Impacts of Surface Hydrophilicity of Carboxylated Polyethersulfone Supports on the Characteristics and Permselectivity of PA-TFC Nanofiltration Membranes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Reagents
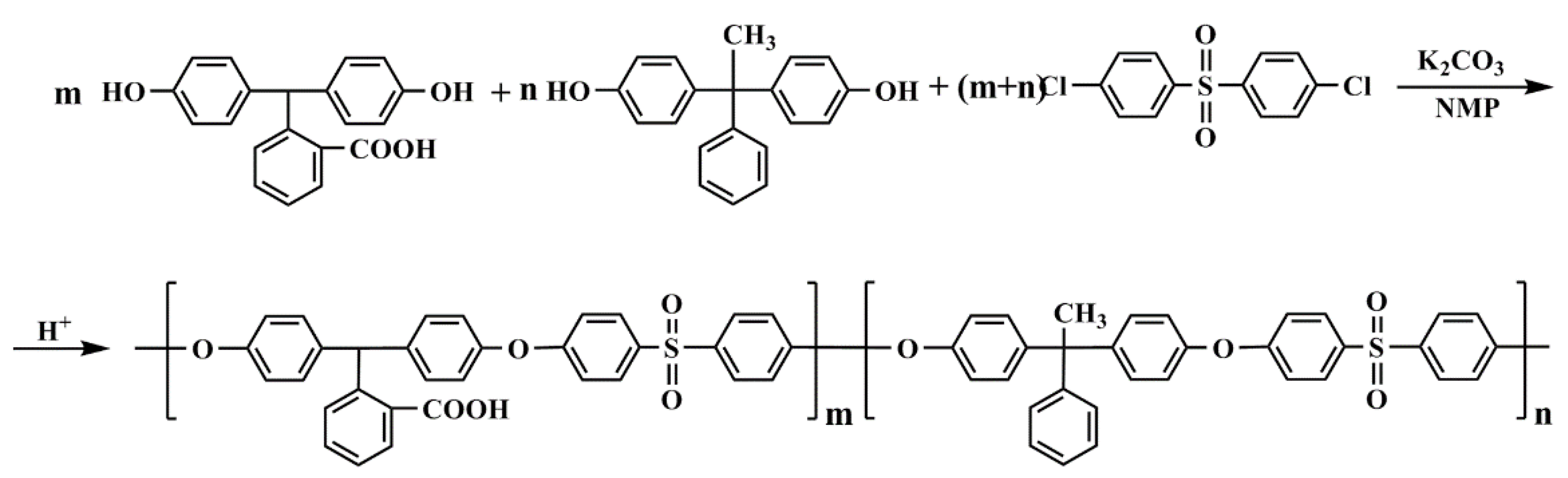
2.2. Preparation of Supports and Synthesis of PA-TFC NF Membranes
2.3. Characterization of Supports and PA-TFC NF Membranes
2.3.1. Thickness of Support
2.3.2. Scanning Electron Microscope (SEM)
2.3.3. Atomic Force Microscope (AFM)
2.3.4. Contact Angle (CA)
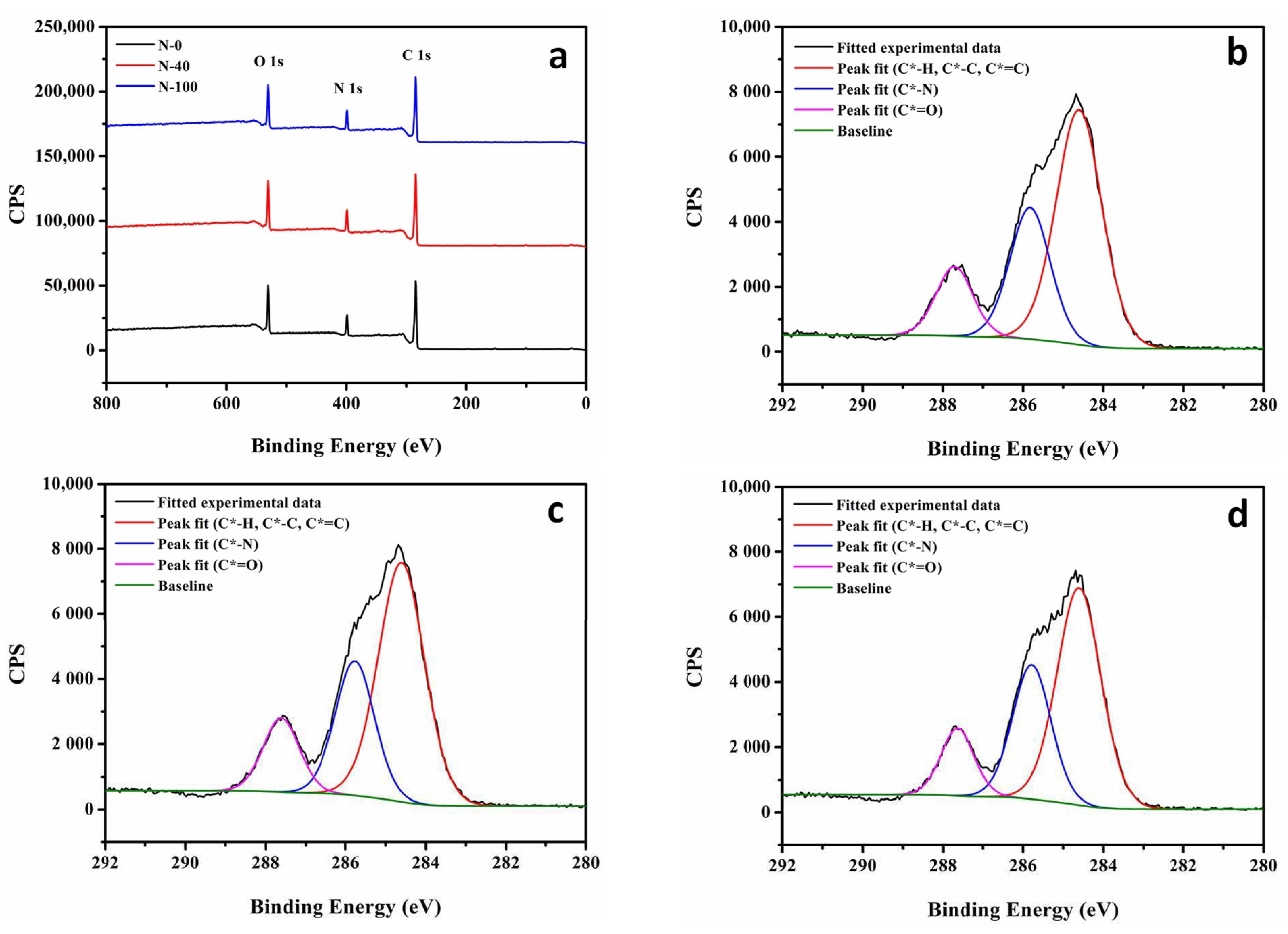
2.3.5. X-ray Photoelectron Spectrometer (XPS)
2.4. Performance Evaluation of Supports and PA-TFC NF Membranes
3. Results and Discussion
3.1. Effects of MR of Carboxyl Units in CPES Copolymers on the Properties of Supports
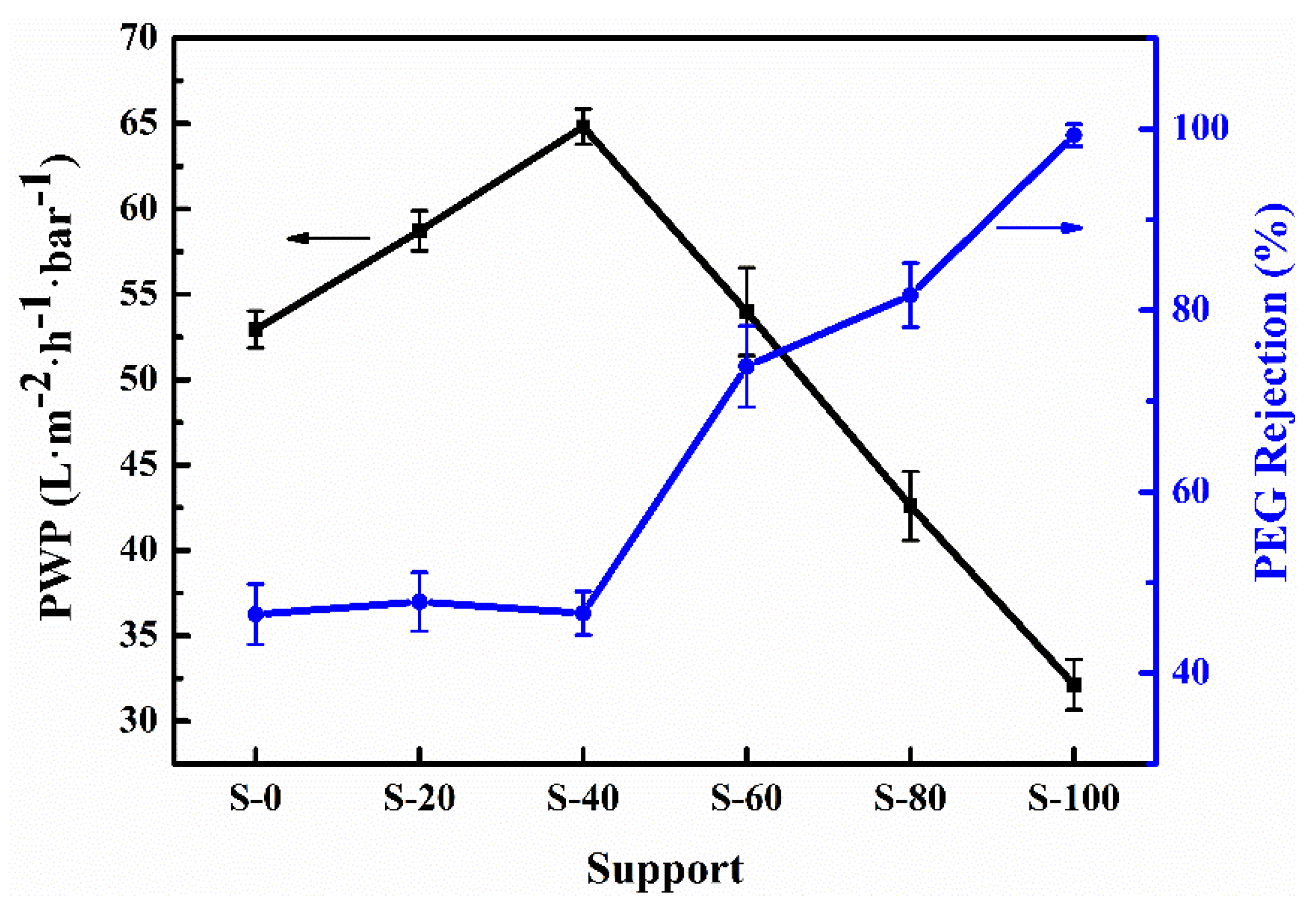
3.2. Effects of Surface Hydrophilicity of Supports on the Characteristics and Permselectivity of PA-TFC NF Membranes
- (1)
- S-0 is featured with a relatively hydrophobic surface (CA: 90.43°).
- (2)
- S-40 possesses an increasing surface hydrophilicity (CA: 87.56°).
- (3)
- S-100 has a more hydrophilic surface (CA: 82.12°).
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Loo, S.L.; Fane, A.G.; Krantz, W.B.; Lim, T.T. Emergency water supply: A review of potential technologies and selection criteria. Water Res. 2012, 46, 3125–3151. [Google Scholar] [CrossRef]
- Elimelech, M.; Phillip, W.A. The future of seawater desalination energy, technology, and the environment. Science 2011, 333, 712–717. [Google Scholar] [CrossRef]
- Logan, B.E.; Elimelech, M. Membrane-based processes for sustainable power generation using water. Nature 2012, 488, 313–319. [Google Scholar] [CrossRef]
- Karan, S.; Jiang, Z.; Livingston, A.G. Sub-10 nm polyamide nanofilms with ultrafast solvent transport for molecular separation. Science 2015, 348, 1347–1351. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Yan, Y.; Wang, H. Recent advances in polymer and polymer composite membranes for reverse and forward osmosis processes. Prog. Polym. Sci. 2016, 61, 104–155. [Google Scholar] [CrossRef] [Green Version]
- Petersen, R.J. Composite reverse osmosis and nanofiltration membranes. J. Membr. Sci. 1993, 83, 81–150. [Google Scholar] [CrossRef]
- Freger, V. Kinetics of film formation by interfacial polycondensation. Langmuir 2005, 21, 1884–1894. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Liu, M.; Liu, X.; Gao, C. Performance enhancement in interfacially synthesized thin-film composite polyamide-urethane reverse osmosis membrane for seawater desalination. J. Membr. Sci. 2009, 342, 313–320. [Google Scholar] [CrossRef]
- Saha, N.K.; Joshi, S.V. Performance evaluation of thin film composite polyamide nanofiltration membrane with variation in monomer type. J. Membr. Sci. 2009, 342, 60–69. [Google Scholar] [CrossRef]
- Kim, I.C.; Jeong, B.R.; Kim, S.J.; Lee, K.H. Preparation of high flux thin film composite polyamide membrane: The effect of alkyl phosphate additives during interfacial polymerization. Desalination 2013, 308, 111–114. [Google Scholar] [CrossRef]
- Xiang, J.; Xie, Z.; Hoang, M.; Zhang, K. Effect of amine salt surfactants on the performance of thin film composite poly(piperazine-amide) nanofiltration membranes. Desalination 2013, 315, 156–163. [Google Scholar] [CrossRef]
- Zou, H.; Jin, Y.; Yang, J.; Dai, H.; Yu, X.; Xu, J. Synthesis and characterization of thin film composite reverse osmosis membranes via novel interfacial polymerization approach. Sep. Purif. Technol. 2010, 72, 256–262. [Google Scholar] [CrossRef]
- Ghosh, A.K.; Jeong, B.H.; Huang, X.; Hoek, E.M.V. Impacts of reaction and curing conditions on polyamide composite reverse osmosis membrane properties. J. Membr. Sci. 2008, 311, 34–45. [Google Scholar] [CrossRef]
- Singh, P.S.; Joshi, S.V.; Trivedi, J.J.; Devmurari, C.V.; Rao, A.P.; Ghosh, P.K. Probing the structural variations of thin film composite RO membranes obtained by coating polyamide over polysulfone membranes of different pore dimensions. J. Membr. Sci. 2006, 278, 19–25. [Google Scholar] [CrossRef]
- Huang, L.; McCutcheon, J.R. Impact of support layer pore size on performance of thin film composite membranes for forward osmosis. J. Membr. Sci. 2015, 483, 25–33. [Google Scholar] [CrossRef]
- Misdan, N.; Lau, W.J.; Ismail, A.F.; Matsuura, T. Formation of thin film composite nanofiltration membrane: Effect of polysulfone substrate characteristics. Desalination 2013, 329, 9–18. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Li, Q.; Fang, W.; Wang, R.; Krantz, W.B. Effects of the support on the characteristics and permselectivity of thin film composite membranes. J. Membr. Sci. 2019, 580, 12–23. [Google Scholar] [CrossRef]
- Maruf, S.H.; Greenberg, A.R.; Ding, Y. Influence of substrate processing and interfacial polymerization conditions on the surface topography and permselective properties of surface-patterned thin-film composite membranes. J. Membr. Sci. 2016, 512, 50–60. [Google Scholar] [CrossRef] [Green Version]
- Ghosh, A.K.; Hoek, E.M.V. Impacts of support membrane structure and chemistry on polyamide-polysulfone interfacial composite membranes. J. Membr. Sci. 2009, 336, 140–148. [Google Scholar] [CrossRef]
- Kim, H.I.; Kim, S.S. Plasma treatment of polypropylene and polysulfone supports for thin film composite reverse osmosis membrane. J. Membr. Sci. 2006, 286, 193–201. [Google Scholar] [CrossRef]
- Cho, Y.H.; Han, J.; Han, S.; Guiver, M.D.; Park, H.B. Polyamide thin-film composite membranes based on carboxylated polysulfone microporous support membranes for forward osmosis. J. Membr. Sci. 2013, 445, 220–227. [Google Scholar] [CrossRef] [Green Version]
- Shi, M.; Yan, W.; Zhou, Y.; Wang, Z.; Liu, L.; Zhao, S.; Ji, Y.; Wang, J.; Gao, C.; Zhang, P.; et al. Combining tannic acid-modified support and a green co-solvent for high performance reverse osmosis membranes. J. Membr. Sci. 2020, 595, 117474. [Google Scholar] [CrossRef]
- Yuan, S.; Li, J.; Zhu, J.; Volodine, A.; Li, J.; Zhang, G.; Van Puyvelde, P.; Van der Bruggen, B. Hydrophilic nanofiltration membranes with reduced humic acid fouling fabricated from copolymers designed by introducing carboxyl groups in the pendant benzene ring. J. Membr. Sci. 2018, 563, 655–663. [Google Scholar] [CrossRef]
- Doan, V.; Ko1ppe, R.; Kasai, P.H. Dimerization of Carboxylic Acids and Salts∶An IR Study in Perfluoropolyether Media. J. Am. Chem. Soc. 1997, 119, 9810–9815. [Google Scholar] [CrossRef]
- Lentsch, S.; Aimar, P.; Orozco, J.L. Separation albumin-PEG: Transmission of PEG through ultrafiltration membranes. Biotechnol. Bioeng. 1993, 41, 1039–1047. [Google Scholar] [CrossRef] [PubMed]
- Kesting, R.E. The four tiers of structure in integrally skinned phase inversion membranes and their relevance to the various separation regimes. J. Appl. Polym. Sci. 1990, 41, 2739–2752. [Google Scholar] [CrossRef]
- Zhenxin, Z.; Matsuura, T. Discussions on the formation mechanism of surface pores in reverse osmosis, ultrafiltration, and microfiltration membranes prepared by phase inversion process. J. Colloid Interface Sci. 1991, 147, 307–315. [Google Scholar] [CrossRef]
- Cassie, A.B.D.; Baxter, S. Wettability of porous surfaces. Trans. Faraday Soc. 1944, 40, 546–551. [Google Scholar] [CrossRef]
- Kim, J.-H.; Lee, K.-H. Effect of PEG additive on membrane formation by phase inversion. J. Membr. Sci. 1998, 138, 153–163. [Google Scholar] [CrossRef]
- Shi, M.; Wang, Z.; Zhao, S.; Wang, J.; Zhang, P.; Cao, X. A novel pathway for high performance RO membrane: Preparing active layer with decreased thickness and enhanced compactness by incorporating tannic acid into the support. J. Membr. Sci. 2018, 555, 157–168. [Google Scholar] [CrossRef]
- Chai, G.; Krantz, W.B. Formation and characterization of polyamide membranes. J. Membr. Sci. 1994, 93, 175–192. [Google Scholar] [CrossRef]
- Freger, V.; Srebnik, S. Mathematical model of charge and density distributions in interfacial polymerization of thin films. J. Appl. Polym. Sci. 2003, 88, 1162–1169. [Google Scholar] [CrossRef]
- Nadler, R.; Srebnik, S. Molecular simulation of polyamide synthesis by interfacial polymerization. J. Membr. Sci. 2008, 315, 100–105. [Google Scholar] [CrossRef]
- Yuan, F.; Wang, Z.; Yu, X.; Wei, Z.; Li, S.; Wang, J.; Wang, S. Visualization of the formation of interfacially polymerized film by an optical contact angle measuring device. J. Phys. Chem. C 2012, 116, 11496–11506. [Google Scholar] [CrossRef]
- Tiraferri, A.; Elimelech, M. Direct quantification of negatively charged functional groups on membrane surfaces. J. Membr. Sci. 2012, 389, 499–508. [Google Scholar] [CrossRef]
- Freger, V. Nanoscale heterogeneity of polyamide membranes formed by interfacial polymerization. Langmuir 2003, 19, 4791–4797. [Google Scholar] [CrossRef]
- Tang, C.Y.; Kwon, Y.-N.; Leckie, J.O. Effect of membrane chemistry and coating layer on physiochemical properties of thin film composite polyamide RO and NF membranes I. FTIR and XPS characterization of polyamide and coating layer chemistry. Desalination 2009, 242, 149–167. [Google Scholar] [CrossRef]
- Yip, N.Y.; Tiraferri, A.; Phillip, W.A.; Schiffman, J.D.; Elimelech, M. High performance thin-film composite forward osmosis membrane. Environ. Sci. Technol. 2010, 44, 3812–3818. [Google Scholar] [CrossRef]
- Lee, A.; Elam, J.W.; Darling, S.B. Membrane materials for water purification: Design, development, and application. Enviorn. Sci. Water Res. 2016, 2, 17–42. [Google Scholar] [CrossRef]
- Wang, J.; Dlamini, D.S.; Mishra, A.K.; Pendergast, M.T.M.; Wong, M.C.Y.; Mamba, B.B.; Freger, V.; Verliefde, A.R.D.; Hoek, E.M.V. A critical review of transport through osmotic membranes. J. Membr. Sci. 2014, 454, 516–537. [Google Scholar] [CrossRef]
- Zhang, Y.; Benes, N.E.; Lammertink, R.G. Visualization and characterization of interfacial polymerization layer formation. Lab Chip 2015, 15, 575–580. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Xu, R.; Yang, F.; Kang, J.; Cao, Y.; Xiang, M. Probing influences of support layer on the morphology of polyamide selective layer of thin film composite membrane. J. Membr. Sci. 2018, 556, 374–383. [Google Scholar] [CrossRef] [Green Version]
- Wong, M.C.Y.; Lin, L.; Coronell, O.; Hoek, E.M.V.; Ramon, G.Z. Impact of liquid-filled voids within the active layer on transport through thin-film composite membranes. J. Membr. Sci. 2016, 500, 124–135. [Google Scholar] [CrossRef] [Green Version]




 ” refers to be dense; “
” refers to be dense; “  ” medium; “
” medium; “  ” loose.
” loose.
 ” refers to be dense; “
” refers to be dense; “  ” medium; “
” medium; “  ” loose.
” loose.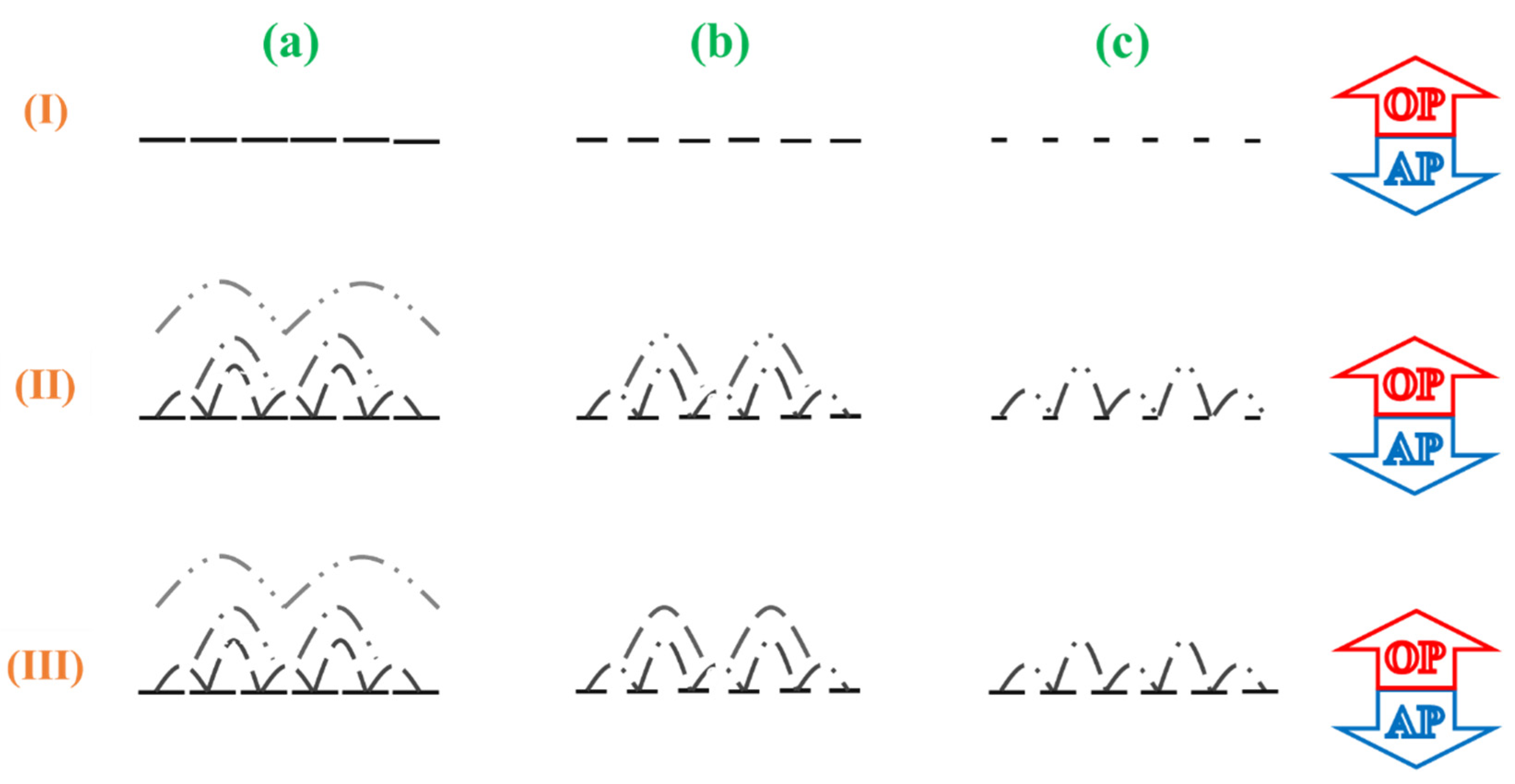
| CPES Copolymer | Support | NF Membrane |
|---|---|---|
| CPES-0 a | S-0 b | N-0 c |
| CPES-20 | S-20 | / |
| CPES-40 | S-40 | N-40 |
| CPES-60 | S-60 | / |
| CPES-80 | S-80 | / |
| CPES-100 | S-100 | N-100 |
| Membrane | Test solution | Concentration (mgL−1) | pH | Temperature (°C) | Pressure (MPa) | MSV a (ms−1) |
|---|---|---|---|---|---|---|
| NF | MgSO4 | 2000 ± 20 | 7.5 ± 0.5 | 25.0 ± 0.2 | 0.60 ± 0.02 | ≥0.45 |
| Support | S-0 | S-20 | S-40 | S-60 | S-80 | S-100 |
|---|---|---|---|---|---|---|
| d (nm) | 9 | 9 | 9 | 9 | 8 | 4 |
| δS (μm) | 48 ± 1 | 50 ± 1 | 48 ± 1 | 49 ± 1 | 48 ± 1 | 48 ± 1 |
| RMS (nm) | 18.3 ± 1.2 | 22.7 ± 2.0 | 27.5 ± 2.1 | 36.6 ± 2.2 | 41.3 ± 2.6 | 49.4 ± 1.4 |
| CA (deg.) | 90.43 ± 0.86 | 87.93 ± 0.44 | 87.56 ± 0.22 | 87.11 ± 0.37 | 86.43 ± 0.92 | 82.12 ± 0.65 |
| NF Membrane | N-0 | N-40 | N-100 |
|---|---|---|---|
| δN (nm) | 262 ± 51 | 96 ± 43 | 72 ± 26 |
| λN (nm) | 282 ± 96 | 195 ± 102 | 142 ± 54 |
| CA (deg.) | 38.41 ± 0.71 | 33.92 ± 1.02 | 18.94 ± 0.75 |
| RMS (nm) | 41.2 ± 1.7 | 78.5 ± 2.4 | 45.0 ± 0.4 |
| NF Membrane | O 1s (%) | N 1s (%) | C 1s (%) | O/N | C*=O (%) |
|---|---|---|---|---|---|
| N-0 | 14.01 ± 0.53 | 9.78 ± 0.82 | 76.21 ± 0.22 | 1.44 ± 0.07 | 13.72 ± 0.61 |
| N-40 | 13.95 ± 0.59 | 10.17 ± 0.74 | 75.87 ± 0.24 | 1.37 ± 0.04 | 14.15 ± 0.65 |
| N-100 | 14.27 ± 0.69 | 10.14 ± 0.86 | 75.59 ± 0.21 | 1.41 ± 0.06 | 13.38 ± 0.72 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lian, Y.; Zhang, G.; Wang, X.; Yang, J. Impacts of Surface Hydrophilicity of Carboxylated Polyethersulfone Supports on the Characteristics and Permselectivity of PA-TFC Nanofiltration Membranes. Nanomaterials 2021, 11, 2470. https://doi.org/10.3390/nano11102470
Lian Y, Zhang G, Wang X, Yang J. Impacts of Surface Hydrophilicity of Carboxylated Polyethersulfone Supports on the Characteristics and Permselectivity of PA-TFC Nanofiltration Membranes. Nanomaterials. 2021; 11(10):2470. https://doi.org/10.3390/nano11102470
Chicago/Turabian StyleLian, Yingfu, Gang Zhang, Xiaojun Wang, and Jie Yang. 2021. "Impacts of Surface Hydrophilicity of Carboxylated Polyethersulfone Supports on the Characteristics and Permselectivity of PA-TFC Nanofiltration Membranes" Nanomaterials 11, no. 10: 2470. https://doi.org/10.3390/nano11102470
APA StyleLian, Y., Zhang, G., Wang, X., & Yang, J. (2021). Impacts of Surface Hydrophilicity of Carboxylated Polyethersulfone Supports on the Characteristics and Permselectivity of PA-TFC Nanofiltration Membranes. Nanomaterials, 11(10), 2470. https://doi.org/10.3390/nano11102470







