1. Introduction
Nuclear Medicine is based on the administration of peculiar compounds, the so-called radiopharmaceuticals, that are composed of a radionuclide (such as
99mTc,
18F,
68Ga,
223Ra,
64Cu, etc.) chelated by a suitable ligand and conjugated to a molecule (such as a metabolic intermediate, a human protein fragment or colloid) acting as a vector able to transport the compound from the injection site to the region of interest. The vector investigated in this study is Nanocolloid Human Serum Albumin (NC), a colloidal suspension of HSA particles. Nanoparticles of HSA labelled with
99mTc ([
99mTc]Tc-NC) are widely used as a radiopharmaceutical in Nuclear Medicine and represent the European gold-standard for the intraoperative detection of the Sentinel Lymph Node (SLN) in many kinds of cancer, mainly breast cancer and melanoma [
1,
2,
3,
4]. The associated favourable γ-emission of
99mTc allows a preoperative check of colloid migration either with dynamic or static studies employing planar scintigraphy or hybrid instruments, such as SPECT-CT (single-photon emission computed tomography—computed tomography scan).
[
99mTc]Tc-NC is helpful in SLN-SPECT because of its ability to map nodes on the tumour drainage pathway, indicating the tumour state of the whole regional lymphatic basin and excluding lymphatic metastatic spread, in case of negative biopsy. NC can be used for the evaluation of pulmonary ventilation in patients with lung damage due to Covid-19 pneumonia [
5,
6].
Even though the intralymphatic kinetics of NC radiopharmaceuticals are not well understood, their ability to visualise the first lymph node of a chain is widely recognised, as in SLN detection for breast-conservating surgery for breast cancer.
Recent developments in precision medicine, such as evaluating regional perfusion in healthy versus cancerous tissues, points to the need for multi-modal contrast agents that can provide both better cross-sectional images via SPECT and positron emission tomography (PET). [
7,
8].
Furthermore, the increasingly common use of robotic surgery has highlighted the need for a specific procedure which can identify the site and contours of the target lesion [
9].
For this reason, the research interest is now increasingly focused on the possibility of labelling commercial kits routinely used with other isotopes, to extend diagnostic accuracy, surgical outcome, follow-up and even therapeutic horizons.
Our previous study concerned the in vitro feasibility of obtaining an instant hybrid NC tracer, radiolabelled with both
99mTc and
68Ga isotopes and tagged with the fluorescent probe Indocyanine Green (ICG) nanocolloids (NC) ([
99mTc]Tc-[
68Ga]Ga-ICG-NC) [
10]. The resulting tri-modal [
99mTc]Tc-[
68Ga]Ga-ICG-NC combined three different imaging system (SPECT, PET and fluorescence) in a single tracer, thus responding to different surgical needs and improving the clinical application of the NC commercial kits.
However, before transferring its use to patients, it is mandatory to increase our knowledge of the chemical bond between NC and the isotopes. Currently, too little is known about the chemical conformation of the NC, even less about the strength and the specificity of the bond with Tc and almost nothing is known about the bond with Ga.
The aim of this study is to investigate and compare the chemical bond between NC and different isotopes. For this purpose, we have primarily studied the kinetic orders of NC labelling with 99mTc and 68Ga, and then compared them with the kinetic orders labelled HSA.
Langmuir isotherms then made it possible to find the maximum concentration of complexable radiometal.
Finally, the bond strength and specificity were indirectly investigated by changing the starting conditions. The bond modification possibility was investigated with the simultaneous presence of the same quantities of the two isotopes. The pH effect on radiolabelling was tested and the stability of the radiometals complex was verified through competition reactions carried out in presence of a different ligand.
Therefore, HSA-based nanocolloids represent an interesting carrier, since a significant amount of radionuclides can be incorporated into the HSA particle, because of the several binding sites of albumin. Moreover, NC catabolism is a fast and easy process that results in innocuous degradation products [
11].
2. Materials and Methods
2.1. Materials
All the pharmaceuticals used in this study have already been commercialised and authorised for clinical use. Nanocolloid (nano-sized human colloidal particles of a ≤80 nm diameter, containing 0.5 mg of human albumin) is a radiopharmaceutical commercially available as NanoAlbumon® (Radiopharmacy Laboratory Ltd, Budaörs, Hungary). The NanoAlbumon® kit is a sterile, non-pyrogenic, lyophilised mixture. Active ingredient: Human Serum Albumin nano sized colloid 0.5 mg. Excipients: stannous (II) chloride dihydrate 0.2 mg, sodium phosphate monobasic and sodium phosphate dibasic 1.0 mg, glucose 15.0 mg. In this article, the NanoAlbumon® kit was generically referred as NC.
Human Serum Albumin is a radiopharmaceutical commercially available as Vasculocis® containing 10 mg of human serum albumin, stannous chloride dihydrate, hydrochloric acid and sodium chloride, under nitrogen atmosphere. (CURIUM IBA Cis-Bio, Gif-sur-Yvette, Paris, France). In this article, Vasculocis® was generically referred as HSA.
Dimercaptosuccinic acid (DMSA) is a radiopharmaceutical commercially available as Renocis®, containing 1 mg of dimercaptosuccinic acid, stannous chloride dihydrate, inositol, ascorbic acid. In this article, the Renocis® kit was generically referred as DMSA.
All kits were labelled with sodium pertechnetate [99mTcO4−], obtained from 99Mo/99mTc Tekcis® generator (CURIUM IBA Cis-Bio, Gif-sur-Yvette, Paris, France) and sodium chloride per injection (0.9%). To perform [68Ga]Ga labelling, 68Ge/68Ga generator (1.1 GBq TiO2-based GalliaPharm® Eckert-Ziegler Isotope Products, Berlin, Germany) was eluted with 8 mL of 0.1 N HCl (Eckert-Ziegler). 0.75 mL of 0.1 N NaOH / phosphate buffer (1 mL) was added to this 0.1N HCl solution of [68Ga] Ga-chloride. Final pH ranged between 4.0 and 4.5.
All chemicals were manufactured by TraceSELECT—UltraPURE from ABX Radensberg, Germany.
2.2. Methods
2.2.1. Quality Controls and Labelling Yield
Quality controls (QC) were performed to verify the labelling yield of [99mTc]Tc-NC and [68Ga]Ga-NC. The percentage of free 99mTc was evaluated by thin-layer chromatography, using ITLC-SG (Varian, Folson, CA, USA) as a stationary phase (10 cm long and 2 cm wide) and CH3OH:H2O 85:15 as a mobile phase. A 10 μL spot of solution containing the sample was applied to a strip roughly 1.5 cm from the bottom edge. The strip was then placed in a separation chamber and the solvent was run for at least 10 cm. Labelled NC remains at the point of application, while free 99mTc pertechnetate migrates with the solvent front. The percentage of free 68Ga was assessed in the same way, using 0.1 M tribasic-citrate solution, adjusted to pH 6 with HCl, as a mobile phase. NC labelled with 68Ga remains at the point of application, while free 68Ga migrates with the solvent front. TLC (thin layer chromatography) strips were cut at 4.5 cm in height, defining a down sample region with point of application, and an up sample with the solvent front. In these two regions, 99mTc and 68Ga activity was determined with a 3″ × 3″ NaI(Tl) pinhole 16 × 40 mm gamma counter (Raytest, Straubenhardt, Germany).
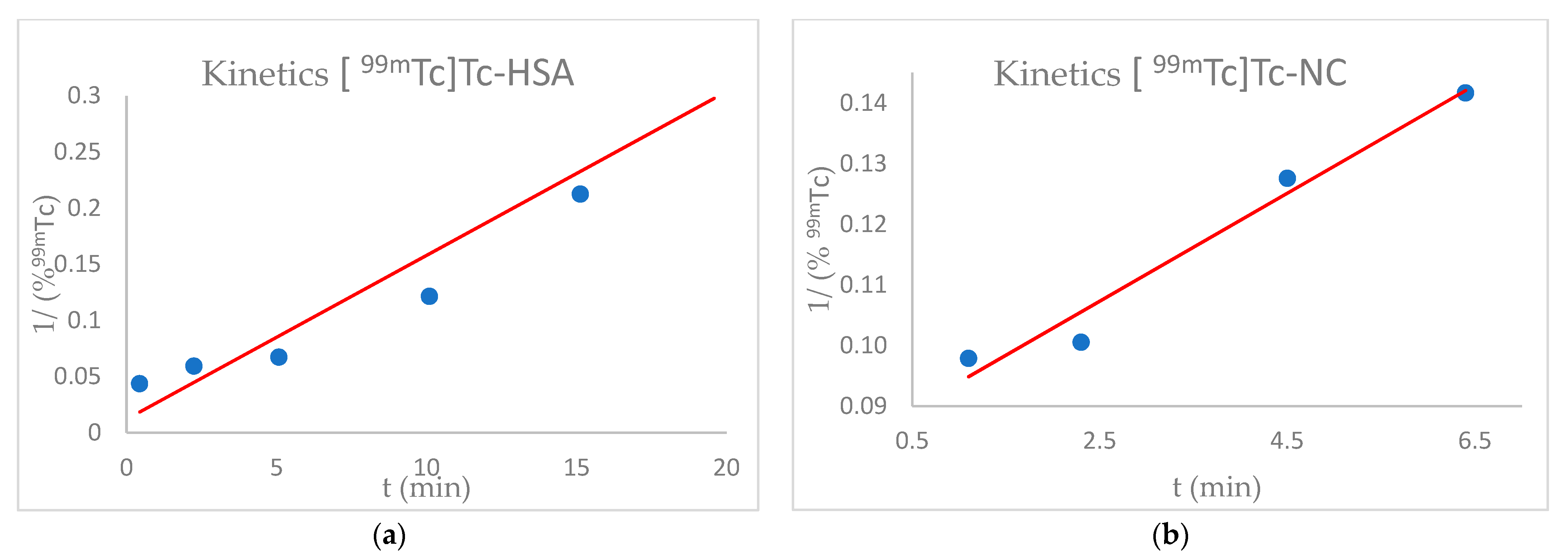
2.2.2. [99mTc]Tc-HSA Labelling Kinetics
According to the manufacturers’ instructions, the HSA kit was reconstituted in 4 mL of saline solution with 2000 MBq of sodium pertechnetate [99mTcO4−] obtained from 99Mo/99mTc Tekcis® generator. The solution was incubated at room temperature. Samples were taken at regular time intervals of 1 min, up to 20 min, and the amount of 99mTc bound to the HSA was measured, according to the QC procedures.
2.2.3. [99mTc]Tc-NC Labelling Kinetics
According to the manufacturers’ instructions, the NC dissolved in the kit was reconstituted with 2000 MBq of sodium pertechnetate [99mTcO4−] obtained from 99Mo/99mTc Tekcis® generator. The solution was incubated at room temperature. Samples were taken at regular time intervals of 1 min, up to 20 min, and the amount of 99mTc bound to the NC was measured, according to the QC procedures.
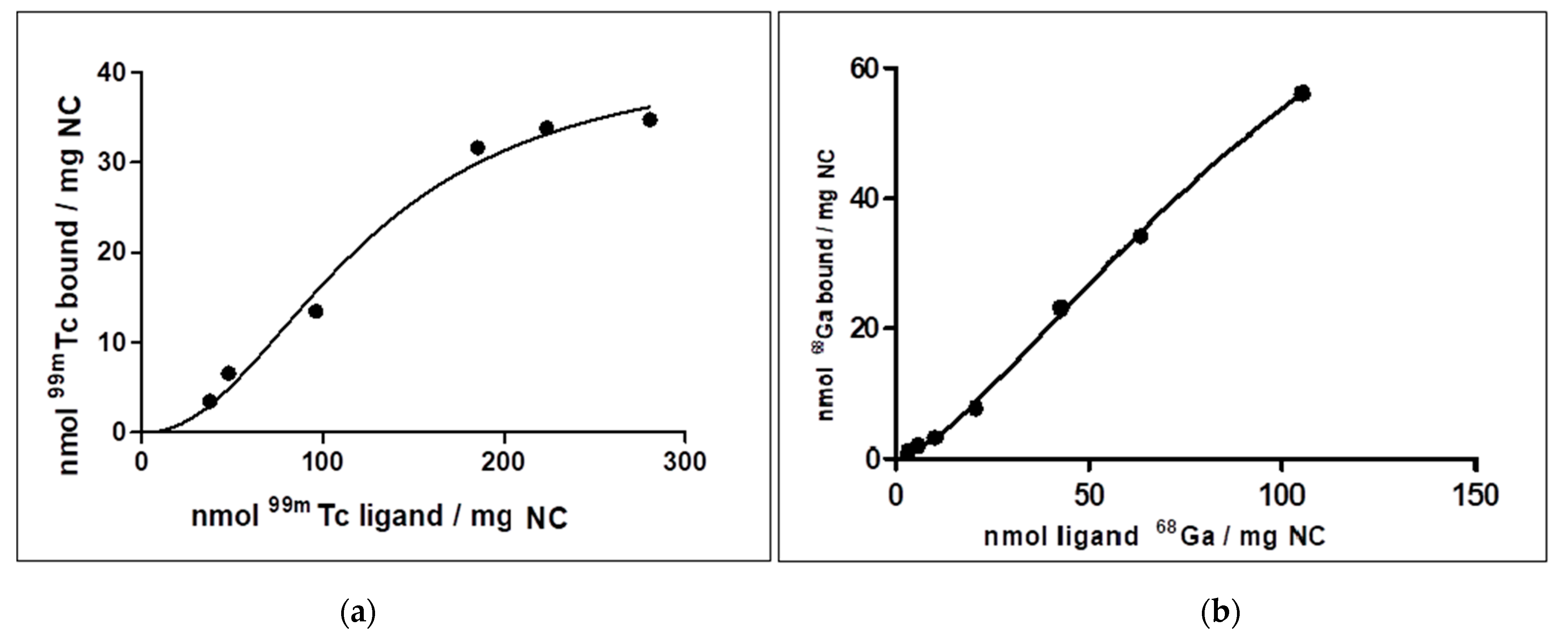
2.2.4. [99mTc]Tc-NC Binding Affinity Studies
Samples containing 50 µg of NC dissolved in 1 mL of deionised water were incubated with increasing concentration of 99mTc to reach values of about 35, 50, 100, 200, 300 nmol 99mTc/mg NC. The incubation time was fixed at 20 min and the reaction was carried out at room temperature. The amount of bound 99mTc was performed as described in the QC procedures. The uptake measurements were expressed in nmol 99mTc bound/mg NC.
2.2.5. [68Ga]Ga-NC Binding Affinity Studies
The procedure described above was replicated to evaluate the binding affinity of NC with [68Ga]Ga. At the fixed incubation times of 20 min, NC samples were incubated with increasing concentration of 68Ga to reach values of about 3, 6, 12, 20, 40, 60, 120 nmol 68Ga /mg NC. The reaction was carried out at 40 °C. The uptake measurements were expressed in nmol 68Ga bound/mg NC.
2.2.6. Competition Evaluation between 68Ga and 99mTc for NC Labelling
To verify the 99mTc and 68Ga competition for the nanocolloid binding sites, a commercial kit of NC was dissolved in 10 mL of saline solution. 18 nmol of 99mTc and 13 nmol of 68Ga were added to 1 mL of this solution and incubated for 20 min at room temperature.
The measurement of the 99mTc vs 68Ga bound was performed after 1 h and 3 h, as described in the QC procedures.
2.2.7. Effects of pH on [99mTc]Tc-NC Labelling
Two methods were used to evaluate the effect of pH on the [99mTc]Tc-NC radiolabelling. A commercial kit of NC was first dissolved in 10 mL of saline solution, then 6 vials were prepared with 100 µL of this suspension and, finally, 300 KBq of 99mTc were added in each vial.
Method 1: the solution was incubated at pH 6 for 20 min at room temperature. At the end of incubation, the pH of 2 vials was adjusted to pH 3 and pH 10, with HCl 5 M and NaOH 2 M, respectively.
Method 2: The pH of 3 vials was immediately adjusted to reach a final pH value of 3, 6 or 10 with HCl 5 M and NaOH 2 M, and then incubated for 20 min at room temperature.
The measurement of 99mTc finally bound was performed as described in the QC procedures.
2.2.8. [99mTc]Tc -HSA Stability Test via DMSA Competition
These experiments were carried out to test the 99mTc binding strength. DMSA was chosen as a highly competitive ligand, due to its ability of heavy metal sequestration. DMSA is therefore commonly used in case of heavy metal poisoning. A high excess of DMSA was used.
Two methods were used to evaluate the stability of [99mTc]Tc-HSA through competition with DMSA.
Method 1: commercial kits of DMSA and HSA previously dissolved in 4 mL of deionised water were mixed. 200 MBq of 99mTc were added, and the resulting solution was incubated at pH 6 for 20 min at room temperature.
Method 2: a commercial kit of HSA was dissolved in 4 mL of deionised water and 200 MBq of 99mTc were added for incubation at pH 6, for 20 min at room temperature. Then a solution of the commercial kit of DMSA dissolved in 4 mL of deionised water was added.
The measurement of 99mTc finally bound to the HSA and to DMSA was performed following the above-described QC procedures.
2.2.9. [99mTc]Tc-NC Stability Test via DMSA Competition
Two methods were used in order to evaluate the amount of 99mTc strongly bound to the NC, via competition with DMSA. A high excess of DMSA was used.
Method 1: commercial kits of DMSA and NC previously dissolved in 4 mL deionised water were mixed. 200 MBq of 99mTc were added, and the resulting solution was incubated at pH 6 for 20 min at room temperature.
Method 2: a commercial kit of NC was dissolved in 4 mL of deionised water and 200 MBq of 99mTc were added for incubation at pH 6, for 20 min at room temperature. Then a solution of the commercial kit of DMSA dissolved in 4 mL of deionised water was added.
The measurement of 99mTc finally bound to the NC and to DMSA was performed following the above-described QC procedures.
5. Conclusions
At the dawn of Nuclear Medicine, NC tracers were developed based on the fundamental role played by albumin proteins. Albumin is an endogenous functional carrier, driving a huge amount of different molecules from human blood to the target district or organ. For this reason, the peculiar function of HSA has often been exploited for pharmacological purposes, to easily carry drugs, including radiotracers. Though the drugs based on HSA are among the most useful probes for diagnosis, the so-called shine-through phenomenon limits preoperative detection with planar scintigraphy 99mTc based SPECT-CT during operative exploration. The use of PET-CT, with better spatial resolution, might circumvent this problem. In addition, the opportunity to quantify the absolute target uptake in terms of SUV (Standardised Uptake Value), allows clinicians to predict intraoperative detectability. Furthermore, the physical characteristics of the 68Ga isotope, given its high percentage of positron emission (89%), relatively short half-life (t½ = 67.71 min) and chemical properties, make it an excellent positron emission isotope with superior resolution, speed and quantification capacity, compared to 99mTc labelling, particularly in guided robot-assisted surgery and robotic-arm PET/CT assisted biopsy.
Since the chemical nature of the 99mTc and 68Ga bond with NC is not fully known, our experience confirmed that pH is an essential parameter influencing Technetium and Gallium binding to NC, with an optimal range between 4.0–6.5. In all likelihood, the protein structure of NC does not favour the binding of the metals in basic conditions. In fact, some chelator groups, such as the SH group, may not be stable under a basic condition (pH 10) and may be easily oxidised, losing their radiolabelling property. Neutralisation of the 68Ge/68Ga generator eluate might gradually increase the affinity of the various Gallium chemical species for NC. Based on our results, we assume that 68Ga strongly binds to cys 34, the only amino acids residue once present in HSA molecules. Due to the slight competition for the same binding sites, dual labelling (99mTc and 68Ga) is possible without problem. The experiments were generally consistent with each other, and the 99mTc -NC show a fair amount of resistance to transchelation, where NC probably incorporate 99mTc in structural pockets. Thus, radiolabelling is possible with excellent efficiency, both for 99mTc and 68Ga, through pseudo-second order kinetics. Knowing well the labelling mechanism of an HSA based drug is essential for making predictions on its behaviour with different radioisotopes. A vast increase of their use is expected in the coming years, thanks to their unique characteristics of in vivo stability, bioavailability, non-toxicity, EPR, and so forth. Further studies will be conducted, focusing on the radiolabelling at physiological pH (7.4) and under the hypoxic condition, that may modify the amino acids’ bonding properties.