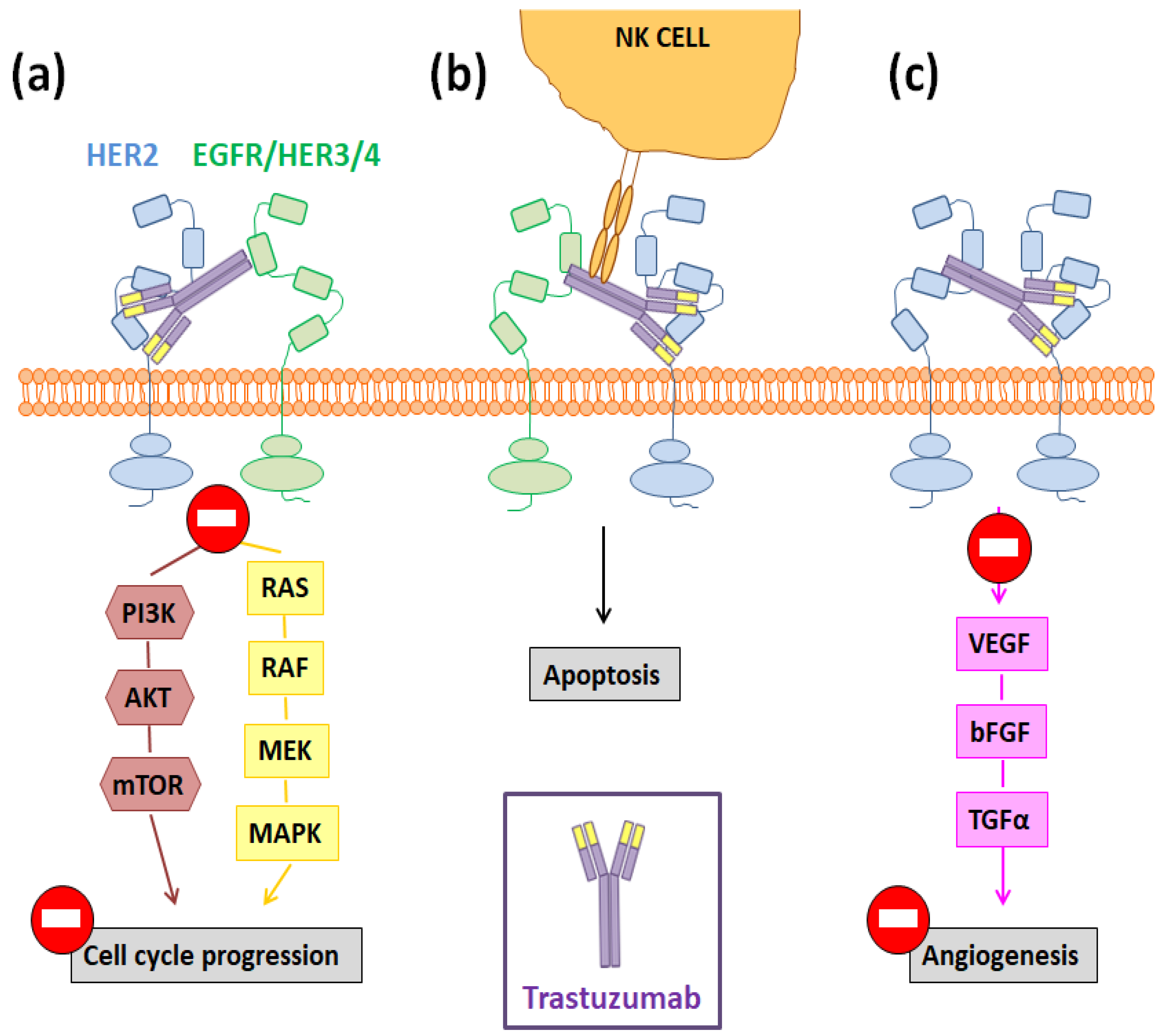
2. Trastuzumab: More Than a Guide for Nanomedicines
3. Nanoparticle and Liposome Functionalization with Trastuzumab: Usual Strategies
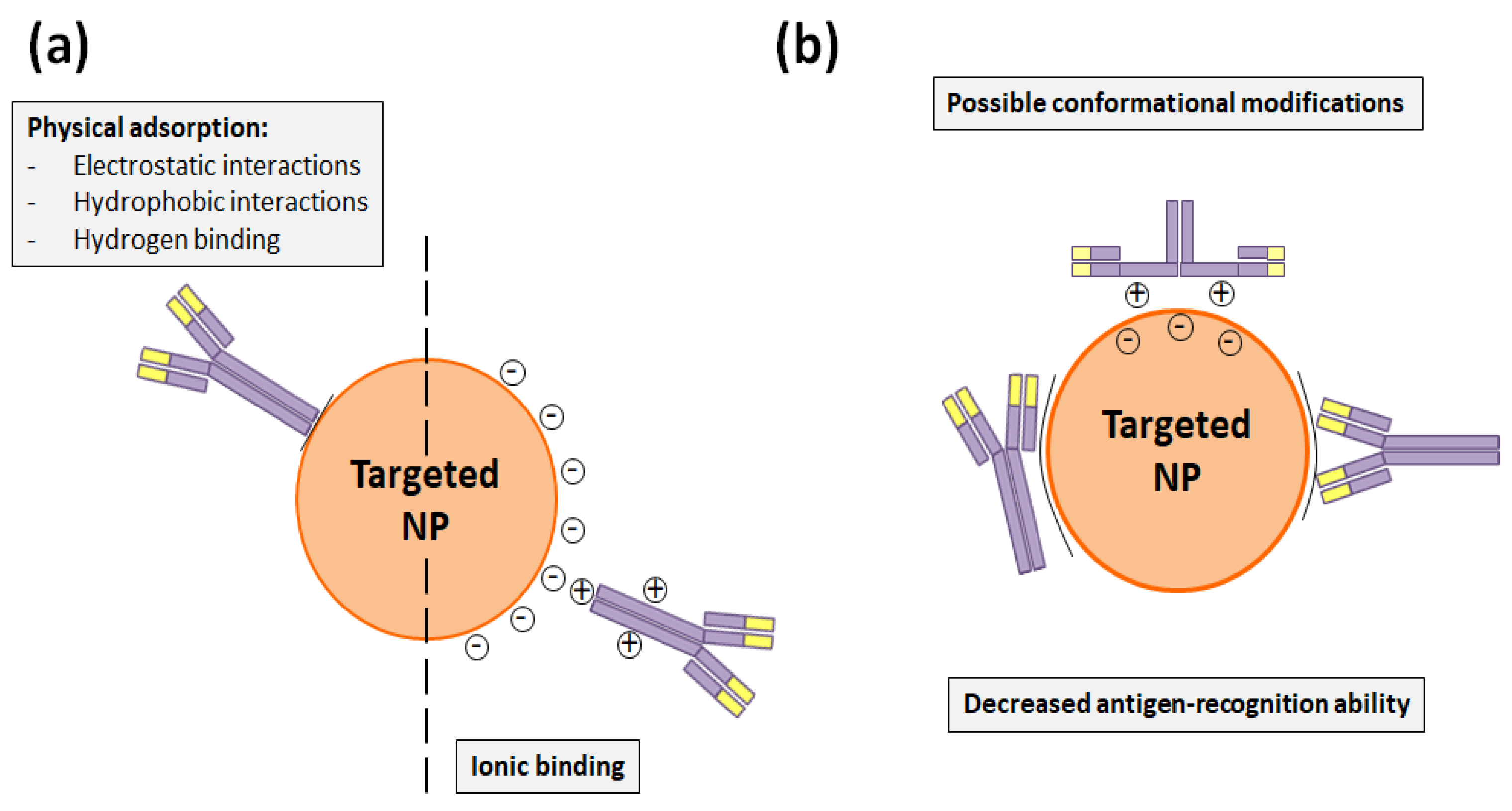
3.1. Functionalization through Trastuzumab Adsorption
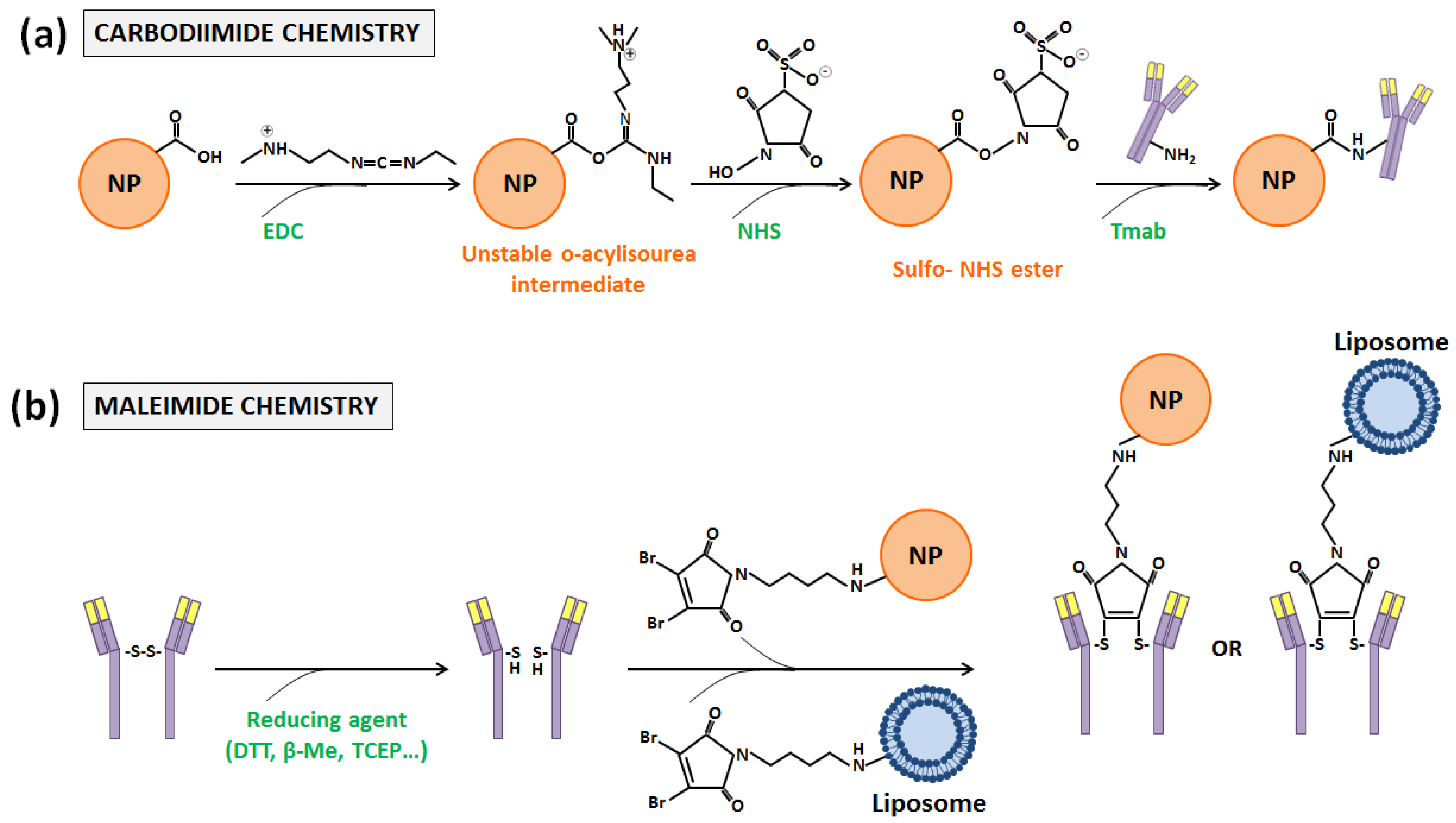
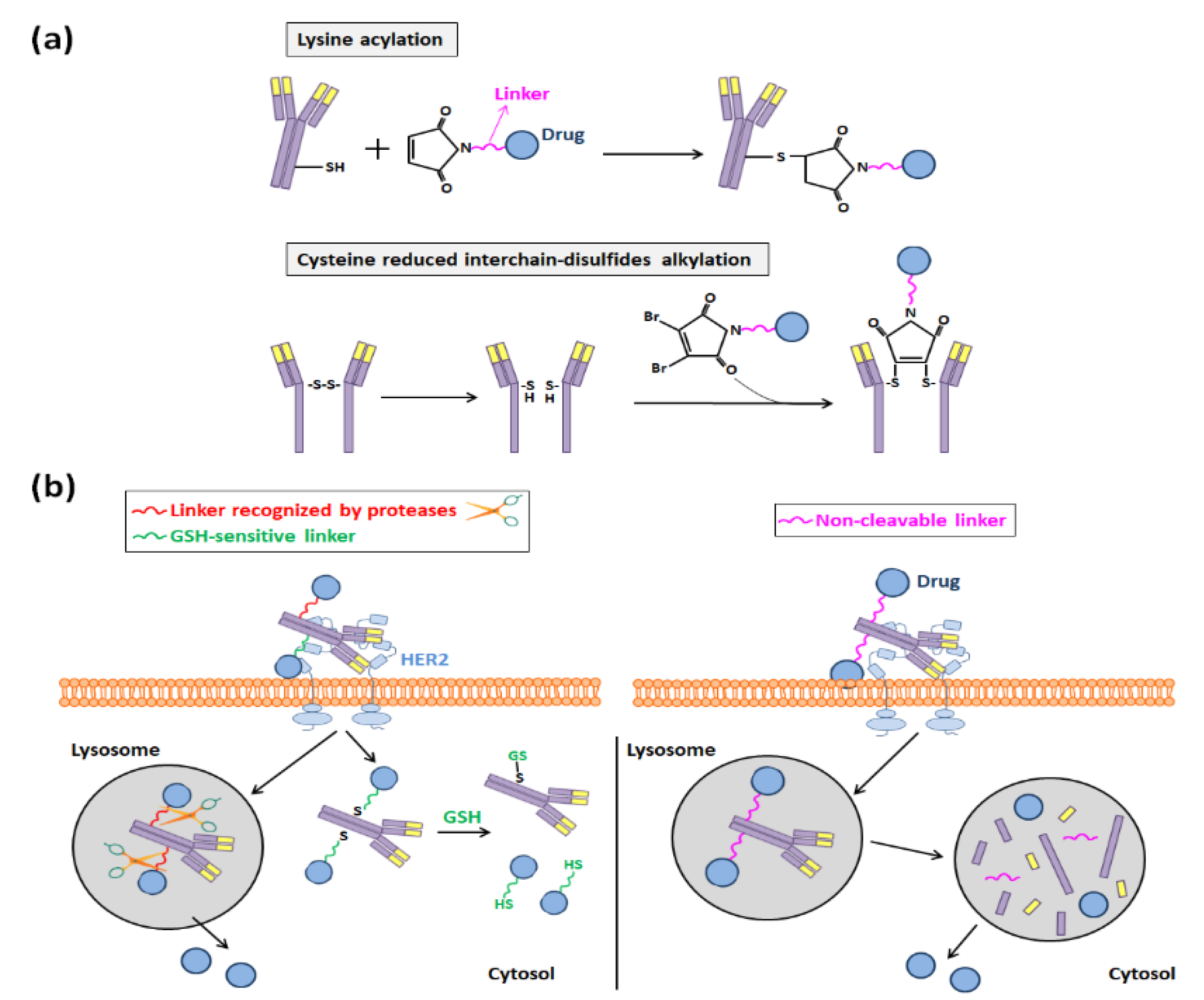
3.2. Trastuzumab-Functionalization through Covalent Bindings
4. Trastuzumab Role in Antibody-Drug Conjugates Development
5. Current and Future Situation of Trastuzumab-Based Nanomedicine
6. Conclusions and Future Directions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADC | Antibody drug conjugate |
| ADCC | Antibody-dependent cellular cytotoxicity |
| AF-HPA | Auristatin F-hydroxypropylamide |
| BCN | Bicyclononyme |
| β-Me | 2-mercaptoethanol |
| DDS | Drug delivery system |
| DENC | Double emulsion nanocapsule |
| DTT | Dithiothreitol |
| EBC | Early breast cancer |
| EDC | 1-ethyl-3-(3-dimethylaminopropyl) |
| EGFR | Epidermal growth factor receptor |
| EMA | European Medicines Agency |
| EPR | Enhanced permeability and retention |
| FDA | U.S. Food and Drug Administration |
| GMBS | 4-maleimidobutyric acid-N hydroxysuccinimide |
| GSH | Glutathione |
| HER | Human epithermal growth factor receptor |
| HSA | Human serum albumin |
| IgG | Immunoglobulin |
| MBC | Metastatic breast cancer |
| MDR | Multidrug resistance |
| MMA | Monomethyl auristatin |
| MRI | Magnetic resonance imaging |
| MTT | Montmorillonite |
| MTX | Methotrexate |
| NGM | Next-generation maleimide |
| NHS | N-hydroxysulfosuccinimide |
| NPs | Nanoparticles |
| NSCLC | Non-small cell lung carcinoma |
| PBD-MA | Pyrrolo[2,1-c][1,4]benzodiazepine monoamide |
| PD | Pyridazinedione |
| PEG | Polyethylene glycol |
| PEI | Polyethylenimine |
| PET | Positron Emission Tomography |
| Phis | Poly-L-histidine |
| PLA | Polylactide |
| PLGA | Poly(lactic-co-glycolic acid) |
| PPɣ | Polypyrrole |
| SMCC | Succinimidyl-4-(N-maleimidomethyl)-cyclohexane-1-carboxylate |
| TCEP | Tris-2-carboxyethylphosphine |
| TCO | Trans-cyclooctene |
| THP | Tumor homing peptide |
| TKI | Tyrosine-kinase inhibitor |
| Tmab | Trastuzumab |
| TPGS | Tocopheryl polyethylene glycol succinate |
| Tz | 1,2,4,5-tetrazine |
| VEGFR | Vascular endothelial growth factor receptor |
| WHO | World Health Organization |
References
- World Health Organization (WHO). Available online: https://www.who.int/news-room/fact-sheets/detail/cancer (accessed on 18 April 2020).
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meisel, J.L.; Zhao, J.; Suo, A.; Zhang, C.; Wei, Z.; Taylor, C.; Aneja, R.; Krishnamurti, U.; Li, Z.; Nahta, R.; et al. Clinicopathologic factors associated with response to neoadjuvant anti-HER2-directed chemotherapy in HER2-positive breast cancer. Clin. Breast Cancer 2020, 20, 19–24. [Google Scholar] [CrossRef] [PubMed]
- Marchiò, C.; Annaratone, L.; Marques, L.; Casorzo, L.; Berrino, E.; Sapino, A. Evolving concepts in HER2 evaluation in breast cancer: Heterogeneity, HER2-low carcinomas and beyond. Semin. Cancer Biol. 2020, in press. [Google Scholar] [CrossRef]
- Jiang, J.; Dong, L.; Wang, L.; Wang, L.; Zhang, J.; Chen, F.; Zhang, X.; Huang, M.; Li, S.; Ma, W.; et al. HER2-targeted antibody drug conjugates for ovarian cancer therapy. Eur. J. Pharm. Sci. 2016, 93, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Mehataa, A.K.; Bhartic, S.; Singhb, P.; Viswanadha, M.K.; Kumaria, L.; Agrawald, P.; Singha, S.; Kochb, B.; Muthua, M.S. Trastuzumab decorated TPGS-g-chitosan nanoparticles for targeted breast cancer therapy. Colloid Surf. B 2019, 173, 366–377. [Google Scholar] [CrossRef] [PubMed]
- Dang, Y.; Guan, J. Nanoparticle-based drug delivery systems for cancer therapy. Smart Mater. Med. 2020, 1, 10–19. [Google Scholar] [CrossRef]
- Khan, H.; Mirzaei, H.R.; Amiri, A.; Akkol, E.K.; Halimi, S.M.A.H.; Mirzaei, H. Glyco-nanoparticles: New drug delivery systems in cancer therapy. Semin. Cancer Biol. 2020, in press. [Google Scholar] [CrossRef]
- Correia, A.L.; Bissell, M.J. The tumor microenvironment is a dominant force in multidrug resistance. Drug Resist. Update 2012, 15, 39–49. [Google Scholar] [CrossRef] [Green Version]
- Raj, S.; Khurana, S.; Choudhari, R.; Kesari, K.K.; Kamal, M.A.; Garg, N.; Ruokolainen, J.; Das, B.C.; Kumar, D. Specific targeting cancer cells with nanoparticles and drug delivery in cancer therapy. Semin. Cancer Biol. 2019, in press. [Google Scholar] [CrossRef]
- Xu, C.H.; Ye, P.J.; Zhou, Y.C.; He, D.X.; Wei, H.; Yu, C.Y. Cell membrane-camouflaged nanoparticles as drug carriers for cancer therapy. Acta Biomater. 2020, 105, 1–14. [Google Scholar] [CrossRef]
- Yih, T.C.; Al-Fandi, M. Engineered nanoparticles as precise drug delivery systems. J. Cell. Biochem. 2006, 97, 1184–1190. [Google Scholar] [CrossRef] [PubMed]
- Elzoghby, A.O.; Samy, W.M.; Elgindy, N.A. Albumin-based nanoparticles as potential controlled release drug delivery system. J. Control. Release 2012, 157, 168–182. [Google Scholar] [CrossRef] [PubMed]
- Duncan, B.; Kim, C.; Rotello, V.M. Gold nanoparticle platforms as drug and biomacromolecule delivery systems. J. Control. Release 2010, 148, 122–127. [Google Scholar] [CrossRef] [Green Version]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, P.; Biswas, S.; Torchilin, V.P. Current trends in the use of liposomes for tumor targeting. Nanomedicine (Lond.) 2013, 8, 1509–1528. [Google Scholar] [CrossRef] [Green Version]
- Marques, A.C.; Costa, P.J.; Velho, S.; Amaral, M.H. Functionalizing nanoparticles with cancer-targeting antibodies: A comparison of strategies. J. Control. Release 2020, 320, 180–200. [Google Scholar] [CrossRef]
- Belfiore, L.; Saunders, D.N.; Ranson, M.; Thurecht, K.J.; Storm, G.; Vine, K.L. Towards clinical translation of ligand-functionalized liposomes in targeted cancer therapy: Challenges and opportunities. J. Control. Release 2018, 277, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nava Andrade, K.; Puebla Pérez, A.M.; Carbajal Arízaga, G.G. Passive and active targeting strategies in hybrid layered doublé hydroxides nanoparticles for tumor bioimaging and therapy. Appl. Clay Sci. 2019, 181, 105214. [Google Scholar] [CrossRef]
- Alibakhshi, A.; Kahaki, F.A.; Ahangarzadeh, S.; Yaghoobi, H.; Yarian, F.; Arezumand, R.; Ranjbari, J.; Mokhtarzadeh, A.; de la Guardia, M. Targeted cancer therapy through antibody fragments-decorated nanomedicines. J. Control. Release 2017, 268, 323–334. [Google Scholar] [CrossRef] [PubMed]
- Reverdatto, S.; Burz, D.S.; Shekhtman, A. Peptide aptamers: Development and applications. Curr. Top. Med. Chem. 2015, 15, 1082–1101. [Google Scholar] [CrossRef] [Green Version]
- Banerjee, J.; Nilsen-Hamilton, M. Aptamers: Multifunctional molecules for biomedical research. J. Mol. Med. 2013, 1, 1333–1342. [Google Scholar] [CrossRef] [PubMed]
- Zhao, P.; Zhanga, Y.; Lia, W.; Jeantya, C.; Xiangb, G.; Dong, Y. Recent advances of antibody drug conjugates for clinical applications. Acta Pharm. Sin. B 2020, in press. [Google Scholar] [CrossRef]
- Parslow, A.C.; Parakh, S.; Lee, F.T.; Gan, H.K.; Scott, A.M. Antibody–drug conjugates for cancer therapy. Biomedicines 2016, 4, 14. [Google Scholar] [CrossRef] [PubMed]
- Horta, E.; Bongiorno, C.; Ezzeddine, M.; Neil, E.C. Neurotoxicity of antibodies in cancer therapy: A review. Clin. Neurol. Neurosurg. 2020, 188, 105566–105574. [Google Scholar] [CrossRef] [PubMed]
- Nahta, R.; Hung, M.C.; Esteva, F.J. The HER2-targeting antibodies trastuzumab and pertuzumab synergistically inhibit the survival of breast cancer cells. Cancer Res. 2004, 64, 2343–2346. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Blackwell, K.; Gligorov, J.; Jacobs, I.; Twelves, C. The global need for a trastuzumab biosimilar for patients with HER2-positive breast cancer. Clin. Breast Cancer 2018, 18, 95–113. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Collins, D.; Hill, A.D.K.; Young, L. Lapatinib: A competitor or companion to trastuzumab? Cancer Treat. Rev. 2009, 35, 574–581. [Google Scholar] [CrossRef] [PubMed]
- Vogel, C.L.; Cobleigh, M.A.; Tripathy, D.; Gutheil, J.C.; Harris, L.N.; Fehrenbacher, L.; Slamon, D.J.; Murphy, M.; Novotny, W.F.; Burchmore, M.; et al. Efficacy and safety of trastuzumab as a single agent in first-line treatment of HER2-overexpressing metastatic breast cancer. J. Clin. Oncol. 2002, 20, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Slamon, D.; Eiermann, W.; Robert, N.; Pienkowski, T.; Martín, M.; Press, M.; Mackey, J.; Glaspy, J.; Chan, A.; Pawlicki, M.; et al. Adjuvant trastuzumab in HER2-positive breast cancer. N. Eng. J. Med. 2011, 365, 1273–1283. [Google Scholar] [CrossRef] [Green Version]
- Zhang, X.; Chen, J.; Weng, Z.; Li, Q.; Zhao, L.; Yu, N.; Deng, L.; Xu, W.; Yang, Y.; Zhu, Z.; et al. A new anti-HER2 antibody that enhances the anti-tumor efficacy of trastuzumab and pertuzumab with a distinct mechanism of action. Mol. Immunol. 2020, 119, 48–58. [Google Scholar] [CrossRef]
- Nahta, R.; Esteva, F.J. Molecular mechanisms of trastuzumab resistance. Breast Cancer Res. 2006, 8, 215. [Google Scholar] [CrossRef] [PubMed]
- Hassan, G.; Du, J.; Afify, S.M.; Seno, A.; Seno, M. Cancer stem cell generation by silenced MAPK enhancing PI3K/AKT signaling. Med. Hypotheses 2020, 141, 109742–109747. [Google Scholar] [CrossRef] [PubMed]
- Dubská, L.; Andӗra, L.; Sheard, M.A. HER2 signaling downregulation by trastuzumab and suppression of the PI3K/Akt pathway: An unexpected effect of TRAIL-induced apoptosis. FEBS Lett. 2005, 579, 4149–4158. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamen, L.; Thakurta, T.; Myneni, S.; Zheng, K.; Chung, S. Development of a kinetic antibody-dependent cellular cytotoxicity assay. J. Immunol. Methods 2019, 468, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Wen, X.F.; Yang, G.; Mao, W.; Thornton, A.; Liu, J.; Bast, R.C.; Le, X.F. HER2 signaling modulates the equilibrium between pro- and antiangiogenic factors via distinct pathways: Implications for HER2-targeted antibody therapy. Oncogene 2006, 25, 6986–6996. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Colombo, M.; Corsi, F.; Foschi, D.; Mazzantini, E.; Mazzucchelle, S.; Morasso, C.; Occhipinti, E.; Polito, L.; Prosperi, D.; Ronchi, S.; et al. HER2-targeting as a two-side strategy for breast cancer diagnosis and treatment: Outlook and recent implications in nanomedical approaches. Pharmacol. Res. 2010, 62, 150–165. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, J.P.; Prado, A.R.; Keijok, W.J.; Antunes, P.W.P.; Yapuchura, E.R.; Guimarães, M.C.C. Impact of conjugation strategies for targeting of antibodies in gold nanoparticles for ultrasensitive detection of 17β-estradiol. Sci. Rep. 2019, 9, 13859. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Feng, S.S. The synergistic effect of Herceptin and docetaxel in polylactide-D-α-tocopheryl polyethylene glycol succinate (PLA-TPGS) nanoparticles. J. Control. Release 2011, 152, e1–e132. [Google Scholar] [CrossRef]
- Yu, K.; Zhao, J.; Zhang, Z.; Gao, Y.; Zhou, Y.; Teng, L.; Li, Y. Enhanced delivery of Paclitaxel using electrostatically-conjugated Herceptin-bearing PEI/PLGA nanoparticles against HER-positive breast cancer cells. Int. J. Pharm. 2016, 497, 78–87. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, J.; Li, X.; Li, F.; Lee, R.J.; Sun, F.; Li, Y.; Liu, Z.; Teng, L. Trastuzumab-coated nanoparticles loaded with docetaxel for breast cancer therapy. Dose-Response 2019, 17, 1–12. [Google Scholar] [CrossRef]
- Sun, B.; Ranganathan, B.; Feng, S.S. Multifunctional poly(D,L-lactide-co-glycolide)/montmorillonite (PLGA/MTT) nanoparticles decorate by Trastuzumab for targeted chemotherapy of breast cancer. Biomaterials 2008, 29, 475–486. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.S.; Janga, W.S.; Parka, J.S. Comparison of adsorption and conjugation of Herceptin on poly(lactic-coglycolic acid) nanoparticles—Effect on cell internalization in breast cancer cells. Mat. Sci. Eng. C Mater. 2018, 92, 496–507. [Google Scholar] [CrossRef] [PubMed]
- Nakajima, N.; Ikada, Y. Mechanism of amide formation by carbodiimide for bioconjugation in aqueous media. Bioconjug. Chem. 1995, 6, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Shirshahi, V.; Shamsipour, F.; Hassan Zarnani, A.; Verdi, J.; Saber, R. Active targeting of HER2-positive breast cancer cells by Herceptin-functionalized organically modified silica nanoparticles. Cancer Nanotechnol. 2013, 4, 27–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hadi Jazayeri, M.; Amani, H.; Akbar Pourfatollah, A.; Pazoki-Toroudi, H.; Sedighimoghaddam, B. Various methods for gold nanoparticles (GNPs) conjugation to antibodies. Sens. Biosens. Res. 2016, 9, 17–22. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Z.; Badkas, A.; Stevenson, M.; Lee, J.Y.; Leung, Y.K. Herceptin conjugated PLGA-PHis-PEG pH sensitive nanoparticles for targeted and controlled drug delivery. Int. J. Pharm. 2015, 487, 81–90. [Google Scholar] [CrossRef] [PubMed]
- Nieto, C.; Centa, A.; Rodríguez-Rodríguez, J.A.; Pandiella, A.; Martín del Valle, E.M. Paclitaxel-trastuzumab mixed nanovehicle to target HER2-overexpressing tumors. Nanomaterials 2019, 9, 948. [Google Scholar] [CrossRef] [Green Version]
- Domínguez-Ríos, R.; Sánchez-Ramírez, D.R.; Ruiz-Saraya, K.; Oceguera-Basurtoa, P.E.; Almadab, M.; Juárez, J.; Zepeda-Moreno, A.; Del Toro-Arreola, A.; Topete, A.; Daneri-Navarro, A. Cisplatin-loaded PLGA nanoparticles for HER2 targeted ovarian cancer therapy. Colloid Surface B 2019, 178, 199–207. [Google Scholar] [CrossRef]
- Arya, G.; Vandana, M.; Acharya, S.; Sahoo, S.K. Enhanced antiproliferative activity of Herceptin (HER2)-conjugated gemcitabine-loaded chitosan nanoparticle in pancreatic cancer therapy. Nanomed. Nanotechnol. 2011, 7, 859–870. [Google Scholar] [CrossRef]
- Haghighia, A.H.; Faghihb, Z.; Taghi Khorasanic, M.; Farjadian, F. Antibody conjugated onto surface modified magnetic nanoparticles for separation of HER2+ breast cancer cells. J. Magn. Magn. Mater. 2019, 490, 165479. [Google Scholar] [CrossRef]
- Dziawer, Ł.; Majkowska-Pilip, A.; Gaweł, D.; Godlewska, M.; Pruszynski, M.; Jastrzebski, J.; Bogdan Was, B.; Bilewicz, A. Trastuzumab-modified gold nanoparticles labeled with 211At as a prospective tool for local treatment of HER2-positive breast cancer. Nanomaterials 2019, 9, 632. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Almaki, J.H.; Nasiri, Z.; Idris, A.; Nasiri, M.; Majid, F.A.A.; Losic, D. Trastuzumab-decorated nanoparticles for in vitro and in vivo tumor-targeting hyperthermia of HER2+ breast cancer. J. Mater. Chem. B 2017, 5, 7369–7383. [Google Scholar] [CrossRef] [PubMed]
- Martín, F.J.; Papahadjopoulos, D. Irreversible coupling of immunoglobulin fragments to performed vesicles. An improved method for liposome targeting. J. Biol. Chem. 1982, 257, 286–288. [Google Scholar] [PubMed]
- Martínez-Jothar, L.; Doulkeridou, S.; Schiffelers, R.M.; Sastre Torano, J.; Oliveira, S.; van Nostrum, C.F.; Hennink, W.E. Insights into maleimide-thiol conjugation chemistry: Conditions for efficient surface functionalization of nanoparticles for receptor targeting. J. Control. Release 2018, 282, 101–109. [Google Scholar] [CrossRef]
- Akkapeddi, P.; Azizi, S.A.; Freedy, A.M.; Cal, P.M.S.D.; Gois, P.M.P.; Bernardes, G.J.L. Construction of homogeneous antibody-drug conjugates using site-selective protein chemistry. Chem. Sci. 2016, 7, 2954–2963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taheri, A.; Dinarvand, R.; Atyabi, F.; Ghahremani, M.H.; Ostad, S.N. Trastuzumab decorated methotrexate–human serum albumin conjugated nanoparticles for targeted delivery to HER2 positive tumor cells. Eur. J. Pharm. Sci. 2012, 47, 331–340. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, H.T.; Tran, T.H.; Thapa, R.K.; Phung, C.D.; Shin, B.S.; Jeong, J.H.; Choi, H.G.; Yong, C.S.; Kim, J.O. Targeted co-delivery of polypyrrole and rapamycin by trastuzumab-conjugated liposomes for combined chemo-photothermal therapy. Int. J. Pharm. 2017, 527, 61–71. [Google Scholar] [CrossRef]
- Amin, M.; Pourshohod, A.; Kheirollah, A.; Afrakhteh, M.; Gholami-Borujeni, F.; Zeinali, M.; Jamalan, M. Specific delivery of idarubicin to HER2-positive breast cancerous cell line by trastuzumab-conjugated liposomes. J. Drug Deliv. Sci. Technol. 2018, 47, 209–214. [Google Scholar] [CrossRef]
- Chiang, C.S.; Hu, S.H.; Liao, B.J.; Chang, Y.C.; Chen, S.Y. Enhancement of cancer therapy efficacy by trastuzumab-conjugated and pH-sensitive nanocapsules with the simultaneous encapsulation of hydrophilic and hydrophobic compounds. Nanomed. Nanotechnol. 2014, 10, 99–107. [Google Scholar] [CrossRef]
- Jang, M.; Yoon, Y.I.; Kwon, Y.S.; Yoon, T.J.; Lee, H.J.; Hwang, S.I.; Yun, B.L.; Kim, S.M. Trastuzumab-conjugated liposome-coated fluorescent magnetic nanoparticles to target breast cancer. Korean J. Radiol. 2014, 15, 411–422. [Google Scholar] [CrossRef]
- Kesavan, A.; Ilaiyaraja, P.; Sofi, Beaula, W.; Veena Kumari, V.; Sugin Lal, J.; Arunkumar, C.; Anjana, G.; Srinivas, S.; Ramesh, A.; Kumar Rayala, S.; et al. Tumor targeting using polyamidoamine dendrimer–cisplatin nanoparticles functionalized with diglycolamic acid and Herceptin. Eur. J. Pharm. Biopharm. 2015, 96, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Steinhauser, I.; Spankuch, B.; Strebhardt, K.; Langer, K. Trastuzumab-modified nanoparticles: Optimization of preparation and uptake in cancer cells. Biomaterials 2016, 27, 4975–4983. [Google Scholar] [CrossRef] [PubMed]
- Kubota, T.; Kuroda, S.; Kanaya, N.; Morihiro, T.; Aoyama, K.; Kakiuchi, Y.; Kikuchi, S.; Nishizaki, M.; Kagawa, S.; Tazawa, H.; et al. HER2-targeted gold nanoparticles potentially overcome resistance to trastuzumab in gastric cancer. Nanomed. Nanotechnol. 2018, 14, 1919–1929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Aaron, J.; Sokolov, K. Directional conjugation of antibodies to nanoparticles for synthesis of multiplexed optical contrast agents with both delivery and targeting moieties. Nat. Protoc. 2008, 3, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Greene, M.K.; Richards, D.A.; Nogueira, J.C.F.; Campbell, K.; Smyth, P.; Fernandez, M.; Scott, C.J.; Chudasama, V. Forming next-generation antibody–nanoparticle conjugates through the oriented installation of non-engineered antibody fragments. Chem. Sci. 2018, 9, 79. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kolb, H.C.; Finn, M.G.; Sharpless, K.B. Click chemistry: Diverse chemical function from a few good reactions. Angew. Chem. Int. Ed. Engl. 2001, 40, 2004–2021. [Google Scholar] [CrossRef]
- Yi, G.; Son, J.; Yoo, J.; Park, C.; Koo, H. Application of click chemistry in nanoparticle modification and its targeted delivery. Biomater. Res. 2018, 22, 13. [Google Scholar] [CrossRef] [Green Version]
- Hein, C.D.; Liu, X.M.; Dong, W. Click chemistry, a powerful tool for pharmaceutical sciences. Pharm. Res. 2008, 25, 2216–2230. [Google Scholar] [CrossRef] [Green Version]
- Takayama, Y.; Kusamori, K.; Nishikawa, M. Click chemistry as a tool for cell engineering and drug delivery. Molecules 2019, 24, 172. [Google Scholar] [CrossRef] [Green Version]
- Devaraj, N.K.; Weissleder, R. Biomedical applications of tetrazine cycloadditions. Acc. Chem. Res. 2011, 44, 816–827. [Google Scholar] [CrossRef] [Green Version]
- Yoo, J.; Choi, S.; Son, J.; Yi, G.; Kim, E.; Koo, H. Click chemistry-mediated tumor-targeting of SN38-loaded nanoparticles using trastuzumab. Biochem. Biophys. Res. Commun. 2019, 515, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Keinänen, O.; Fung, K.; Pourat, J.; Jallinoja, V.; Vivier, D.; Kishore Pillarsetty, N.; Airaksinen, A.J.; Lewis, J.S.; Zeglis, B.M.; Sarparanta, M. Pretargeting of internalizing trastuzumab and cetuximab with a 18F-tetrazine tracer in xenograft models. EJMMI Res. 2017, 7, 95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.T.W.; Maruani, A.; Richards, D.A.; Baker, J.R.; Caddick, S.; Chudasama, V. Enabling the controlled assembly of antibody conjugates with a loading of two modules without antibody engineering. Chem. Sci. 2017, 8, 2056. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sievers, E.L.; Senter, P.D. Antibody-drug conjugates in cancer therapy. Annu. Rev. Med. 2013, 64, 15–29. [Google Scholar] [CrossRef] [PubMed]
- Alley, S.C.; Okeley, N.M.; Senter, P.D. Antibody-drug conjugates: Targeted drug delivery for cancer. Curr. Opin. Chem. Biol. 2010, 14, 529–537. [Google Scholar] [CrossRef] [PubMed]
- Shen, B.Q.; Xu, K.; Liu, L.; Raab, H.; Bhakta, S.; Kenrick, M.; Parsons-Reponte, K.L.; Tien, J.; Yu, S.F.; Mai, E.; et al. Conjugation site modulates the in vivo stability and therapeutic activity of antibody-drug conjugates. Nat. Biotechnol. 2012, 30, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Panawski, S.; Bhakta, S.; Raab, H.; Polakis, P.; Junutula, J.R. Site-specific antibody drug conjugates for cancer therapy. mAbs 2014, 6, 34–45. [Google Scholar] [CrossRef] [Green Version]
- Erickson, H.K.; Park, P.U.; Widdison, W.C.; Kovtun, Y.V.; Garrett, L.M.; Hoffman, K.; Lutz, R.J.; Goldmacher, V.S.; Blättler, W.A. Antibody-maytansinoid conjugates are activated in targeted cancer cells by lysosomal degradation and linker-dependent intracellular processing. Cancer Res. 2006, 66, 4426–4433. [Google Scholar] [CrossRef] [Green Version]
- Lewis Phillips, G.D.; Li, G.; Dugger, D.L.; Crocker, L.M.; Parsons, K.L.; Mai, E.; Blättler, W.A.; Lambert, J.M.; Chari, R.V.J.; Lutz, R.J.; et al. Targeting HER2-positive breast cancer with trastuzumab-DM1, an antibody–cytotoxic drug conjugate. Cancer Res. 2008, 68, 9280–9290. [Google Scholar] [CrossRef] [Green Version]
- Lopus, M. Antibody-DM1 conjugates as cancer therapeutics. Cancer Lett. 2011, 307, 113–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambert, J.M.; Chary, R.V.J. Ado-trastuzumab emtansine (T-DM1): An antibody−drug conjugate (ADC) for HER2-positive breast cancer. J. Med. Chem. 2014, 57, 6949–6964. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Guo, D.; Jiang, Z.; Tong, R.; Jiang, P.; Bai, L.; Chen, L.; Zhu, Y.; Guo, C.; Shi, J.; et al. Novel HER2-targeting antibody-drug conjugates of trastuzumab beyond T-DM1 in breast cancer: Trastuzumab deruxtecan (DS-8201a) and (Vic-)trastuzumab duocarmazine (SYD985). Eur. J. Med. Chem. 2019, 183, 111682. [Google Scholar] [CrossRef] [PubMed]
- Nunes, J.P.M.; Morais, M.; Vassileva, V.; Robinson, E.; Rajkumar, V.S.; Smith, M.E.B.; Pedley, R.B.; Caddick, S.; Baker, J.R.; Chudasama, V. Functional native disulfide bridging enables delivery of a potent, stable and targeted antibody–drug conjugate (ADC). Chem. Commun. 2015, 51, 10624. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robinson, E.; Nunes, J.P.M.; Vassileva, V.; Maruani, A.; Nogueira, J.C.F.; Smith, M.E.B.; Pedley, R.B.; Caddick, S.; Baker, J.R.; Chudasama, V. Pyridazinediones deliver potent, stable, targeted and efficacious antibody–drug conjugates (ADCs) with a controlled loading of 4 drugs per antibody. RSC Adv. 2017, 7, 9073. [Google Scholar] [CrossRef] [Green Version]
- Schumacher, F.F.; Nunes, J.P.M.; Maruani, A.; Chudasama, V.; Smith, M.E.B.; Chester, K.A.; Baker, J.R.; Caddick, S. Next generation maleimides enable the controlled assembly of antibody–drug conjugates via native disulfide bond bridging. Org. Biomol. Chem. 2014, 12, 7261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elgersma, R.C.; Coumans, R.G.E.; Huijbregts, T.; Menge, W.M.P.B.; Joosten, J.A.F.; Spijker, H.J.; de Groot, F.M.H.; van der Lee, M.M.C.; Ubink, R.; van den Dobbelsteen, D.J.; et al. Design, synthesis, and evaluation of linker-duocarmycin payloads: Toward selection of HER2-targeting antibody−drug conjugate SYD985. Mol. Pharm. 2015, 12, 1813–1835. [Google Scholar] [CrossRef]
- Ogitani, Y.; Aida, T.; Hagihara, K.; Yamaguchi, J.; Ishii, C.; Harada, N.; Soma, M.; Okamoto, H.; Oitate, M.; Arakawa, S.; et al. DS-8201a, a novel HER2-targeting ADC with a novel DNA topoisomerase I inhibitor, demonstrates a promising antitumor efficacy with differentiation from T-DM1. Clin. Cancer Res. 2016, 22, 5097–5108. [Google Scholar] [CrossRef] [Green Version]
- Rinnerthaler, G.; Gampenrieder, S.P.; Greil, R. HER2 directed antibody-drug-conjugates beyond T-DM1 in breast cancer. Int. J. Mol. Sci. 2019, 20, 1115. [Google Scholar] [CrossRef] [Green Version]
- Gradishar, W.J. Emerging approaches for treating HER2-positive metastatic breast cancer beyond trastuzumab. Ann. Oncol. 2013, 24, 2492–2500. [Google Scholar] [CrossRef]
- Leyland-Jones, B.; Arnold, A.; Gelmon, K.; Verma, S.; Ayoub, J.P.; Seidman, A.; Dias, R.; Howell, J.; Rakhit, A. Pharmacologic insights into the future of trastuzumab B. Ann. Oncol. 2001, 12, S43–S47. [Google Scholar] [CrossRef]
- Wang, J.; Xu, B. Targeted therapeutic options and future perspectives for HER2-positive breast cancer. Signal Transduct. Target. Ther. 2019, 4, 34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, X.; Zheng, X.; Pang, X.; Zhang, Z.; Zhang, Q. Incorporation of lapatinib into human serum albumin nanoparticles with enhanced anti-tumor effects in HER2-positive breast cancer. Colloids Surf. B 2015, 136, 817–827. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhang, S.; Ruan, S.B.; Zhang, Q.Y.; He, Q.; Gao, H.L. Lapatinib-incorporated lipoprotein-like nanoparticles: Preparation and a proposed breast cancer-targeting mechanism. Acta Pharmacol. Sin. 2014, 35, 846–852. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mobasseri, R.; Karimi, M.; Tian, L.; Naderi-Manesh, H.; Ramakrishna, S. Hydrophobic lapatinib encapsulated dextran-chitosan nanoparticles using a toxic solvent free method: Fabrication, release property & in vitro anti-cancer activity. Mater. Sci. Eng. C Mater. 2017, 74, 413–421. [Google Scholar]
- Singh, M.K.; Pindiprolu, S.K.S.S.; Sanapolli, B.K.R.; Yele, V.; Ganesh, G.N.K. HER2 targeted biological macromolecule modified liposomes for improved efficacy of capecitabine in breast cancer. Int. J. Biol. Macromol. 2020, 150, 631–636. [Google Scholar] [CrossRef]
- Espelin, C.W.; Leonard, S.C.; Geretti, E.; Wickham, T.; Hendriks, B.S. Dual HER2 targeting with trastuzumab and liposomal-encapsulated doxorubicin (MM-302) demonstrates synergistic antitumor activity in breast and gastric cancers. Cancer Res. 2016, 76, 1517:1527. [Google Scholar] [CrossRef] [Green Version]
- Miller, K.; Cortes, J.; Hurvitz, S.A.; Krop, I.E.; Tripathy, D.; Verma, S.; Riahi, K.; Reynolds, J.G.; Wickham, T.J.; Molnar, I.; et al. HERMIONE: A randomized phase 2 trial of MM-302 plus trastuzumab versus chemotherapy of physician’s choice plus trastuzumab in patients with previously treated, anthracycline-naïve, HER2-positive, locally advanced/metastatic breast cancer. BMC Cancer 2016, 16, 352–363. [Google Scholar] [CrossRef] [Green Version]
- Hamblett, K.J. HER2-Targeted ADCs: At the forefront of ADC technology development. In Innovations for Next-Generation Antibody-Drug Conjugates, 1st ed.; Damelin, M., Ed.; Humana Press: Cham, Switzerland, 2018; pp. 163–186. [Google Scholar]
- Yao, X.; Jiang, J.; Wang, X.; Huang, C.; Li, D.; Xie, K.; Xu, Q.; Li, H.; Li, Z.; Lou, L.; et al. A novel humanized anti-HER2 antibody conjugated with MMAE exerts potent anti-tumor activity. Breast Cancer Res. Treat. 2015, 153, 123–133. [Google Scholar] [CrossRef]
- Li, H.; Yu, C.; Jiang, J.; Huang, C.; Yao, X.; Xu, Q.; Yu, F.; Lou, L.; Fang, J. An anti-HER2 antibody conjugated with monomethyl auristatin E is highly effective in HER2-positive human gastric cancer. Cancer Biol. Ther. 2016, 17, 346–354. [Google Scholar] [CrossRef] [Green Version]
- Humphreys, C.R.C.; Kirtely, J.; Hewit, A.; Biroc, S.; Knudsen, N.; Skidmore, L.; Wahl, A. Abstract 639: Site specific conjugation of ARX-788, an antibody drug conjugate (ADC) targeting HER2, generates a potent and stable targeted therapeutic for multiple cancers. Cancer Res. 2015, 75, 639. [Google Scholar]
- Li, J.Y.; Perry, S.R.; Muniz-Medina, V.; Wang, X.; Wetzel, L.K.; Rebelatto, M.C.; Hinrichs, M.J.; Bezabeh, B.Z.; Fleming, R.L.; Dimasi, N.; et al. A biparatopic HER2-targeting antibody-drug conjugate induces tumor regression in primary models refractory to or ineligible for HER2-targeted therapy. Cancer Cell 2016, 29, 117–129. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clinical Trials. Available online: https://clinicaltrials.gov/ct2/home (accessed on 21 August 2020).
- Costa, R.L.B.; Czerniecki, B.J. Clinical development of immunotherapies for HER2+ breast cancer: A review of HER2-directed monoclonal antibodies and beyond. NPJ Breast Cancer 2020, 6, 10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mahjub, R.; Jatana, S.; Ern Lee, S.; Qin, Z.; Pauli, G.; Soleimani, M.; Madadi, S.; Li, S.D. Recent advances in applying nanotechnologies for cancer immunotherapy. J. Control. Release 2018, 288, 239–263. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.; Ma, W.; Yang, Y.; He, M.; Li, A.; Bai, L.; Yu, B.; Yu, Z. Cancer nanotechnology: Enhancing tumor cell response to chemotherapy for hepatocellular carcinoma therapy. Asian J. Pharm. Sci. 2019, 14, 581–594. [Google Scholar] [CrossRef]
- Kim, K.Y. Nanotechnology platforms and physiological challenges for cancer therapeutics. Nanomed. Nanotechnol. 2007, 3, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Matos, A.I.; Carreira, B.; Peres, C.; Moura, L.I.F.; Conniot, J.; Fourniols, T.; Scomparin, A.; Martínez-Barriocanal, A.; Arango, D.; Conde, J.P.; et al. Nanotechnology is an important strategy for combinational innovative chemo-immunotherapies against colorectal cancer. J. Control. Release 2019, 307, 108–138. [Google Scholar] [CrossRef]

| Strategy | Type of DDS | Payload | Targeted Type of HER2+ Cancer | Reference |
|---|---|---|---|---|
| Physical adsorption | NPs (PLGA-TPGS) | Docetaxel | Breast | Liu et al. [39] |
| NPs (PEI/PLGA) | Paclitaxel | Breast | Yu et al. [40] | |
| NPs (PEI/PLGA) | Docetaxel | Breast | Zhang et al. [41] | |
| NPs (PLGA/MTT) | Paclitaxel | Breast | Sun et al. [42] | |
| Physical and ionic adsorption and carbodiimide chem. | NPs (PLGA) | Docetaxel | Breast | Choi et al. [43] |
| Carbodiimide chemistry | NPs (PLGA-Phis-PEG) | Doxorubicin | Breast | Zhou et al. [47] |
| NPs (TPGS-g-chitosan) | Docetaxel | Breast | Mehata et al. [6] | |
| NPs (Alginate-piperazine) | Paclitaxel | Breast, ovarian | Nieto et al. [31] | |
| NPs (PLGA) | Cisplatin | Ovarian | Domínguez-Ríos et al. [49] | |
| NPs (Chitosan) | Gemcitabine | Pancreatic | Arya et al. [50] | |
| NPs (Magnetic) | - | Breast | Almaki et al. [53] | |
| Maleimide chemistry | NPs (HSA) | Methotrexate | Breast | Taheri et al. [57] |
| Liposomes | Rapamycin, PPɣ NPs | Breast | Nguyen et al. [58] | |
| Liposomes | Idarubicin | Breast | Amin et al. [59] | |
| DENCs | Paclitaxel, doxorubicin | Breast | Chiang et al. [60] | |
| Liposomes | Magnetic NPs | Breast | Jang et al. [61] | |
| NPs (Polyamidoamine dendrimers) | Cisplatin | Ovarian | Kesavan et al. [62] | |
| NPs (HSA) | - | Anyone | Steinhauser et al. [63] | |
| NPs (Gold) | - | Gastric | Kubota et al. [64] | |
| Click-chemistry (SPAAC) | NPs (PLGA) | - | Breast | Greene et al. [66] |
| Click-chemistry (iEDDA) | Liposomes | SN38 | Anyone | Yoo et al. [72] |
| - | Fluorine-18 | Breast | Keinänen et al. [73] |
| ADC | IgG | Payload | Clinical Trial Phase 5 | Indication | Developer |
|---|---|---|---|---|---|
| RC48-ADC 2–4 | IgG1 (Hertuzumab) | MMAE | Phase I | Solid tumors | Regemen |
| ARX788 2,3,6 | Engineered IgG1 | MMAF | Phase I | Breast, stomach cancers | Ambrx |
| TAK-522 (XMT-1522) 2,3,6 | IgG1 (HT19) | AF-HPA | Phase I | NSCLC, breast, gastric cancers | Mersana |
| A116 2 | Not disclosed | Not disclosed | Phase I | Breast cancer | Klus Pharma |
| Tmab Duocarmazine (SYD985) 1–3,6 | IgG1 | Seco-DUBA | Phase II | Endometrial cancer | Synthon |
| ALT-P7 2 | IgG1 (HM2, Tmab biobetter) | MMAE | Phase I | Breast cancer | Altrogen |
| DHES0815A 2 | IgG1 | PBD-MA | Phase I | Breast cancer | Genentech |
| MEDI4276 2,3 | Bi-specific IgG1 (Tmab ScFv) | AZ13599185 | Phase I/II | Breast, gastric cancers | MedImmune |
| Tmab Deruxtecan (DS-8201a) 1–3,6 | IgG1 | DXd | Phase II | Breast cancer | Daiichi Sankyo |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nieto, C.; Vega, M.A.; Martín del Valle, E.M. Trastuzumab: More than a Guide in HER2-Positive Cancer Nanomedicine. Nanomaterials 2020, 10, 1674. https://doi.org/10.3390/nano10091674
Nieto C, Vega MA, Martín del Valle EM. Trastuzumab: More than a Guide in HER2-Positive Cancer Nanomedicine. Nanomaterials. 2020; 10(9):1674. https://doi.org/10.3390/nano10091674
Chicago/Turabian StyleNieto, Celia, Milena A. Vega, and Eva M. Martín del Valle. 2020. "Trastuzumab: More than a Guide in HER2-Positive Cancer Nanomedicine" Nanomaterials 10, no. 9: 1674. https://doi.org/10.3390/nano10091674
APA StyleNieto, C., Vega, M. A., & Martín del Valle, E. M. (2020). Trastuzumab: More than a Guide in HER2-Positive Cancer Nanomedicine. Nanomaterials, 10(9), 1674. https://doi.org/10.3390/nano10091674







