Flowery ln2MnSe4 Novel Electrocatalyst Developed via Anion Exchange Strategy for Efficient Water Splitting
Abstract
:1. Introduction
2. Experimental Segment
2.1. Reagents
2.2. Fabrication of In2MnSe4
2.3. Characterizations
2.4. Ink Preparation and Electrochemical Measurements
3. Results and Discussion
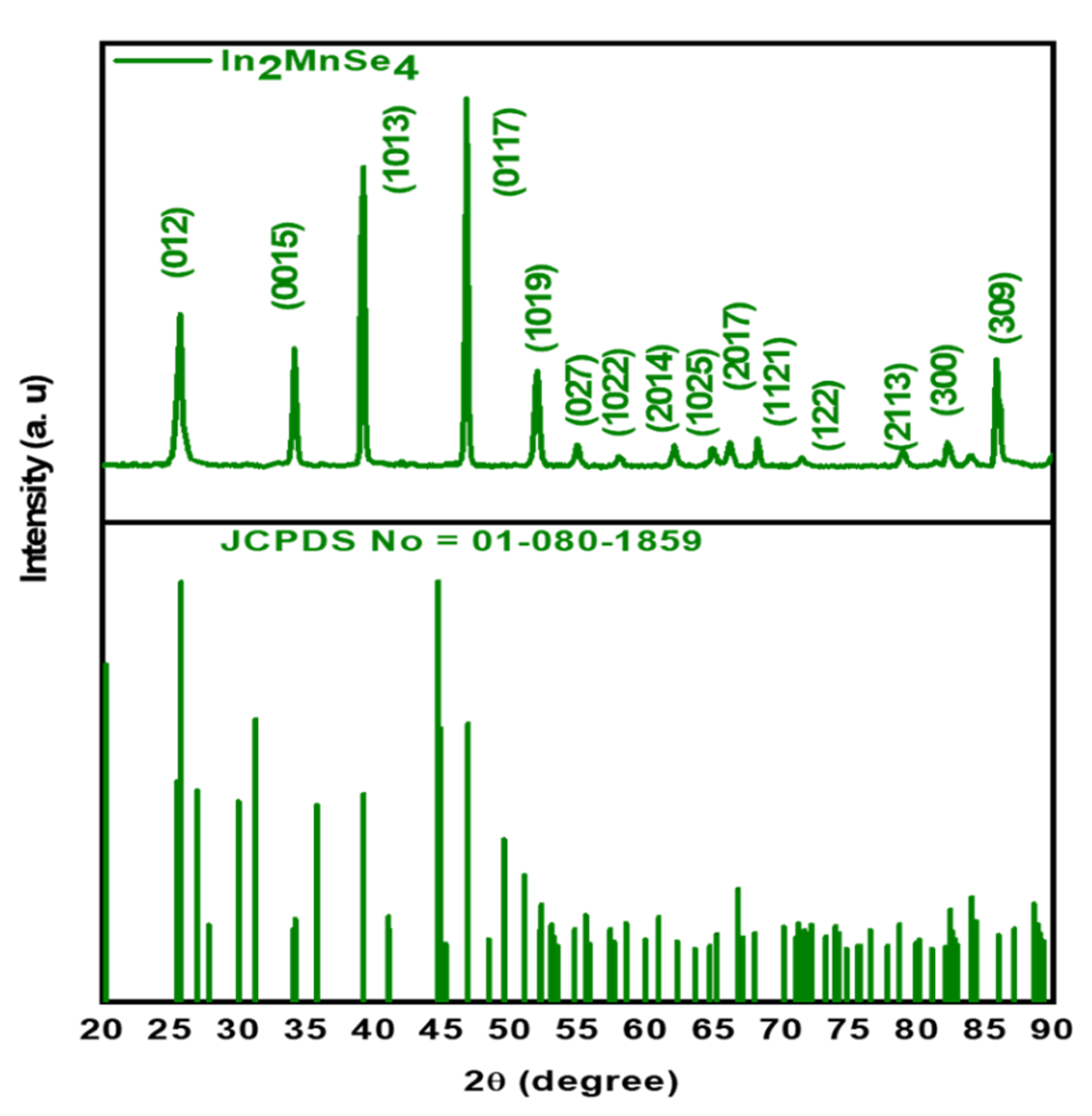
3.1. Structural Analysis
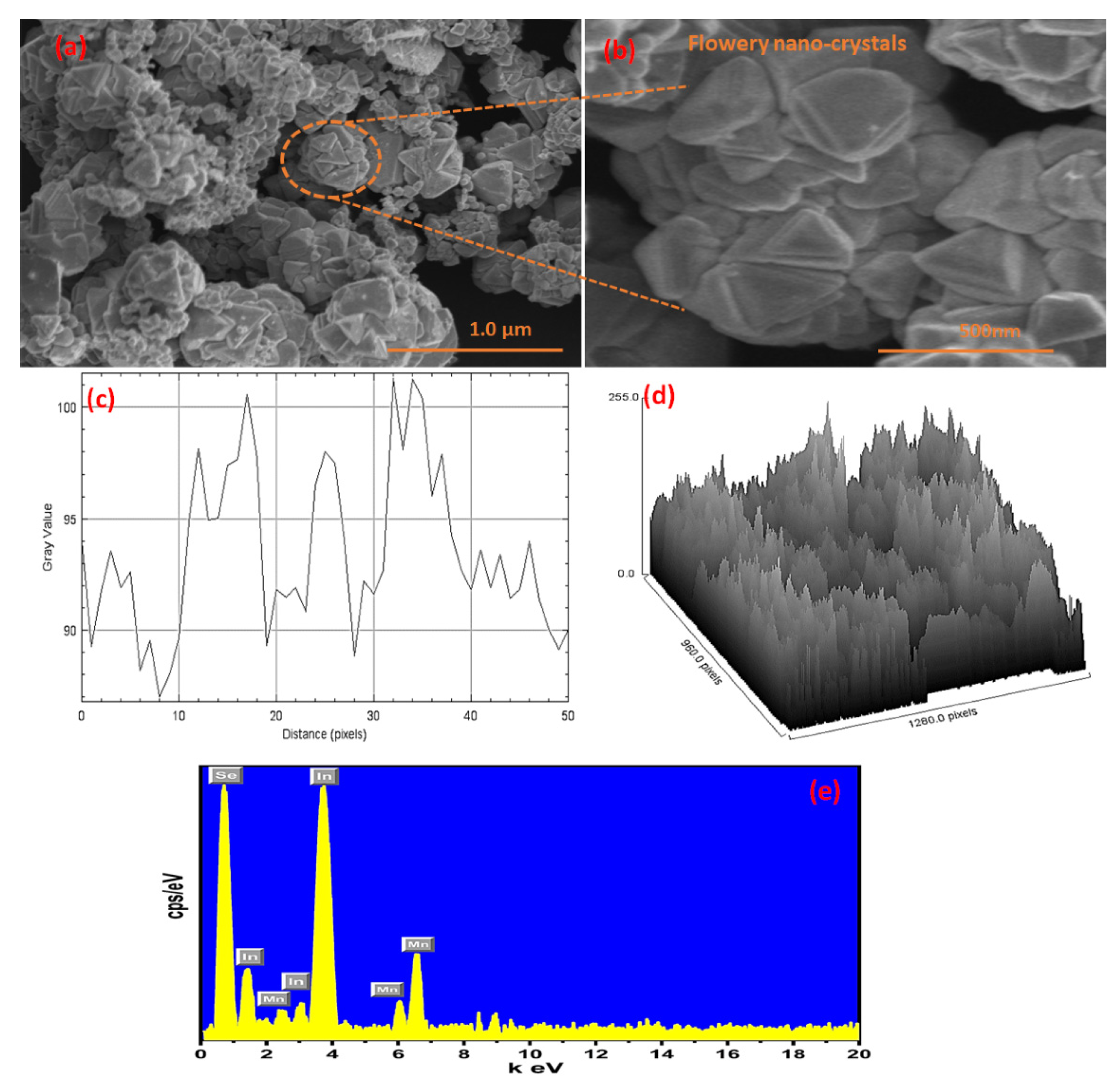
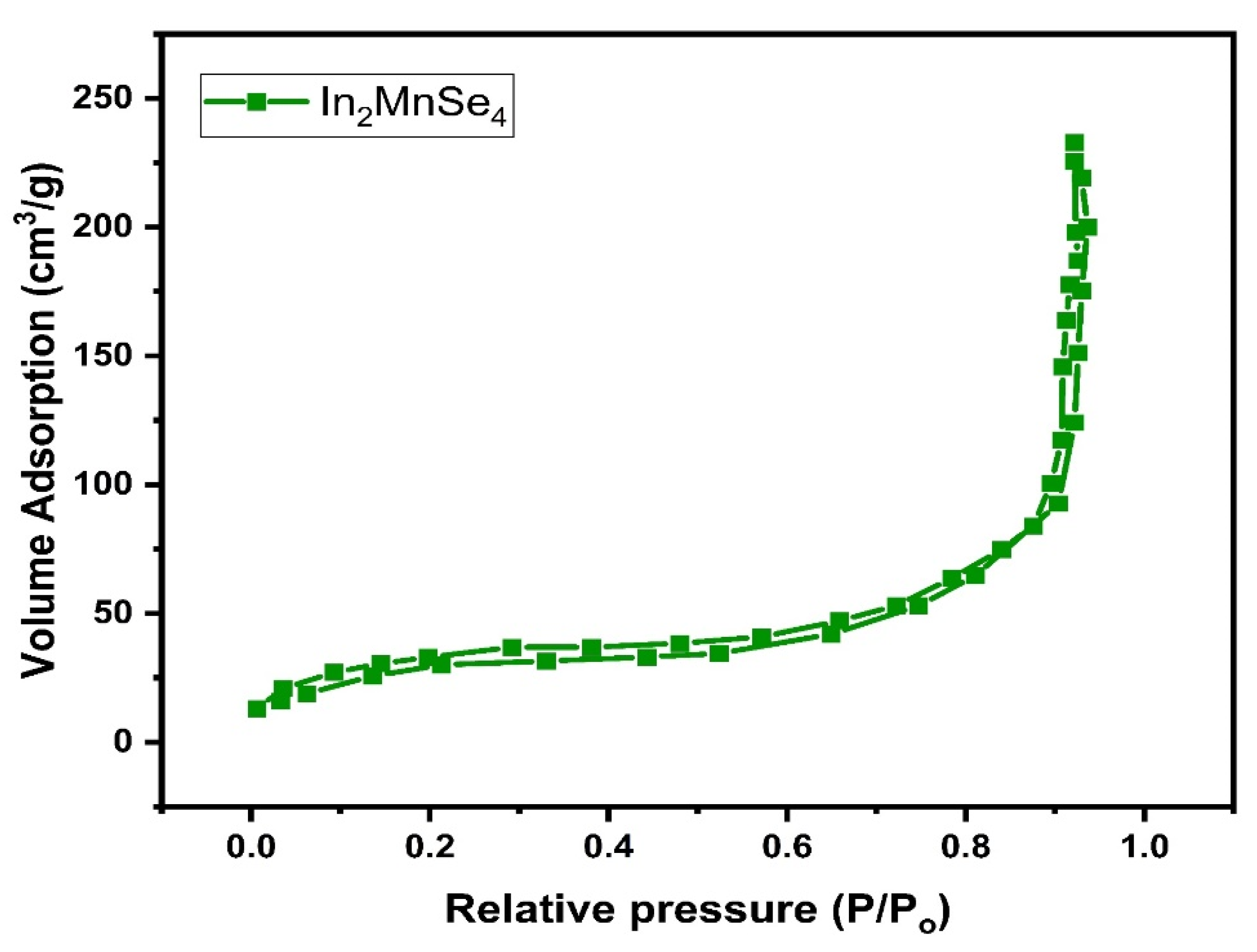
3.2. Morphological, Elemental, and Textural Analysis
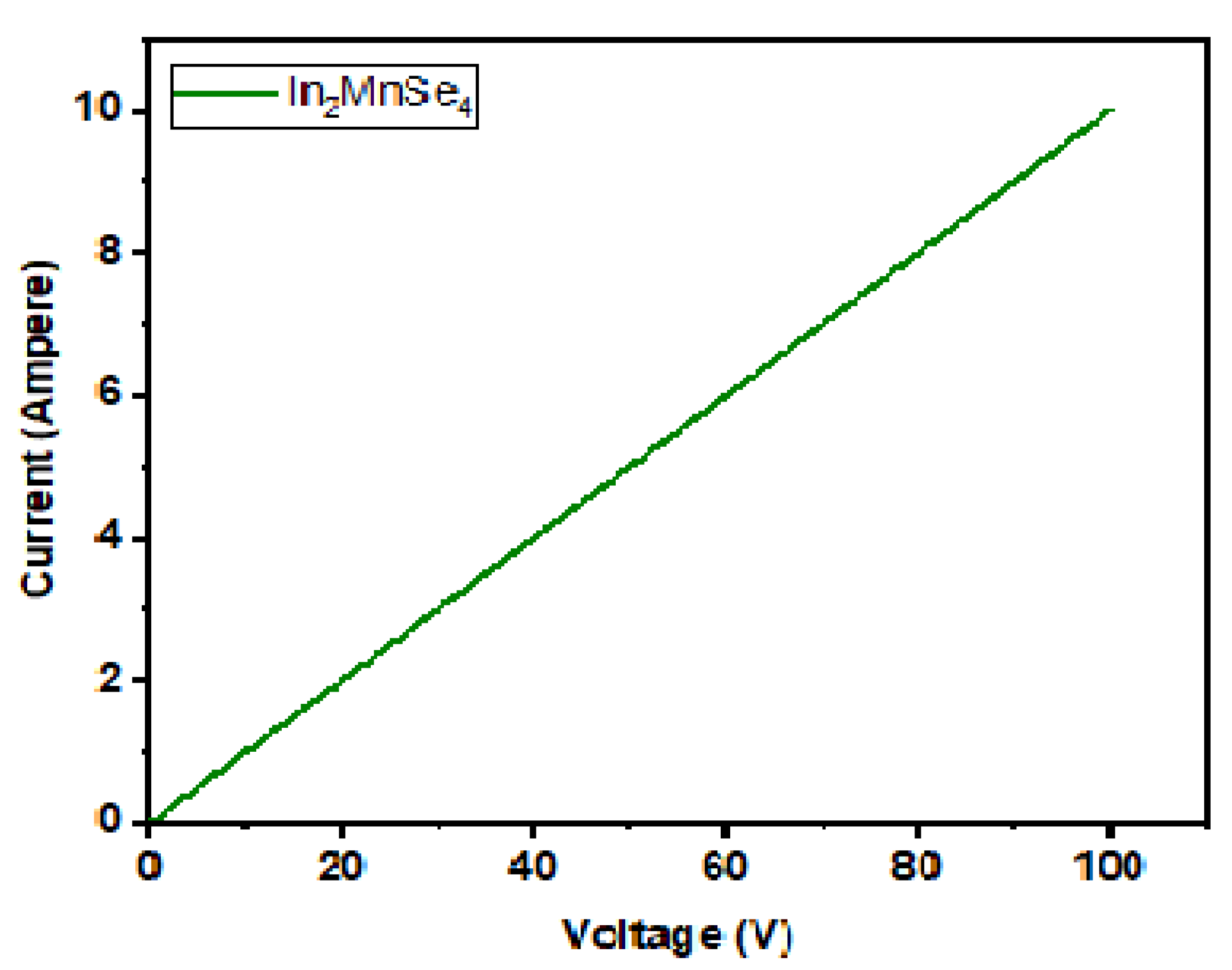
3.3. Electrical Measurement
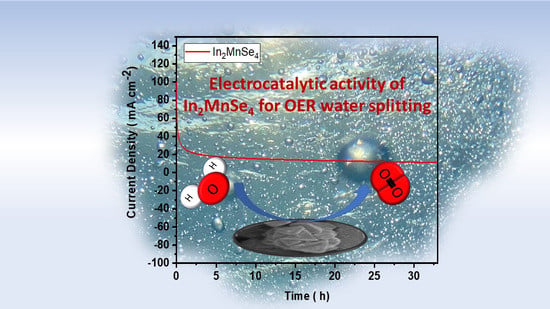
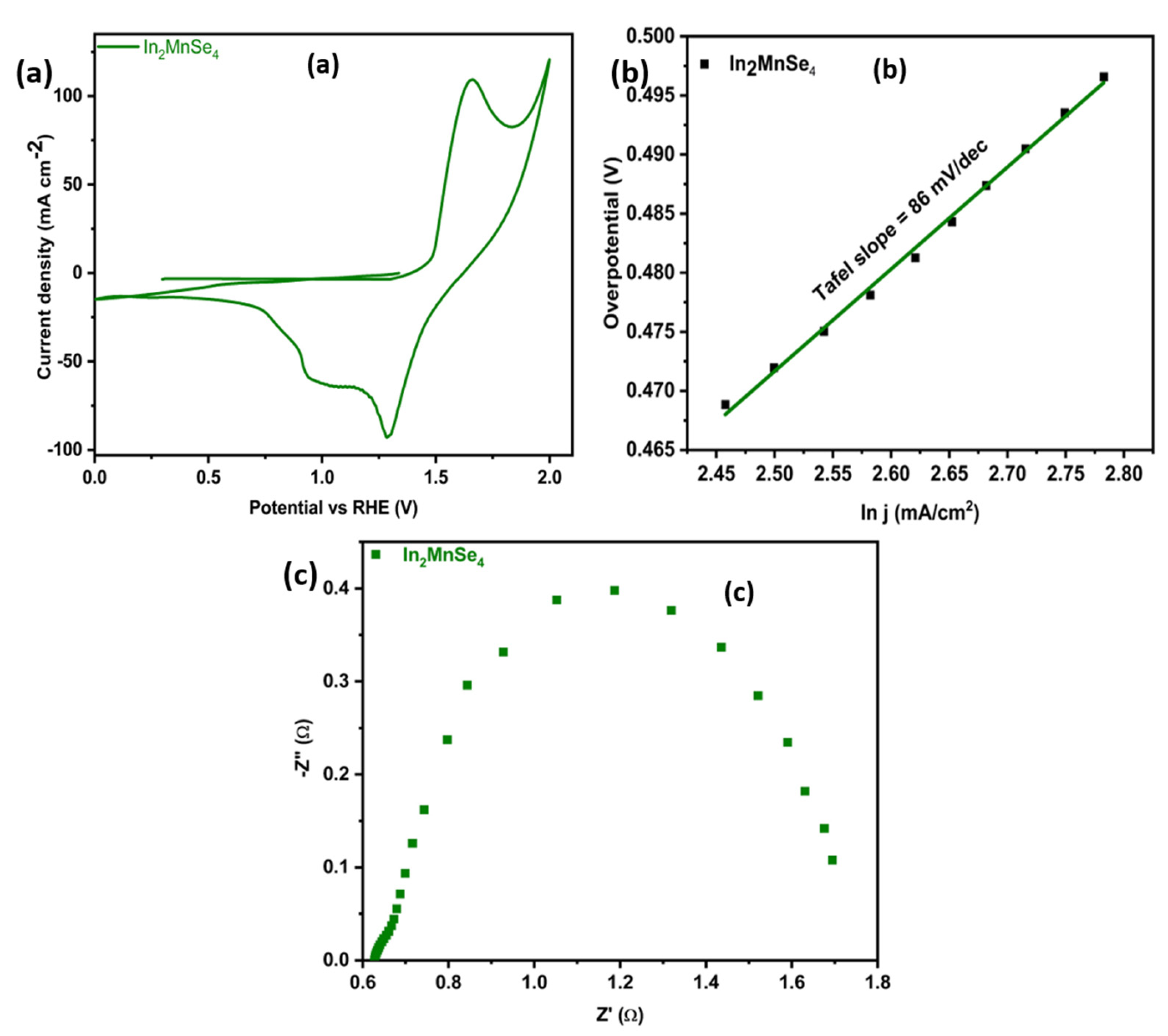
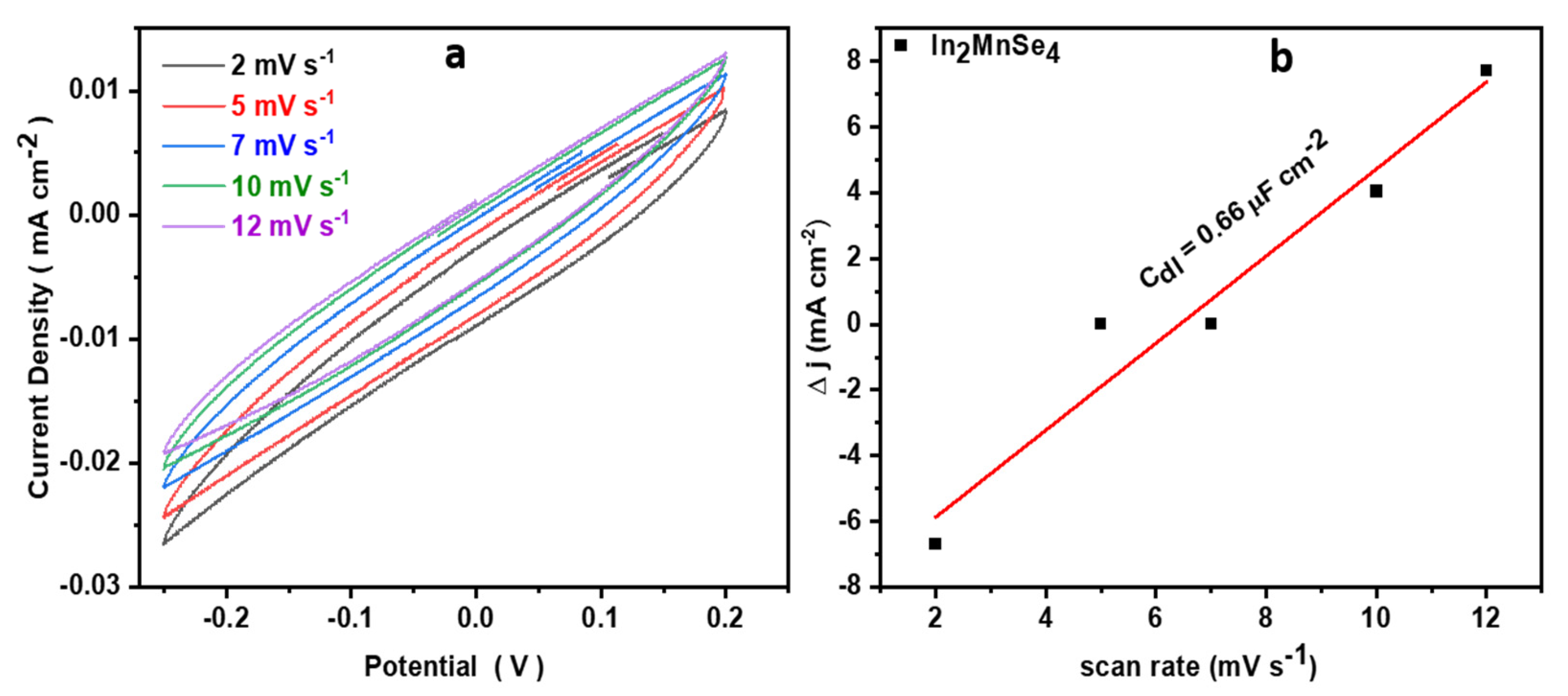
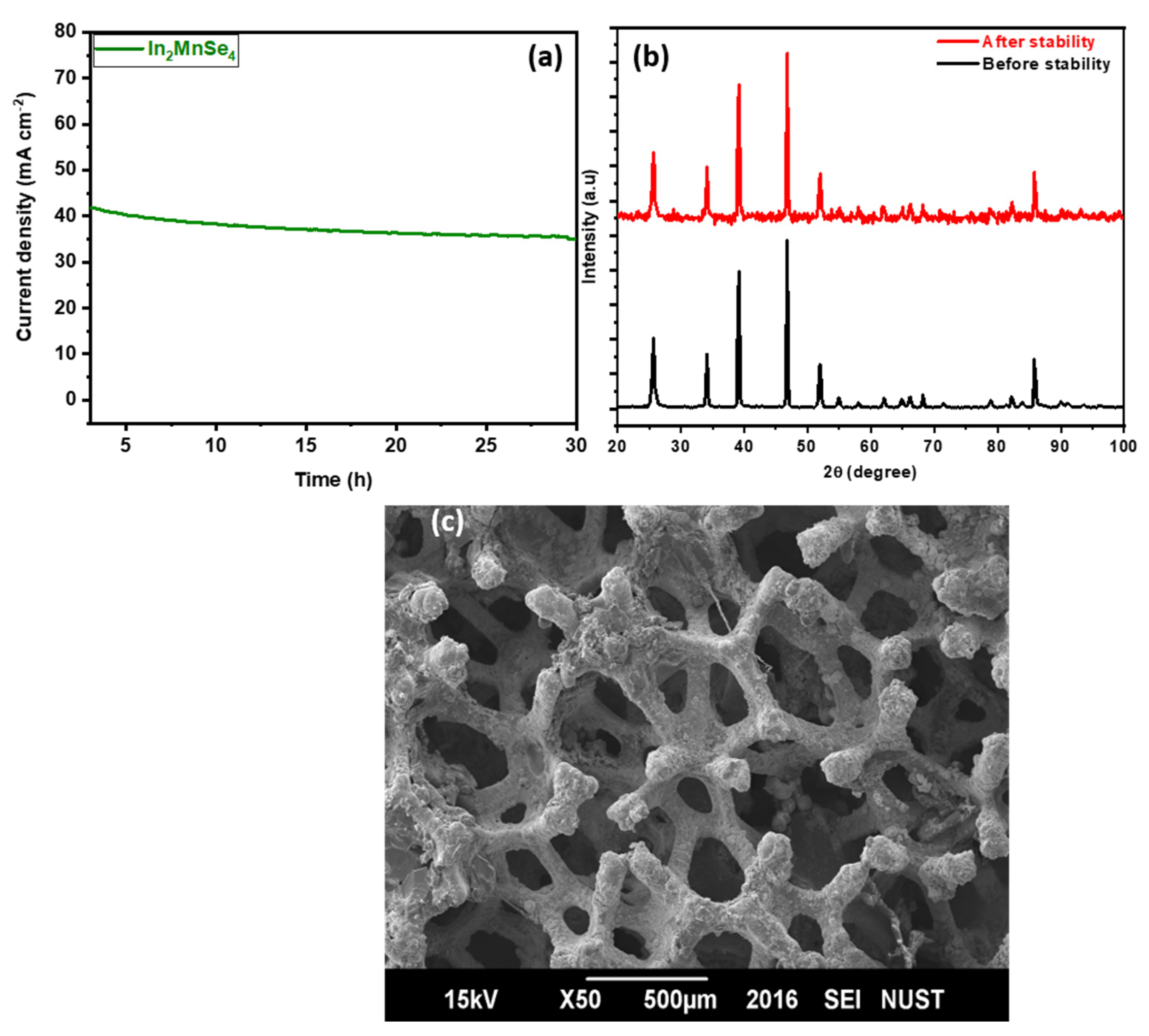
3.4. Electrochemical Study
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stambouli, A.B. Fuel cells: The expectations for an environmental-friendly and sustainable source of energy. Renew. Sust. Energy Rev. 2011, 15, 4507–4520. [Google Scholar] [CrossRef]
- Lin, Z.; Xiao, B.; Huang, M.; Yan, L.; Wang, Z.; Huang, Y.; Shen, S.; Zhang, Q.; Gu, L.; Zhong, W. Realizing negatively charged metal atoms through controllable d-electron transfer in ternary Ir1−xRhxSb intermetallic alloy for hydrogen evolution reaction. Adv. Energy Mater. 2022, 2200855. [Google Scholar] [CrossRef]
- Yang, L.; Yang, L.; Ding, L.; Deng, F.; Luo, X.-B.; Luo, S.L. Principles for the application of nanomaterials in environmental pollution control and resource reutilization. In Nanomaterials for the Removal of Pollutants and Resource Reutilization, Micro and Nano Technolgies; Elsevier: Amsterdam, The Netherlands, 2019; pp. 1–23. [Google Scholar] [CrossRef]
- Shen, S.; Hu, Z.; Zhang, H.; Song, K.; Wang, Z.; Lin, Z.; Zhang, Q.; Gu, L.; Zhong, W. Highly active Si sites enabled by negative valent Ru for electrocatalytic hydrogen evolution in LaRuSi. Angew. Chem. 2022. [Google Scholar] [CrossRef]
- Opoku, F.; Govender, K.K.; van Sittert, C.G.C.E.; Govender, P.P. Tuning the electronic structures, work functions, optical properties and stability of bifunctional hybrid graphene oxide/V–doped NaNbO3 type–II heterostructures: A promising photocatalyst for H2 production. Carbon 2018, 136, 187–195. [Google Scholar] [CrossRef]
- Hu, E.; Ning, J.; He, B.; Li, Z.; Zheng, C.; Zhong, Y.; Zhang, Z.; Hu, Y. Unusual formation of tetragonal microstructures from nitrogen-doped carbon nanocapsules with cobalt nanocores as a bi-functional oxygen electrocatalyst. J. Mater. Chem. A 2017, 5, 2271–2279. [Google Scholar] [CrossRef]
- Zhang, Q.; Guan, J. Applications of atomically dispersed oxygen reduction catalysts in fuel cells and zinc–air batteries. Energy Environ. Mater. 2021, 4, 307–335. [Google Scholar] [CrossRef]
- Najafi, L.; Oropesa-Nuñez, R.; Bellani, S.; Martín-García, B.; Pasquale, L.; Serri, M.; Drago, F.; Luxa, J.; Sofer, Z.K.; Sedmidubský, D. Topochemical transformation of two-dimensional VSe2 into metallic nonlayered VO2 for water splitting reactions in acidic and alkaline media. ACS Nano 2022, 16, 351–367. [Google Scholar] [CrossRef]
- Tang, T.; Wang, Z.; Guan, J. A review of defect engineering in two-dimensional materials for electrocatalytic hydrogen evolution reaction. Chin. J. Catal. 2022, 43, 636–678. [Google Scholar] [CrossRef]
- Seh, Z.; Kibsgaard, J.; Dickens, C.; Chorkendorff, I.B.; Nørskov, J.K.; Jaramillo, T.F. Combining theory and experiment in electrocatalysis: Insights into materials design. Science 2017, 355, eaad4998. [Google Scholar] [CrossRef] [Green Version]
- Morales-Guio, C.G.; Stern, L.-A.; Hu, X. Nanostructured hydrotreating catalysts for electrochemical hydrogen evolution. Chem. Soc. Rev. 2014, 43, 6555–6569. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Guan, J. Applications of single-atom catalysts. Nano Res. 2022, 15, 38. [Google Scholar] [CrossRef]
- Li, Z.; Lu, X.; Teng, J.; Zhou, Y.; Zhuang, W. Nonmetal-doping of noble metal-based catalysts for electrocatalysis. Nanoscale 2021, 13, 11314–11324. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Yan, J.; Wu, H.; Zhang, Y.; Liu, S. One-pot fabrication of NiFe2O4 nanoparticles on α-Ni(OH)2 nanosheet for enhanced water oxidation. J. Power Sources 2016, 324, 499–508. [Google Scholar] [CrossRef]
- Zhu, P.; Xiong, X.; Wang, D. Regulations of active moiety in single atom catalysts for electrochemical hydrogen evolution reaction. Nano Res. 2022, 1–24. [Google Scholar] [CrossRef]
- Fan, K.; Ji, Y.; Zou, H.; Zhang, J.; Zhu, B.; Chen, H.; Daniel, Q.; Luo, Y.; Yu, J.; Sun, L. Hollow iron–vanadium composite spheres: A highly efficient iron-based water oxidation electrocatalyst without the need for nickel or cobalt. Angew. Chem. Int. Ed. 2017, 56, 3289–3293. [Google Scholar] [CrossRef]
- Bai, X.; Wang, L.; Nan, B.; Tang, T.; Niu, X.; Guan, J. Atomic manganese coordinated to nitrogen and sulfur for oxygen evolution. Nano Res. 2022, 1–7. [Google Scholar] [CrossRef]
- Tian, T.; Huang, L.; Ai, L.; Jiang, J. Surface anion-rich NiS2 hollow microspheres derived from metal–organic frameworks as a robust electrocatalyst for the hydrogen evolution reaction. J. Mater. Chem. A 2017, 5, 20985–20992. [Google Scholar] [CrossRef]
- Kuang, P.; Tong, T.; Fan, K.; Yu, J. In situ fabrication of Ni–Mo bimetal sulfide hybrid as an efficient electrocatalyst for hydrogen evolution over a wide pH range. ACS Catal. 2017, 7, 6179–6187. [Google Scholar] [CrossRef]
- Xu, D.; Xia, T.; Xu, H.; Fan, W.; Shi, W. Synthesis of ternary spinel MCo2O4 (M = Mn, Zn)/BiVO4 photoelectrodes for photolectrochemical water splitting. Chem. Eng. J. 2020, 392, 124838. [Google Scholar] [CrossRef]
- Digraskar, R.V.; Sapner, V.S.; Mali, S.M.; Narwade, S.S.; Ghule, A.V.; Sathe, B.R. CZTS decorated on graphene oxide as an efficient electrocatalyst for high-performance hydrogen evolution reaction. ACS Omega 2019, 4, 7650–7657. [Google Scholar] [CrossRef]
- Wang, J.; Cui, W.; Liu, Q.; Xing, Z.; Asiri, A.M.; Sun, X. Recent progress in cobalt-based heterogeneous catalysts for electrochemical water splitting. Adv. Mater. 2016, 28, 215–230. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Zhou, Y.; He, J. Two unexplored two-dimensional MSe2 (M = Cd, Zn) structures as the photocatalysts of water splitting and the enhancement of their performances by strain. Vacuum 2020, 182, 109728. [Google Scholar] [CrossRef]
- Weng, B.; Xu, F.; Wang, C.; Meng, W.; Grice, C.R.; Yan, Y. A layered Na1−xNiyFe1−yO2 double oxide oxygen evolution reaction electrocatalyst for highly efficient water-splitting. Energy Environ. Sci. 2017, 10, 121–128. [Google Scholar] [CrossRef]
- Ito, R.; Akatsuka, M.; Ozawa, A.; Kato, Y.; Kawaguchi, Y.; Yamamoto, M.; Tanabe, T.; Yoshida, T. Photocatalytic activity of Ga2O3 supported on Al2O3 for water splitting and CO2 reduction. ACS Omega 2019, 4, 5451–5458. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jessl, S.; Rongé, J.; Copic, D.; Jones, M.A.; Martens, J.; De Volder, M. Honeycomb-shaped carbon nanotube supports for BiVO4 based solar water splitting. Nanoscale 2019, 11, 22964–22970. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-Y.; Yang, J.-S.; Wu, J.-J. Three-dimensional undoped crystalline SnO2 nanodendrite arrays enable efficient charge separation in BiVO4/SnO2 heterojunction photoanodes for photoelectrochemical water splitting. ACS Appl. Energy Mater. 2018, 1, 2143–2149. [Google Scholar] [CrossRef]
- Ghosh, K.; Zhang, W.; Tassinari, F.; Mastai, Y.; Lidor-Shalev, O.; Naaman, R.; Moöllers, P.; Nuürenberg, D.; Zacharias, H.; Wei, J. Controlling chemical selectivity in electrocatalysis with chiral CuO-coated electrodes. J. Phys. Chem. 2019, 123, 3024–3031. [Google Scholar] [CrossRef]
- Abid, A.G.; Manzoor, S.; Usman, M.; Munawar, T.; Nisa, M.U.; Iqbal, F.; Ashiq, M.N.; Najam-ul-Haq, M.; Shah, A.; Imran, M. Scalable synthesis of Sm2O3/Fe2O3 hierarchical oxygen vacancy-based gyroid-inspired morphology: With enhanced electrocatalytic activity for oxygen evolution performance. Energy Fuel. 2021, 35, 17820–17832. [Google Scholar] [CrossRef]
- Sumesh, C.K.; Peter, S.C. Two-dimensional semiconductor transition metal based chalcogenide based heterostructures for water splitting applications. Dalton Trans. 2019, 48, 12772–12802. [Google Scholar] [CrossRef]
- Volokh, M.; Peng, G.; Barrio, J.; Shalom, M. Carbon nitride materials for water splitting photoelectrochemical cells. Angew. Chem. Int. Ed. 2019, 58, 6138–6151. [Google Scholar] [CrossRef]
- Chen, S.; Ma, G.; Wang, Q.; Sun, S.; Hisatomi, T.; Higashi, T.; Wang, Z.; Nakabayashi, M.; Shibata, N.; Pan, Z. Metal selenide photocatalysts for visible-light-driven Z-scheme pure water splitting. J. Mater. Chem. A 2019, 7, 7415–7422. [Google Scholar] [CrossRef] [Green Version]
- Sadaqat, M.; Manzoor, S.; Nisar, L.; Hassan, A.; Tyagi, D.; Shah, J.H.; Ashiq, M.N.; Joya, K.S.; Alshahrani, T.; Najam-ul-Haq, M. Iron doped nickel ditelluride hierarchical nanoflakes arrays directly grown on nickel foam as robust electrodes for oxygen evolution reaction. Electrochim. Acta 2021, 371, 137830. [Google Scholar] [CrossRef]
- Sadaqat, M.; Nisar, L.; Hussain, F.; Ashiq, M.N.; Shah, A.; Ehsan, M.F.; Najam-Ul-Haq, M.; Joya, K. Zinc-telluride nanospheres as an efficient water oxidation electrocatalyst displaying a low overpotential for oxygen evolution. J. Mater. Chem. A 2019, 7, 26410–26420. [Google Scholar] [CrossRef]
- Xiong, X.; You, C.; Liu, Z.; Asiri, A.M.; Sun, X. Co-doped CuO nanoarray: An efficient oxygen evolution reaction electrocatalyst with enhanced activity. ACS Sust. Chem. Eng. 2018, 6, 2883–2887. [Google Scholar] [CrossRef]
- Panda, C.; Menezes, P.W.; Zheng, M.; Orthmann, S.; Driess, M. In situ formation of nanostructured core–shell Cu3N–CuO to promote alkaline water electrolysis. ACS Energy Lett. 2019, 4, 747–754. [Google Scholar] [CrossRef]
- Liu, E.; Zhang, X.; Xue, P.; Fan, J.; Hu, X. Carbon membrane bridged ZnSe and TiO2 nanotube arrays: Fabrication and promising application in photoelectrochemical water splitting. Int. J. Hydrogen Energy 2020, 45, 9635–9647. [Google Scholar] [CrossRef]
- Chakraborty, B.; Beltrán-Suito, R.; Hlukhyy, V.; Schmidt, J.; Menezes, P.W.; Driess, M. Crystalline copper selenide as a reliable non-noble electro (pre) catalyst for overall water splitting. ChemSusChem 2020, 13, 3222–3229. [Google Scholar] [CrossRef]
- Wang, P.; Pu, Z.; Li, W.; Zhu, J.; Zhang, C.; Zhao, Y.; Mu, S. Coupling NiSe2-Ni2P heterostructure nanowrinkles for highly efficient overall water splitting. J. Catal. 2019, 377, 600–608. [Google Scholar] [CrossRef]
- Wu, L.L.; Wang, Q.S.; Li, J.; Long, Y.; Liu, Y.; Song, S.Y.; Zhang, H. Co9S8 Nanoparticles-embedded N/S-codoped carbon nanofibers derived from metal–organic framework-wrapped CdS nanowires for efficient oxygen evolution reaction. Small 2018, 14, 1704035. [Google Scholar] [CrossRef]
- Wang, Y.; Kong, B.; Zhao, D.; Wang, H.; Selomulya, C. Strategies for developing transition metal phosphides as heterogeneous electrocatalysts for water splitting. Nano Today 2017, 15, 26–55. [Google Scholar] [CrossRef]
- Kroon, R. Nanoscience and the Scherrer equation versus the’Scherrer-Gottingen equation. S. Afr. J. Sci. 2013, 109, 1–2. [Google Scholar] [CrossRef] [Green Version]
- Shao, D.; Li, P.; Zhang, R.; Zhao, C.; Wang, D.; Zhao, C. One-step preparation of Fe-doped Ni3S2/rGO@ NF electrode and its superior OER performances. Int. J. Hydrogen Energy 2019, 44, 2664–2674. [Google Scholar] [CrossRef]
- Liu, T.; Liang, Y.; Liu, Q.; Sun, X.; He, Y.; Asiri, A.M. Electrodeposition of cobalt-sulfide nanosheets film as an efficient electrocatalyst for oxygen evolution reaction. Electrochem. Commun. 2015, 60, 92–96. [Google Scholar] [CrossRef]
- Trotochaud, L.; Ranney, J.K.; Williams, K.N.; Boettcher, S.W. Solution-cast metal oxide thin film electrocatalysts for oxygen evolution. J. Amer. Chem. Soc. 2012, 134, 17253–17261. [Google Scholar] [CrossRef] [PubMed]
- Sun, M.; Gao, R.-T.; Liu, X.; Gao, R.; Wang, L. Manganese-based oxygen evolution catalysts boosting stable solar-driven water splitting: MnSe as an intermetallic phase. J. Mater. Chem. A 2020, 8, 25298–25305. [Google Scholar] [CrossRef]
- Liang, H.; Meng, F.; Cabán-Acevedo, M.; Li, L.; Forticaux, A.; Xiu, L.; Wang, Z.; Jin, S. Hydrothermal continuous flow synthesis and exfoliation of NiCo layered double hydroxide nanosheets for enhanced oxygen evolution catalysis. Nano Lett. 2015, 15, 1421–1427. [Google Scholar] [CrossRef]
- Wang, L.; Wu, Y.; Cao, R.; Ren, L.; Chen, M.; Feng, X.; Zhou, J.; Wang, B. Interfaces, Fe/Ni metal–organic frameworks and their binder-free thin films for efficient oxygen evolution with low overpotential. ACS Appl. Mater. Interf. 2016, 8, 16736–16743. [Google Scholar] [CrossRef]
- Dutta, A.; Samantara, A.K.; Dutta, S.K.; Jena, B.K.; Pradhan, N. Surface-oxidized dicobalt phosphide nanoneedles as a nonprecious, durable, and efficient OER catalyst. ACS Energy Lett. 2016, 1, 169–174. [Google Scholar] [CrossRef]
- Chen, X.; Wang, H.; Meng, R.; Xia, B.; Ma, Z. Cadmium hydroxide: A missing non-noble metal hydroxide electrocatalyst for the oxygen evolution reaction. ACS Appl. Energy Mater. 2020, 3, 1305–1310. [Google Scholar] [CrossRef]
- Lu, X.-F.; Liao, P.-Q.; Wang, J.-W.; Wu, J.-X.; Chen, X.-W.; He, C.-T.; Zhang, J.-P.; Li, G.-R.; Chen, X.-M. An alkaline-stable, metal hydroxide mimicking metal–organic framework for efficient electrocatalytic oxygen evolution. J. Amer. Chem. Soc. 2016, 138, 8336–8339. [Google Scholar] [CrossRef]
- Dai, F.; Fan, W.; Bi, J.; Jiang, P.; Liu, D.; Zhang, X.; Lin, H.; Gong, C.; Wang, R.; Zhang, L. A lead–porphyrin metal–organic framework: Gas adsorption properties and electrocatalytic activity for water oxidation. Dalton Trans. 2016, 45, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Majhi, K.; Karfa, P.; De, S.; Madhuri, R. Hydrothermal synthesis of zinc cobalt telluride nanorod towards oxygen evolution reaction (OER). IOP Conf. Ser. Mater. Sci. Eng. 2019, 577, 012076. [Google Scholar] [CrossRef]
- Li, Z.; Wang, X.; Wang, X.; Lin, Y.; Meng, A.; Yang, L.; Li, Q. Mn-Cd-S@ amorphous-Ni3S2 hybrid catalyst with enhanced photocatalytic property for hydrogen production and electrocatalytic OER. Appl. Surf. Sci. 2019, 491, 799–806. [Google Scholar] [CrossRef]

| Sr. No. | Material Name | Overpotential mV | Tafel mV dec−1 | Electrolyte | Electrode Type | Ref. |
|---|---|---|---|---|---|---|
| 1 | Sm2O3/Fe2O3 | 272 | 75 | 1.0M KOH | Graphite Pencil | [29] |
| 2 | Fe dopedNi2S3/rGO | 247 | 63 | 1.0M KOH | Ni-foam | [43] |
| 3 | Co-S/Ti-mesh | 361 | 64 | 1.0M KOH | Graphite | [44] |
| 4 | CoOx film | 403 | 42 | 1.0 MKOH | Glass electrode | [45] |
| 5 | MnFeSe | 247 | 35 | 1.0 MKOH | Ni foam | [46] |
| 6 | NiCo LDH | 367 | 40 | 1.0 MKOH | Carbon paper | [47] |
| 7 | Fe/Ni-BTC@NF | 270 | 47 | 0.1M KOH | Ni-foam | [48] |
| 8 | Co2P nanoneedles | 310 | 50 | 1.0M KOH | Glassy Carbon | [49] |
| 9 | Cd(OH)2 | 266 | 47 | 1.0M KOH | Ni-foam | [50] |
| 10 | MAFX27-OH | 387 | 60 | 1.0M KOH | Glassy Carbon | [51] |
| 11 | Pb-TCPP | 470 | 106 | 1.0M KOH | Glassy Carbon | [52] |
| 12 | ZnCoTe | 221 | 91 | 1.0M KOH | Graphite pencil | [53] |
| 13 | Mn-Cd-S@Ni3S2 | 333 | 150 | 1.0M KOH | Ni-foam | [54] |
| 14 | In2MnSe4 | 259 | 86 | 1.0M KOH | NF | Present work |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manzoor, S.; Trukhanov, S.V.; Ansari, M.N.; Abdullah, M.; Alruwaili, A.; Trukhanov, A.V.; Khandaker, M.U.; Idris, A.M.; El-Nasser, K.S.; Taha, T.A. Flowery ln2MnSe4 Novel Electrocatalyst Developed via Anion Exchange Strategy for Efficient Water Splitting. Nanomaterials 2022, 12, 2209. https://doi.org/10.3390/nano12132209
Manzoor S, Trukhanov SV, Ansari MN, Abdullah M, Alruwaili A, Trukhanov AV, Khandaker MU, Idris AM, El-Nasser KS, Taha TA. Flowery ln2MnSe4 Novel Electrocatalyst Developed via Anion Exchange Strategy for Efficient Water Splitting. Nanomaterials. 2022; 12(13):2209. https://doi.org/10.3390/nano12132209
Chicago/Turabian StyleManzoor, Sumaira, Sergei V. Trukhanov, Mohammad Numair Ansari, Muhammad Abdullah, Atalah Alruwaili, Alex V. Trukhanov, Mayeen Uddin Khandaker, Abubakr M. Idris, Karam S. El-Nasser, and Taha AbdelMohaymen Taha. 2022. "Flowery ln2MnSe4 Novel Electrocatalyst Developed via Anion Exchange Strategy for Efficient Water Splitting" Nanomaterials 12, no. 13: 2209. https://doi.org/10.3390/nano12132209
APA StyleManzoor, S., Trukhanov, S. V., Ansari, M. N., Abdullah, M., Alruwaili, A., Trukhanov, A. V., Khandaker, M. U., Idris, A. M., El-Nasser, K. S., & Taha, T. A. (2022). Flowery ln2MnSe4 Novel Electrocatalyst Developed via Anion Exchange Strategy for Efficient Water Splitting. Nanomaterials, 12(13), 2209. https://doi.org/10.3390/nano12132209