Preparation of Lambda-Cyhalothrin-Loaded Chitosan Nanoparticles and Their Bioactivity against Drosophila suzukii
Abstract
:1. Introduction
2. Materials and Methods
2.1. CS-LC Nanoparticles
2.1.1. Materials
2.1.2. Experimental Design
2.1.3. Preparation of CS-LC Nanoparticles
2.2. Characterization of Nanoparticles
2.2.1. Physical Properties
Yield Percentage, Pesticide Loading and Entrapment Efficiency
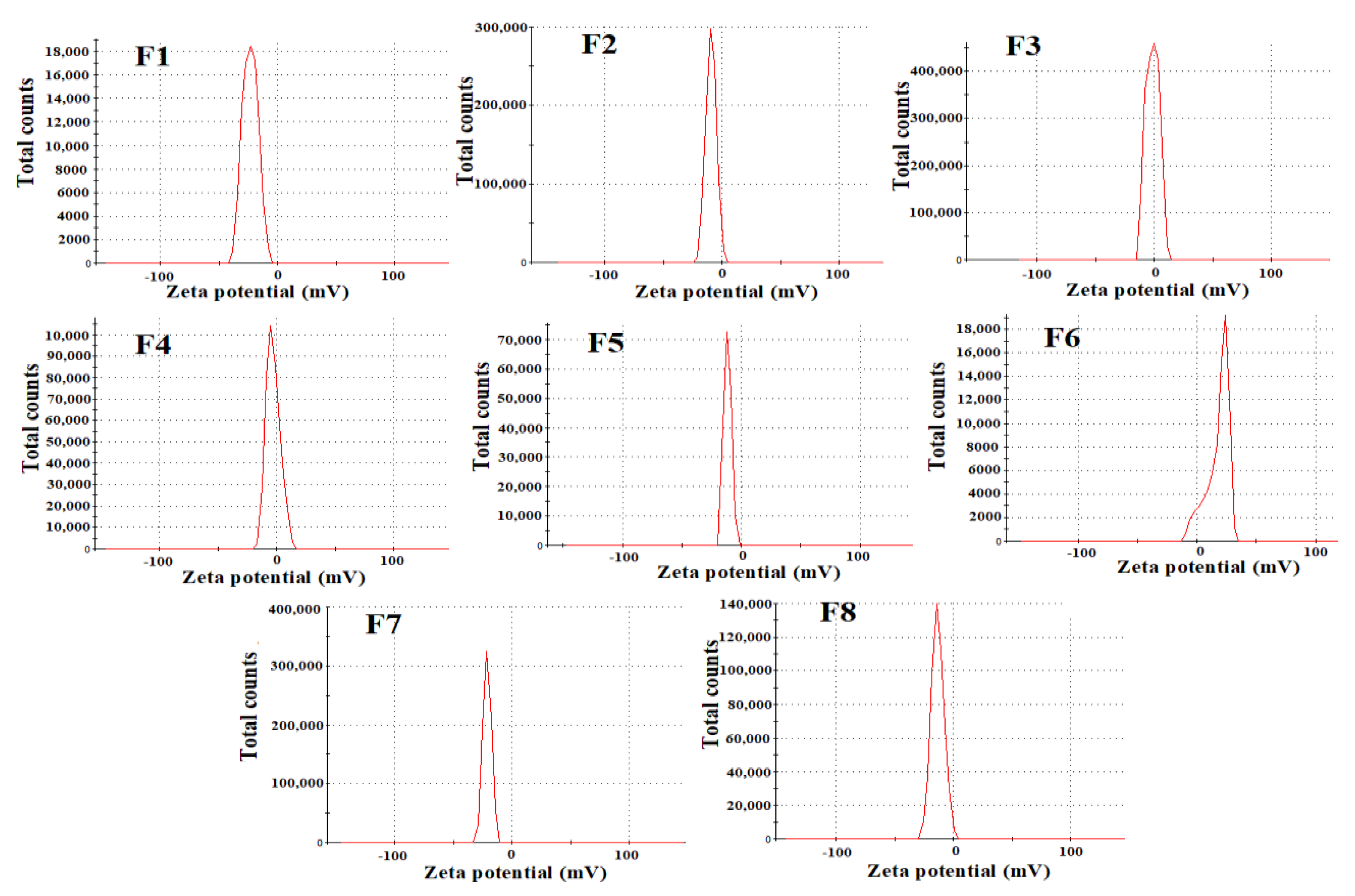
Particle Size, Polydispersity Index and Zeta Potential
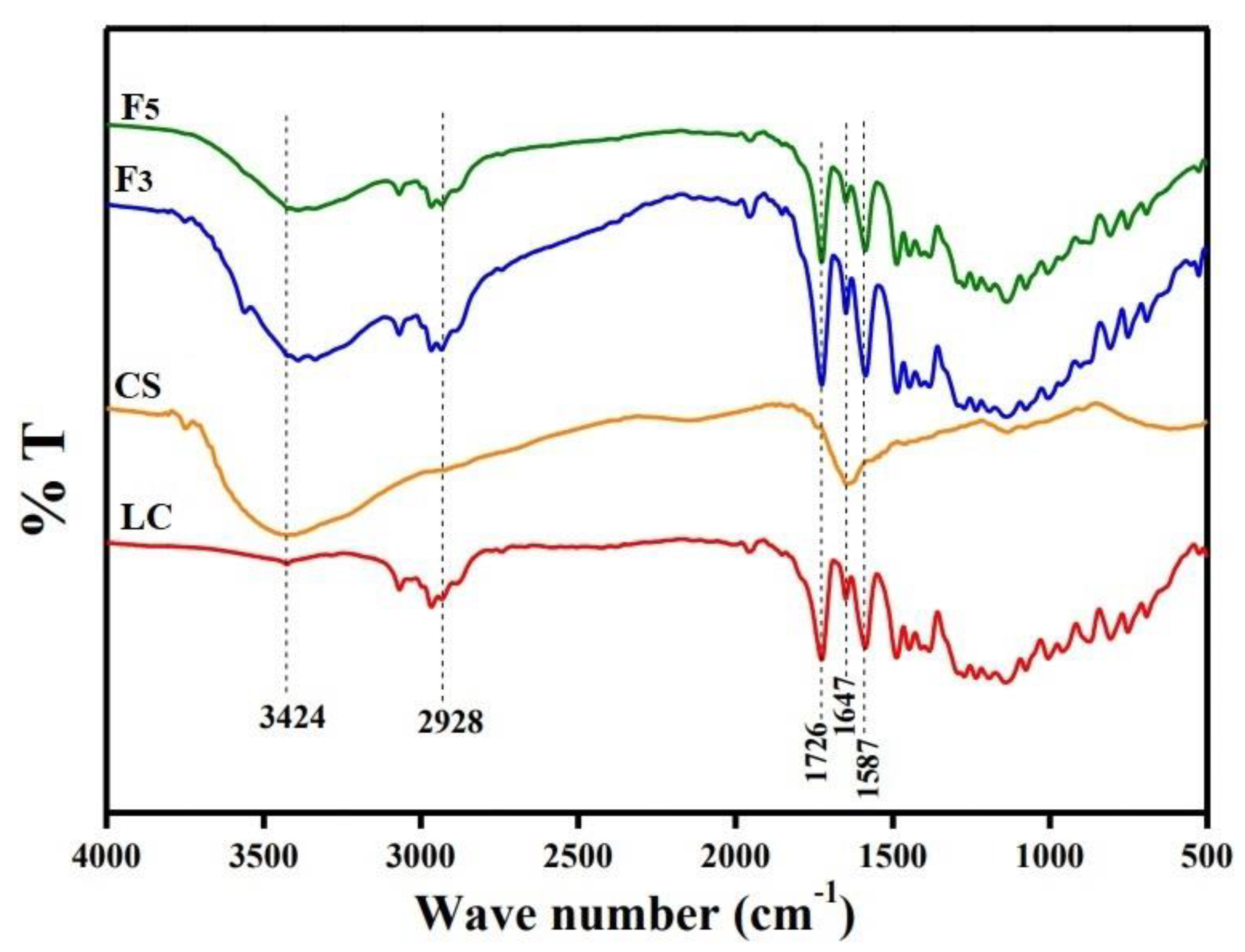
2.2.2. Morphological and FTIR Examination
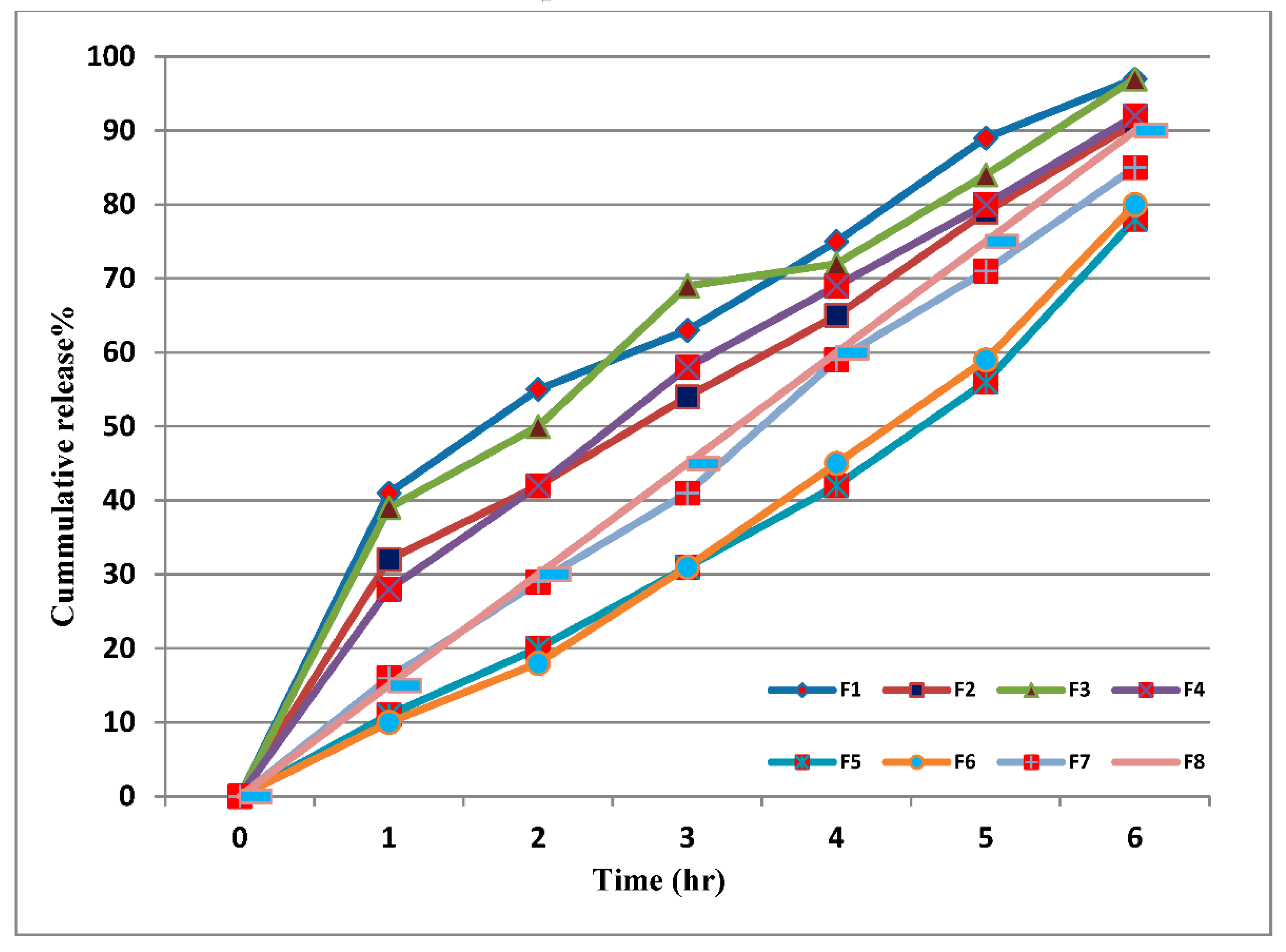
2.3. In Vitro Release Study
2.4. Bioactivity of Nanoparticles
2.4.1. Laboratory SWD Colony
2.4.2. Laboratory Bioassays
2.5. Statistical Analysis
3. Results
3.1. Characterizations of Nanoparticles
3.1.1. Physical Properties
3.1.2. Morphological Characterization
3.1.3. FTIR Analysis
3.2. In Vitro Release Study
3.3. Bioactivity of Nanoparticles
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sharma, S.; Kooner, R.; Arora, R. Insect pests and crop losses. In Breeding Insect Resistant Crops for Sustainable Agriculture; Springer: Berlin/Heidelberg, Germany, 2017; pp. 45–66. [Google Scholar]
- Ebadollahi, A.; Jalali Sendi, J.; Setzer, W.N.; Changbunjong, T. Encapsulation of Eucalyptus largiflorens Essential Oil by Mesoporous Silicates for Effective Control of the Cowpea Weevil, Callosobruchus maculatus (Fabricius) (Coleoptera: Chrysomelidae). Molecules 2022, 27, 3531. [Google Scholar] [CrossRef] [PubMed]
- Ebadollahi, A.; Naseri, B.; Abedi, Z.; Setzer, W.N. Chemical Profiles and Insecticidal Potential of Essential Oils Isolated from Four Thymus Species against Rhyzopertha dominica (F.). Plants 2022, 11, 1567. [Google Scholar] [CrossRef] [PubMed]
- Haye, T.; Girod, P.; Cuthbertson, A.; Wang, X.; Daane, K.; Hoelmer, K.; Baroffio, C.; Zhang, J.; Desneux, N. Current SWD IPM tactics and their practical implementation in fruit crops across different regions around the world. J. Pest Sci. 2016, 89, 643–651. [Google Scholar] [CrossRef]
- Shawer, R.; Donati, I.; Cellini, A.; Spinelli, F.; Mori, N. Insecticidal Activity of Photorhabdus luminescens against Drosophila suzukii. Insects 2018, 9, 148. [Google Scholar] [CrossRef] [PubMed]
- Shawer, R. Chemical control of Drosophila suzukii. In Drosophila suzukii Management; Springer: Berlin/Heidelberg, Germany, 2020; pp. 133–142. [Google Scholar]
- Shawer, R.; El-Shazly, M.M.; Khider, A.M.; Baeshen, R.S.; Hikal, W.M.; Kordy, A.M. Botanical Oils Isolated from Simmondsia chinensis and Rosmarinus officinalis Cultivated in Northern Egypt: Chemical Composition and Insecticidal Activity against Sitophilus oryzae (L.) and Tribolium castaneum (Herbst). Molecules 2022, 27, 4383. [Google Scholar] [CrossRef]
- Enkerli, J.; Widmer, F.; Keller, S. Long-term field persistence of Beauveria brongniartii strains applied as biocontrol agents against European cockchafer larvae in Switzerland. Biol. Control 2004, 29, 115–123. [Google Scholar] [CrossRef]
- Sahayaraj, K.; Namasivayam, S.K.R. Mass production of entomopathogenic fungi using agricultural products and by products. Afr. J. Biotechnol. 2008, 7, 1907–1910. [Google Scholar]
- Karungi, J.; Kyamanywa, S.; Adipala, E.; Erbaugh, M. Pesticide utilisation, regulation and future prospects in small scale horticultural crop production systems in a developing country. In Pesticides in the Modern World-Pesticides Use and Management; InTech: Rijeka, Croatia, 2011. [Google Scholar] [CrossRef]
- Shawer, R.; Abou-Elnasar, H.; El-Dardiry, M.M.; Kordy, A.M. Biochemical and Toxicological Effects of Emamectin Bonzoate against Spodoptera littoralis (Boisd.)(Lepidoptera: Noctuidae). Egypt. Acad. J. Biol. Sci. F. Toxicol. Pest Control 2022, 14, 1–11. [Google Scholar] [CrossRef]
- Ismail, I.A.; Qari, S.H.; Shawer, R.; Elshaer, M.M.; Dessoky, E.S.; Youssef, N.H.; Hamad, N.A.; Abdelkhalek, A.; Elsamra, I.A.; Behiry, S.I. The Application of Pomegranate, Sugar Apple, and Eggplant Peel Extracts Suppresses Aspergillus flavus Growth and Aflatoxin B1 Biosynthesis Pathway. Horticulturae 2021, 7, 558. [Google Scholar] [CrossRef]
- Shawer, R.; El-Shazly, M.M.; Khider, A.M.; Kordy, A.M. Chemical Composition of Five Botanical Powders and Their Insecticidal Activity Against the Rice Weevil, Sitophilus oryzae on Wheat Crop. Egypt. Acad. J. Biol. Sci. G. Microbiol. 2020, 12, 125–136. [Google Scholar] [CrossRef]
- Sundar, N.S.; Karthi, S.; Sivanesh, H.; Stanley-Raja, V.; Chanthini, K.M.-P.; Ramasubramanian, R.; Ramkumar, G.; Ponsankar, A.; Narayanan, K.R.; Vasantha-Srinivasan, P. Efficacy of Precocene I from Desmosstachya bipinnata as an Effective Bioactive Molecules against the Spodoptera litura Fab. and Its Impact on Eisenia fetida Savigny. Molecules 2021, 26, 6384. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.; Mukherjee, A.; Jaiswal, D.K.; de Araujo Pereira, A.P.; Prasad, R.; Sharma, M.; Kuhad, R.C.; Shukla, A.C.; Verma, J.P. Advances and future prospects of pyrethroids: Toxicity and microbial degradation. Sci. Total Environ. 2022, 829, 154561. [Google Scholar] [CrossRef] [PubMed]
- Chrustek, A.; Hołyńska-Iwan, I.; Dziembowska, I.; Bogusiewicz, J.; Wróblewski, M.; Cwynar, A.; Olszewska-Słonina, D. Current research on the safety of pyrethroids used as insecticides. Medicina 2018, 54, 61. [Google Scholar] [CrossRef] [PubMed]
- KB, A.A.; HM, A.R. Lambda, the pyrethroid insecticide as a mutagenic agent in both somatic and germ cells. J. Am. Sci. 2010, 6, 317–326. [Google Scholar]
- Guida, Y.; Pozo, K.; de Carvalho, G.O.; Capella, R.; Targino, A.C.; Torres, J.P.M.; Meire, R.O. Occurrence of pyrethroids in the atmosphere of urban areas of Southeastern Brazil: Inhalation exposure and health risk assessment. Environ. Pollut. 2021, 290, 118020. [Google Scholar] [CrossRef]
- Zaim, M.; Aitio, A.; Nakashima, N. Safety of pyrethroid-treated mosquito nets. Med. Vet. Entomol. 2000, 14, 1–5. [Google Scholar] [CrossRef]
- Gnanguenon, V.; Agossa, F.R.; Badirou, K.; Govoetchan, R.; Anagonou, R.; Oke-Agbo, F.; Azondekon, R.; AgbanrinYoussouf, R.; Attolou, R.; Tokponnon, F.T. Malaria vectors resistance to insecticides in Benin: Current trends and mechanisms involved. Parasites Vectors 2015, 8, 223. [Google Scholar] [CrossRef]
- He, L.-M.; Troiano, J.; Wang, A.; Goh, K. Environmental chemistry, ecotoxicity, and fate of lambda-cyhalothrin. In Reviews of Environmental Contamination and Toxicology; Springer: Berlin/Heidelberg, Germany, 2008; pp. 71–91. [Google Scholar]
- IRAC, Insecticide Resistance Action Committee, IRAC Mode of Action Classification Scheme MOA CLASSIFICATION, Ver. 10.3. 2022. Available online: https://irac-online.org/ (accessed on 1 June 2022).
- Cui, B.; Feng, L.; Pan, Z.; Yu, M.; Zeng, Z.; Sun, C.; Zhao, X.; Wang, Y.; Cui, H. Evaluation of stability and biological activity of solid nanodispersion of lambda-cyhalothrin. PLoS ONE 2015, 10, e0135953. [Google Scholar] [CrossRef]
- Kah, M.; Hofmann, T. Nanopesticide research: Current trends and future priorities. Environ. Int. 2014, 63, 224–235. [Google Scholar] [CrossRef]
- Balaure, P.C.; Gudovan, D.; Gudovan, I. Nanopesticides: A new paradigm in crop protection. In New Pesticides and Soil Sensors; Elsevier: Amsterdam, The Netherlands, 2017; pp. 129–192. [Google Scholar]
- Bharani, R.A.; Namasivayam, S.K.R.; Shankar, S.S. Biocompatible Chitosan Nanoparticles Incorporated Pesticidal Protein Beauvericin (Csnp-Bv) Preparation for the Improved Pesticidal Activity Against Major Groundnut Defoliator Spodoptera Litura (Fab.)(Lepidoptera; Noctuidae). Int. J. Chem.Tech. Res. 2014, 6, 5007–5012. [Google Scholar]
- Scott, N.R. Nanotechnology Opportunities in Agriculture and Food Systems; Biological & Environmental Engineering, University NSF Cornell Nanoscale Science & Engineering Grantees Conference: Arlington, VA, USA, 2007. [Google Scholar]
- Bouwmeester, H.; Dekkers, S.; Noordam, M.Y.; Hagens, W.I.; Bulder, A.S.; De Heer, C.; Ten Voorde, S.E.; Wijnhoven, S.W.; Marvin, H.J.; Sips, A.J. Review of health safety aspects of nanotechnologies in food production. Regul. Toxicol. Pharmacol. 2009, 53, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, P.L.; Xiang, X.; Heiden, P. Chitosan nanoparticle based delivery systems for sustainable agriculture. Int. J. Biol. Macromol. 2015, 77, 36–51. [Google Scholar] [CrossRef] [PubMed]
- Abd El-Monaem, E.M.; Eltaweil, A.S.; Elshishini, H.M.; Hosny, M.; Abou Alsoaud, M.M.; Attia, N.F.; El-Subruiti, G.M.; Omer, A.M. Sustainable adsorptive removal of antibiotic residues by chitosan composites: An insight into current developments and future recommendations. Arab. J. Chem. 2022, 15, 103743. [Google Scholar] [CrossRef]
- George, M.; Abraham, T.E. Polyionic hydrocolloids for the intestinal delivery of protein drugs: Alginate and chitosan—A review. J. Control. Release 2006, 114, 1–14. [Google Scholar] [CrossRef]
- Stoica, R.; Şomoghi, R.; Ion, R. Preparation of Chitosan-Tripolyphosphate Nanoparticles for the Encapsulation of Polyphenols Extracted from Rose Hips. Dig. J. Nanomater. Biostructures (DJNB) 2013, 8, 955–963. [Google Scholar]
- Hoang, N.H.; Le Thanh, T.; Sangpueak, R.; Treekoon, J.; Saengchan, C.; Thepbandit, W.; Papathoti, N.K.; Kamkaew, A.; Buensanteai, N. Chitosan Nanoparticles-Based Ionic Gelation Method: A Promising Candidate for Plant Disease Management. Polymers 2022, 14, 662. [Google Scholar] [CrossRef] [PubMed]
- Pedroso-Santana, S.; Fleitas-Salazar, N. Ionotropic gelation method in the synthesis of nanoparticles/microparticles for biomedical purposes. Polym. Int. 2020, 69, 443–447. [Google Scholar] [CrossRef]
- Eltaweil, A.S.; Abd El-Monaem, E.M.; Elshishini, H.M.; El-Aqapa, H.G.; Hosny, M.; Abdelfatah, A.M.; Ahmed, M.S.; Hammad, E.N.; El-Subruiti, G.M.; Fawzy, M. Recent developments in alginate-based adsorbents for removing phosphate ions from wastewater: A review. RSC Adv. 2022, 12, 8228–8248. [Google Scholar] [CrossRef]
- Bolda, M.P.; Goodhue, R.E.; Zalom, F.G. Spotted wing drosophila: Potential economic impact of a newly established pest. Agric. Resour. Econ. Update 2010, 13, 5–8. [Google Scholar]
- Walsh, D.B.; Bolda, M.P.; Goodhue, R.E.; Dreves, A.J.; Lee, J.; Bruck, D.J.; Walton, V.M.; O’Neal, S.D.; Zalom, F.G. Drosophila suzukii (Diptera: Drosophilidae): Invasive pest of ripening soft fruit expanding its geographic range and damage potential. J. Integr. Pest Manag. 2011, 2, G1–G7. [Google Scholar] [CrossRef]
- Goodhue, R.E.; Bolda, M.; Farnsworth, D.; Williams, J.C.; Zalom, F.G. Spotted wing drosophila infestation of California strawberries and raspberries: Economic analysis of potential revenue losses and control costs. Pest Manag. Sci. 2011, 67, 1396–1402. [Google Scholar] [CrossRef] [PubMed]
- Deprá, M.; Poppe, J.L.; Schmitz, H.J.; De Toni, D.C.; Valente, V.L. The first records of the invasive pest Drosophila suzukii in the South American continent. J. Pest Sci. 2014, 87, 379–383. [Google Scholar] [CrossRef]
- EPPO, EPPO Global Database. Drosophila Suzukii. 2018. Available online: https://gd.eppo.int/taxon/DROSSU/distribution (accessed on 18 May 2020).
- Cini, A.; Ioriatti, C.; Anfora, G. A review of the invasion of Drosophila suzukii in Europe and a draft research agenda for integrated pest management. Bull. Insectology 2012, 65, 149–160. [Google Scholar]
- Asplen, M.K.; Anfora, G.; Biondi, A.; Choi, D.-S.; Chu, D.; Daane, K.M.; Gibert, P.; Gutierrez, A.P.; Hoelmer, K.A.; Hutchison, W.D. Invasion biology of spotted wing Drosophila (Drosophila suzukii): A global perspective and future priorities. J. Pest Sci. 2015, 88, 469–494. [Google Scholar] [CrossRef]
- Ioriatti, C.; Boselli, M.; Caruso, S.; Galassi, T.; Gottardello, A.; Grassi, A.; Tonina, L.; Vaccari, G.; Mori, N. Approccio integrato per la difesa dalla Drosophila suzukii. Riv. Di Fruttic. E Di Ortofloric. 2015, 77, 32–37. [Google Scholar]
- Mori, N.; Tonina, L.; Sancassani, M.; Colombari, F.; Dall’Ara, P.; Cero, M.d.; Marchesini, E. Integrated pest management approaches against Drosophila suzukii. Italus Hortus 2019, 26, 67–74. [Google Scholar] [CrossRef]
- Beers, E.H.; Van Steenwyk, R.A.; Shearer, P.W.; Coates, W.W.; Grant, J.A. Developing Drosophila suzukii management programs for sweet cherry in the western United States. Pest Manag. Sci. 2011, 67, 1386–1395. [Google Scholar] [CrossRef]
- Bruck, D.J.; Bolda, M.; Tanigoshi, L.; Klick, J.; Kleiber, J.; DeFrancesco, J.; Gerdeman, B.; Spitler, H. Laboratory and field comparisons of insecticides to reduce infestation of Drosophila suzukii in berry crops. Pest Manag. Sci. 2011, 67, 1375–1385. [Google Scholar] [CrossRef]
- Van Timmeren, S.; Isaacs, R. Control of spotted wing drosophila, Drosophila suzukii, by specific insecticides and by conventional and organic crop protection programs. Crop Prot. 2013, 54, 126–133. [Google Scholar] [CrossRef]
- Shawer, R.; Tonina, L.; Gariberti, E.; Mori, N. Efficacy of insecticides against Drosophila suzukii on cherries. In XVIII. International Plant Protection Congress; DPG: Berlin, Germany, 2015. [Google Scholar]
- Shawer, R.; Tonina, L.; Tirello, P.; Duso, C.; Mori, N. Laboratory and field trials to identify effective chemical control strategies for integrated management of Drosophila suzukii in European cherry orchards. Crop Prot. 2018, 103, 73–80. [Google Scholar] [CrossRef]
- Shawer, R. Impact of Traditional Pesticides and New Controlled Release Formulations on Drosophila suzukii. Ph.D. Thesis, Padova University, Padova, Italy, 2017. [Google Scholar]
- Haviland, D.R.; Beers, E.H. Chemical control programs for Drosophila suzukii that comply with international limitations on pesticide residues for exported sweet cherries. J. Integr. Pest Manag. 2012, 3, F1–F6. [Google Scholar] [CrossRef]
- Wiman, N.G.; Dalton, D.T.; Anfora, G.; Biondi, A.; Chiu, J.C.; Daane, K.M.; Gerdeman, B.; Gottardello, A.; Hamby, K.A.; Isaacs, R. Drosophila suzukii population response to environment and management strategies. J. Pest Sci. 2016, 89, 653–665. [Google Scholar] [CrossRef]
- de Pinho Neves, A.L.; Milioli, C.C.; Müller, L.; Riella, H.G.; Kuhnen, N.C.; Stulzer, H.K. Factorial design as tool in chitosan nanoparticles development by ionic gelation technique. Colloids Surf. A Physicochem. Eng. Asp. 2014, 445, 34–39. [Google Scholar] [CrossRef]
- Montgomery, J.D. Social networks and labor-market outcomes: Toward an economic analysis. Am. Econ. Rev. 1991, 81, 1408–1418. [Google Scholar]
- Mohammadpour Dounighi, N.; Eskandari, R.; Avadi, M.; Zolfagharian, H.; Mir Mohammad Sadeghi, A.; Rezayat, M. Preparation and in vitro characterization of chitosan nanoparticles containing Mesobuthus eupeus scorpion venom as an antigen delivery system. J. Venom. Anim. Toxins Incl. Trop. Dis. 2012, 18, 44–52. [Google Scholar] [CrossRef]
- Werle, M.; Takeuchi, H.; Bernkop-Schnürch, A. Modified chitosans for oral drug delivery. J. Pharm. Sci. 2009, 98, 1643–1656. [Google Scholar] [CrossRef]
- Calvo, P.; Remunan-Lopez, C.; Vila-Jato, J.L.; Alonso, M. Novel hydrophilic chitosan-polyethylene oxide nanoparticles as protein carriers. J. Appl. Polym. Sci. 1997, 63, 125–132. [Google Scholar] [CrossRef]
- Calvo, P.; Remuñan-López, C.; Vila-Jato, J.L.; Alonso, M.J. Chitosan and chitosan/ethylene oxide-propylene oxide block copolymer nanoparticles as novel carriers for proteins and vaccines. Pharm. Res. 1997, 14, 1431–1436. [Google Scholar] [CrossRef]
- Gazori, T.; Khoshayand, M.R.; Azizi, E.; Yazdizade, P.; Nomani, A.; Haririan, I. Evaluation of Alginate/Chitosan nanoparticles as antisense delivery vector: Formulation, optimization and in vitro characterization. Carbohydr. Polym. 2009, 77, 599–606. [Google Scholar] [CrossRef]
- Leelapornpisid, P.; Leesawat, P.; Natakarnkitkul, S.; Rattanapanadda, P. Application of chitosan for preparation of arbutin nanoparticles as skin whitening. J. Met. Mater. Miner. 2010, 20, 101–105. [Google Scholar]
- Masalova, O.; Kulikouskaya, V.; Shutava, T.; Agabekov, V. Alginate and Chitosan gel nanoparticles for efficient protein entrapment. Phys. Procedia 2013, 40, 69–75. [Google Scholar] [CrossRef]
- Açikgöz, M.; Kaş, H.; Orman, M.; Hincal, A. Chitosan microspheres of diclofenac sodium: I. application of factorial design and evaluation of release kinetics. J. Microencapsul. 1996, 13, 141–159. [Google Scholar] [CrossRef] [PubMed]
- Barzegar-Jalali, M. Kinetic analysis of drug release from nanoparticles. J. Pharm. Pharm. Sci. 2008, 11, 167–177. [Google Scholar] [CrossRef]
- England, C.G.; Miller, M.C.; Kuttan, A.; Trent, J.O.; Frieboes, H.B. Release kinetics of paclitaxel and cisplatin from two and three layered gold nanoparticles. Eur. J. Pharm. Biopharm. 2015, 92, 120–129. [Google Scholar] [CrossRef]
- Cuthbertson, A.G.; Collins, D.A.; Blackburn, L.F.; Audsley, N.; Bell, H.A. Preliminary screening of potential control products against Drosophila suzukii. Insects 2014, 5, 488–498. [Google Scholar] [CrossRef] [PubMed]
- Hosny, M.; Fawzy, M.; El-Fakharany, E.M.; Omer, A.M.; Abd El-Monaem, E.M.; Khalifa, R.E.; Eltaweil, A.S. Biogenic synthesis, characterization, antimicrobial, antioxidant, antidiabetic, and catalytic applications of platinum nanoparticles synthesized from Polygonum salicifolium leaves. J. Environ. Chem. Eng. 2022, 10, 106806. [Google Scholar] [CrossRef]
- Shen, Y.; Zhu, H.; Cui, J.; Wang, A.; Zhao, X.; Cui, B.; Wang, Y.; Cui, H. Construction of lambda-cyhalothrin nano-delivery system with a high loading content and controlled-release property. Nanomaterials 2018, 8, 1016. [Google Scholar] [CrossRef]
- Pan, Z.; Cui, B.; Zeng, Z.; Feng, L.; Liu, G.; Cui, H.; Pan, H. Lambda-cyhalothrin nanosuspension prepared by the melt emulsification-high pressure homogenization method. J. Nanomater. 2015, 2015, 263. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Cui, B.; Guo, L.; Wang, A.; Zhao, X.; Wang, Y.; Sun, C.; Zeng, Z.; Zhi, H.; Chen, H. Fabrication and evaluation of lambda-cyhalothrin nanosuspension by one-step melt emulsification technique. Nanomaterials 2019, 9, 145. [Google Scholar] [CrossRef]
- Severino, P.; Fangueiro, J.F.; Chaud, M.V.; Cordeiro, J.; Silva, A.M.; Souto, E.B. Advances in nanobiomaterials for topical administrations: New galenic and cosmetic formulations. In Nanobiomaterials in Galenic Formulations and Cosmetics; Elsevier: Amsterdam, The Netherlands, 2016; pp. 1–23. [Google Scholar]
- Fàbregas, A.; Miñarro, M.; García-Montoya, E.; Pérez-Lozano, P.; Carrillo, C.; Sarrate, R.; Sánchez, N.; Ticó, J.; Suñé-Negre, J. Impact of physical parameters on particle size and reaction yield when using the ionic gelation method to obtain cationic polymeric chitosan–tripolyphosphate nanoparticles. Int. J. Pharm. 2013, 446, 199–204. [Google Scholar] [CrossRef]
- Yang, H.-C.; Hon, M.-H. The effect of the molecular weight of chitosan nanoparticles and its application on drug delivery. Microchem. J. 2009, 92, 87–91. [Google Scholar] [CrossRef]
- Guan, H.; Chi, D.; Yu, J.; Li, X. A novel photodegradable insecticide: Preparation, characterization and properties evaluation of nano-Imidacloprid. Pestic. Biochem. Physiol. 2008, 92, 83–91. [Google Scholar] [CrossRef]
- Qian, K.; Shi, T.; Tang, T.; Zhang, S.; Liu, X.; Cao, Y. Preparation and characterization of nano-sized calcium carbonate as controlled release pesticide carrier for validamycin against Rhizoctonia solani. Microchim. Acta 2011, 173, 51–57. [Google Scholar] [CrossRef]
- Paulraj, M.G.; Ignacimuthu, S.; Gandhi, M.R.; Shajahan, A.; Ganesan, P.; Packiam, S.M.; Al-Dhabi, N.A. Comparative studies of tripolyphosphate and glutaraldehyde cross-linked chitosan-botanical pesticide nanoparticles and their agricultural applications. Int. J. Biol. Macromol. 2017, 104, 1813–1819. [Google Scholar] [CrossRef]
- Rajkumar, V.; Gunasekaran, C.; Paul, C.A.; Dharmaraj, J. Development of encapsulated peppermint essential oil in chitosan nanoparticles: Characterization and biological efficacy against stored-grain pest control. Pestic. Biochem. Physiol. 2020, 170, 104679. [Google Scholar] [CrossRef]
- Wang, A.; Wang, Y.; Sun, C.; Wang, C.; Cui, B.; Zhao, X.; Zeng, Z.; Yao, J.; Yang, D.; Liu, G. Fabrication, characterization, and biological activity of avermectin nano-delivery systems with different particle sizes. Nanoscale Res. Lett. 2018, 13, 2. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, N.; Barbole, R.S.; Banerjee, S.S.; Chate, G.P.; Biradar, A.V.; Khandare, J.J.; Giri, A.P. Budding trends in integrated pest management using advanced micro-and nano-materials: Challenges and perspectives. J. Environ. Manag. 2016, 184, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Melanie, M.; Miranti, M.; Kasmara, H.; Malini, D.M.; Husodo, T.; Panatarani, C.; Joni, I.M.; Hermawan, W. Nanotechnology-based bioactive antifeedant for plant protection. Nanomaterials 2022, 12, 630. [Google Scholar] [CrossRef]
- Amidi, M.; Romeijn, S.G.; Borchard, G.; Junginger, H.E.; Hennink, W.E.; Jiskoot, W. Preparation and characterization of protein-loaded N-trimethyl chitosan nanoparticles as nasal delivery system. J. Control. Release 2006, 111, 107–116. [Google Scholar] [CrossRef]
- Grenha, A.; Grainger, C.I.; Dailey, L.A.; Seijo, B.; Martin, G.P.; Remuñán-López, C.; Forbes, B. Chitosan nanoparticles are compatible with respiratory epithelial cells in vitro. Eur. J. Pharm. Sci. 2007, 31, 73–84. [Google Scholar] [CrossRef]
- Grillo, R.; Pereira, A.E.; Nishisaka, C.S.; De Lima, R.; Oehlke, K.; Greiner, R.; Fraceto, L.F. Chitosan/tripolyphosphate nanoparticles loaded with paraquat herbicide: An environmentally safer alternative for weed control. J. Hazard. Mater. 2014, 278, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Silva, M.; Concenza, D.; Grillo, R.; Melo, N.; Tonello, P.; Oliveira, L.; Cassimiro, D.; Rosa, A.; Fraceto, L. Paraquat-loaded alginate/chitosan nanoparticles: Preparation, characterization and soil sorption studies. J. Hazard. Mater. 2011, 190, 366–374. [Google Scholar] [CrossRef] [PubMed]

| Independent Variables | Levels | |
|---|---|---|
| Low | High | |
| CS type (0.4% w/v) | Low MW | High MW |
| Cross-linking agent | TPP (0.3% w/v) | ALG (0.2% w/v) |
| LC concentration (% w/v) | 1 | 1.5 |
| Stirring rate (rpm) | 500 | 1500 |
| Formula Code | CS MW | Cross-Linking Agent (% w/v) | Pesticide Conc (% w/v) | Stirring Rate (rpm) |
|---|---|---|---|---|
| F1 | High | TPP (0.3) | 1.5 | 1500 |
| F2 | High | TPP (0.3) | 1 | 500 |
| F3 | Low | TPP (0.3) | 1.5 | 1500 |
| F4 | Low | TPP (0.3) | 1 | 500 |
| F5 | High | Alginate (0.2) | 1.5 | 1500 |
| F6 | High | Alginate (0.2) | 1 | 500 |
| F7 | Low | Alginate (0.2) | 1.5 | 1500 |
| F8 | Low | Alginate (0.2) | 1 | 500 |
| Formula | %Yield | %PL | %EE | Particle Size (nm) | PDI | Z-Potential (mv) | Desirability |
|---|---|---|---|---|---|---|---|
| F1 | 79.64 ± 2.1 | 10.86 ± 0.3 | 53.12 ± 3.5 | 346.1 ± 5.59 | 0.528 ± 0.05 | −23.1 ± 0.5 | 0.5511 |
| F2 | 58.44 ± 3.1 | 10.15 ± 0.9 | 68.93 ± 2.9 | 414.3 ± 14.2 | 0.448 ± 0.08 | −9.13 ± 0.3 | 0.5506 |
| F3 | 61.62 ± 1.7 | 9.8 ± 0.1 | 46.53 ± 3.9 | 308.4 ± 19.5 | 0.341 ± 0.03 | −1.21 ± 0.3 | 0.5511 |
| F4 | 57.31 ± 2.0 | 3.9 ± 0.2 | 24.95 ± 1.3 | 363 ± 27.7 | 0.457 ± 0.06 | −2.84 ± 0.3 | 0.5410 |
| F5 | 68.55 ± 3.2 | 8.41 ± 0.1 | 59.56 ± 2.1 | 353.3 ± 24.9 | 0.390 ± 0.05 | −11.3 ± 0.2 | 0.4801 |
| F6 | 57.48 ± 1.9 | 10.72 ± 0.4 | 73.56 ± 4.3 | 415.7 ± 22.8 | 0.447 ± 0.05 | −19.8 ± 0.6 | 0.4668 |
| F7 | 65.60 ± 2.5 | 4.15 ± 0.3 | 29.32 ± 1.2 | 278 ± 15.1 | 0.472 ± 0.01 | −21.1 ± 1.2 | 0.4967 |
| F8 | 59.29 ± 4.0 | 5.31 ± 0.1 | 36.12 ± 1.1 | 289 ± 19.1 | 0.453 ± 0.03 | −13.2 ± 0.5 | 0.4665 |
| Formula | Percent Mortality | |||
|---|---|---|---|---|
| 2 h | 16 h | |||
| Male | Female | Male | Female | |
| F1 | 74.0 ± 5.8 a | 78.0 ± 8.7 ab | 100 ± 0.0 a | 98.0 ± 2.0 a |
| F2 | 74.0 ± 7.9 ab | 86.0 ± 5.2 a | 100 ± 0.0 a | 100 ± 0.0 a |
| F3 | 84.0 ± 6.0 ab | 79.0 ± 7.0 ab | 100 ± 0.0 a | 100 ± 0.0 a |
| F4 | 82.0 ± 6.3 ab | 70.9 ± 6.3 ab | 100 ± 0.0 a | 100 ± 0.0 a |
| F5 | 70.0 ± 9.1 ab | 60.0 ± 10.3 b | 100 ± 0.0 a | 100 ± 0.0 a |
| F6 | 68.0 ± 9.0 ab | 80.0 ± 4.2 a | 100 ± 0.0 a | 100 ± 0.0 a |
| F7 | 86.0 ± 4.3 a | 84.0 ± 5.0 a | 100 ± 0.0 a | 100 ± 0.0 a |
| F8 | 78.0 ± 6.3 ab | 68.0 ± 7.4 ab | 100 ± 0.0 a | 100 ± 0.0 a |
| Karate-zeon | 64.0 ± 7.8 b | 74.0 ± 6.0 ab | 98.0 ±2.0 a | 90.0 ± 5.4 b |
| Control | 2.0 ± 2.0 c | 0.0 ± 0.0 c | 12.0 ± 4.4 b | 10.0 ± 3.3 c |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shawer, R.; El-Leithy, E.S.; Abdel-Rashid, R.S.; Eltaweil, A.S.; Baeshen, R.S.; Mori, N. Preparation of Lambda-Cyhalothrin-Loaded Chitosan Nanoparticles and Their Bioactivity against Drosophila suzukii. Nanomaterials 2022, 12, 3110. https://doi.org/10.3390/nano12183110
Shawer R, El-Leithy ES, Abdel-Rashid RS, Eltaweil AS, Baeshen RS, Mori N. Preparation of Lambda-Cyhalothrin-Loaded Chitosan Nanoparticles and Their Bioactivity against Drosophila suzukii. Nanomaterials. 2022; 12(18):3110. https://doi.org/10.3390/nano12183110
Chicago/Turabian StyleShawer, Rady, Eman S. El-Leithy, Rania S. Abdel-Rashid, Abdelazeem S. Eltaweil, Rowida S. Baeshen, and Nicola Mori. 2022. "Preparation of Lambda-Cyhalothrin-Loaded Chitosan Nanoparticles and Their Bioactivity against Drosophila suzukii" Nanomaterials 12, no. 18: 3110. https://doi.org/10.3390/nano12183110






