1. Introduction
Amorphous structures have been prepared in the binary Ni-X systems, while Ni-Nb system is one of the best binary metallic glass forming systems due to their unique properties including excellent properties, high corrosion resistance and various magnetic properties [
1,
2,
3]. Ni-Nb alloys exhibited a wide composition range (40–60%) to form amorphous structures. The best forming composition of the amorphous phase was Ni = 32% and Nb = 38% [
4,
5,
6], which could be well confirmed by cluster formulas for stable liquids [
7]. However, amorphous alloys possessed very low ductility as per-experiment study according to the reference [
8]. Besides, it is hard for researchers to fabricate amorphous alloys to ensure its thermal stability that is another drawback. Under the heating condition, the free energy would be reduced through crystallization for the amorphous alloys. Nevertheless, several properties of amorphous alloys with the nanoscale crystal structure were found to be fundamentally different from, and often superior to, those of the conventional polycrystals and amorphous solids. For instance, nano-crystalline may enhance the strength, and ductility of materials and so on [
9,
10], which could remedy the above drawbacks. These nanocrystalline materials could have many potential applications, such as for surface protection and in nano-devices [
11,
12]. Pioneer explorations to study methods for nano-crystallization from amorphous state of Ni-Nb system was mainly included mechanical alloying [
9,
13,
14], magnetron sputtering method [
15,
16] with the starting materials in the solid, powder or gaseous states. All of these methods have their inherent disadvantages for fabricating the amorphous alloys. The thin amorphous films were successfully prepared onto the substrate material through the sputtering method. However, there were the poor cohesion and adhesion between the film and substrate, limiting their potential biomedical and other applications. Mechanical alloying is only suitable for preparing powder materials. Therefore, searching for the suitable method to overcome these problems could be regarded as a better way.
The above problems could be solved by the formation of the alloying layer with amorphous structures in nanoscale size on the substrate. The alloying layer could be achieved by High current pulsed electron beam (HCPEB) irradiation as an innovative processing technique, which has been developed in recent years. Generally, this technique with many characteristics, such as short-duration pulse, high energy etc., was adopted to complete surface treatment. As inputting extremely intensive energy onto the topmost surface, the material surface was melted quickly and then solidified owing to the heat extraction towards the sub-surface. Under the effect of ultrafast melting and solidification, refined grains, even nano-grains and amorphous phase were formed which could be realized to promote the improvement of material properties [
17,
18,
19]. HCPEB irradiation is an appropriate method for forming amorphous phase onto the surface layer. According to the reference [
20], HCPEB irradiation promoted the formation of Ni-Nb amorphous layer onto the substrate to protect the substrate from corrosion. However, for the films/substrate system, the liquid-phase mutual diffusion impeded the amorphization process of melting layer and then resulted in the formation of nanocrystalline in the solidification process [
21]. Meanwhile, supersaturated solid solution would be decomposed to form particles in nanoscale at the substrate [
22,
23]. Therefore, amorphous phase mixed nanostructures could be obtained by HCPEB technology, finally promoting a huge application of material. According to the previous literatures [
17,
18,
19,
20,
21,
22,
23,
24,
25], investigation about amorphous mixed nanostructures and its evolution mechanism under HCPEB technology was few.
Therefore, in present research, the new Ni-Nb amorphous alloys have been prepared onto the GH3039 alloys by HCPEB irradiation. The aim of the present work was to investigate the microstructure evolution of Ni-Nb alloying layer for different number of pulses and their corresponding hardness and corrosion property.
3. Results
Figure 2 gives the typical cross-section BSE images of initial and irradiated Ni-Nb coating. As shown in
Figure 2a, the Ni-Nb coating with an average thickness of 1.9 μm was successfully prepared on the surface of GH3039 substrate. The as-fabricated coatings with tiny pore-structures were relatively dense. Besides, Nb particles were clearly observed, indicating that the Ni and Nb elements were no uniformly distributed in the coating. The probable reason was the short milling time and high energy collision to result into insufficient energy input during the ball milling. In addition, cold welding occurred due to the short milling time. The clear interface was formed between the as fabricating a coating and the substrate. After 10-pulsed irradiation, the top layer was re-melted and the thickness of which was only about 0.64 μm as compared to the initial coating, as shown in
Figure 2b. With an increment of the pulses number, the thickness of HCPEB alloyed layer was increased from 1.5 μm for 20-pulsed sample (
Figure 2c) to about 2.5 μm for the 30-pulsed sample. One can see that the morphology of all the irradiated samples exhibited a relatively white layer from the contrast. There was excellent metallurgical bonding that increased the coating density and reduced the interface defect for the irradiated samples. Especially, for the 30-pulsed samples, the alloying layer can be clearly divided into three layers: re-melted layer, heat affected zone and the substrate. From the line scanning result inserted in
Figure 2d, there was obvious Nb-enriched element at the beginning of the line scanning, which was corresponded to the re-melted layer. Besides, it could be seen that the Nb element was present in the heat affected layer that was resulted from the dissolution of the Nb. Therefore, it is considered that the Ni-Nb alloying layer was formed onto the substrate after HCPEB irradiation.
Figure 3 depicts a typical XRD pattern of the initial Ni-Nb coating and after HCPEB irradiation. The reasons of phase transformation were allowed to make several assumptions due to surface treatment. Firstly, the diffractograms exhibit some peaks of the Ni, Nb and Ni
3Nb phase in the irradiated layer. Noticeably, Nb peak at 2ϴ = 38° with low intensity still existed in the XRD pattern after 10-pulsed irradiation. With pulses increased to 20, the Nb peak was disappeared, which suggests the formation of Ni
3Nb or Ni(Nb) solid solution. Additionally, Ni diffraction peaks have shifted to low angle after 20-pulsed irradiation comparing to the initial sample, meaning that the Ni lattices were expanded, which indicates that Ni(Nb) solid solution was formed. For the 30-pulsed samples, a minor shift of Ni peaks to high angle occurred. The possible reason was that the Nb atoms were decomposed from the Ni(Nb) solid solution to form Ni
3Nb phase. Especially in the pattern of 30-pulsed Ni-Nb samples, the δ-Ni
3Nb phase was clearly identified. Besides, the broader diffractograms peaks of Ni were resulted from the formation of amorphous phase due to the distorted lattice. Certainly, all of the grain refining, dislocation density increasing and micro stress accumulating resulted into broader peaks. The specific reason needs further detailed characterization by TEM to elaborate in the following.
Figure 4 shows the SAED patterns of local area for Ni-Nb coating with different number of pulses. From
Figure 4a–c, it could be considered that an amorphous phase was formed in the irradiated samples, but the atomic range order of which was different. After 10-pulsed irradiation, the SAED pattern gives three broader rings with low diffusion (
Figure 4a), suggesting the atom ordered arrangement of crystal structure still existed in the characterized local area. Namely, the degree of amorphization was low. With the pulse number increased to 20, only two diffraction rings were observed in the SAED pattern (
Figure 4b), indicating the ordered arrangement of only the nearest neighbor and next nearest neighbor and the disordered arrangement of the third adjacent atoms. Therefore, it could decide that these structures have been completely amorphous. However, after HCPEB irradiation with 30 pulses, the SAED pattern in
Figure 4c exhibits that the amorphous halo coexists with several weak diffraction speckles, suggesting that the nano-particles were separated out within the measured area. Therefore, the structural transformation in the Ni-Nb layer could be described as from partial amorphous to fully amorphous and then to nano-crystalline from the perspective of the SAED patterns due to HCPEB irradiation.
In addition, a large number of nano black particles and blocky-shaped structures with about 30 nm in size were formed in some area, as shown in
Figure 5c, the SAED (taken from the green circle in the
Figure 5c) of which demonstrated that the Ni phase was coexisted with the Ni
3Nb phase (
Figure 5d). For further examining the morphology of Ni and Ni
3Nb phase, the HRTEM image (magnification of the region 1) and the Fast-flourier transformation (FFT) pattern of enlarged image was exhibited in the
Figure 5e. For the A zone, the d value of the measure area correspond well with d(020) = 0.212 nm for the δ-Ni
3Nb, and for the B zone, d(111) = 0.203 nm and d(200) = 0.176 nm for the FCC Ni phase. Therefore, it could be concluded that nano black particles were the mixture of γ−Ni particles and Ni
3Nb particles with the blocky-shaped structures.
Figure 6a,b give the bright field and HRTEM images of 20-pulsed Ni-Nb layer. Although the SAED of 20 pulsed samples indicates that the matrix was composed of the amorphous phase (
Figure 4b), the very fine clusters crystals with about 3 nm in size were still formed in the amorphous layer on the topmost matrix, as shown in
Figure 6a. From the HRTEM image, the individual nanoparticles were separated by envelopes of the amorphous matrix. The FFT pattern taken from the
Figure 6b indicates that the d value was equal with 0.2023 nm, which was Ni or Ni
3Nb clusters.
Figure 6c,d exhibit the images of 20-pulsed Ni-Nb coating through high angle angular field (HAADF) and the corresponding elements distribution. It could be concluded from the results of HAADF image that the element with large atomic number segregated at the spherical microstructures, as shown in
Figure 6c. From
Figure 6d, the spectrum analysis result indicates that the spherical microstructure was consisted of the Nb element dispersed in the Ni matrix. Therefore, combined of the HRTEM image, these results confirmed that the spherical microstructures were Ni
3Nb particles. In addition, different from the 10-pulsed samples, no large size Nb particles were observed in the 20-pulsed layer, which suggests that Nb was completely dissolved into the Ni amorphous matrix.
After 30-pulsed irradiations, a little change in microstructure took place in the alloying layer. There was also consisted of amorphous matrix and the nano particles embedded in it, but the size of which increased to about ~8 nm, as shown in
Figure 7a. The SAED pattern exhibits the diffuse ring along with several pairs of diffraction spots that confirmed the presence of a homogeneous amorphous structure with amounts of finely-dispersed nanocrystalline Ni
3Nb. Noticeably, faint diffraction speckles were observed in
Figure 7b, which suggested the formation of ultrafine Nb particles that were the product of precipitation from the γ-Ni. Besides, the HRTEM image (magnification of the region 2) was used to demonstrate the presence of Ni
3Nb and Nb particles, as shown in
Figure 7c. The FFT and inverse FFT patterns corresponding to the A zone was indexed as the Nb phase, and that of the B zone was confirmed as Ni
3Nb phase. Therefore, it can be concluded that the alloying layer was consisted of nano Νb and Ni
3Nb particles that were uniformly dispersed into the Ni-Nb amorphous matrix after 30-pulsed irradiation.
Based on the experimental results and observations at different stages of HCPEB irradiation, a microstructure evolution is proposed for HCPEB irradiation, as shown by a schematic in
Figure 8. Under as-milled condition, all the atoms are uniformly arranged due to short milled time, as shown in
Figure 8a. After 10-pulsed irradiation, the amorphous phase was formed in the Ni-Nb layer, which could be got from
Figure 4a. The rapid cooling is responsible for the amorphization of the Ni
60Nb
40 coatings due to HCPEB irradiation. However, from
Figure 5, there were still the Ni, Nb particles and intermetallic Ni
3Nb with large size dispersed in the amorphous Ni-Nb layer (
Figure 8b). Hence, the stage I was identified as partial amorphization stage. With increasing the pulses number to 20, the residual Nb particles were continuously dissolved into the amorphous matrix to aggravate the amorphization. Yet, it is inevitable that Ni
3Nb clusters were formed and uniformly distributed in the amorphous Ni-Nb layer (see
Figure 5). Therefore, the stage II could be called fully amorphization process comparing to the stage I (
Figure 8c). The more energy inputting is responsible for the fully amorphous of the Ni
62Nb
38 layer, which have reduced the diffusivity and retarded rearrangement of all atoms after proceeding uniform mixing of Ni and Nb element. Meanwhile, the number of nucleation sites could be increased during 20-pulsed irradiation, restraining the coarsening of Ni
3Nb clusters at this stage. In addition, the atoms with the difference in the atomic size changed in the order Nb (0.143 nm) > Ni (0.125 nm), together with the highly negative mixing enthalpy in the Ni-Nb (−30 kJ/mol) [
26,
27]. All of the above factors could make it hard to arrange the Ni and Nb element on a long-range scale under the ultra-rapid cooling rates condition during the HCPEB irradiation. Therefore, during the stage II of HCPEB irradiation, the Ni
3Nb clusters have grown to only ~3 nm in size, as shown in
Figure 6a. Actually, it indicates that the onset of primary crystallization has begun in the process of fully amorphization.
On base of the stage II, the size of nano-Ni
3Nb clusters coarsened up to 8 nm in size with increasing pulses number. Meanwhile, a large number of Nb nano-particles has already been formed during this stage. This strongly supported that the chemical fluctuation to break the Ni/Nb component ratio (Ni:Nb = 62:38) in the local area. It is considered that the growth of Ni
3Nb particles is due to composition fluctuation existed in the liquid metal [
28]. Owing to the ultra-short time (1.5 μs) of HCPEB irradiation, it is hard to grow up for the Nb and Ni
3Nb nano-particles, as shown in
Figure 7c. Therefore, nano-crystallization (stage III) from a fully amorphous state during HCPEB irradiation enforced crystallization of locally enriched regions under rapid cooling rates (
Figure 8d). In addition, with increasing thickness of Ni-Nb coating, Ni element from the GH3039 matrix has dissolved into the Ni
62Nb
38 alloying layer also to break the Ni/Nb component ratio (the best forming amorphous ratio of Ni to Nb is 62:38), which makes it to deviate proper component of amorphous phase. Finally, the formation of nano-crystalline inevitably took place to decrease the content of amorphous phase for 30-pulsed Ni-Nb layer. Therefore, it could be concluded that the microstructure evolution proposed here was partial amorphous to fully amorphous and then to nano-crystalline during HCPEB irradiation.
To investigate the mechanical property of Ni-Nb coating, the microhardness testing was performed.
Figure 9 gives the hardness values for the Ni-Nb coating with the different pulses. For the initial sample, the hardness was about 375 HV; while the hardness value was 425, 520 and 458 HV after HCPEB irradiation, corresponding to 10, 20 and 30 pulses, respectively. It can be seen that the microhardness values had an increasing trend with increasing up to 20 pulses and then dropped slightly after 30 pulses. On the whole, the hardness was dramatically improved after HCPEB irradiation. The mainly reason for increasing hardness was the formation of mixing amorphous and nanocrystalline structures. Vickers hardness suggested the bonding strengthen inside alloys, and was referred to one of intrinsic parameters to characterize the mechanical properties of alloys [
29]. The number of metallic bonding between adjacent atoms was increased in Ni-Nb layer to result in the increasing the number of atoms connected by metallic bonding. Then the higher hardness value could be obtained due to the presence of no directed bonding in the binary Ni-Nb amorphous layer. Therefore, the fully amorphized structures for 20-pulsed samples together with the uniformly distributed Ni
3Nb particles resulted in the increasing strengthen, enabling the Ni-Nb layer hardening. It was understandable that the decreasing hardness of 30-pulsed samples was owing to the precipitation of Nb nano-particles, damaging reduction of amorphous phase strengthen. Meanwhile, the coarsening of dispersed Ni
3Nb nanocrystals occurred during HCPEB irradiation. So, these factors were making hardness for the 30-pulsed samples to decrease. In conclusion, samples hardening was mainly attributed to the metal-metal interatomic bonding strengthen, nanocrystalline strengthen and so on [
23,
30].
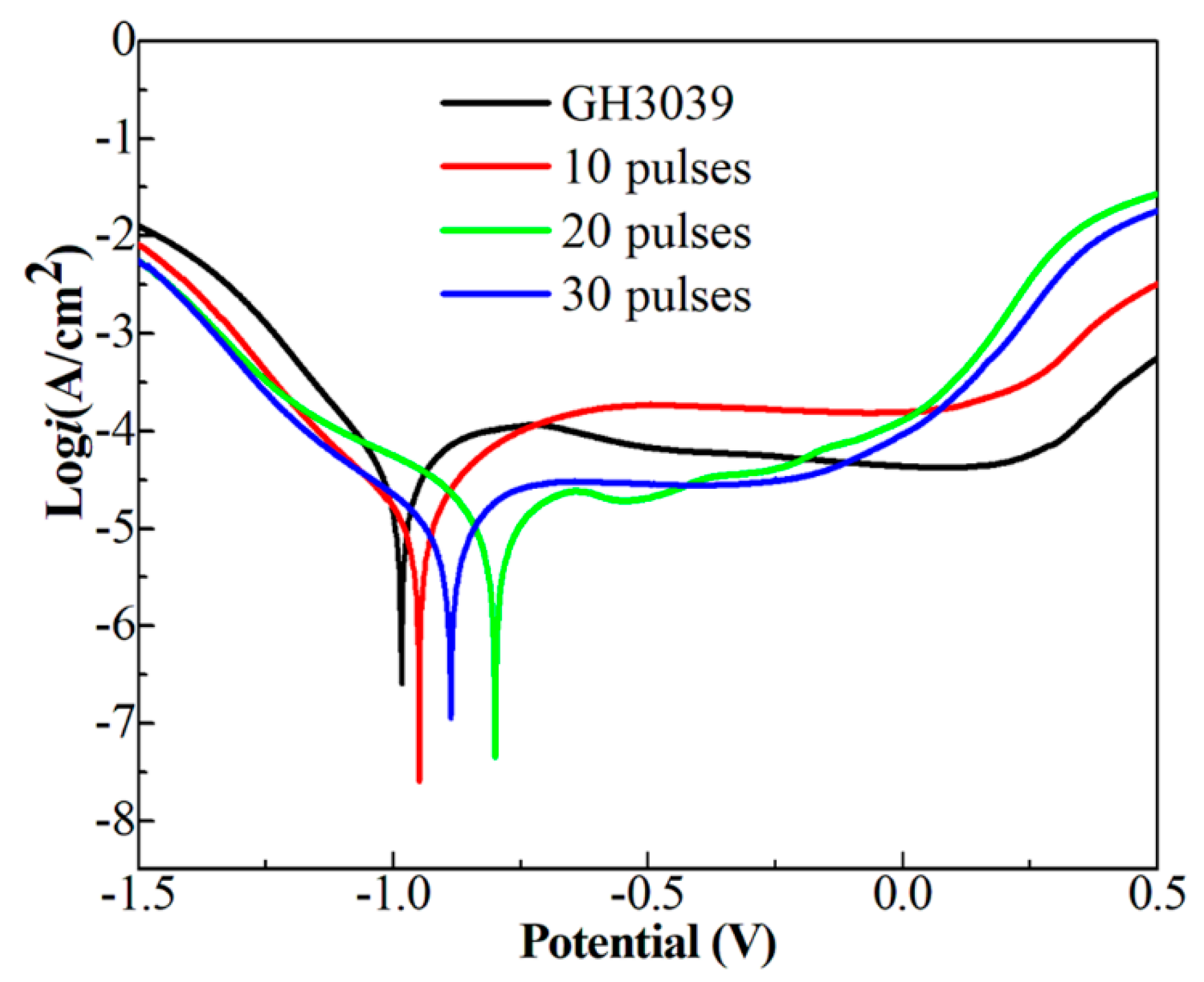
Figure 10 gives the polarization curves of the Ni-Nb coating before and after HCPEB irradiation and the corresponding corrosion current ((i
corr) and corrosion potential (E
corr) were calculated and tabulated in
Table 1. From
Figure 10, it can be observed that the polarization curve is divided into four parts: active region, passive transition zone, passive stabilization zone and active passive zone [
31]. At the early stage, there were the crystal defects seen in the many HCPEB treated metals [
20,
25] with energy concentration inside microstructure at the topmost layer, which was easy to be corroded preferentially, forming the active region. Then hydrolysis of corrosion products promoted the formation of dense passivation film, resulting in a stable passivation area. The E
corr and the I
corr of the initial samples were −0.990 V and 28.90 μA·cm
−2, respectively, as shown in
Table 1. After HCPEB irradiation, the E
corr and I
corr of specimens were changed apparently. The Ecorr of 10- and 20-pulsed samples was increased to −0.904 V and −0.799 V and I
corr decreased to 10.2 μA/cm
2 and 6.98 μA/cm
2, respectively. As increasing the pulses number up to 30, the I
corr increased to 7.83 μA/cm
2 and the E
corr decreased to −0.887 V. Therefore, a drastic enhancement of corrosion resistance was obtained for the Ni-Nb layer with 20 pulses compared to that of other samples.
Generally, the corrosion resistance of amorphous alloys was superior to the crystalline metal material. However, the Ni-Nb alloys only with amorphous structure have a characterization of the cracks in the reference [
20]. It destroyed the surface integrity and aggravated the damage of the surface of the Ni-Nb alloy. However, in present paper, these cracks could be released or eliminated under the circumstance of the amorphous layer with the nanoscale crystal structure, which improved the toughness and plasticity [
9,
10]. Therefore, the enhancement in corrosion resistance of Ni-Nb layer was naturally connected with the formation of nanocrystalline and amorphous phase. The 20-pulsed samples have the best corrosion resistance in the test. In addition, the corrosion resistance for the different pulses samples also has been affected by the formation of crystal defects, deformed structures, solid solution precipitation and intermetallic compounds as reported in the references [
32,
33], which will be confirmed by further characterization in the future.