Tribological Performance Investigation of a Commercial Engine Oil Incorporating Reduced Graphene Oxide as Additive
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Consumables
2.2. Materials’ Preparation Procedure
2.2.1. Preparation of rGO
2.2.2. Preparation of the Engine Oil (5W-40) Samples Incorporating rGO as an Additive
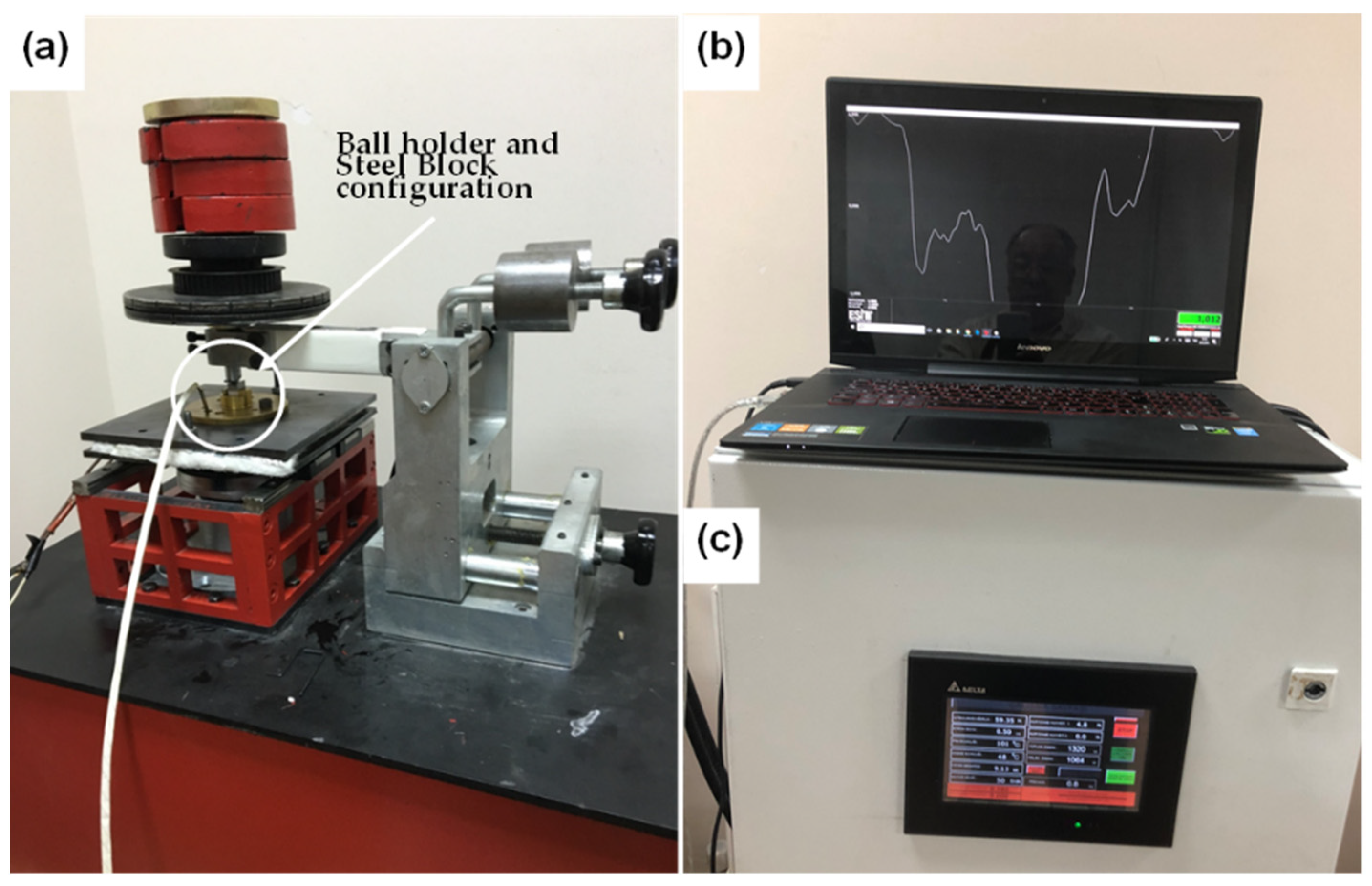
2.3. Tribological Characterization
2.4. Characterization Methods
3. Results and Discussions
3.1. rGO Analysis
3.2. Evaluation of 5W-40 Oil Samples Incorporating rGO
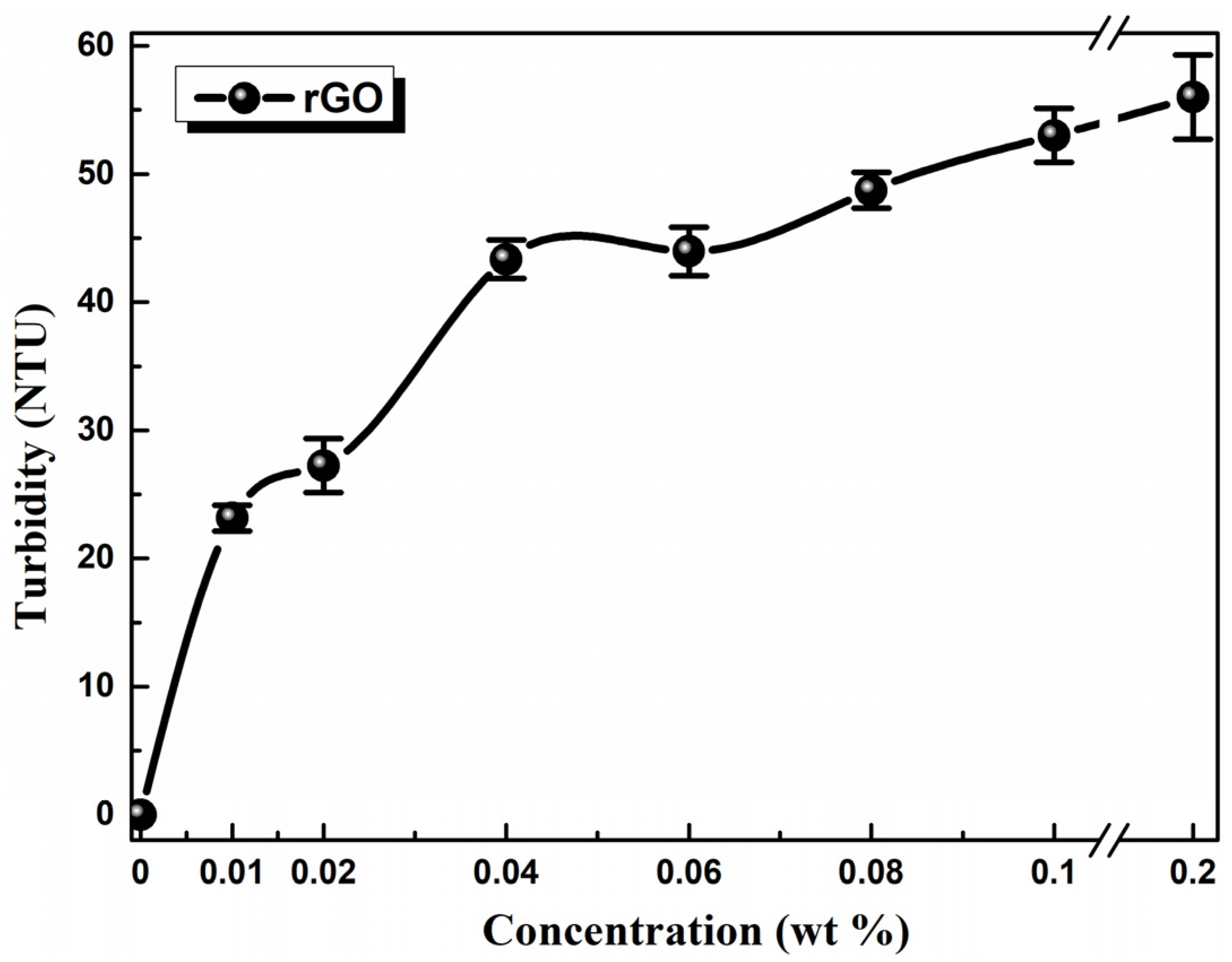
3.2.1. Turbidimetry Measurements
3.2.2. Size and Zeta Potential Measurements
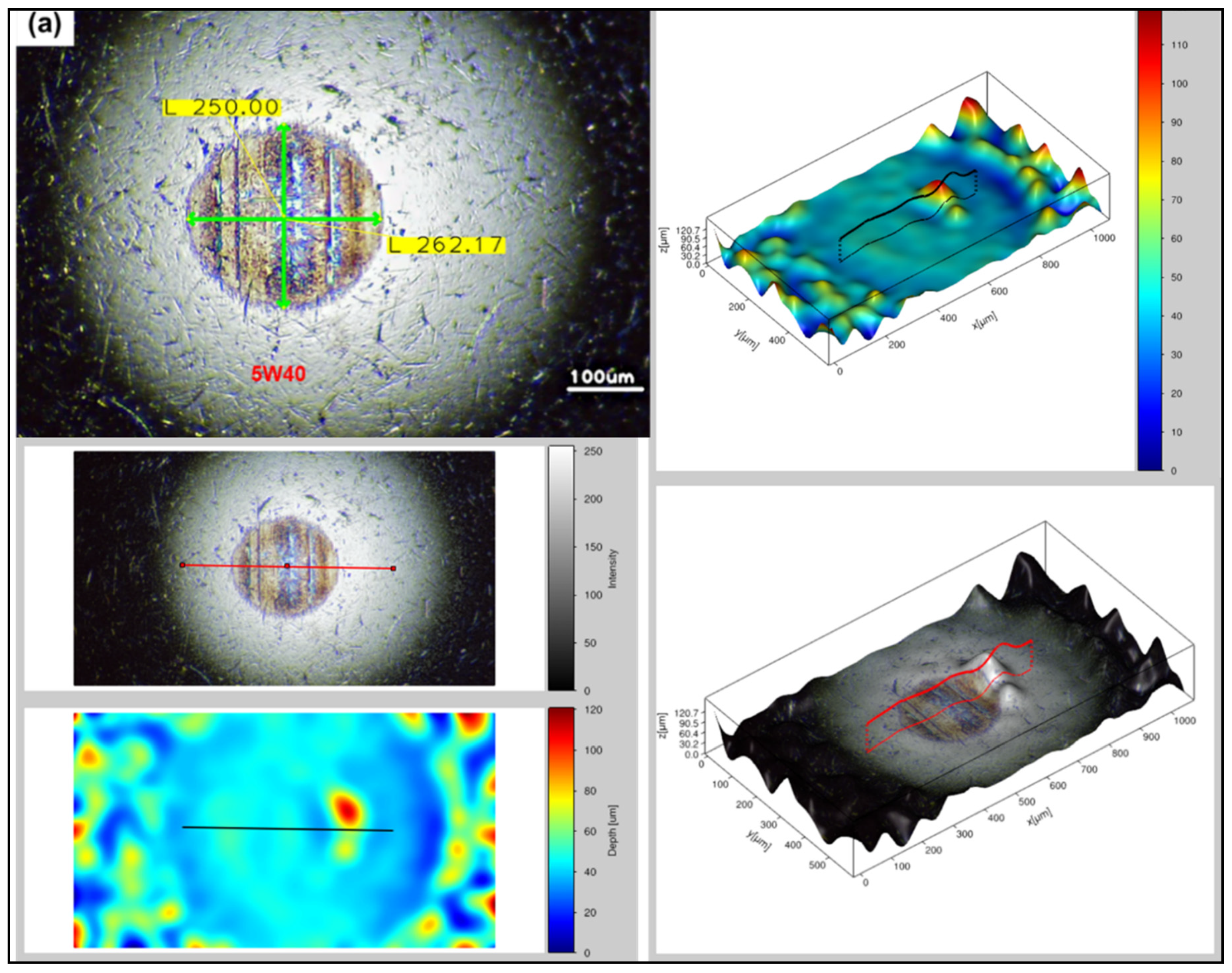
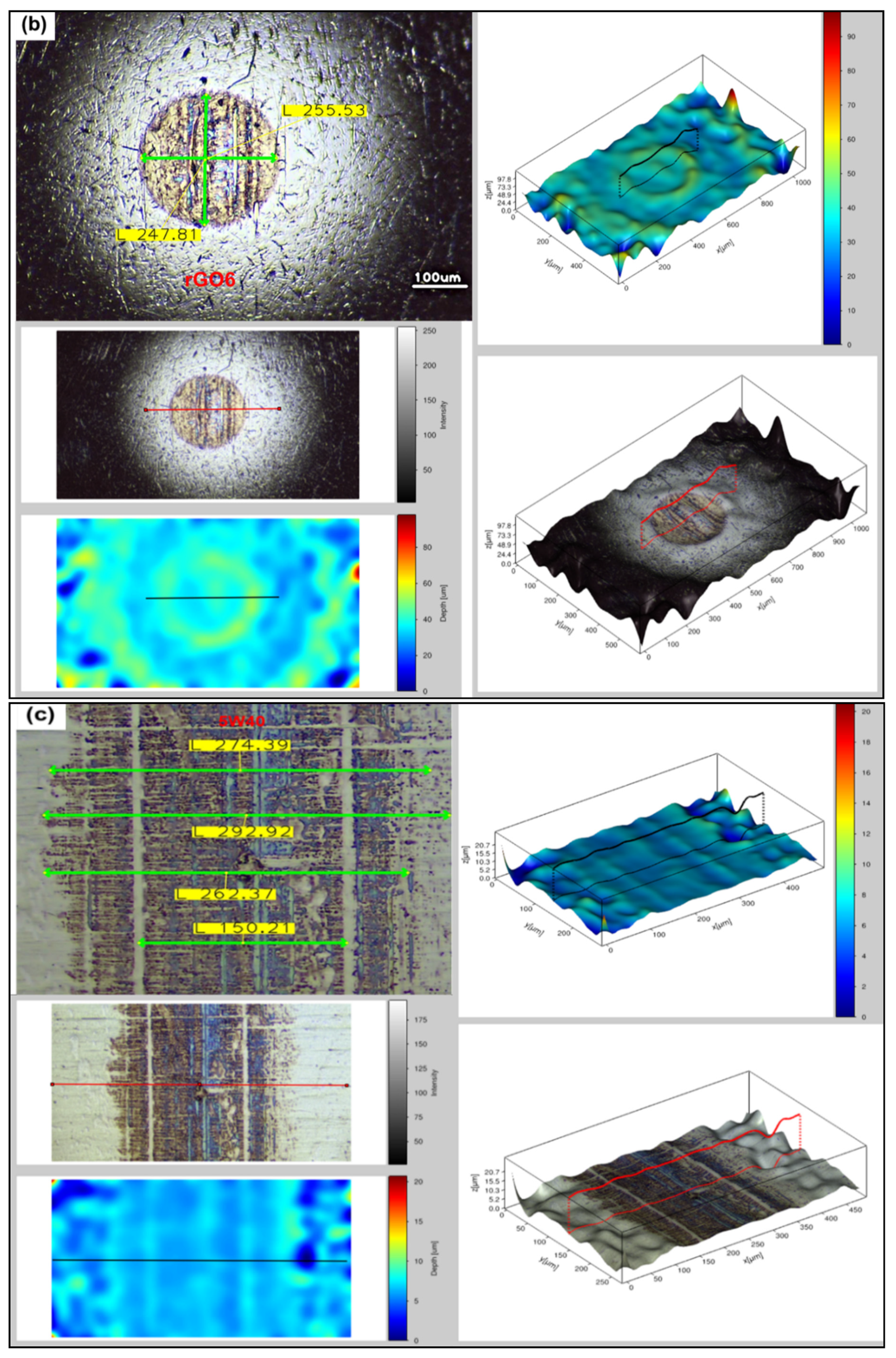
3.3. Surface Analysis
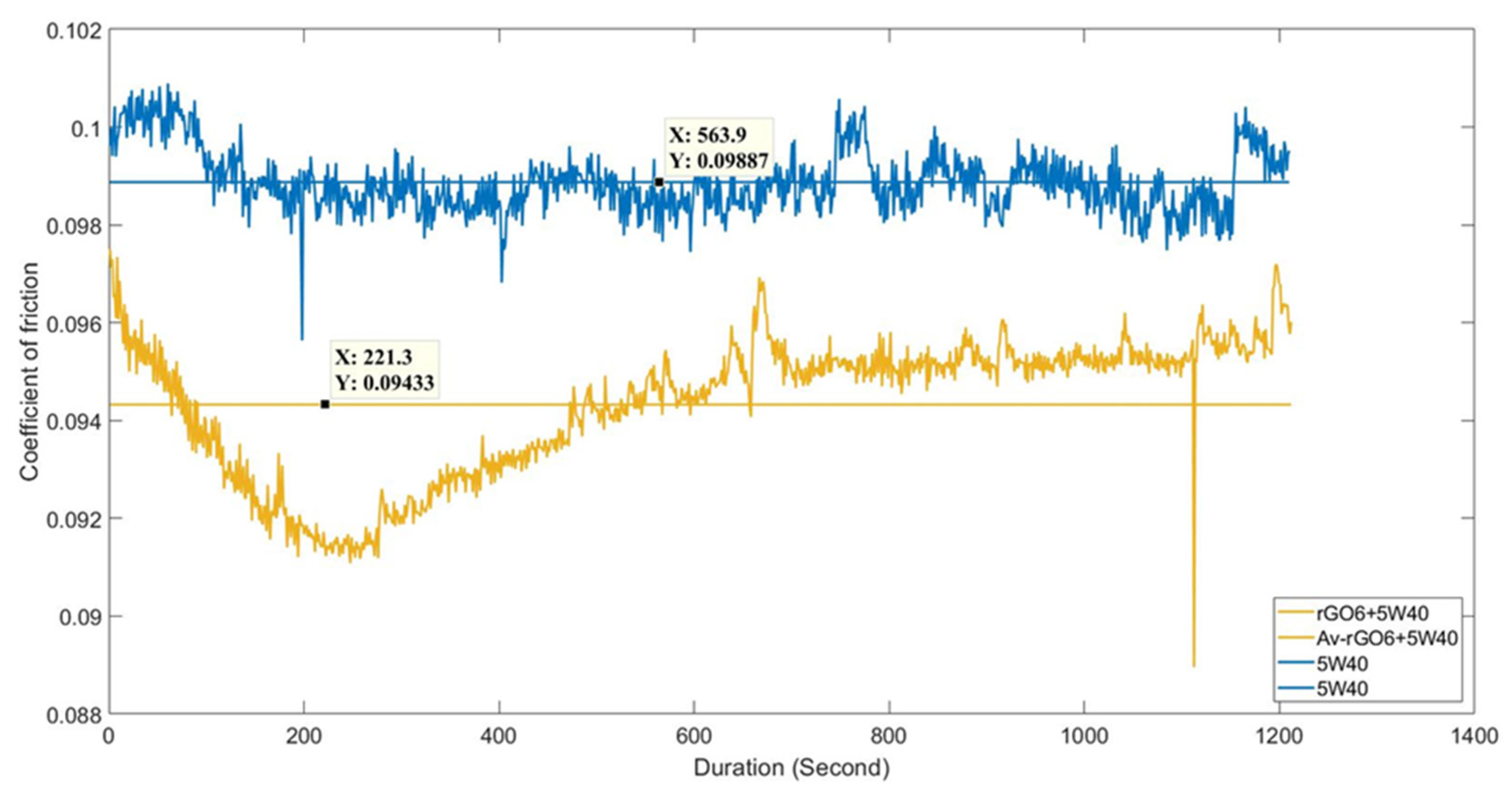
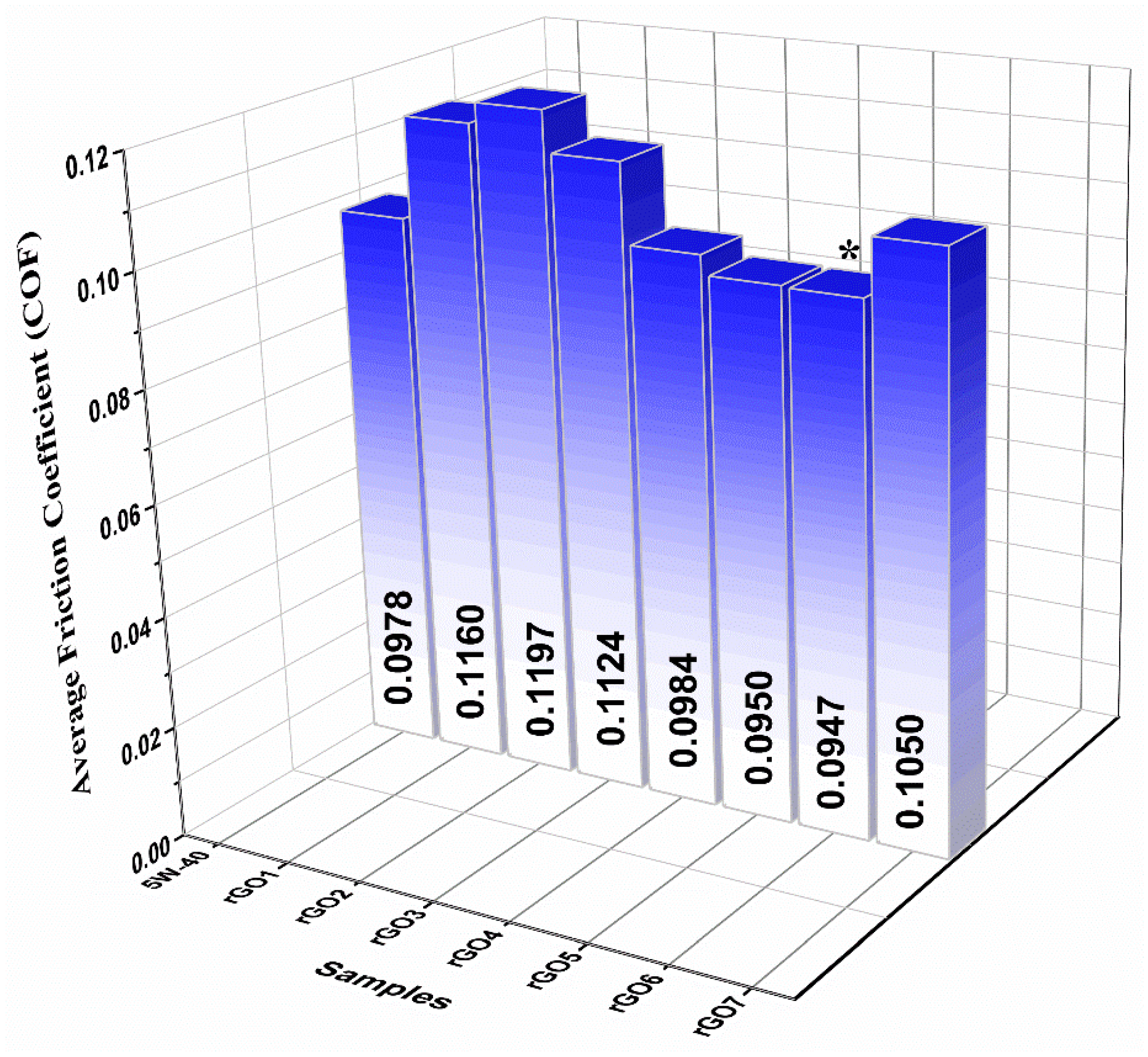
3.4. Tribological Characterization
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kohn, E.M. A theory on the role of lubricants in metal cutting at low speeds and in boundary lubrication. Wear 1965, 8, 43. [Google Scholar] [CrossRef]
- Erdemir, A. Handbook of Modern Tribology; Bhushan, B., Ed.; CRC Press: Boca Raton, FL, USA, 2001; pp. 787–818. [Google Scholar]
- Liu, X.; Lu, Z.; Dong, H.; Cao, Y.; Qian, X. Friction and Wear Characteristics of Microporous Interface Filled with Mixed Lubricants of M50 Steel at Different Loads. Materials 2020, 13, 2934. [Google Scholar] [CrossRef] [PubMed]
- Diana, B.; Ali, E.; Anirudha, V.S. Graphene: A new emerging lubricant. Mater. Today 2014, 17, 31. [Google Scholar] [CrossRef] [Green Version]
- Li, Z.; Xu, C.; Xiao, G.; Zhang, J.; Chen, Z.; Yi, M. Lubrication Performance of Graphene as Lubricant Additive in 4-n-pentyl-4′-cyanobiphyl Liquid Crystal (5CB) for Steel/Steel Contacts. Materials 2018, 11, 2110. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, L.; Zhou, M.; Jin, L.; Li, L.; Mo, Y.; Su, G.; Li, X.; Zhu, H.; Tian, Y. Recent advances in friction and lubrication of graphene and other 2D materials: Mechanisms and applications. Friction 2019, 7, 199. [Google Scholar] [CrossRef] [Green Version]
- Nine, M.J.; Cole, M.A.; Tran, D.N.H.; Losic, D. Graphene: A multipurpose material for protective coatings. J. Mater. Chem. A 2015, 3, 12580. [Google Scholar] [CrossRef]
- Smolyanitsky, A.; Killgore, J.P.; Tewary, V.K. Effect of elastic deformation on frictional properties of few-layer graphene. Phys. Rev. 2012, 85, 035412. [Google Scholar] [CrossRef] [Green Version]
- Feng, X.; Kwon, S.; Park, J.Y.; Salmeron, M. Superlubric Sliding of Graphene Nanoflakes on Graphene. ACS Nano 2013, 7, 1718. [Google Scholar] [CrossRef]
- Lee, C.; Li, Q.; Kalb, W.; Liu, X.Z.; Berger, H.; Carpick, R.W.; Hone, J. Frictional characteristics of atomically thin sheets. Science 2010, 328, 76. [Google Scholar] [CrossRef] [Green Version]
- Kim, H.; Kim, D. Water Lubrication of Stainless Steel using Reduced Graphene Oxide Coating. Sci. Rep. 2015, 5, 17034. [Google Scholar] [CrossRef]
- Kaleli, H. Evaluation of additive’s layer formation in engine crankcase oil using two different types of tribological test rigs. Ind. Lub. Tribol. 2004, 56, 158. [Google Scholar] [CrossRef]
- Restuccia, P.; Righi, M.C. Tribochemistry of graphene on iron and its possible role in lubrication of steel. Carbon 2019, 141, 794. [Google Scholar] [CrossRef]
- Fan, X.; Xia, Y.; Wang, L.; Li, W. Multilayer Graphene as a Lubricating Additive in Bentone Grease. Tribol. Lett. 2014, 55, 455. [Google Scholar] [CrossRef]
- Gupta, B.; Kumar, N.; Panda, K.; Dash, S.; Tyagi, A.K. Energy efficient reduced graphene oxide additives: Mechanism of effective lubrication and antiwear properties. Sci. Rep. 2016, 6, 18372. [Google Scholar] [CrossRef] [Green Version]
- Ali, M.K.A.; Xianjun, H.; Mai, L.; Qingping, C.; Turkson, R.F.; Bicheng, C. Improving the tribological characteristics of piston ring assembly in automotive engines using Al2O3 and TiO2 nanomaterials as nano-lubricant additives. Tribol. Inter. 2016, 103, 540. [Google Scholar] [CrossRef]
- Rajkumar, K.; Aravindan, S. Tribological behavior of microwave processed copper–nanographite composites. Tribol. Inter. 2013, 57, 282. [Google Scholar] [CrossRef]
- Rylski, A.; Siczek, K. The Effect of Addition of Nanoparticles, Especially ZrO2-Based, on Tribological Behavior of Lubricants. Lubricants 2020, 8, 23. [Google Scholar] [CrossRef] [Green Version]
- Ali, M.K.A.; Xianjun, H.; Mohamed, A.A.; Abdelkaree, M.A.A.; Elsheikh, A.H. Role of Nanolubricants Formulated in Improving Vehicle Engines Performance. IOP Conf. Ser. Mater. Sci. Eng. 2019, 563, 022015. [Google Scholar] [CrossRef]
- Bao, T.; Wang, Z.; Zhao, Y.; Wang, Y.; Yi, X. Long-term stably dispersed functionalized graphene oxide as an oil additive. RSC Adv. 2019, 9, 39230. [Google Scholar] [CrossRef] [Green Version]
- Berman, D.; Erdemir, A.; Zinovev, A.V.; Sumant, A.V. Nanoscale friction properties of graphene and graphene oxide. Diam. Relat. Mater. 2015, 54, 91. [Google Scholar] [CrossRef] [Green Version]
- Ou, J.; Wang, J.; Liu, S.; Mu, B.; Ren, J.; Wang, H.; Yang, S. Tribology Study of Reduced Graphene Oxide Sheets on Silicon Substrate Synthesized via Covalent Assembly. Langmuir 2010, 26, 15830. [Google Scholar] [CrossRef] [PubMed]
- Eswaraiah, V.; Sankaranarayanan, V.; Ramaprabhu, S. Graphene-Based Engine Oil Nanofluids for Tribological Applications. ACS Appl. Mater. Interfaces 2011, 3, 4221. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Xie, Z.; Gu, L.; Song, B.; Wang, L. Investigation of the tribological behavior of graphene oxide nanoplates as lubricant additives for ceramic/steel contact. Tribol. Int. 2018, 128, 113. [Google Scholar] [CrossRef]
- Konios, D.; Stylianakis, M.M.; Stratakis, E.; Kymakis, E. Dispersion behaviour of graphene oxide and reduced graphene oxide. J. Colloid Interfaces Sci. 2014, 430, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Iliut, M.; Leordean, C.; Canpean, V.; Teodorescu, C.-M.; Astilean, S. A new green, ascorbic acid-assisted method for versatile synthesis of Au–graphene hybrids as efficient surface-enhanced Raman scattering platforms. J. Mater. Chem. C 2013, 1, 4094. [Google Scholar] [CrossRef]
- Anagnostou, K.; Stylianakis, M.M.; Atsalakis, G.; Kosmidis, D.M.; Skouras, A.; Stavrou, I.J.; Petridis, K.; Kymakis, E. An Extensive Case Study on the Dispersion Parameters of HI-assisted Reduced Graphene Oxide and its Graphene Oxide Precursor. J. Colloid Interfaces Sci. 2020, 580, 332. [Google Scholar] [CrossRef]
- Tarcan, R.; Handrea-Dragan, M.; Todor-Boer, O.; Petrovai, I.; Farcau, C.; Rusu, M.; Vulpoi, A.; Todea, M.; Astilean, S.; Botiz, I. A new, fast and facile synthesis method for reduced graphene oxide in N,N-dimethylformamide. Synth. Met. 2020, 269, 116576. [Google Scholar] [CrossRef]
- Li, W.; Tang, X.-Z.; Zhang, H.-B.; Jiang, Z.-G.; Yu, Z.-Z.; Du, X.-S.; Mai, Y.-W. Simultaneous surface functionalization and reduction of graphene oxide with octadecylamine for electrically conductive polystyrene composites. Carbon 2011, 49, 4724. [Google Scholar] [CrossRef]
- Kim, H.-S.; Yang, B.; Stylianakis, M.M.; Kymakis, E.; Zakeeruddin, S.M.; Grätzel, M.; Hagfeldt, A. Reduced Graphene Oxide Improves Moisture and Thermal Stability of Perovskite Solar Cells. Cell. Rep. Phys. Sci. 2020, 1, 100053. [Google Scholar] [CrossRef]
- Stylianakis, M.M.; Viskadouros, G.; Polyzoidis, C.; Veisakis, G.; Kenanakis, G.; Kornilios, N.; Petridis, K.; Kymakis, E. Updating the Role of Reduced Graphene Oxide Ink on Field Emission Devices in Synergy with Charge Transfer Materials. Nanomaterials 2019, 9, 137. [Google Scholar] [CrossRef] [Green Version]
- Tarcan, R.; Todor-Boer, O.; Petrovai, I.; Leordean, C.; Astilean, S.; Botiz, I. Reduced graphene oxide today. J. Mater. Chem. C 2020, 8, 1198. [Google Scholar] [CrossRef]
- Petridis, C.; Konios, D.; Stylianakis, M.M.; Kakavelakis, G.; Sygletou, M.; Savva, K.; Tzourmpakis, P.; Krassas, M.; Vaenas, N.; Stratakis, E.; et al. Solution processed reduced graphene oxide electrodes for organic photovoltaics. Nanoscale Horiz. 2016, 1, 375. [Google Scholar] [CrossRef] [PubMed]
- Wisniewska, M.; Terpilowski, K.; Chibowski, S.; Urban, T.; Zarko, V.I.; Gunko, V.M. Stability of Colloidal Silica Modified by Macromolecular Polyacrylic Acid (PAA)-Application of Turbidimetry Method. J. Macromol. Sci. A Pure Appl. Chem. 2013, 50, 639. [Google Scholar] [CrossRef]
- Malard, L.M.; Pimenta, M.A.; Dresselhaus, G.; Dresselhaus, M.S. Raman spectroscopy in graphene. Phys. Rep. 2009, 473, 51. [Google Scholar] [CrossRef]
- Some, S.; Kim, Y.; Yoon, Y.; Yoo, H.; Park, Y.; Lee, H. High-Quality Reduced Graphene Oxide by a Dual-Function Chemical Reduction and Healing Process. Sci. Rep. 2013, 3, 1929. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, M.; Nan, J.; Chen, T. Effect of particle size distribution on turbidity under various water quality levels during flocculation processes. Desalination 2014, 354, 116. [Google Scholar] [CrossRef]
- Bhattacharjee, S. DLS and zeta potential—What they are and what they are not? J. Control. Release 2016, 235, 337. [Google Scholar] [CrossRef]
- Fan, K.; Chen, X.; Wang, X.; Liu, X.; Liu, Y.; Lai, W.; Liu, X. Toward Excellent Tribological Performance as Oil-Based Lubricant Additive: Particular Tribological Behavior of Fluorinated Graphene. ACS Appl. Mater. Interf. 2018, 10, 28828. [Google Scholar] [CrossRef]
- Demirtas, S.; Kaleli, H.; Khadem, M.; Kim, D.-E. Characterization of the friction and wear effects of graphene nanoparticles in oil on the ring/cylinder liner of internal combustion engine. Ind. Lub. Tribol. 2019, 71, 642. [Google Scholar] [CrossRef]
- Wong, V.W.; Tung, S.C. Overview of automotive engine friction and reduction trends—Effects of surface, material, and lubricant-additive technologies. Friction 2016, 4, 1. [Google Scholar] [CrossRef] [Green Version]




| Properties | |
|---|---|
| Total Base Number (TBN) | 11.28 mg KOH/g |
| Flash point | 217 °C |
| Viscosity @100 °C | 13.59 cSt |
| Viscosity @40 °C | 84.27 cSt |
| Sample | Oil (ml) | Oil (g) | rGO (mg) | rGO (wt %) |
|---|---|---|---|---|
| R | 20 | 10 | 0 | 0 |
| 1 | 20 | 10 | 20 | 0.2 |
| 2 | 20 | 10 | 10 | 0.1 |
| 3 | 20 | 10 | 8 | 0.08 |
| 4 | 20 | 10 | 6 | 0.06 |
| 5 | 20 | 10 | 4 | 0.04 |
| 6 | 20 | 10 | 2 | 0.02 |
| 7 | 20 | 10 | 1 | 0.01 |
| Spectrum No | Sample Name | C | O | Al | Si | P | Ca | Mn | Fe | Zn | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 64 | 5W-40 ball | 5.49 | 0.86 | 0 | 0.25 | 0.1 | 0.01 | 0.49 | 92.44 | 0.36 | 100 |
| 65 | 6.06 | 4.69 | 0.09 | 0.21 | 0.72 | 1.36 | 0.51 | 84.03 | 2.33 | 100 | |
| 66 | 5.94 | 2.95 | 0.04 | 0.25 | 0.46 | 0.81 | 0.59 | 87.66 | 1.29 | 100 | |
| 73 | rGO6 ball | 8.94 | 7.54 | 0.07 | 0.18 | 1.28 | 1.81 | 0.39 | 76.02 | 3.78 | 100 |
| 74 | 11.16 | 5.52 | 0.13 | 0.28 | 0.43 | 1.65 | 0.54 | 79.16 | 1.13 | 100 | |
| 75 | 7.61 | 4.73 | 0.02 | 0.23 | 0.42 | 1.53 | 0.42 | 83.66 | 1.38 | 100 | |
| 124 | 5W-40 block | 6.44 | 3.76 | 0.08 | 0.34 | 0.55 | 0.92 | 1.34 | 83.47 | 3.1 | 100 |
| 127 | 7.69 | 3.67 | 0.07 | 0.37 | 0.42 | 0.86 | 1.82 | 82.01 | 3.09 | 100 | |
| 128 | 7.72 | 3.75 | 0.08 | 0.36 | 0.51 | 0.82 | 2.04 | 81.69 | 3.02 | 100 | |
| 130 | rGO6 block | 9.85 | 3.73 | 0.14 | 0.31 | 0.3 | 0.86 | 1.76 | 81.58 | 1.47 | 100 |
| 131 | 11 | 3.7 | 0.11 | 0.28 | 0.22 | 0.75 | 1.75 | 80.47 | 1.72 | 100 | |
| 132 | 11.53 | 3.74 | 0.07 | 0.27 | 0.24 | 0.89 | 2.13 | 79.76 | 1.37 | 100 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaleli, H.; Demirtaş, S.; Uysal, V.; Karnis, I.; Stylianakis, M.M.; Anastasiadis, S.H.; Kim, D.-E. Tribological Performance Investigation of a Commercial Engine Oil Incorporating Reduced Graphene Oxide as Additive. Nanomaterials 2021, 11, 386. https://doi.org/10.3390/nano11020386
Kaleli H, Demirtaş S, Uysal V, Karnis I, Stylianakis MM, Anastasiadis SH, Kim D-E. Tribological Performance Investigation of a Commercial Engine Oil Incorporating Reduced Graphene Oxide as Additive. Nanomaterials. 2021; 11(2):386. https://doi.org/10.3390/nano11020386
Chicago/Turabian StyleKaleli, Hakan, Selman Demirtaş, Veli Uysal, Ioannis Karnis, Minas M. Stylianakis, Spiros H. Anastasiadis, and Dae-Eun Kim. 2021. "Tribological Performance Investigation of a Commercial Engine Oil Incorporating Reduced Graphene Oxide as Additive" Nanomaterials 11, no. 2: 386. https://doi.org/10.3390/nano11020386
APA StyleKaleli, H., Demirtaş, S., Uysal, V., Karnis, I., Stylianakis, M. M., Anastasiadis, S. H., & Kim, D.-E. (2021). Tribological Performance Investigation of a Commercial Engine Oil Incorporating Reduced Graphene Oxide as Additive. Nanomaterials, 11(2), 386. https://doi.org/10.3390/nano11020386











