Tyrosine-Modification of Polypropylenimine (PPI) and Polyethylenimine (PEI) Strongly Improves Efficacy of siRNA-Mediated Gene Knockdown
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Chemical Synthesis of Tyrosine-Modified PPI
2.3. Complex Preparation and Characterization
2.4. Luciferase Assay and Flow Cytometry
2.5. RT-qPCR
2.6. Proliferation and LDH Release
2.7. Erythrocyte Aggregation and Hemolysis
2.8. Blood Serum Markers and Immunostimulation
2.9. Statistics
3. Results
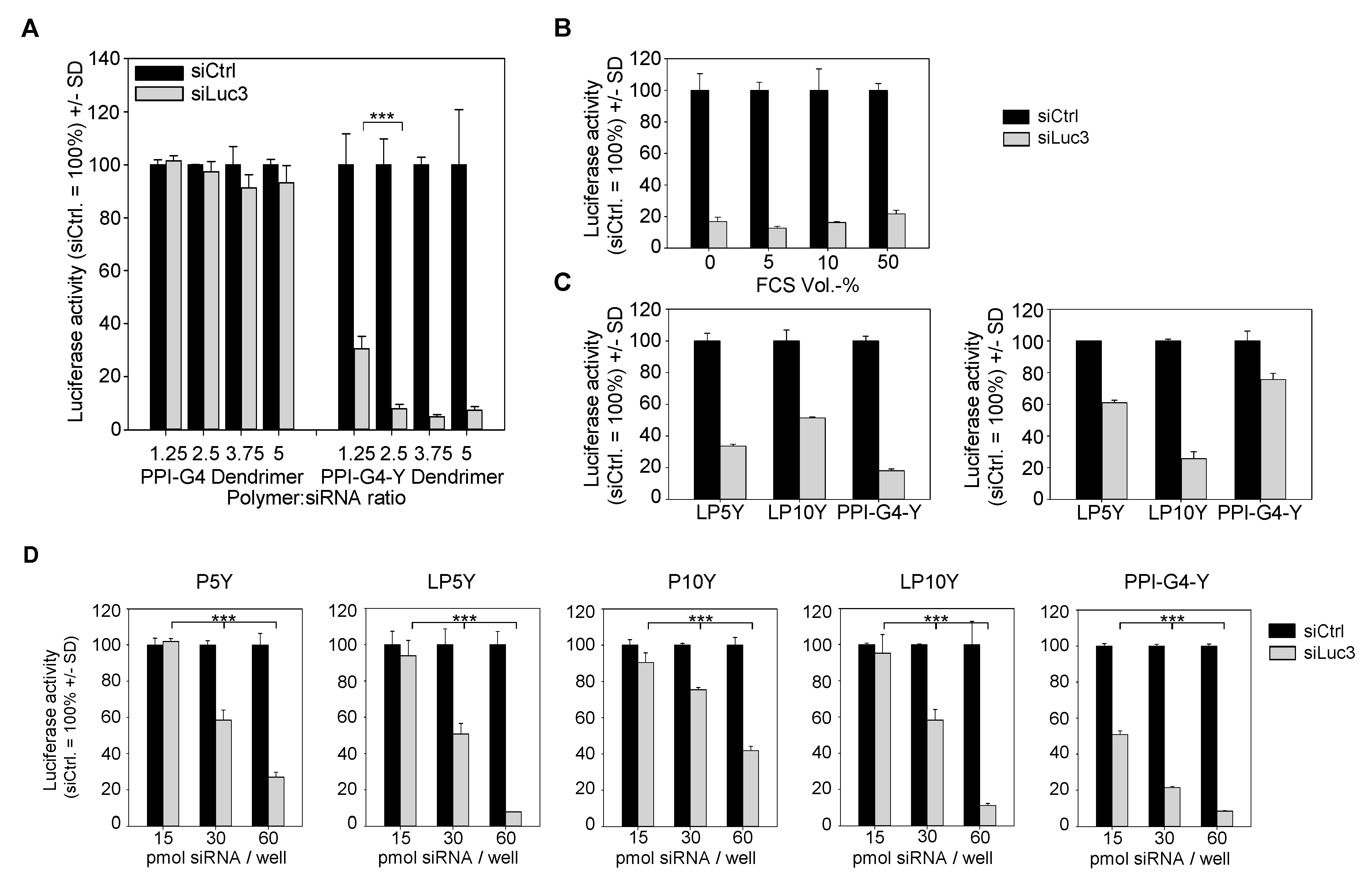
3.1. Generation and Analysis of Tyrosine-Modified PPI-G4 and (L)PEIs, and Their Corresponding siRNA Complexes
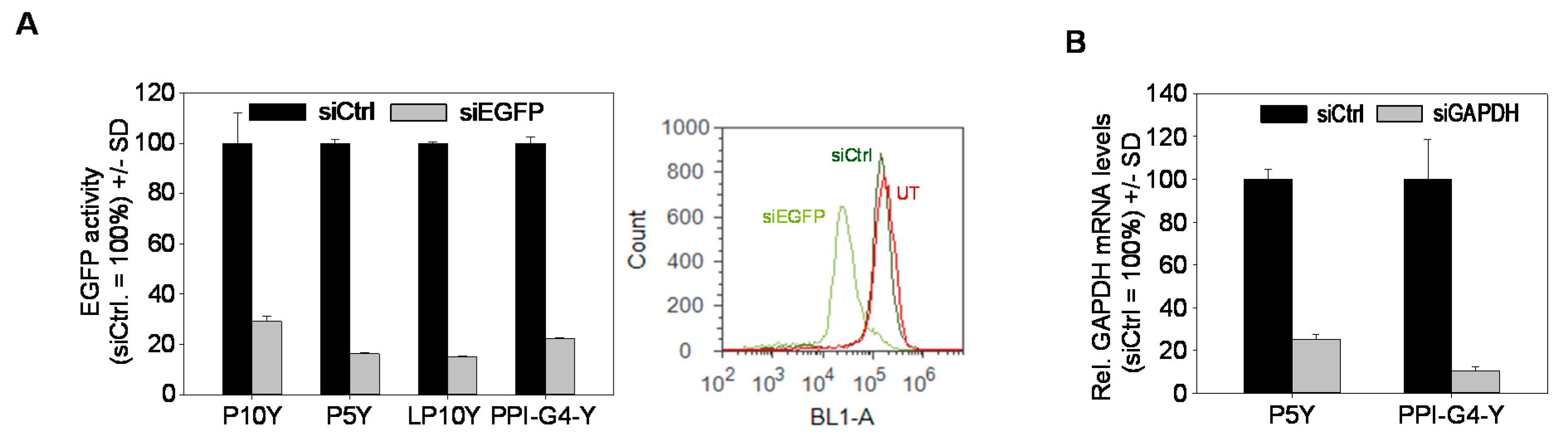
3.2. Biological Efficacies of siRNA Complexes Based on PPI-G4-Y- or Various Tyrosine-Modified PEIs
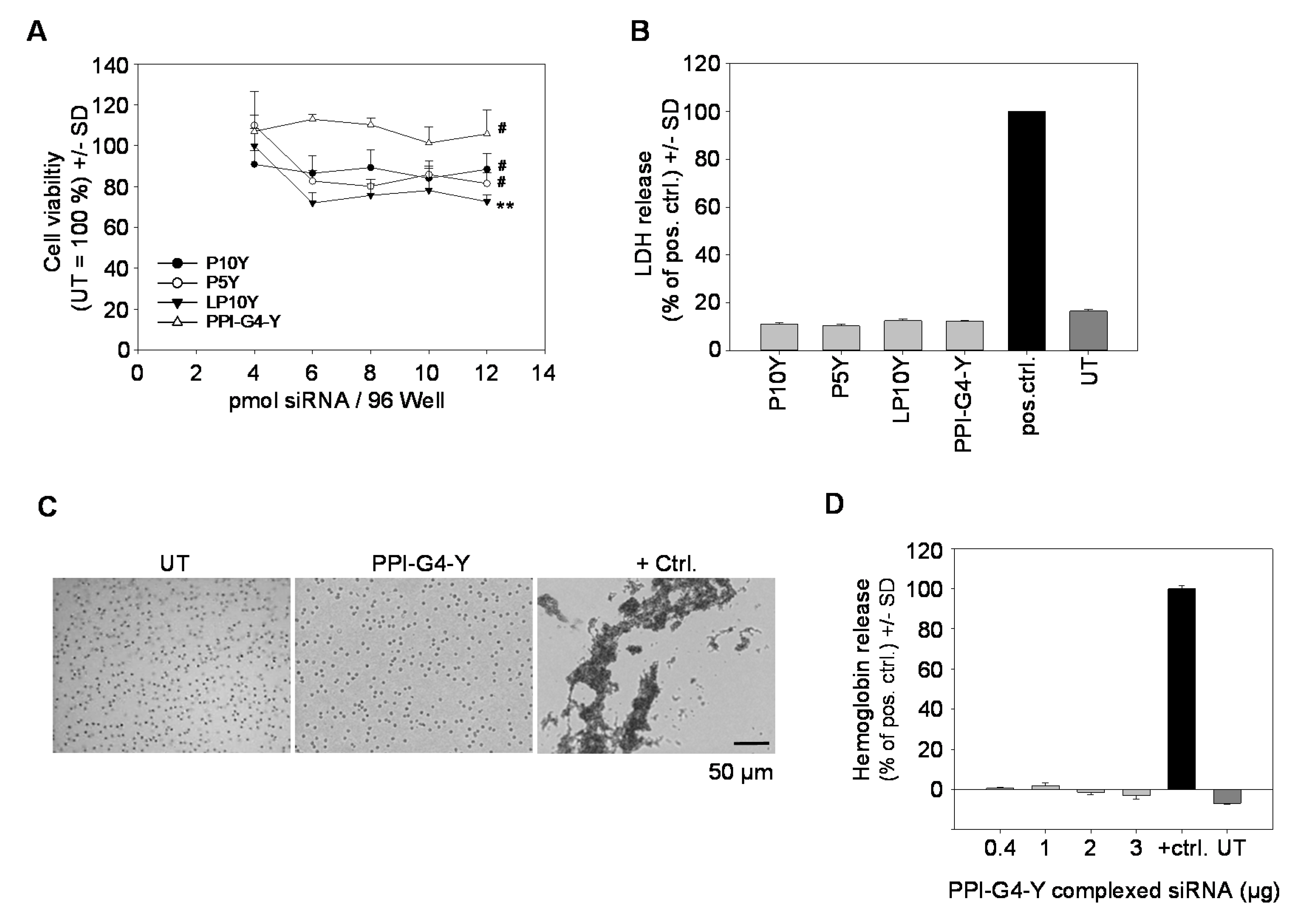
3.3. Biocompatibility of siRNA Complexes Based on PPI-G4-Y- or Various Tyrosine-Modified PEIs
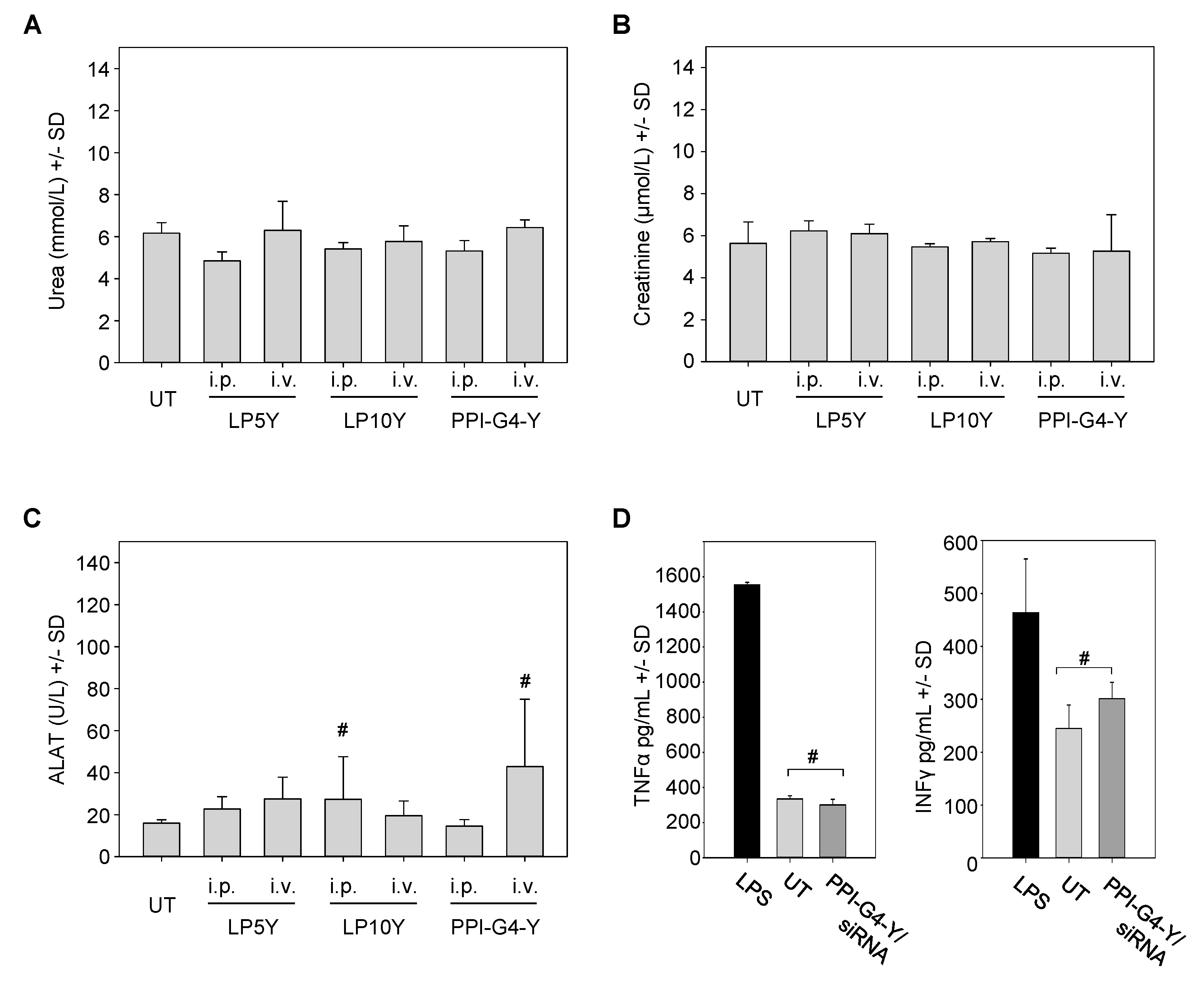
3.4. In Vivo Biocompatibility of siRNA Complexes Based on PPI-G4-Y- or Various Tyrosine-Modified PEIs
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chakraborty, C.; Sharma, A.R.; Sharma, G.; Doss, C.G.P.; Lee, S.-S. Therapeutic miRNA and siRNA: Moving from Bench to Clinic as Next Generation Medicine. Mol. Ther. Nucleic Acids 2017, 8, 132–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Setten, R.L.; Rossi, J.J.; Han, S.-P. The current state and future directions of RNAi-based therapeutics. Nat. Rev. Drug Discov. 2019, 18, 421–446. [Google Scholar] [CrossRef] [PubMed]
- Fakhr, E.; Zare, F.; Teimoori-Toolabi, L. Precise and efficient siRNA design: A key point in competent gene silencing. Cancer Gene Ther. 2016, 23, 73–82. [Google Scholar] [CrossRef] [PubMed]
- Friedrich, M.; Wiedemann, K.; Reiche, K.; Puppel, S.-H.; Pfeifer, G.; Zipfel, I.; Binder, S.; Köhl, U.; Müller, G.A.; Engeland, K.; et al. The Role of lncRNAs TAPIR-1 and -2 as Diagnostic Markers and Potential Therapeutic Targets in Prostate Cancer. Cancers 2020, 12, 1122. [Google Scholar] [CrossRef]
- Benizri, S.; Gissot, A.; Martin, A.; Vialet, B.; Grinstaff, M.W.; Barthélémy, P. Bioconjugated Oligonucleotides: Recent Developments and Therapeutic Applications. Bioconjug. Chem. 2019, 30, 366–383. [Google Scholar] [CrossRef]
- Gooding, M.; Malhotra, M.; Evans, J.C.; Darcy, R.; O’Driscoll, C.M. Oligonucleotide conjugates—Candidates for gene silencing therapeutics. Eur. J. Pharm. Biopharm. 2016, 107, 321–340. [Google Scholar] [CrossRef]
- Kuboyama, T.; Yagi, K.; Naoi, T.; Era, T.; Yagi, N.; Nakasato, Y.; Yabuuchi, H.; Takahashi, S.; Shinohara, F.; Iwai, H.; et al. Simplifying the Chemical Structure of Cationic Lipids for siRNA-Lipid Nanoparticles. ACS Med. Chem. Lett. 2019, 10, 749–753. [Google Scholar] [CrossRef]
- Kulkarni, J.A.; Darjuan, M.M.; Mercer, J.E.; Chen, S.; van der Meel, R.; Thewalt, J.L.; Tam, Y.Y.C.; Cullis, P.R. On the Formation and Morphology of Lipid Nanoparticles Containing Ionizable Cationic Lipids and siRNA. ACS Nano 2018, 12, 4787–4795. [Google Scholar] [CrossRef] [Green Version]
- Lin, Z.; Bao, M.; Yu, Z.; Xue, L.; Ju, C.; Zhang, C. The development of tertiary amine cationic lipids for safe and efficient siRNA delivery. Biomater. Sci. 2019, 7, 2777–2792. [Google Scholar] [CrossRef]
- Peng, L.; Wagner, E. Polymeric Carriers for Nucleic Acid Delivery: Current Designs and Future Directions. Biomacromolecules 2019, 20, 3613–3626. [Google Scholar] [CrossRef]
- Ulkoski, D.; Bak, A.; Wilson, J.T.; Krishnamurthy, V.R. Recent advances in polymeric materials for the delivery of RNA therapeutics. Expert Opin. Drug Deliv. 2019, 16, 1149–1167. [Google Scholar] [CrossRef] [PubMed]
- Conde, J.; Ambrosone, A.; Hernandez, Y.; Tian, F.; McCully, M.; Berry, C.C.; Baptista, P.V.; Tortiglione, C.; de La Fuente, J.M. 15 years on siRNA delivery: Beyond the State-of-the-Art on inorganic nanoparticles for RNAi therapeutics. Nano Today 2015, 10, 421–450. [Google Scholar] [CrossRef] [Green Version]
- Steinbacher, J.L.; Landry, C.C. Adsorption and release of siRNA from porous silica. Langmuir 2014, 30, 4396–4405. [Google Scholar] [CrossRef] [PubMed]
- Krasheninina, O.A.; Apartsin, E.K.; Fuentes, E.; Szulc, A.; Ionov, M.; Venyaminova, A.G.; Shcharbin, D.; de La Mata, F.J.; Bryszewska, M.; Gόmez, R. Complexes of Pro-Apoptotic siRNAs and Carbosilane Dendrimers: Formation and Effect on Cancer Cells. Pharmaceutics 2019, 11, 25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.-C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L.; et al. Patisiran, an RNAi Therapeutic, for Hereditary Transthyretin Amyloidosis. N. Engl. J. Med. 2018, 379, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Aseguinolaza, G. Givosiran—Running RNA Interference to Fight Porphyria Attacks. N. Engl. J. Med. 2020, 382, 2366–2367. [Google Scholar] [CrossRef]
- Balwani, M.; Sardh, E.; Ventura, P.; Aguilera Peiró, P.; Rees, D.C.; Stölzel, U.; Bissell, M.D.; Bonkovsky, H.; Windyga, J.; Anderson, K.E.; et al. Phase 3 Trial of RNAi Therapeutic Givosiran for Acute Intermittent Porphyria. N. Engl. J. Med. 2020, 382, 2289–2301. [Google Scholar] [CrossRef]
- Lorenzer, C.; Dirin, M.; Winkler, A.-M.; Baumann, V.; Winkler, J. Going beyond the liver: Progress and challenges of targeted delivery of siRNA therapeutics. J. Control. Release 2015, 203, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Zuckerman, J.E.; Davis, M.E. Clinical experiences with systemically administered siRNA-based therapeutics in cancer. Nat. Rev. Drug Discov. 2015, 14, 843–856. [Google Scholar] [CrossRef]
- Dominska, M.; Dykxhoorn, D.M. Breaking down the barriers: siRNA delivery and endosome escape. J. Cell Sci. 2010, 123, 1183–1189. [Google Scholar] [CrossRef] [Green Version]
- Whitehead, K.A.; Langer, R.; Anderson, D.G. Knocking down barriers: Advances in siRNA delivery. Nat. Rev. Drug Discov. 2009, 8, 129–138. [Google Scholar] [CrossRef] [PubMed]
- Boussif, O.; Lezoualc’h, F.; Zanta, M.A.; Mergny, M.D.; Schermann, D.; Demeneix, B.; Behr, J.-P. A versatile vector for gene and oligo transfer into cells in culture and in vivo: Polyethylenimine. Proc. Natl. Acad. Sci. USA 1995, 92, 7297–7301. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Höbel, S.; Aigner, A. Polyethylenimines for siRNA and miRNA delivery in vivo. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2013, 5, 484–501. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Chen, J.; Li, Z.; Wang, Z.; Zhang, W.; Liu, J. Recent advances in the development of polyethylenimine-based gene vectors for safe and efficient gene delivery. Expert Opin. Drug Deliv. 2019, 16, 363–376. [Google Scholar] [CrossRef]
- Neu, M.; Fischer, D.; Kissel, T. Recent advances in rational gene transfer vector design based on poly(ethylene imine) and its derivatives. J. Gene Med. 2005, 7, 992–1009. [Google Scholar] [CrossRef]
- Palmerston Mendes, L.; Pan, J.; Torchilin, V.P. Dendrimers as Nanocarriers for Nucleic Acid and Drug Delivery in Cancer Therapy. Molecules 2017, 22, 1401. [Google Scholar] [CrossRef] [Green Version]
- Dzmitruk, V.; Apartsin, E.; Ihnatsyeu-Kachan, A.; Abashkin, V.; Shcharbin, D.; Bryszewska, M. Dendrimers Show Promise for siRNA and microRNA Therapeutics. Pharmaceutics 2018, 10, 126. [Google Scholar] [CrossRef] [Green Version]
- Zinselmeyer, B.H.; Mackay, S.P.; Schätzlein, A.; Uchegbu, I. The Lower-Generation Polypropylenimine Dendrimers Are Effective Gene-Transfer Agents. Pharm. Res. 2002, 19, 960–967. [Google Scholar] [CrossRef]
- Taratula, O.; Savla, R.; He, H.; Minko, T. Poly(propyleneimine) dendrimers as potential siRNA delivery nanocarrier: From structure to function. IJNT 2011, 8, 36. [Google Scholar] [CrossRef]
- De La Fuente, M.; Jones, M.-C.; Santander-Ortega, M.J.; Mirenska, A.; Marimuthu, P.; Uchegbu, I.; Schätzlein, A. A nano-enabled cancer-specific ITCH RNAi chemotherapy booster for pancreatic cancer. Nanomedicine 2015, 11, 369–377. [Google Scholar] [CrossRef] [Green Version]
- Malloggi, C.; Pezzoli, D.; Magagnin, L.; de Nardo, L.; Mantovani, D.; Tallarita, E.; Candiani, G. Comparative evaluation and optimization of off-the-shelf cationic polymers for gene delivery purposes. Polym. Chem. 2015, 6, 6325–6339. [Google Scholar] [CrossRef] [Green Version]
- Kafil, V.; Omidi, Y. Cytotoxic impacts of linear and branched polyethylenimine nanostructures in A431 cells. BioImpacts 2011, 1, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Russ, V.; Günther, M.; Halama, A.; Ogris, M.; Wagner, E. Oligoethylenimine-grafted polypropylenimine dendrimers as degradable and biocompatible synthetic vectors for gene delivery. J. Control. Release 2008, 132, 131–140. [Google Scholar] [CrossRef] [PubMed]
- Somani, S.; Laskar, P.; Altwaijry, N.; Kewcharoenvong, P.; Irving, C.; Robb, G.; Pickard, B.S.; Dufès, C. PEGylation of polypropylenimine dendrimers: Effects on cytotoxicity, DNA condensation, gene delivery and expression in cancer cells. Sci. Rep. 2018, 8, 9410. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stasko, N.A.; Johnson, C.B.; Schoenfisch, M.H.; Johnson, T.A.; Holmuhamedov, E.L. Cytotoxicity of polypropylenimine dendrimer conjugates on cultured endothelial cells. Biomacromolecules 2007, 8, 3853–3859. [Google Scholar] [CrossRef]
- Taratula, O.; Garbuzenko, O.B.; Kirkpatrick, P.; Pandya, I.; Savla, R.; Pozharov, V.P.; He, H.; Minko, T. Surface-engineered targeted PPI dendrimer for efficient intracellular and intratumoral siRNA delivery. J. Control. Release 2009, 140, 284–293. [Google Scholar] [CrossRef] [Green Version]
- Sideratou, Z.; Kontoyianni, C.; Drossopoulou, G.I.; Paleos, C.M. Synthesis of a folate functionalized PEGylated poly(propylene imine) dendrimer as prospective targeted drug delivery system. Bioorg. Med. Chem. Lett. 2010, 20, 6513–6517. [Google Scholar] [CrossRef]
- Tietze, S.; Schau, I.; Michen, S.; Ennen, F.; Janke, A.; Schackert, G.; Aigner, A.; Appelhans, D.; Temme, A. A Poly(Propyleneimine) Dendrimer-Based Polyplex-System for Single-Chain Antibody-Mediated Targeted Delivery and Cellular Uptake of SiRNA. Small 2017, 13. [Google Scholar] [CrossRef]
- Hashemi, M.; Fard, H.S.; Farzad, S.A.; Parhiz, H.; Ramezani, M. Gene transfer enhancement by alkylcarboxylation of poly(propylenimine). Nanomed. J. 2013, 1, 55–62. [Google Scholar]
- Liu, H.; Wang, Y.; Wang, M.; Xiao, J.; Cheng, Y. Fluorinated poly(propylenimine) dendrimers as gene vectors. Biomaterials 2014, 35, 5407–5413. [Google Scholar] [CrossRef]
- Byrne, M.; Victory, D.; Hibbitts, A.; Lanigan, M.; Heise, A.; Cryan, S.-A. Molecular weight and architectural dependence of well-defined star-shaped poly(lysine) as a gene delivery vector. Biomater. Sci. 2013, 1, 1223–1234. [Google Scholar] [CrossRef] [PubMed]
- Gu, J.; Hao, J.; Fang, X.; Sha, X. Factors influencing the transfection efficiency and cellular uptake mechanisms of Pluronic P123-modified polypropyleneimine/pDNA polyplexes in multidrug resistant breast cancer cells. Colloid Surface B 2016, 140, 83–93. [Google Scholar] [CrossRef] [PubMed]
- Aldawsari, H.; Edrada-Ebel, R.; Blatchford, D.R.; Tate, R.J.; Tetley, L.; Dufès, C. Enhanced gene expression in tumors after intravenous administration of arginine-, lysine- and leucine-bearing polypropylenimine polyplex. Biomaterials 2011, 32, 5889–5899. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-I.; Baek, J.-U.; Zhe Bai, C.; Park, J.-S. Arginine-conjugated polypropylenimine dendrimer as a non-toxic and efficient gene delivery carrier. Biomaterials 2007, 28, 2061–2067. [Google Scholar] [CrossRef] [PubMed]
- Hashemi, M.; Tabatabai, S.M.; Parhiz, H.; Milanizadeh, S.; Amel Farzad, S.; Abnous, K.; Ramezani, M. Gene delivery efficiency and cytotoxicity of heterocyclic amine-modified PAMAM and PPI dendrimers. Mater. Sci. Eng. C 2016, 61, 791–800. [Google Scholar] [CrossRef]
- Khan, O.F.; Zaia, E.W.; Jhunjhunwala, S.; Xue, W.; Cai, W.; Yun, D.S.; Barnes, C.M.; Dahlman, J.E.; Dong, Y.; Pelet, J.M.; et al. Dendrimer-Inspired Nanomaterials for the in Vivo Delivery of siRNA to Lung Vasculature. Nano Lett. 2015, 15, 3008–3016. [Google Scholar] [CrossRef] [Green Version]
- Chen, A.M.; Taratula, O.; Wei, D.; Yen, H.-I.; Thomas, T.; Thomas, T.J.; Minko, T.; He, H. Labile catalytic packaging of DNA/siRNA: Control of gold nanoparticles “out” of DNA/siRNA complexes. ACS Nano 2010, 4, 3679–3688. [Google Scholar] [CrossRef]
- Shen, W.; Wang, Q.; Shen, Y.; Gao, X.; Li, L.; Yan, Y.; Wang, H.; Cheng, Y. Green Tea Catechin Dramatically Promotes RNAi Mediated by Low-Molecular-Weight Polymers. ACS Cent. Sci. 2018, 4, 1326–1333. [Google Scholar] [CrossRef]
- Maitz, M.F. Applications of synthetic polymers in clinical medicine. Biosurf. Biotribol. 2015, 1, 161–176. [Google Scholar] [CrossRef] [Green Version]
- Ewe, A.; Przybylski, S.; Burkhardt, J.; Janke, A.; Appelhans, D.; Aigner, A. A novel tyrosine-modified low molecular weight polyethylenimine (P10Y) for efficient siRNA delivery in vitro and in vivo. J. Control. Release 2016, 230, 13–25. [Google Scholar] [CrossRef]
- Ewe, A.; Höbel, S.; Heine, C.; Merz, L.; Kallendrusch, S.; Bechmann, I.; Merz, F.; Franke, H.; Aigner, A. Optimized polyethylenimine (PEI)-based nanoparticles for siRNA delivery, analyzed in vitro and in an ex vivo tumor tissue slice culture model. Drug Deliv. Transl. Res. 2017, 7, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Ewe, A.; Noske, S.; Karimov, M.; Aigner, A. Polymeric Nanoparticles Based on Tyrosine-Modified, Low Molecular Weight Polyethylenimines for siRNA Delivery. Pharmaceutics 2019, 11, 600. [Google Scholar] [CrossRef] [Green Version]
- Tauhardt, L.; Kempe, K.; Knop, K.; Altuntaş, E.; Jäger, M.; Schubert, S.; Fischer, D.; Schubert, U.S. Linear Polyethyleneimine: Optimized Synthesis and Characterization—On the Way to “Pharmagrade” Batches. Macromol. Chem. Phys. 2011, 212, 1918–1924. [Google Scholar] [CrossRef]
- Karimov, M.; Schulz, M.; Kahl, T.; Noske, S.; Kubczak, M.; Gockel, I.; Thieme, R.; Büch, T.; Reinert, A.; Ionov, M.; et al. Tyrosine-modified linear PEIs for highly efficacious and biocompatible siRNA delivery in vitro and in vivo. Nano Res. 2020, Submitted. [Google Scholar]
- Höbel, S.; Koburger, I.; John, M.; Czubayko, F.; Hadwiger, P.; Vornlocher, H.-P.; Aigner, A. Polyethylenimine/small interfering RNA-mediated knockdown of vascular endothelial growth factor in vivo exerts anti-tumor effects synergistically with Bevacizumab. J. Gene Med. 2010, 12, 287–300. [Google Scholar] [CrossRef]
- Troiber, C.; Edinger, D.; Kos, P.; Schreiner, L.; Kläger, R.; Herrmann, A.; Wagner, E. Stabilizing effect of tyrosine trimers on pDNA and siRNA polyplexes. Biomaterials 2013, 34, 1624–1633. [Google Scholar] [CrossRef]
- Creusat, G.; Thomann, J.-S.; Maglott, A.; Pons, B.; Dontenwill, M.; Guérin, E.; Frisch, B.; Zuber, G. Pyridylthiourea-grafted polyethylenimine offers an effective assistance to siRNA-mediated gene silencing in vitro and in vivo. J. Control. Release 2012, 157, 418–426. [Google Scholar] [CrossRef]
- Creusat, G.; Rinaldi, A.-S.; Weiss, E.; Elbaghdadi, R.; Remy, J.-S.; Mulherkar, R.; Zuber, G. Proton sponge trick for pH-sensitive disassembly of polyethylenimine-based siRNA delivery systems. Bioconjug. Chem. 2010, 21, 994–1002. [Google Scholar] [CrossRef] [Green Version]
- Dougherty, D.A. Cation-π Interactions in Chemistry and Biology- A New View of Benzene, Phe, Tyr, and Trp. Science 1996, 271, 163–168. [Google Scholar] [CrossRef]
- Edwards-Gayle, C.J.C.; Greco, F.; Hamley, I.W.; Rambo, R.P.; Reza, M.; Ruokolainen, J.; Skoulas, D.; Iatrou, H. Self-Assembly of Telechelic Tyrosine End-Capped PEO Star Polymers in Aqueous Solution. Biomacromolecules 2018, 19, 167–177. [Google Scholar] [CrossRef]
- Roviello, G.N.; Roviello, V.; Autiero, I.; Saviano, M. Solid phase synthesis of TyrT, a thymine-tyrosine conjugate with poly(A) RNA-binding ability. RSC Adv. 2016, 6, 27607–27613. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fröhlich, E. The role of surface charge in cellular uptake and cytotoxicity of medical nanoparticles. Int. J. Nanomed. 2012, 7, 5577–5591. [Google Scholar] [CrossRef] [PubMed] [Green Version]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Noske, S.; Karimov, M.; Aigner, A.; Ewe, A. Tyrosine-Modification of Polypropylenimine (PPI) and Polyethylenimine (PEI) Strongly Improves Efficacy of siRNA-Mediated Gene Knockdown. Nanomaterials 2020, 10, 1809. https://doi.org/10.3390/nano10091809
Noske S, Karimov M, Aigner A, Ewe A. Tyrosine-Modification of Polypropylenimine (PPI) and Polyethylenimine (PEI) Strongly Improves Efficacy of siRNA-Mediated Gene Knockdown. Nanomaterials. 2020; 10(9):1809. https://doi.org/10.3390/nano10091809
Chicago/Turabian StyleNoske, Sandra, Michael Karimov, Achim Aigner, and Alexander Ewe. 2020. "Tyrosine-Modification of Polypropylenimine (PPI) and Polyethylenimine (PEI) Strongly Improves Efficacy of siRNA-Mediated Gene Knockdown" Nanomaterials 10, no. 9: 1809. https://doi.org/10.3390/nano10091809




