Polyethylene Glycol Coated Magnetic Nanoparticles: Hybrid Nanofluid Formulation, Properties and Drug Delivery Prospects
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
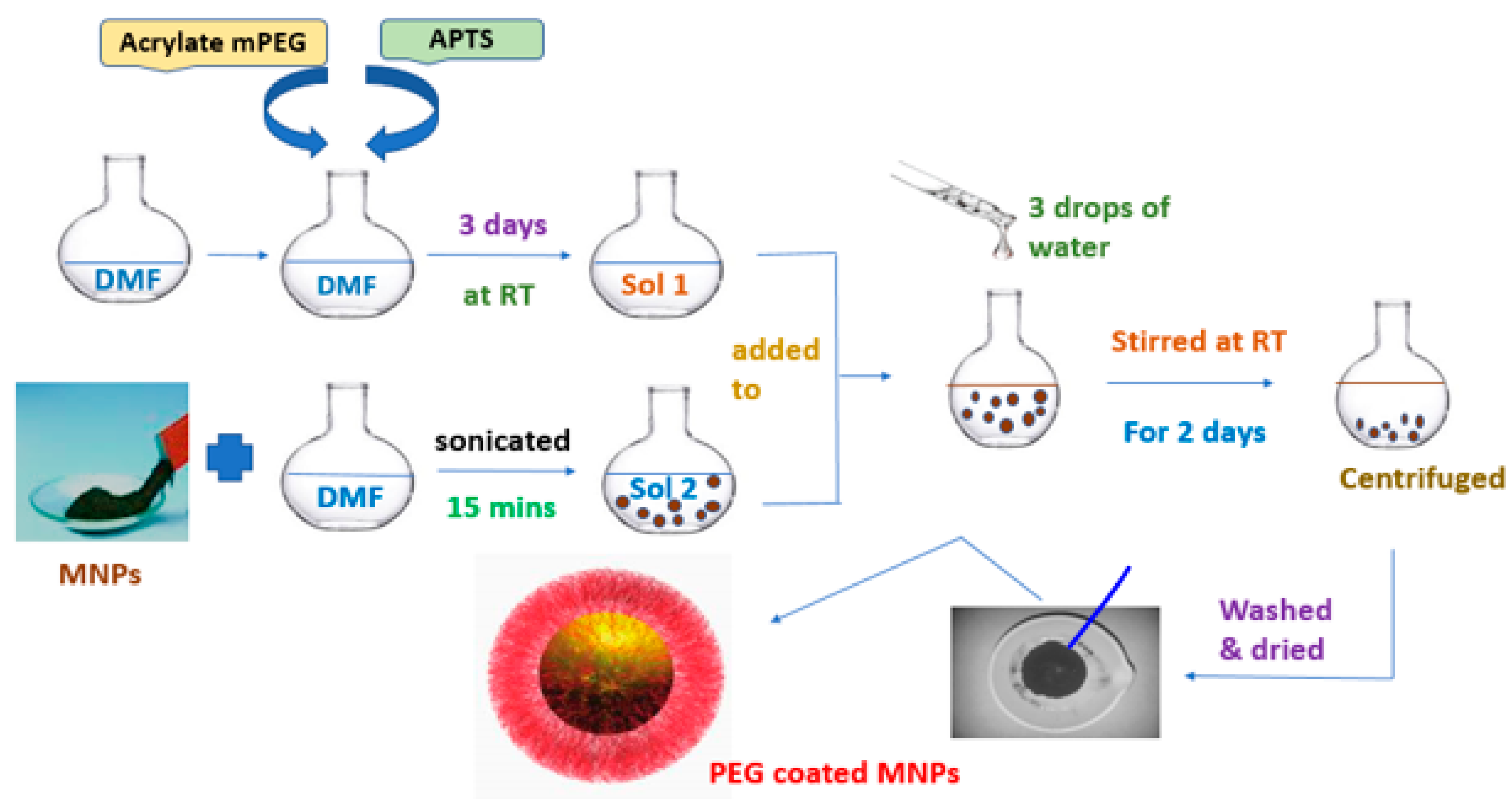
2.2. Modification of MNP with mPEG
2.3. MNF Preparation
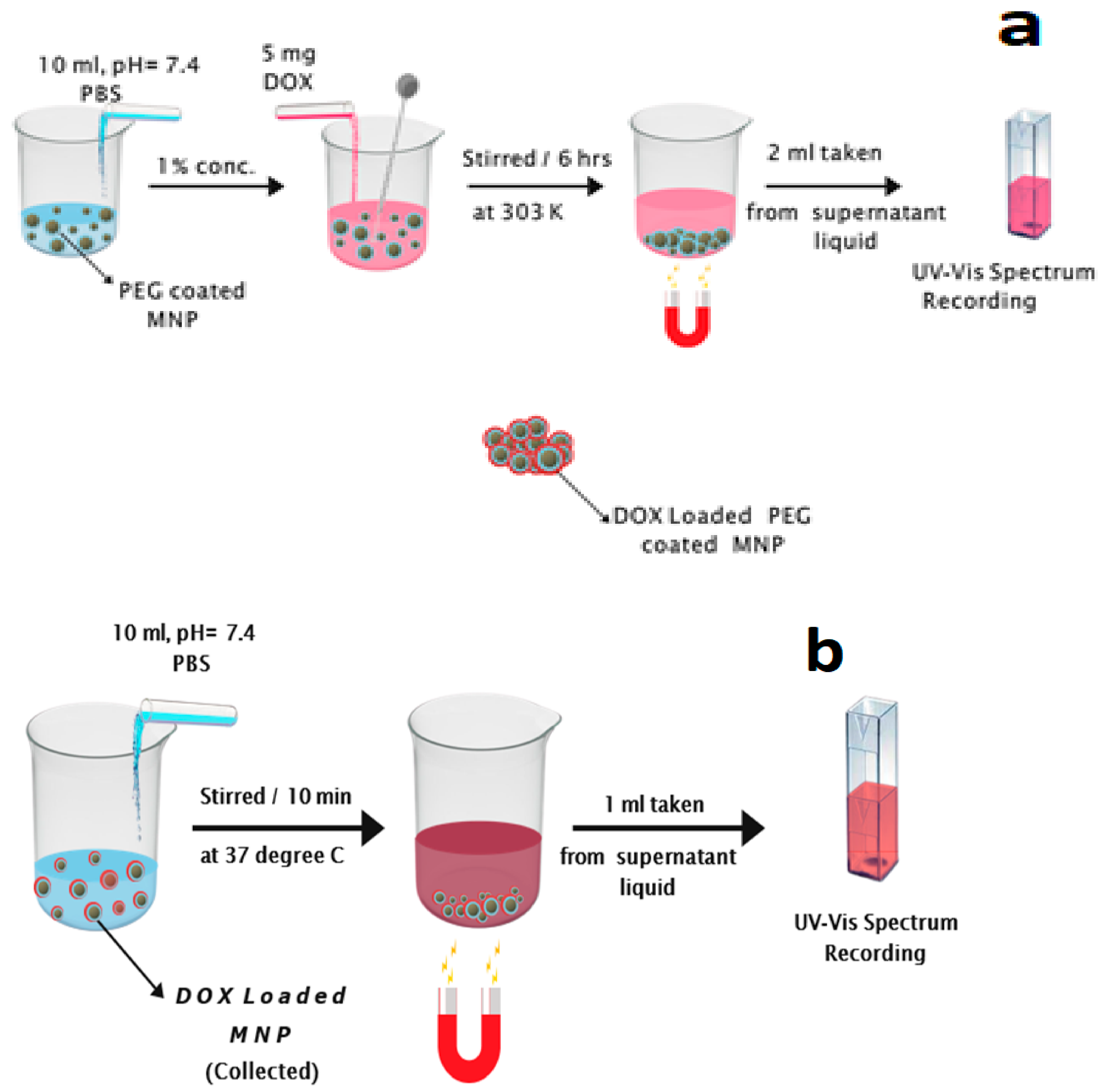
2.4. DOX Loading/Release in MNF with PBS as the Base Fluid
2.4.1. DOX Loading
2.4.2. DOX Release
2.5. Characterization
3. Results and Discussion
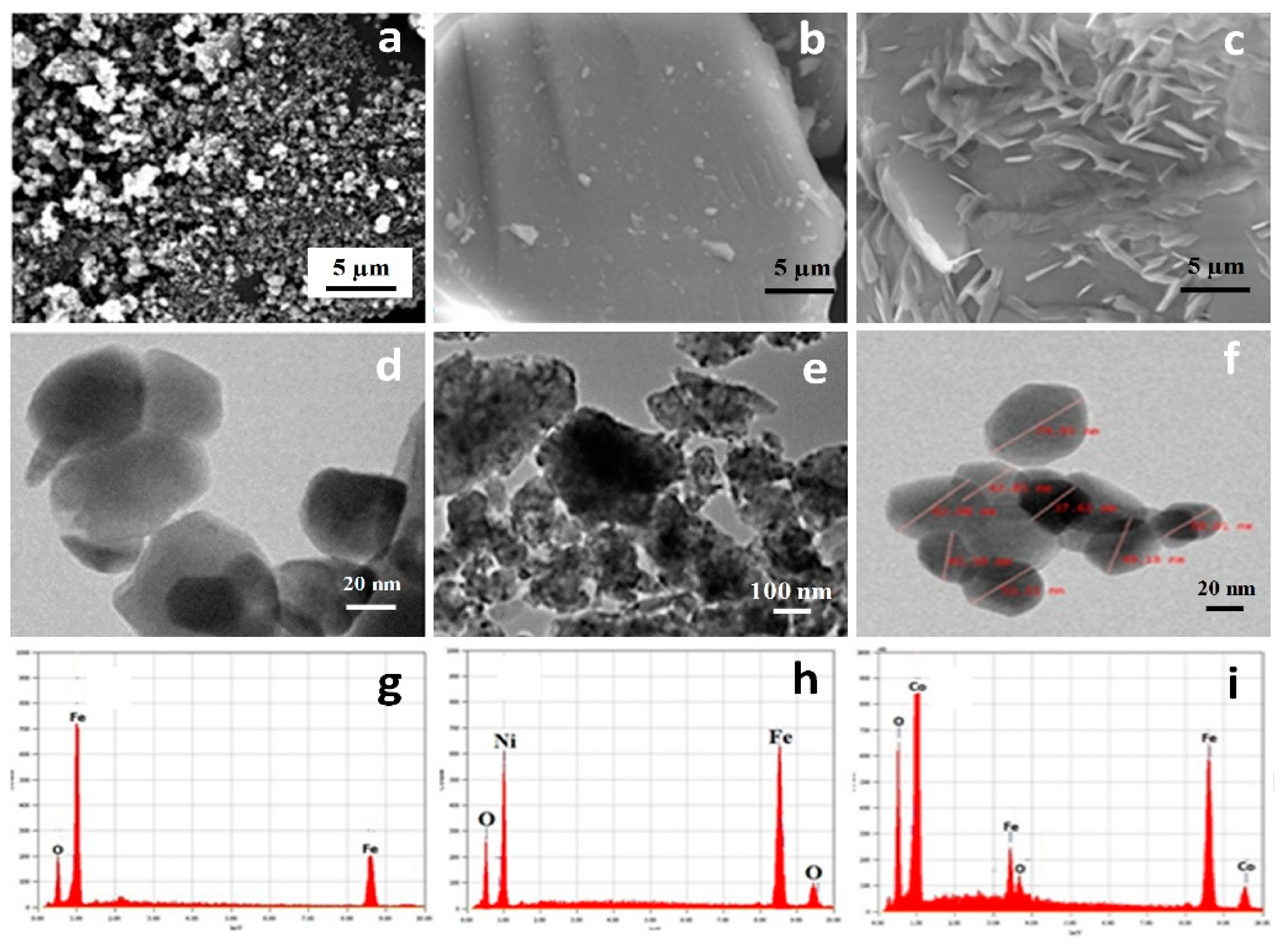
3.1. Particle Size and Morphology of MNPs
3.2. Phase Structure of Pristine MNPs and PEG-Coated MNPs
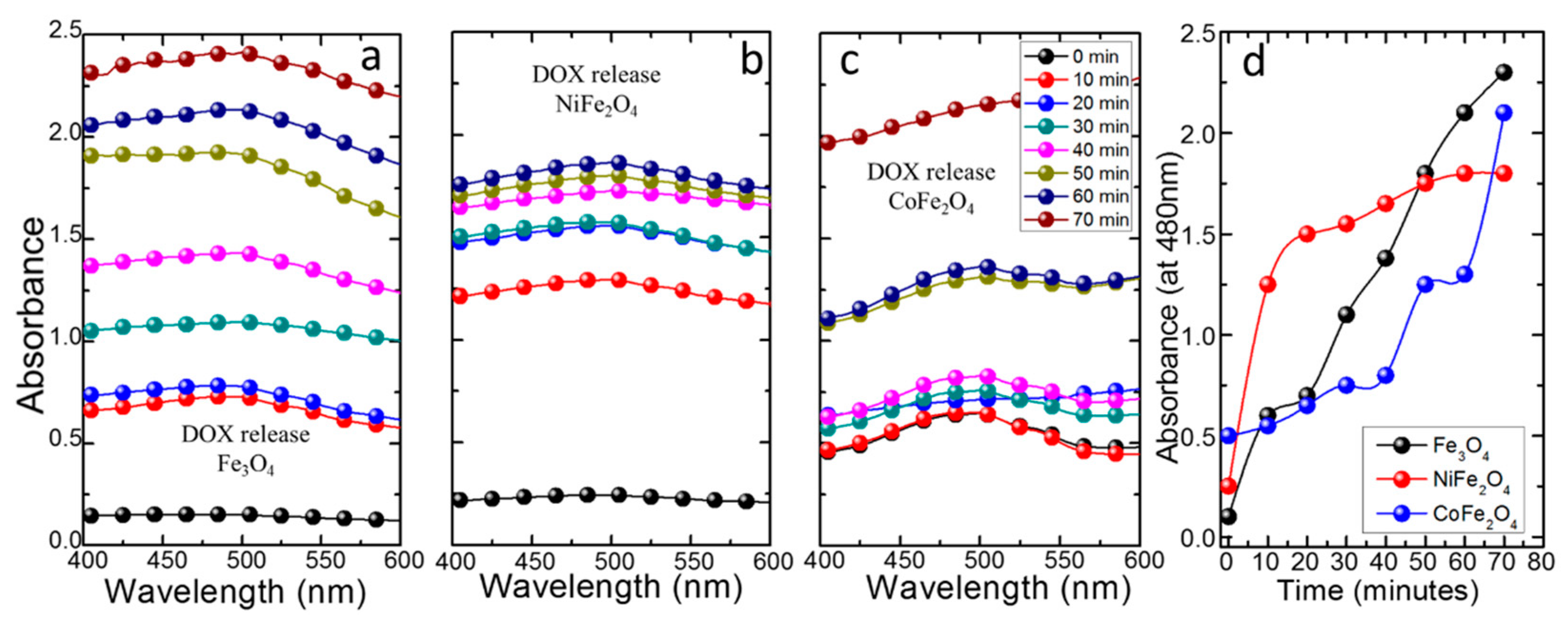
3.3. DOX Uptake/Release Characteristics of MNFs
3.3.1. DOX Loading
3.3.2. DOX Release Kinetics
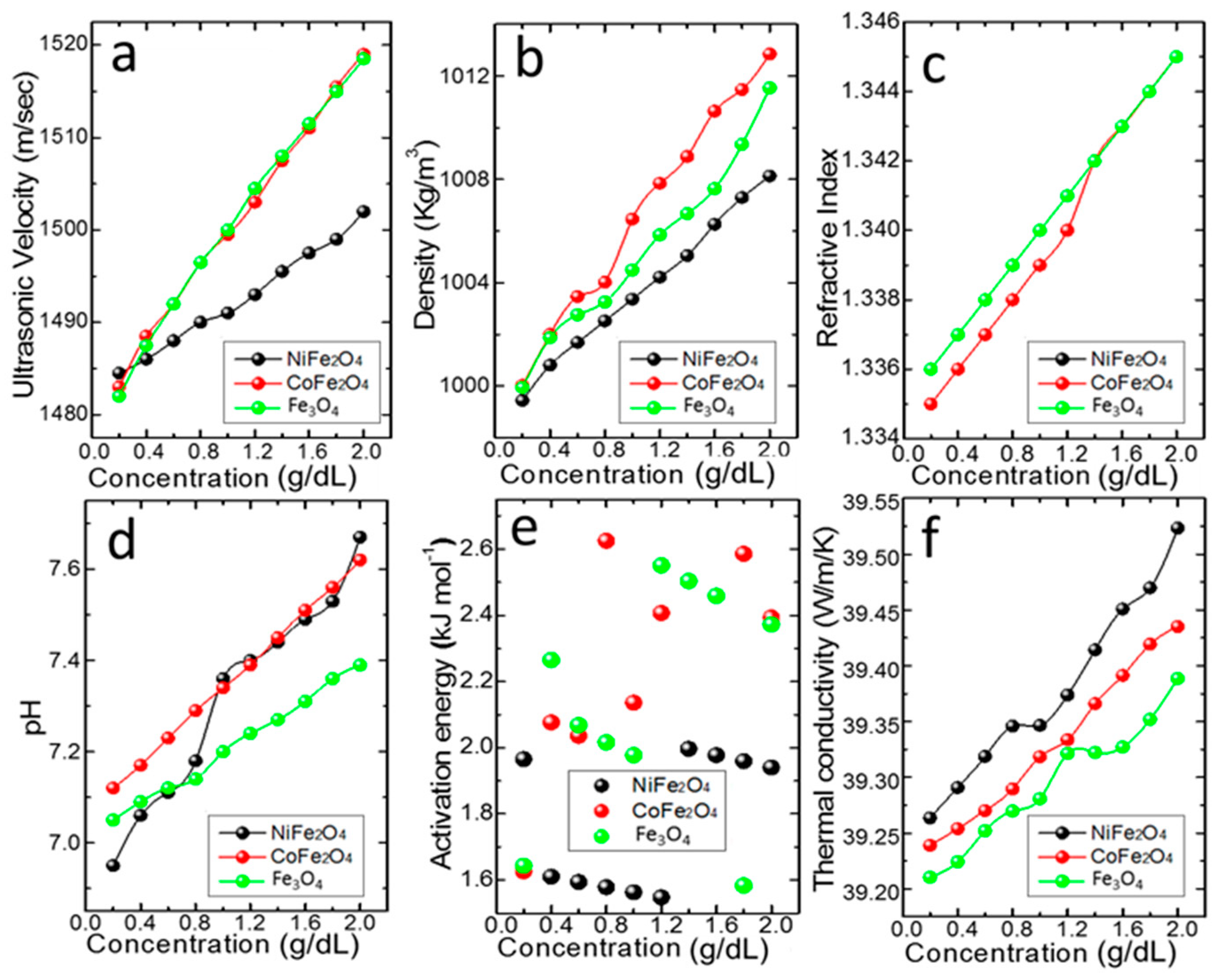
3.4. Thermo-Physical Properties
3.4.1. Effective Velocity and Density
3.4.2. Effective Viscosity and Activation Energy
3.4.3. Effective Refractive Index
3.4.4. Effective Thermal Conductivity
3.4.5. Effective Stability and pH
3.4.6. Effective Magnetic Susceptibility
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- McNamara, K.; TofailS, A.M. Nanosystems: The use of nanoalloys, metallic, bimetallic, and magnetic nanoparticles in biomedical applications. Phys. Chem. Chem. Phys. 2015, 17, 27981–27995. [Google Scholar] [CrossRef] [PubMed]
- Karimi, Z.; Karimi, L.; Shokrollahi, H. Nanomagnetic Particles Used in Biomedicine: Core and Coating Materials. Mater. Sci. Eng. C 2013, 33, 2465–2475. [Google Scholar] [CrossRef] [PubMed]
- McNamara, K.; Tofail, S.A.M. Nanoparticles in biomedical applications. Adv. Phys. X 2017, 2, 54–88. [Google Scholar] [CrossRef]
- Saianand, G.; Gopalan, A.-I.; Lee, J.-C.; Sathish, C.; Gopalakrishnan, K.; Unni, G.E.; Shanbag, D.; Venkata Dasireddy, D.B.C.; Yi, J.; Xie, S.; et al. Mixed Copper/Copper-Oxide Anchored Mesoporous Fullerene Nanohybrids as Superior Electrocatalysts toward Oxygen Reduction Reaction. Small 2020, 16, 1903937. [Google Scholar] [CrossRef] [PubMed]
- Benzigar, M.R.; Stalin, J.; Saianand, G.; Gopalan, A.-I.; Sarkar, S.; Srinivasan, S.; Park, D.-H.; Kim, S.; Talapaneni, S.N.; Ramadass, K.; et al. Highly ordered iron oxide-mesoporous fullerene nanocomposites for oxygen reduction reaction and supercapacitor applications. Microporous Mesoporous Mater. 2019, 285, 21–31. [Google Scholar] [CrossRef]
- Lee, J.-C.; Gopalan, A.-I.; Saianand, G.; Lee, K.-P.; Kim, W.-J. Manganese and Graphene Included Titanium Dioxide Composite Nanowires: Fabrication, Characterization and Enhanced Photocatalytic Activities. Nanomaterials 2020, 10, 456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sridara, T.; Upan, J.; Saianand, G.; Tuantranont, A.; Karuwan, C.; Jakmunee, J. Non-Enzymatic Amperometric Glucose Sensor Based on Carbon Nanodots and Copper Oxide Nanocomposites Electrode. Sensors 2020, 20, 808. [Google Scholar] [CrossRef] [Green Version]
- Rashmi, M.; Padmanaban, R.; Vaithinathan Karthikeyan, D.; Vellaisamy Roy, A.L.; Gopalan, A.-I.; Saianand, G.; Kim, W.-J.; Kannan, V. A Comparative Evaluation of Physicochemical Properties and Photocatalytic Efficiencies of Cerium Oxide and Copper Oxide Nanofluids. Catalysts 2020, 10, 34. [Google Scholar]
- Rümenapp, C.; Gleich, B.; Haase, A. Magnetic nanoparticles in magnetic resonance imaging and diagnostics. Pharm. Res. 2012, 29, 1165–1179. [Google Scholar] [CrossRef]
- Chaitali, D.; Arup, G.; Manisha, A.; Ajay, G.; Madhuri, M. Improvement of anticancer drug release by cobalt ferrite magnetic nanoparticles through combined pH and temperature responsive technique. ChemPhysChem Eur. J. Chem. Phys. Phys. Chem. 2018, 19, 1–18. [Google Scholar]
- Radoń, A.; Łoński, S.; Kądziołka-Gaweł, M.; Gębara, P.; Lis, M.; Łukowiec, D.; Babilas, R. Influence of magnetite nanoparticles surface dissolution, stabilization and functionalization by malonic acid on the catalytic activity, magnetic and electrical properties. Colloids Surf. A Physicochem. Eng. Asp. 2020, 607, 125446. [Google Scholar] [CrossRef]
- Radoń, A.; Kubacki, J.; Kądziołka-Gaweł, M.; Gębara, P.; Hawełek, Ł.; Topolska, S.; Łukowiec, D. Structure and magnetic properties of ultrafine superparamagnetic Sn-doped magnetite nanoparticles synthesized by glycol assisted co-precipitation method. J. Phys. Chem. Solids 2020, 145, 109530. [Google Scholar] [CrossRef]
- Gopalan, A.-I.; Komathi, S.; Muthuchamya, N.; Lee, K.-P.; Whitcombe, M.J.; Dhana, L.; Gopalan, S.-A. Functionalized conjugated polymers for sensing and molecular imprinting applications. Prog. Polym. Sci. 2019, 88, 1–129. [Google Scholar]
- Covaliu, C.I.; Jitaru, I.; Paraschiv, G.; Vasile, E.; Biris, S.-S.; Diamandescu, L.; Ionita, V.; Iovu, H. Core–shell hybrid nanomaterials based on CoFe2O4 particles coated with PVP or PEG biopolymers for applications in biomedicine. Powder Technol. 2013, 237, 415–426. [Google Scholar] [CrossRef]
- Lee, S.-W.; Choi, K.-J.; Kang, B.-H.; Lee, J.-S.; Kim, S.-W.; Kwon, J.-B.; Gopalan, S.-A.; Bae, J.-H.; Kim, E.-S.; Kwon, D.-H.; et al. Low dark current and improved detectivity of hybrid ultraviolet photodetector based on carbon-quantum-dots/zinc-oxide-nanorod composites. Org. Electron. 2016, 39, 250–257. [Google Scholar] [CrossRef]
- Alam, S.R.; Shah, A.S.; Richards, J.; Lang, N.N.; Barnes, G.; Joshi, N.; MacGillivray, T.; McKillop, G.; Mirsadraee, S.; Payne, J.; et al. Ultrasmall superparamagnetic particles of iron oxide in patients with acute myocardial infarction: Early clinical experience. Circ. Cardiovasc. Imaging 2012, 5, 559–565. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Topel, S.D.; Topel, O.; Bostancıoglu, R.B.; Koparal, A.T. Synthesis and characterization of Bodipy functionalized magnetic iron oxide nanoparticles for potential bioimaging applications. Colloid Surf. B Biointerfaces 2015, 128, 245–253. [Google Scholar] [CrossRef]
- Barrow, M.; Taylor, A.; Murray, P.; Rosseinsky, M.J.; Adams, D.J. Design considerations for the synthesis of polymer coated iron oxide nanoparticles for stem cell labelling and tracking using MRI. Chem. Soc. Rev. 2015, 44, 6733–6748. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barick, K.C.; Singh, S.; Bahadur, D.; Lawande, M.A.; Patkar, D.P.; Hassan, P.A. Stimuli Responsive Polymeric Nanocarriers for Drug Delivery Applications, 1st ed.; Woodhead Publishing: Cambridge, UK, 2018; Volume 2, pp. 1–647. [Google Scholar]
- Ebrahim, S.A.; Ashtari, A.; Pedram, M.Z.; Ebrahim, N.A. Publication Trends in Drug Delivery and Magnetic Nanoparticles. Nanoscale Res. Lett. 2019, 14, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jia, Y.; Yuan, M.; Yuan, H.; Huang, X.; Sui, X.; Cui, X.; Tang, F.; Peng, J.; Chen, J.; Lu, S.; et al. Co-encapsulation of magnetic Fe3O4 nanoparticles and doxorubicin into biodegradable PLGA nanocarriers for intratumoral drug delivery. Int. J. Nanomed. 2012, 7, 1697–1708. [Google Scholar]
- Tatyana, I.S.; Olga, I.V.; Vladimir, P.S.; Mikhail, Y.M. Magnetic Nanoparticles for Biomedical Purposes: Modern Trends and Prospects. Magnetochemistry 2020, 6, 30. [Google Scholar]
- Gopalan, S.-A.; Sivanesan, A.; Benzigar, M.R.; Singh, G.; Gopalan, A.-I.; Baskar, A.V.; Ilbeygi, H.; Ramadass, K.; Kambala, V.; Vinu, A. Recent Progress on the Sensing of Pathogenic Bacteria Using Advanced Nanostructures. Bull. Chem. Soc. Jpn. 2019, 92, 216–244. [Google Scholar]
- Warner, C.L.; Chouyyok, W.; Mackie, K.E.; Neiner, D.; Saraf, L.V.; Droubay, T.C.; Warner, M.G.; Addleman, S. Manganese Doping of Magnetic Iron Oxide Nanoparticles: Tailoring Surface Reactivity for a Regenerable Heavy Metal Sorbent. Langmuir 2012, 28, 3931–3937. [Google Scholar] [CrossRef]
- Kharat, P.B.; Shisode, M.V.; Birajdar, S.D.; Bhoyar, D.N.; Jadhav, K.M. Synthesis and Characterization of Water Based NiFe2O4 Ferrofluid. AIP Conf. Proc. 2017, 1832, 1–3. [Google Scholar]
- Coskun, M.; Citoglu, S.; Korkmaz, M.; Firat, T. The Magnetic Anisotropy Effectiveness on NiFe2O4 and NiFe2O4@SiO2 Nanoparticles for Hyperthermia Applications. Cumhur. Sci. J. 2017, 38, 193–205. [Google Scholar]
- Wong, K.V.; De Leon, O. Review Article Applications of Nanofluids: Current and Future. Adv. Mech. Eng. 2010, 2010, 1–11. [Google Scholar]
- Bica, D.; Vekas, L.; Avdeev, M.V. Sterically stabilized water based magnetic fluids: Synthesis, structure and properties. J. Magn. Magn. Mater. 2007, 311, 17–21. [Google Scholar] [CrossRef]
- Gopalan, S.-A.; Gopalan, A.-I.; Vinu, A.; Lee, K.-P.; Kang, S.-W. A new optical-electrical integrated buffer layer design based on gold nanoparticles tethered thiol containing sulfonated polyaniline towards enhancement of solar cell performance. Sol. Energy Mater. Sol. Cells 2018, 174, 112–123. [Google Scholar] [CrossRef]
- Wang, G.; Ma, Y.; Mu, J.; Zhang, Z.; Zhang, X.; Zhang, L.; Zhang, H.; Che, Y.; Bai, J.H.; Xie, H. Monodisperse polyvinylpyrrolidone-coated CoFe2O4 nanoparticles: Synthesis, characterization and cytotoxicity study. Appl. Surf. Sci. 2016, 365, 114–119. [Google Scholar] [CrossRef]
- Mirzaee, S.; Shayesteh, S.F. Ultrasound induced strain in ultrasmall CoFe2O4@polyvinyl alcohol nanocomposites. Ultrason. Sonochem. 2018, 40, 583–586. [Google Scholar] [CrossRef]
- Gopalan, S.-A.; Gopalan, A.-I.; Kang, S.-W.; Lee, K.-P. Fabrication of Gold Nanoflower Anchored Conducting Polymer Hybrid Film Electrode by Pulse Potentiostatic Deposition. IEEE Electron Device Lett. 2013, 34, 1065–1067. [Google Scholar]
- Gopalan, S.-A.; Gopalan, A.-I.; Kang, S.-W.; Komathi, S.; Lee, K.-P. One Pot Synthesis of New Gold Nanoparticles Dispersed Poly (2-aminophenyl boronic acid) Composites for Fabricating an Affinity Based Electrochemical Detection of Glucose. Sci. Adv. Mater. 2014, 6, 1356–1364. [Google Scholar]
- Gopalan, S.-A.; Franklin, P.M.; Lee, K.-P.; Kang, S.-W.; Gopalan, A.-I. Facile Electrodeposition of Flower Like Gold Nanostructures on a Conducting Polymer Support. J. Nanosci. Nanotechnol. 2014, 14, 3256–3261. [Google Scholar]
- Gopalan, S.-A.; Baoyin, H.; Kang, B.-H.; Kim, S.-W.; Lee, S.-W.; Lee, J.-S.; Jeong, H.-M.; Kang, S.-W. Incorporation of Gold Nanodots Into Poly(3,4-ethylenedioxythiophene): Poly(styrene sulfonate) for an Efficient Anode Interfacial Layer for Improved Plasmonic Organic Photovoltaics. J. Nanosci. Nanotechnol. 2015, 15, 7092–7098. [Google Scholar]
- Chiang, P.C.; Hung, D.S.; Wang, J.W.; Ho, C.S.; Yao, Y.D. Engineering water-dispersible FePt nanoparticles for biomedical applications. IEEE Trans. Magn. 2007, 43, 2445–2447. [Google Scholar] [CrossRef]
- Vekas, L.; Bica, D.; Avdeev, M.V. Magnetic nanoparticles and concentrated magnetic nanofluids: Synthesis, properties and some applications. China Particuol. 2007, 5, 43–49. [Google Scholar] [CrossRef]
- Yadav, N.; Jaiswal, A.K.; Singh, S.; Singh, D.K.; Yadav, R.R. Acoustical Investigations in NiFe2O4 Nanofluid. Int. J. Sci. Res. 2013, 193–194. [Google Scholar]
- Kesavamoorthi, R.; Vigneshwaran, A.N.; Sanyal, V.; Raja, R.C. Synthesis and characterization of nickel ferrite nanoparticles by sol-gel auto combustion method. J. Chem. Pharm. Sci. 2016, 9, 160–162. [Google Scholar]
- Udhaya, A.P.; Meena, M. Albumen Assisted Green Synthesis of NiFe2O4 Nanoparticles and Their Physico-Chemical Properties. Mater. Today Proc. 2019, 9, 528–534. [Google Scholar] [CrossRef]
- Gingas, D.; Mindru, I.; Patron, L.; Calderon-Moreno, J.M.; Mocioiu, O.C.; Preda, S.; Stanica, N.; Nita, S.; Dobre, N.; Popa, M.; et al. Green Synthesis Methods of CoFe2O4 and Ag-CoFe2O4 Nanoparticles Using Hibiscus Extracts and Their Antimicrobial Potential. J. Nanomater. 2016, 2016, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Pathak, A.; Pramanik, P. Nano-particles of oxides through chemical methods. Proc. Indian Natl. Sci. Acad. 2001, 67, 47–70. [Google Scholar]
- Jia, Z.; Ren, D.; Zhu, R. Synthesis, characterization and magnetic properties of CoFe2O4 nanorods. Mater. Lett. 2012, 66, 128–131. [Google Scholar] [CrossRef]
- Amiri, S.; Shokrollahi, H. The role of cobalt ferrite magnetic nanoparticles in medical science. Mater. Sci. Eng. C 2013, 33, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Chieh, J.-J.; Huang, K.-W.; Shi, J.-C. Sub-Tesla-Field magnetization of vibrated magnetic nanoreagents for screening tumor markers. Appl. Phys. Lett. 2015, 106, 1–4. [Google Scholar] [CrossRef]
- Onar, K.; Yakinci, M.E. Synthesis of Fe3O4 nanoparticles for biomedical applications. J. Phys. Conf. Ser. 2015, 667, 1–5. [Google Scholar]
- Wilson, M.W.; Kerlan, R.K.; Fidelman, N.A.; Venook, A.P.; LaBerge, J.M.; Koda, J.; Gordon, R.L. Hepatocellular carcinoma: Regional therapy with a magnetic targeted carrier bound to doxorubicin in a dual MR imaging/conventional angiography suite-initial experience with four patients. Radiology 2004, 230, 287–293. [Google Scholar] [CrossRef]
- Carvalho, S.M.; Leone, A.G.; Mansur, A.P.; Carvalho, I.C.; Krambrockb, K.; Mansur, H.S. Bifunctional Magnetopolymersomes of Iron Oxide Nanoparticles and Carboxymethylcellulose Conjugated with Doxorubicin for Hyperthermo-Chemotherapy of Brain Cancer Cells. Biomater. Sci. 2019, 7, 2102–2122. [Google Scholar] [CrossRef]
- Meerod, S.; Tumcharern, G.; Wichai, U.; Rutnakornpituk, M. Magnetite nanoparticles stabilized with polymeric bilayer of poly (ethylene glycol) methyl ether–poly(ɛ-caprolactone) copolymers. Polymer 2008, 49, 3950–3956. [Google Scholar] [CrossRef]
- Ramimoghadam, D.; Bagheri, S.; Abd Hamid, S.B. In-situ precipitation of ultra-stable nano-magnetite slurry. J. Magn. Magn. Mater. 2015, 379, 74–79. [Google Scholar] [CrossRef]
- Tai, M.F.; Lai, C.W.; Abdul Hamid, S.B. Facile Synthesis Polyethylene Glycol Coated Magnetite Nanoparticles for High Colloidal Stability. J. Nanomater. 2016, 2016, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Higuchi, T. Rate of release of medicaments from ointment bases containing drugs in suspension. J. Pharm. Sci. 1961, 50, 874–875. [Google Scholar] [CrossRef]
- Korsmeyer, R.W.; Peppas, N.A. Solute and penetrant diffusion in swellable polymers. III. Drug release from glassy poly(HEMA-co-NVP) copolymers. J. Control. Release 1983, 1, 89–98. [Google Scholar] [CrossRef]
- Singhvi, G.; Singh, M. Review: In-Vitro Drug Release Characterization Models. Int. J. Pharm. Stud. Res. 2011, 2, 77–84. [Google Scholar]
- Kalam, A.M.; Humayun, M.; Parvez, N.; Yadav, S.; Garg, A.; Amin, S.; Sultana, Y.; Ali, A. Release Kinetics of Modified Pharmaceutical Dosage Forms: A Review. Cont. J. Pharm. Sci. 2007, 1, 30–35. [Google Scholar]
- Gupta, S.; Kaur, H. Prediction of in vitro Drug Release Mechanisms from Extended Release Matrix Tablets using SSR/R2 Technique. Trends Appl. Sci. Res. 2011, 6, 400–409. [Google Scholar]
- Keum, C.G.; Noh, Y.W.; Baek, J.S.; Lim, J.H.; Hwang, C.J.; Na, Y.G.; Shin, S.C.; Cho, C.W. Practical preparation procedures for docetaxel-loaded nanoparticles using polylactic acid-co-glycolic acid. Int. J. Nanomed. 2011, 6, 2225–2234. [Google Scholar]
- Paarakh, M.P.; Preethy, A.J.; Setty, C.; Christoper, P.G.V. Release Kinetics—Concepts and Applications. Int. J. Pharm. Res. Technol. 2018, 8, 1–9. [Google Scholar]
- Omidirad, R.; Hosseinpour, R.F.; Vasheghani, F.B. Preparation and in vitro drug delivery response of doxorubicin loaded PAA coated magnetite nanoparticles. J. Serb. Chem. Soc. 2013, 78, 1609–1616. [Google Scholar] [CrossRef]
- Mhlanga, N.; Suprakas Sinha, R. Kinetic models for the release of the anticancer drug doxorubicin from biodegradable polylactide/metal oxide-based hybrids. Int. J. Biol. Macromol. 2015, 72, 1301–1307. [Google Scholar] [CrossRef] [PubMed]
- Bajpai, A.K.; Shukla, S.; Bhanu, S.; Kankane, S. Responsive Polymers in Controlled Drug Delivery. Prog. Polym. Sci. 2008, 33, 1088–1118. [Google Scholar] [CrossRef]
- Prokopowicz, M. Formulation, characterisation and in vitro studies of doxorubicin-loaded silica–polydimethylsiloxane granules. Eur. J. Pharm. Sci. 2015, 66, 10–19. [Google Scholar] [CrossRef]
- Nam, J.H.; Kim, S.; Seong, H. Investigation on Physicochemical Characteristics of a Nanoliposome-Based System for Dual Drug Delivery. Nanoscale Res. Lett. 2018, 13, 101. [Google Scholar] [CrossRef]
- Kharat, P.B.; More, S.D.; Somvanshi, S.B. Exploration of thermoacoustics behavior of water-based nickel ferrite nanofluids by ultrasonic velocity method. J. Mater. Sci. Mater. Electron. 2019, 30, 6564–6574. [Google Scholar] [CrossRef]
- Kiruba, R.; Gopalakrishnan, M.; Mahalingam, T.; Kingson Solomon, J.A. The Effect of Temperature on the Ultrasonic Properties of Carbon Nanotubes Incorporated in Zinc Oxide Nanofluids. J. Nanofluids 2013, 2, 50–54. [Google Scholar] [CrossRef]
- Vekas, L. Magnetic Nanofluids Properties and Some Applications. Rom. J. Phys. 2004, 49, 707–721. [Google Scholar]
- Many, E.; Sahin, B.; Mandev, E.; Ates, I.; Yetim, T. Determination of Thermo physical properties of water based Magnetic Nanofluids. Int. J. Chem. Mol. Eng. 2016, 10, 1089–1095. [Google Scholar]
- Malekzadeh, A.; Pouranfard, A.R.; Hatami, N.; Kazemnejad Banari, A.; Rahimi, M.R. Experimental Investigations on the Viscosity of Magnetic Nanofluids under the Influence of Temperature, Volume Fractions of Nanoparticles and External Magnetic Field. J. Appl. Fluid Mech. 2016, 9, 693–697. [Google Scholar] [CrossRef]
- Moore, W.R.; Brown, A.M. Viscosity-temperature relationships for dilute solutions of cellulose derivatives: Temperature dependence of solution viscosities of ethyl cellulose. J. Colloid Sci. 1959, 14, 1–12. [Google Scholar] [CrossRef]
- Ijaz Khan, M.; Tehreem, N.; Hayat, T.; Niaz Khan, B.; Alsaedi, A. Binary chemical reaction with activation energy in rotating flow subject to nonlinear heat flux and heat source/sink. J. Comput. Des. Eng. 2020, 7, 279–286. [Google Scholar] [CrossRef]
- Cao, Q.; Zhang, Z.; Yu, J.; Cui, H.; He, X.; Li, D. Preparation and Characterization of a New Low Refractive Index Ferrofluid. Materials 2019, 12, 1658. [Google Scholar] [CrossRef] [Green Version]
- Altan, C.L.; Elkatmis, A.; Yuksel, M.; Aslan, N.; Bucak, S. Enhancement of thermal conductivity upon application of magnetic field to Fe3O4 nanofluids. J. Appl. Phys. 2011, 110, 1–8. [Google Scholar] [CrossRef]
- Ashok Singh, K.; Vijay Raykar, S. Experimental Investigation of Thermal Conductivity and Effusivity of Ferrite Based Nanofluids under Magnetic Field. ISRN Nanotechnol. 2013, 2013, 1–5. [Google Scholar] [CrossRef]
- Evans, W. Role of Brownian motion hydrodynamics on nanofluid thermal conductivity. Appl. Phys. Lett. 2006, 88. [Google Scholar] [CrossRef] [Green Version]
- Patel, H.E.; Sundararajan, T.; Das, S.K. An experimental investigation into the thermal conductivity enhancement in oxide and metallic nanofluids. J. Nanoparticle Res. 2010, 12, 1015–1031. [Google Scholar] [CrossRef]
- Ravi Babu, S.; Ramesh Babu, P.; Rambabu, V. Effects of Some Parameters on Thermal Conductivity of Nanofluids and Mechanisms of Heat Transfer Improvement. Int. J. Eng. Res. Appl. 2013, 3, 2136–2140. [Google Scholar]
- Prashant Kharat, B.; Swapnil Jadhav, A.; Suraj Deshmukh, S.; Keche, A.P.; More, S.D.; Sarnaik, M.N.; Jadhav, K.M. Evaluation of thermal conductivity of the NiFe2O4 ferrofluids under influence of magnetic field. AIP Conf. Proc. 2019, 2115, 1. [Google Scholar]
- Karimi, A.; Afghahi, S.S.S.; Shariatmadar, H.; Ashjaee, M. Experimental investigation on thermal conductivity of MFe2O4 (M = Fe and Co) magnetic nanofluids under influence of magnetic field. Thermochim. Acta 2014, 598, 59–67. [Google Scholar] [CrossRef]
- Wei, Y.; Xie, H. A Review on Nanofluids: Preparation, Stability Mechanisms, and Applications. J. Nanomater. 2012, 2012, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Wang, X.-J.; Li, X.; Yang, S. Influence of pH and SDBS on the Stability and Thermal Conductivity of Nanofluids. Energy Fuels 2009, 23, 2684–2689. [Google Scholar] [CrossRef]
- Sharifi, I.; Shokrollahi, H.; Amiri, S. Ferrite-based magnetic nanofluids used in hyperthermia applications. J. Magn. Magn. Mater. 2012, 324, 903–915. [Google Scholar] [CrossRef]
- Roszko, A.; Fornalik-Wajs, E. Evaluation of diamagnetic nanofluid ability to heat transfer in the strong magnetic field. J. Phys. Conf. Ser. 2016, 745, 1–10. [Google Scholar] [CrossRef]


| Drug Released | Kinetic Models with Derived Parameter | ||||
|---|---|---|---|---|---|
| Medium | Parameters | Zero-Order | First-Order | Higuchi Model | Korsmeyer–Peppa’s Model |
| Polyethylene glycol (PEG) Coated | R2 | 0.9719 | 0.9718 | 0.9735 | 0.9839 |
| CoFe2O4 | Coefficient | 0.9858 | −0.9858 | 0.9867 | 0.9919 |
| Slope, n | 1.2239 | −1.2239 | 10.9105 | 0.5475 | |
| Intercept, log K | - | - | - | 0.9723 | |
| K | - | - | - | 9.3834 | |
| PEG Coated | R2 | 0.9623 | 0.9623 | 0.806 | 0.9727 |
| Fe3O4 | Coefficient | 0.9809 | −0.9809 | 0.8978 | 0.9862 |
| Slope, n | 1.3575 | −1.3575 | 11.0658 | 1.1015 | |
| Intercept, log K | - | - | - | −0.0987 | |
| K | - | - | - | 0.7966 | |
| PEG Coated | R2 | 0.9897 | 0.9897 | 0.896 | 0.9688 |
| NiFe2O4 | Coefficient | 0.9948 | −0.9948 | 0.9465 | 0.9843 |
| Slope, n | 1.3342 | −1.3342 | 11.0431 | 0.8249 | |
| Intercept, | - | - | - | 0.4275 | |
| log K | |||||
| K | - | - | - | 2.6765 | |
| Concentration (%) | Relative Viscosity (cP) | |||||
|---|---|---|---|---|---|---|
| 303 K | 308 K | |||||
| NiFe2O4 | CoFe2O4 | Fe3O4 | NiFe2O4 | CoFe2O4 | Fe3O4 | |
| 0.2 | 0.83 | 1 | 1 | 0.82 | 0.99 | 0.99 |
| 0.4 | 1.01 | 1.02 | 1.02 | 1 | 1.01 | 1.01 |
| 0.6 | 1.02 | 1.04 | 1.04 | 1.01 | 1.03 | 1.03 |
| 0.8 | 1.03 | 1.06 | 1.06 | 1.02 | 1.05 | 1.05 |
| 1 | 1.04 | 1.07 | 1.07 | 1.03 | 1.06 | 1.06 |
| 1.2 | 1.05 | 1.09 | 1.09 | 1.04 | 1.07 | 1.07 |
| 1.4 | 1.06 | 1.11 | 1.11 | 1.05 | 1.1 | 1.1 |
| 1.6 | 1.07 | 1.13 | 1.13 | 1.06 | 1.12 | 1.12 |
| 1.8 | 1.08 | 1.14 | 1.14 | 1.07 | 1.13 | 1.13 |
| 2 | 1.09 | 1.16 | 1.16 | 1.08 | 1.15 | 1.15 |
| Concentration (%) | Relative Viscosity (cP) | |||||
|---|---|---|---|---|---|---|
| 313 K | 318 K | |||||
| NiFe2O4 | CoFe2O4 | Fe3O4 | NiFe2O4 | CoFe2O4 | Fe3O4 | |
| 0.2 | 0.81 | 0.98 | 0.98 | 0.8 | 0.97 | 0.97 |
| 0.4 | 0.99 | 1 | 1 | 0.98 | 0.98 | 0.98 |
| 0.6 | 1 | 1.02 | 1.02 | 0.99 | 1 | 1 |
| 0.8 | 1.01 | 1.03 | 1.03 | 1 | 1.01 | 1.01 |
| 1 | 1.02 | 1.04 | 1.04 | 1.01 | 1.03 | 1.03 |
| 1.2 | 1.03 | 1.06 | 1.06 | 1.02 | 1.04 | 1.04 |
| 1.4 | 1.04 | 1.08 | 1.08 | 1.02 | 1.06 | 1.06 |
| 1.6 | 1.05 | 1.1 | 1.1 | 1.03 | 1.08 | 1.08 |
| 1.8 | 1.06 | 1.1 | 1.1 | 1.04 | 1.09 | 1.09 |
| 2 | 1.07 | 1.13 | 1.13 | 1.05 | 1.11 | 1.11 |
| Concentration (%) | Magnetic Susceptibility (Xm × 10−10) | ||
|---|---|---|---|
| NiFe2O4 | CoFe2O4 | Fe3O4 | |
| 0.2 | 1.2885 | 0.0558 | 0.9936 |
| 0.4 | 1.1293 | 0.1120 | 1.3034 |
| 0.6 | 1.4631 | 0.1619 | 1.6317 |
| 0.8 | 1.0682 | 0.0581 | 0.3617 |
| 1 | 1.8417 | 0.1299 | 13.174 |
| 1.2 | 0.1801 | 0.0622 | 0.3638 |
| 1.4 | 0.1345 | 0.067 | 0.6257 |
| 1.6 | 0.1553 | 0.0207 | 0.9722 |
| 1.8 | 0.1102 | 0.0105 | 0.6037 |
| 2 | 0.0259 | 0.2051 | 0.822 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mannu, R.; Karthikeyan, V.; Velu, N.; Arumugam, C.; Roy, V.A.L.; Gopalan, A.-I.; Saianand, G.; Sonar, P.; Lee, K.-P.; Kim, W.-J.; et al. Polyethylene Glycol Coated Magnetic Nanoparticles: Hybrid Nanofluid Formulation, Properties and Drug Delivery Prospects. Nanomaterials 2021, 11, 440. https://doi.org/10.3390/nano11020440
Mannu R, Karthikeyan V, Velu N, Arumugam C, Roy VAL, Gopalan A-I, Saianand G, Sonar P, Lee K-P, Kim W-J, et al. Polyethylene Glycol Coated Magnetic Nanoparticles: Hybrid Nanofluid Formulation, Properties and Drug Delivery Prospects. Nanomaterials. 2021; 11(2):440. https://doi.org/10.3390/nano11020440
Chicago/Turabian StyleMannu, Rashmi, Vaithinathan Karthikeyan, Nandakumar Velu, Chandravadhana Arumugam, Vellaisamy A. L. Roy, Anantha-Iyengar Gopalan, Gopalan Saianand, Prashant Sonar, Kwang-Pill Lee, Wha-Jung Kim, and et al. 2021. "Polyethylene Glycol Coated Magnetic Nanoparticles: Hybrid Nanofluid Formulation, Properties and Drug Delivery Prospects" Nanomaterials 11, no. 2: 440. https://doi.org/10.3390/nano11020440
APA StyleMannu, R., Karthikeyan, V., Velu, N., Arumugam, C., Roy, V. A. L., Gopalan, A.-I., Saianand, G., Sonar, P., Lee, K.-P., Kim, W.-J., Lee, D.-E., & Kannan, V. (2021). Polyethylene Glycol Coated Magnetic Nanoparticles: Hybrid Nanofluid Formulation, Properties and Drug Delivery Prospects. Nanomaterials, 11(2), 440. https://doi.org/10.3390/nano11020440













