A Comparative Assessment of the Chronic Effects of Micro- and Nano-Plastics on the Physiology of the Mediterranean Mussel Mytilus galloprovincialis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Lysosomal Parameters
2.2.1. Lysosomal Membrane Stability (LMS)
2.2.2. Histopathological Alterations
2.3. Biochemical and Enzymatic Biomarkers
2.4. Immunological Parameters
2.4.1. Lysozyme Enzymatic Assay
2.4.2. Phagocytosis
2.5. Statistical Analysis and Data Integration
3. Results
3.1. Effects of PS-MP and PS-NP on Lysosomal Parameters of General Stress
3.2. Effects of PS-MP and PS-NP on Levels of LF and MDA
3.3. Effects of PS-MP and PS-NP on the Activities of GST and CAT
3.4. Effects of PS-MP and PS-NP on Immunological and Neurological Parameters
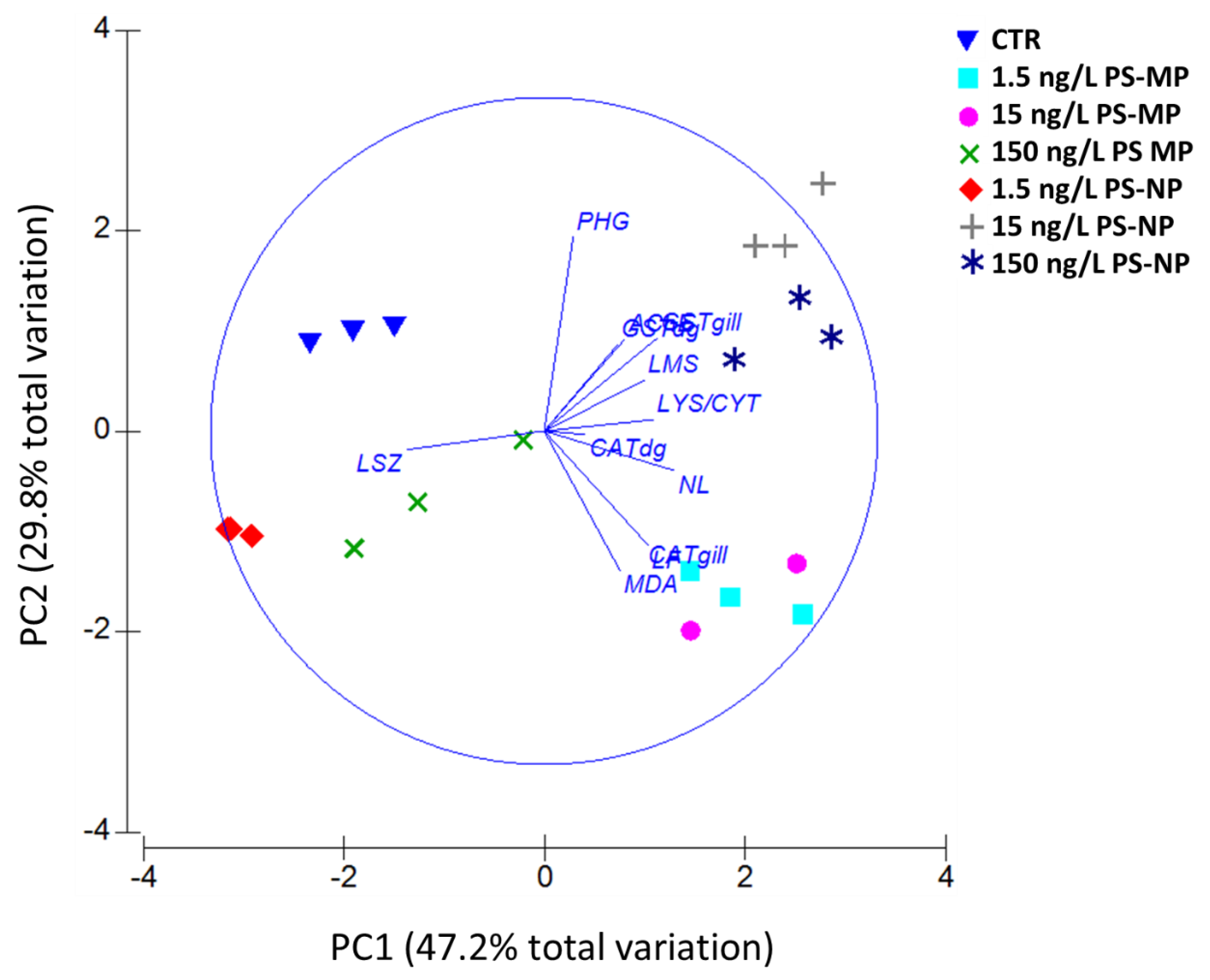
3.5. PCA and MES Data Integration
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- PlasticsEurope. Plastics Europe—The Facts 2020: An Analysis of European Plastics Production, Demand and Waste Data; PlasticsEurope: Brussels, Belgium, 2020; pp. 1–64. [Google Scholar]
- United Nations Environment Programme. Marine Plastic Debris and Microplastics—Global Lessons and Research to Inspire Action and Guide Policy Change; United Nations Environment Programme: Nairobi, Kenya, 2016; pp. 1–252. ISBN 978-92-807-3580-6. [Google Scholar]
- Pinto Da Costa, J.; Rocha-santos, T.; Duarte, A.C. The Environmental Impacts of Plastics and Micro-Plastics Use, Waste and Pollution: EU and National Measures—Requested by the Peti Committee; European Union—PE 658.279; European Union: Brussels, Belgium, 2020; pp. 1–72. [Google Scholar]
- Galloway, T.S.; Cole, M.; Lewis, C. Interactions of microplastic debris throughout the marine ecosystem. Nat. Ecol. Evol. 2017, 1, 0116. [Google Scholar] [CrossRef] [PubMed]
- Gunaalan, K.; Fabbri, E.; Capolupo, M. The hidden threat of plastic leachates: A critical review on their impacts on aquatic organisms. Water Res. 2020, 184, 116170. [Google Scholar] [CrossRef] [PubMed]
- GESAMP The Joint Group of Experts on Scientific Aspects of Marine Protection. Sources, Fate and Effects of Microplastics in the Marine Environment: Part Two of a Global Assessment; Kershaw, P.J., Rochman, C.M., Eds.; International Maritime Organization: London, UK, 2016; p. 220. [Google Scholar]
- Guzzetti, E.; Sureda, A.; Tejada, S.; Faggio, C. Microplastic in marine organism: Environmental and toxicological effects. Environ. Toxicol. Pharmacol. 2018, 64, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Setälä, O.; Lehtiniemi, M.; Coppock, R.; Cole, M. Microplastics in Marine Food Webs. In Microplastic Contamination in Aquatic Environments—An Emerging Matter of Environmental Urgency; Zeng, E., Ed.; Elsevier: Amsterdam, The Netherlands, 2018; pp. 339–363. [Google Scholar] [CrossRef]
- Foekema, E.M.; De Gruijter, C.; Mergia, M.T.; van Franeker, J.A.; Murk, A.J.; Koelmans, A.A. Plastic in North Sea fish. Environ. Sci. Technol. 2013, 47, 8818–8824. [Google Scholar] [CrossRef]
- Nelms, S.E.; Duncan, E.M.; Broderick, A.C.; Galloway, T.S.; Godfrey, M.H.; Hamann, M.; Lindeque, P.K.; Godley, B.J. Plastic and marine turtles: A review and call for research. ICES J. Mar. Sci. 2016, 73, 165–181. [Google Scholar] [CrossRef]
- Lu, Y.; Zhang, Y.; Deng, Y.; Jiang, W.; Zhao, Y.; Geng, J.; Ding, L.; Ren, H. Uptake and accumulation of polystyrene microplastics in zebrafish (Danio rerio) and toxic effects in liver. Environ. Sci. Technol. 2016, 50, 4054–4060. [Google Scholar] [CrossRef]
- Rochman, C.M.; Hoh, E.; Kurobe, T.; Teh, S.J. Ingested plastic transfers hazardous chemicals to fish and induces hepatic stress. Sci. Rep. 2013, 3, 3263. [Google Scholar] [CrossRef]
- Cole, M.; Lindeque, P.; Fileman, E.; Halsband, C.; Galloway, T.S. The Impact of Polystyrene Microplastics on Feeding, Function and Fecundity in the Marine Copepod Calanus helgolandicus. Environ. Sci. Technol. 2015, 49, 1130–1137. [Google Scholar] [CrossRef]
- Piarulli, S.; Vanhove, B.; Comandini, P.; Scapinello, S.; Moens, T.; Vrielinck, H.; Sciutto, G.; Prati, S.; Mazzeo, R.; Booth, A.M.; et al. Do different habits affect microplastics contents in organisms? A trait-based analysis on salt marsh species. Mar. Pollut. Bull. 2020, 153, 110983. [Google Scholar] [CrossRef]
- Wright, S.L.; Rowe, D.; Thompson, R.C.; Galloway, T.S. Microplastic ingestion decreases energy reserves in marine worms. Curr. Biol. 2013, 23, R1031–R1033. [Google Scholar] [CrossRef] [Green Version]
- Capolupo, M.; Franzellitti, S.; Valbonesi, P.; Lanzas, C.S.; Fabbri, E. Uptake and transcriptional effects of polystyrene microplastics in larval stages of the Mediterranean mussel Mytilus galloprovincialis. Environ. Pollut. 2018, 241, 1038–1047. [Google Scholar] [CrossRef]
- Franzellitti, S.; Capolupo, M.; Wathsala, R.H.G.R.; Valbonesi, P.; Fabbri, E. The Multixenobiotic resistance system as a possible protective response triggered by microplastic ingestion in Mediterranean mussels (Mytilus galloprovincialis): Larvae and adult stages. Comp. Biochem. Physiol. Part C 2019, 219, 50–58. [Google Scholar] [CrossRef]
- Murano, C.; Agnisola, C.; Caramiello, D.; Castellano, I.; Casotti, R.; Corsi, I.; Palumbo, A. How sea urchins face microplastics: Uptake, tissue distribution and immune system response. Environ. Pollut. 2020, 264, 114685. [Google Scholar] [CrossRef]
- Stapleton, P.A. Toxicological considerations of nano-sized plastics. AIMS Environ. Sci. 2019, 6, 367–378. [Google Scholar] [CrossRef] [PubMed]
- Hollóczki, O.; Gehrke, S. Can Nanoplastics Alter Cell Membranes? ChemPhysChem 2020, 21, 9–12. [Google Scholar] [CrossRef] [Green Version]
- Sun, X.; Chen, B.; Li, Q.; Liu, N.; Xia, B.; Zhu, L.; Qu, K. Science of the Total Environment Toxicities of polystyrene nano- and microplastics toward marine bacterium Halomonas alkaliphila. Sci. Total Environ. 2018, 642, 1378–1385. [Google Scholar] [CrossRef] [PubMed]
- Bergami, E.; Pugnalini, S.; Vannuccini, M.L.; Manfra, L.; Faleri, C.; Savorelli, F.; Dawson, K.A.; Corsi, I. Long-term toxicity of surface-charged polystyrene nanoplastics to marine planktonic species Dunaliella tertiolecta and Artemia franciscana. Aquat. Toxicol. 2017, 189, 159–169. [Google Scholar] [CrossRef]
- Sendra, M.; Carrasco-Braganza, M.I.; Yeste, P.M.; Vila, M.; Blasco, J. Immunotoxicity of polystyrene nanoplastics in different hemocyte subpopulations of Mytilus galloprovincialis. Sci. Rep. 2020, 10, 8637. [Google Scholar] [CrossRef] [PubMed]
- Auguste, M.; Lasa, A.; Balbi, T.; Pallavicini, A.; Vezzulli, L.; Canesi, L. Impact of nanoplastics on hemolymph immune parameters and microbiota composition in Mytilus galloprovincialis. Mar. Environ. Res. 2020, 159, 105017. [Google Scholar] [CrossRef]
- Brandts, I.; Teles, M.; Gonçalves, A.P.; Barreto, A.; Franco-martinez, L.; Tvarijonaviciute, A. Science of the Total Environment Effects of nanoplastics on Mytilus galloprovincialis after individual and combined exposure with carbamazepine. Sci. Total Environ. 2018, 643, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Viarengo, A.; Lowe, D.; Bolognesi, C.; Fabbri, E.; Koehler, A. The use of biomarkers in biomonitoring: A 2-tier approach assessing the level of pollutant-induced stress syndrome in sentinel organisms. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2007, 146, 281–300. [Google Scholar] [CrossRef] [PubMed]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Martínez-Gómez, C.; Bignell, J.; Lowe, D. Lysosomal Membrane Stability in Mussels; ICES Techniques in Marine Environmental Sciences; International Council for the Exploration of the Sea (ICES): Copenhagen, Denmark, 2015; pp. 1–41. ISBN 9788774821663. [Google Scholar]
- United Nations Environment Programme/RAMOGE. Manual on the Biomarkers Recommended for the Med Pol Biomonitoring Programme; United Nations Environment Programme: Athens, Greece, 1999; ISBN 928071788X. [Google Scholar]
- Capolupo, M.; Franzellitti, S.; Kiwan, A.; Valbonesi, P.; Dinelli, E.; Pignotti, E.; Birke, M.; Fabbri, E. A comprehensive evaluation of the environmental quality of a coastal lagoon (Ravenna, Italy): Integrating chemical and physiological analyses in mussels as a biomonitoring strategy. Sci. Total Environ. 2017, 598, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Martín-Díaz, M.L.; Gagné, F.; Blaise, C. The use of biochemical responses to assess ecotoxicological effects of Pharmaceutical and Personal Care Products (PPCPs) after injection in the mussel Elliptio complanata. Environ. Toxicol. Pharmacol. 2009, 28, 237–242. [Google Scholar] [CrossRef]
- Franzellitti, S.; Buratti, S.; Capolupo, M.; Du, B.; Haddad, S.P.; Chambliss, C.K.; Brooks, B.W.; Fabbri, E. An exploratory investigation of various modes of action and potential adverse outcomes of fluoxetine in marine mussels. Aquat. Toxicol. 2014, 151, 14–26. [Google Scholar] [CrossRef] [PubMed]
- Capolupo, M.; Valbonesi, P.; Kiwan, A.; Buratti, S.; Franzellitti, S.; Fabbri, E. Use of an integrated biomarker-based strategy to evaluate physiological stress responses induced by environmental concentrations of caffeine in the Mediterranean mussel Mytilus galloprovincialis. Sci. Total Environ. 2016, 563–564, 538–548. [Google Scholar] [CrossRef]
- Valbonesi, P.; Sartor, G.; Fabbri, E. Characterization of cholinesterase activity in three bivalves inhabiting the North Adriatic sea and their possible use as sentinel organisms for biosurveillance programmes. Sci. Total Environ. 2003, 312, 79–88. [Google Scholar] [CrossRef]
- Chu, F.L.E.; La Peyre, J.F. Effect of environmental factors and parasitism on hemolymph lysozyme and protein of American oysters (Crassostrea virginica). J. Invertebr. Pathol. 1989, 54, 224–232. [Google Scholar] [CrossRef]
- Canesi, L.; Betti, M.; Lorusso, L.C.; Ciacci, C.; Gallo, G. In vivo effects of Bisphenol A in Mytilus hemocytes: Modulation of kinase-mediated signalling pathways. Aquat. Toxicol. 2005, 71, 73–84. [Google Scholar] [CrossRef] [PubMed]
- Dagnino, A.; Allen, J.I.; Moore, M.N.; Broeg, K.; Canesi, L.; Viarengo, A. Development of an expert system for the integration of biomarker responses in mussels into an animal health index. Biomarkers 2007, 12, 155–172. [Google Scholar] [CrossRef]
- Martínez-Gómez, C.; Robinson, C.D.; Burgeot, T.; Gubbins, M.; Halldorsson, H.P.; Albentosa, M.; Bignell, J.P.; Hylland, K.; Vethaak, A.D. Biomarkers of general stress in mussels as common indicators for marine biomonitoring programmes in Europe: The ICON experience. Mar. Environ. Res. 2017, 124, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Moore, M.N.; Allen, J.I.; McVeigh, A.; Shaw, J. Lysosomal and autophagic reactions as predictive indicators of environmental impact in aquatic animals. Autophagy 2006, 2, 217–220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Franzellitti, S.; Canesi, L.; Auguste, M.; Wathsala, R.H.G.R.; Fabbri, E. Microplastic exposure and effects in aquatic organisms: A physiological perspective. Environ. Toxicol. Pharmacol. 2019, 68, 37–51. [Google Scholar] [CrossRef]
- Ringwood, A.H.; Levi-Polyachenko, N.; Carroll, D.L. Fullerene exposures with oysters: Embryonic, adult, and cellular responses. Environ. Sci. Technol. 2009, 43, 7136–7141. [Google Scholar] [CrossRef]
- Wang, F.; Salvati, A.; Boya, P. Lysosome-dependent cell death and deregulated autophagy induced by amine-modified polystyrene nanoparticles. Open Biol. 2018, 8, 170271. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dailianis, S. Environmental impact of anthropogenic activities: The use of mussels as a reliable tool for monitoring marine pollution. In Mussels: Anatomy, Habitat and Environmental Impact; McGevin, L.E., Ed.; Nova Science Publishers: Hauppauge, NY, USA, 2010; pp. 1–30. ISBN 9781617617638. [Google Scholar]
- Orbea, A.; Garmendia, L.; Marigómez, I.; Cajaraville, M.P. Effects of the “Prestige” oil spill on cellular biomarkers in intertidal mussels: Results of the first year of studies. Mar. Ecol. Prog. Ser. 2006, 306, 177–189. [Google Scholar] [CrossRef]
- Gaspar, T.R.; Chi, R.J.; Parrow, M.W.; Ringwood, A.H. Cellular bioreactivity of micro- and nano-plastic particles in oysters. Front. Mar. Sci. 2018, 5, 1–8. [Google Scholar] [CrossRef]
- Avio, C.G.; Gorbi, S.; Milan, M.; Benedetti, M.; Fattorini, D.; Pauletto, M.; Bargelloni, L.; Regoli, F. Pollutants bioavailability and toxicological risk from microplastics to marine mussels. Environ. Pollut. 2015, 198, 211–222. [Google Scholar] [CrossRef]
- Moore, M. Cytochemical responses of the lysosomal system and NADPH-ferrihemoprotein reductase in molluscan digestive cells to environmental and experimental exposure to xenobiotics. Mar. Ecol. Prog. Ser. 1988, 46, 81–89. [Google Scholar] [CrossRef]
- Lu, L.; Wan, Z.; Luo, T.; Fu, Z.; Jin, Y. Polystyrene microplastics induce gut microbiota dysbiosis and hepatic lipid metabolism disorder in mice. Sci. Total Environ. 2018, 631–632, 449–458. [Google Scholar] [CrossRef]
- Auguste, M.; Balbi, T.; Montagna, M.; Fabbri, R.; Sendra, M.; Blasco, J.; Canesi, L. In vivo immunomodulatory and antioxidant properties of nanoceria (nCeO2) in the marine mussel Mytilus galloprovincialis. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2019, 219, 95–102. [Google Scholar] [CrossRef]
- Gomes, T.; Pereira, C.G.; Cardoso, C.; Pinheiro, J.P.; Cancio, I.; Bebianno, M.J. Accumulation and toxicity of copper oxide nanoparticles in the digestive gland of Mytilus galloprovincialis. Aquat. Toxicol. 2012, 118–119, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Koehler, A.; Marx, U.; Broeg, K.; Bahns, S.; Bressling, J. Effects of nanoparticles in Mytilus edulis gills and hepatopancreas—A new threat to marine life? Mar. Environ. Res. 2008, 66, 12–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Moore, M.N. Autophagy as a second level protective process in conferring resistance to environmentally-induced oxidative stress. Autophagy 2008, 4, 254–256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woods, M.N.; Stack, M.E.; Fields, D.M.; Shaw, S.D.; Matrai, P.A. Microplastic fiber uptake, ingestion, and egestion rates in the blue mussel (Mytilus edulis). Mar. Pollut. Bull. 2018, 137, 638–645. [Google Scholar] [CrossRef]
- Regoli, F.; Giuliani, M.E. Oxidative pathways of chemical toxicity and oxidative stress biomarkers in marine organisms. Mar. Environ. Res. 2014, 93, 106–117. [Google Scholar] [CrossRef]
- Bergami, E.; Krupinski Emerenciano, A.; González-Aravena, M.; Cárdenas, C.A.; Hernández, P.; Silva, J.R.M.C.; Corsi, I. Polystyrene nanoparticles affect the innate immune system of the Antarctic sea urchin Sterechinus neumayeri. Polar Biol. 2019, 42, 743–757. [Google Scholar] [CrossRef]
- Greven, A.C.; Merk, T.; Karagöz, F.; Mohr, K.; Klapper, M.; Jovanović, B.; Palić, D. Polycarbonate and polystyrene nanoplastic particles act as stressors to the innate immune system of fathead minnow (Pimephales promelas). Environ. Toxicol. Chem. 2016, 35, 3093–3100. [Google Scholar] [CrossRef]
- Hamed, M.; Soliman, H.A.M.; Osman, A.G.M.; Sayed, A.E.D.H. Assessment the effect of exposure to microplastics in Nile Tilapia (Oreochromis niloticus) early juvenile: I. blood biomarkers. Chemosphere 2019, 228, 345–350. [Google Scholar] [CrossRef]
- Yong, C.Q.Y.; Valiyaveetill, S.; Tang, B.L. Toxicity of microplastics and nanoplastics in Mammalian systems. Int. J. Environ. Res. Public Health 2020, 17, 1509. [Google Scholar] [CrossRef] [Green Version]
- Canesi, L.; Ciacci, C.; Bergami, E.; Monopoli, M.P.; Dawson, K.A.; Papa, S.; Canonico, B.; Corsi, I. Evidence for immunomodulation and apoptotic processes induced by cationic polystyrene nanoparticles in the hemocytes of the marine bivalve Mytilus. Mar. Environ. Res. 2015, 111, 34–40. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Parisi, M.G.; Toubiana, M.; Cammarata, M.; Roch, P. Lysozyme gene expression and hemocyte behaviour in the Mediterranean mussel, Mytilus galloprovincialis, after injection of various bacteria or temperature stresses. Fish Shellfish Immunol. 2008, 25, 143–152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Callewaert, L.; Van Herreweghe, J.M.; Vanderkelen, L.; Leysen, S.; Voet, A.; Michiels, C.W. Guards of the great wall: Bacterial lysozyme inhibitors. Trends Microbiol. 2012, 20, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Rong, J.; Guan, X.; Zha, S.; Shi, W.; Han, Y.; Du, X.; Wu, F.; Huang, W.; Liu, G. Immunotoxicity of microplastics and two persistent organic pollutants alone or in combination to a bivalve species. Environ. Pollut. 2020, 258, 113845. [Google Scholar] [CrossRef] [PubMed]
- Browne, M.A.; Dissanayake, A.; Galloway, T.S.; Lowe, D.M.; Thompson, R.C. Ingested Microscopic Plastic Translocates to the Circulatory System of the Mussel, Mytilus edulis (L.). Environ. Sci. Technol. 2008, 42, 5026–5031. [Google Scholar] [CrossRef] [PubMed]
- Paul-Pont, I.; Lacroix, C.; González Fernández, C.; Hégaret, H.; Lambert, C.; Le Goïc, N.; Frère, L.; Cassone, A.L.; Sussarellu, R.; Fabioux, C.; et al. Exposure of marine mussels Mytilus spp. to polystyrene microplastics: Toxicity and influence on fluoranthene bioaccumulation. Environ. Pollut. 2016, 216, 724–737. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alnajar, N.; Jha, A.N.; Turner, A. Impacts of microplastic fibres on the marine mussel, Mytilus galloprovinciallis. Chemosphere 2021, 262, 128290. [Google Scholar] [CrossRef] [PubMed]
- Bringer, A.; Cachot, J.; Prunier, G.; Dubillot, E.; Clérandeau, C.; Thomas, H. Experimental ingestion of fluorescent microplastics by pacific oysters, Crassostrea gigas, and their effects on the behaviour and development at early stages. Chemosphere 2020, 254, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Silva, C.J.M.; Patrício Silva, A.L.; Campos, D.; Machado, A.L.; Pestana, J.L.T.; Gravato, C. Oxidative damage and decreased aerobic energy production due to ingestion of polyethylene microplastics by Chironomus riparius (Diptera) larvae. J. Hazard. Mater. 2021, 402, 123775. [Google Scholar] [CrossRef] [PubMed]
- González-Soto, N.; Hatfield, J.; Katsumiti, A.; Duroudier, N.; Lacave, J.M.; Bilbao, E.; Orbea, A.; Navarro, E.; Cajaraville, M.P. Impacts of dietary exposure to different sized polystyrene microplastics alone and with sorbed benzo[a]pyrene on biomarkers and whole organism responses in mussels Mytilus galloprovincialis. Sci. Total Environ. 2019, 684, 548–566. [Google Scholar] [CrossRef] [PubMed]
- Colovic, M.B.; Krstic, D.Z.; Lazarevic-Pasti, T.D.; Bondzic, A.M.; Vasic, V.M. Acetylcholinesterase inhibitors: Pharmacology and toxicology. Curr. Neuropharmacol. 2013, 11, 315–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valbonesi, P.; Brunelli, F.; Mattioli, M.; Rossi, T.; Fabbri, E. Cholinesterase activities and sensitivity to pesticides in different tissues of silver European eel, Anguilla anguilla. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2011, 154, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, F.; Garcia, A.R.; Pereira, B.P.; Fonseca, M.; Mestre, N.C.; Fonseca, T.G.; Ilharco, L.M.; Bebianno, M.J. Microplastics effects in Scrobicularia plana. Mar. Pollut. Bull. 2017, 122, 379–391. [Google Scholar] [CrossRef]
- Barboza, L.G.A.; Vieira, L.R.; Branco, V.; Figueiredo, N.; Carvalho, F.; Carvalho, C.; Guilhermino, L. Microplastics cause neurotoxicity, oxidative damage and energy-related changes and interact with the bioaccumulation of mercury in the European seabass, Dicentrarchus labrax (Linnaeus, 1758). Aquat. Toxicol. 2018, 195, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Fonte, E.; Ferreira, P.; Guilhermino, L. Temperature rise and microplastics interact with the toxicity of the antibiotic cefalexin to juveniles of the common goby (Pomatoschistus microps): Post-exposure predatory behaviour, acetylcholinesterase activity and lipid peroxidation. Aquat. Toxicol. 2016, 180, 173–185. [Google Scholar] [CrossRef] [PubMed]
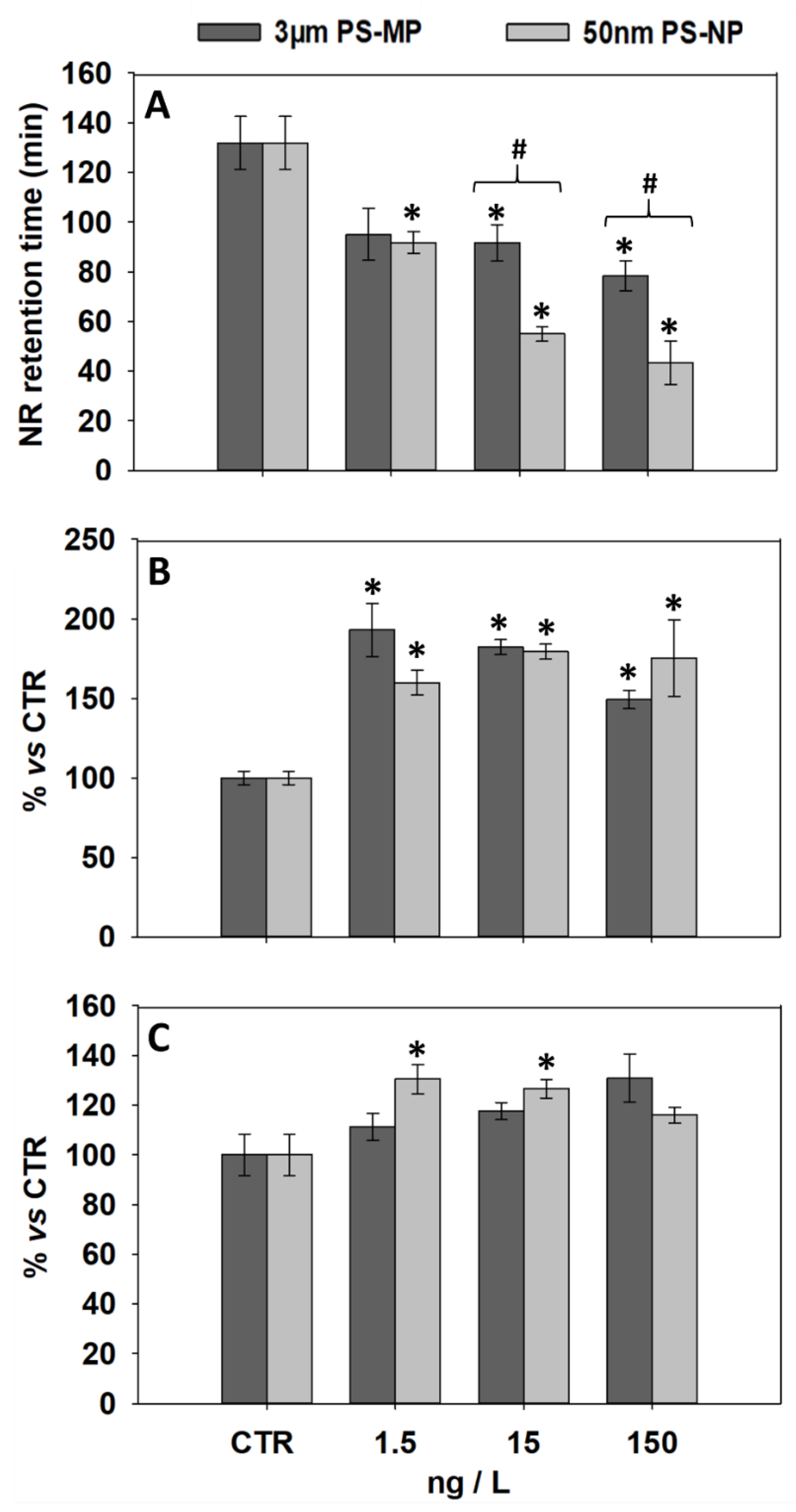
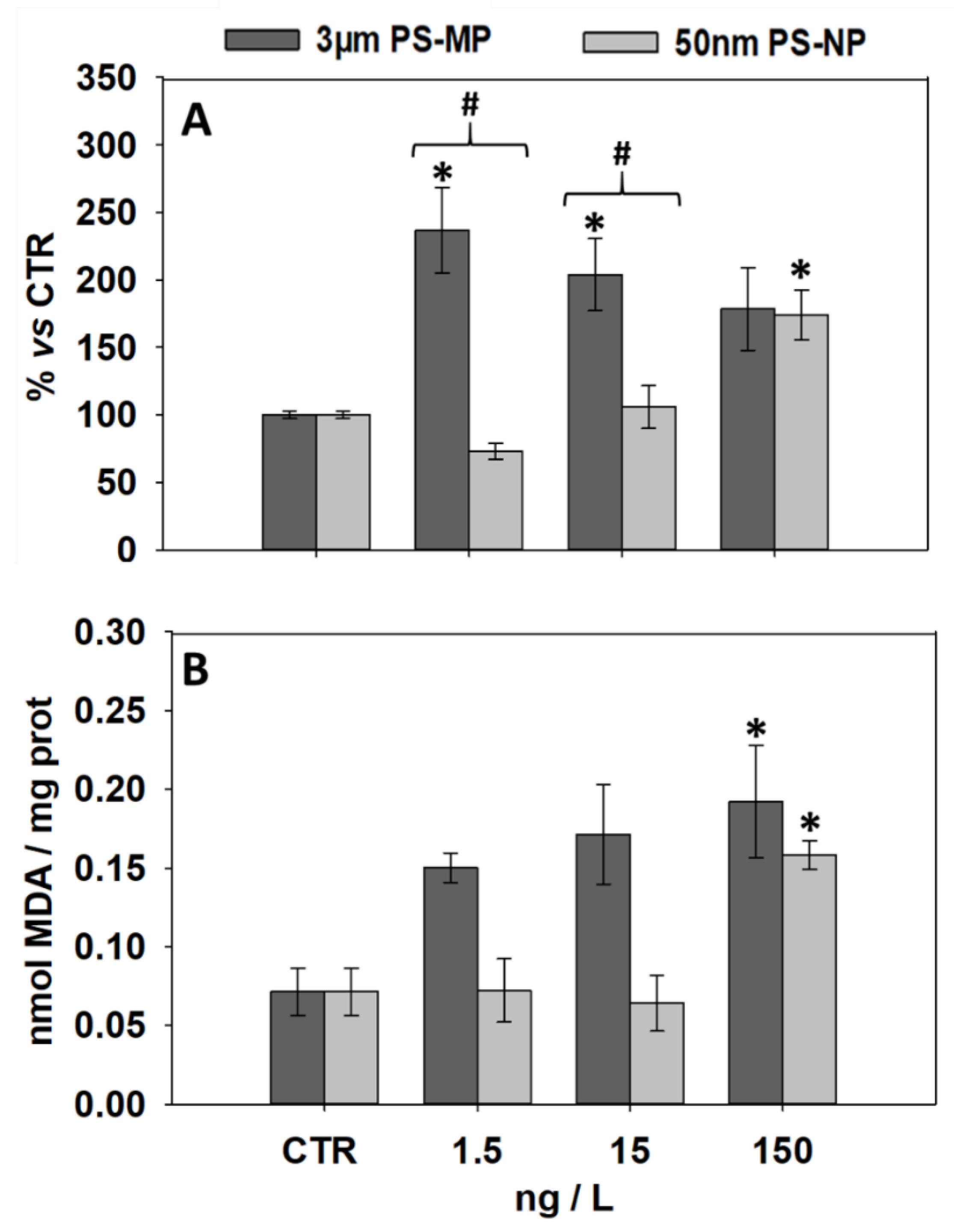
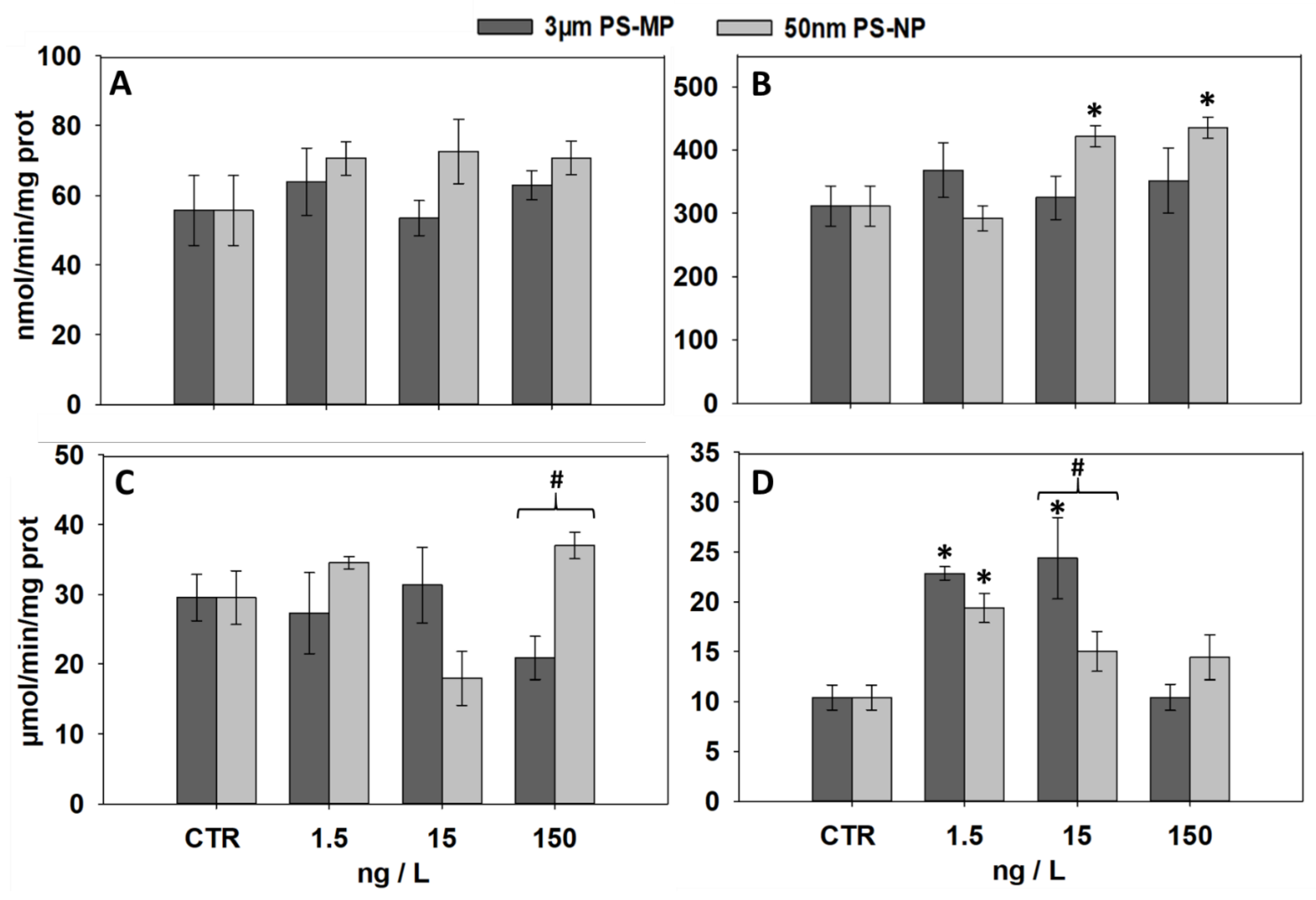
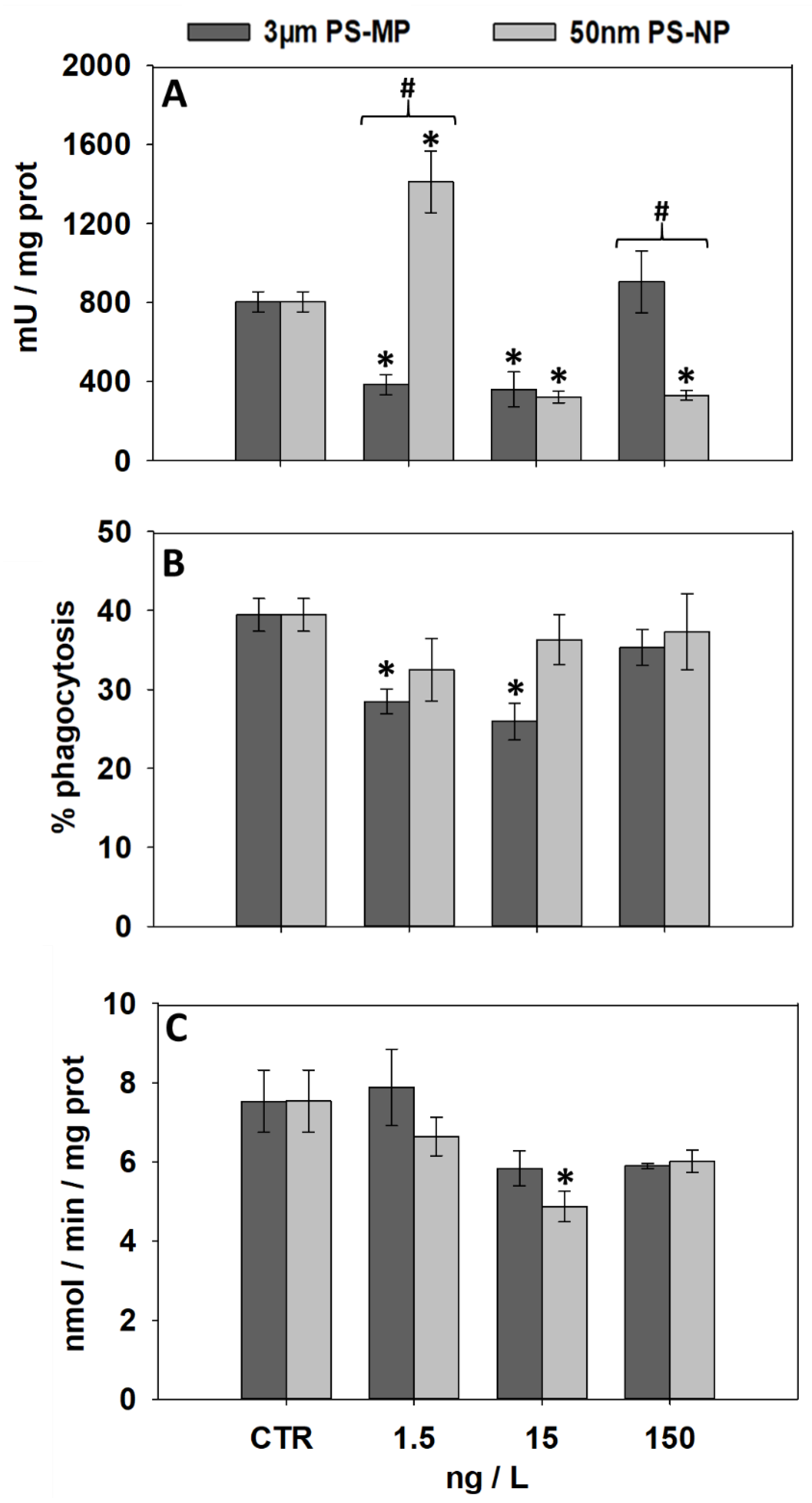
| Biomarker | 1.5 ng/L MP | 15 ng/L MP | 150 ng/L MP | 1.5 ng/L NP | 15 ng/L NP | 150 ng/L NP | |
|---|---|---|---|---|---|---|---|
| Cell | |||||||
| LMSGP | AF | NV | 0.70 * | 0.59 * | 0.70 * | 0.42 * | 0.33 * |
| AL | NV | - | - | - | -- | -- | |
| NL | AF | 1.93 * | 1.82 * | 1.49 * | 1.60 * | 1.79 * | 1.75 * |
| AL | + | + | + | + | + | + | |
| LF | AF | 2.37 * | 2.04 * | 1.78 | 0.73 | 1.06 | 1.74 * |
| AL | + | NV | NV | NV | NV | + | |
| MDA | AF | 2.08 | 2.38 | 2.67 * | 1.00 | 0.89 | 2.19 * |
| AL | NV | NV | ++ | NV | NV | ++ | |
| GSTdg | AF | 1.15 | 0.96 | 1.13 | 1.27 | 1.30 | 1.27 |
| AL | NV | NV | NV | NV | NV | NV | |
| GSTg | AF | 1.18 | 1.04 | 1.13 | 0.94 | 1.35 * | 1.40 * |
| AL | NV | NV | NV | NV | + | + | |
| CATdg | AF | 0.93 | 1.06 | 0.71 | 1.17 | 0.61 | 1.25 |
| AL | NV | NV | NV | NV | NV | NV | |
| CATg | AF | 2.19 * | 2.24 * | 1.00 | 1.86 * | 1.44 | 1.38 |
| AL | ++ | ++ | NV | + | NV | NV | |
| AChE | AF | 1.05 | 0.77 | 0.78 | 0.88 | 0.65 * | 0.80 |
| AL | NV | NV | NV | NV | - | NV | |
| Lysozyme | AF | 0.48 * | 0.45 * | 1.13 | 1.75 * | 0.40 | 0.41 |
| AL | NV | NV | NV | + | NV | NV | |
| Phagocythosis | AF | 0.72 * | 0.66 * | 0.90 | 0.82 | 0.92 | 0.95 |
| AL | NV | NV | NV | NV | NV | NV | |
| Tissue | |||||||
| LYS/CYT | AF | 1.11 | 1.18 | 1.31 | 1.30 * | 1.27 * | 1.16 |
| AL | NV | NV | NV | + | + | NV | |
| Organism | |||||||
| Survival | AF | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 | 1.00 |
| AL | NV | NV | NV | NV | NV | NV | |
| HSI | A (Healthy) | B (Low stress) | B (Low stress) | C (Moderate stress) | D (High stress) | C (Moderate stress) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capolupo, M.; Valbonesi, P.; Fabbri, E. A Comparative Assessment of the Chronic Effects of Micro- and Nano-Plastics on the Physiology of the Mediterranean Mussel Mytilus galloprovincialis. Nanomaterials 2021, 11, 649. https://doi.org/10.3390/nano11030649
Capolupo M, Valbonesi P, Fabbri E. A Comparative Assessment of the Chronic Effects of Micro- and Nano-Plastics on the Physiology of the Mediterranean Mussel Mytilus galloprovincialis. Nanomaterials. 2021; 11(3):649. https://doi.org/10.3390/nano11030649
Chicago/Turabian StyleCapolupo, Marco, Paola Valbonesi, and Elena Fabbri. 2021. "A Comparative Assessment of the Chronic Effects of Micro- and Nano-Plastics on the Physiology of the Mediterranean Mussel Mytilus galloprovincialis" Nanomaterials 11, no. 3: 649. https://doi.org/10.3390/nano11030649






