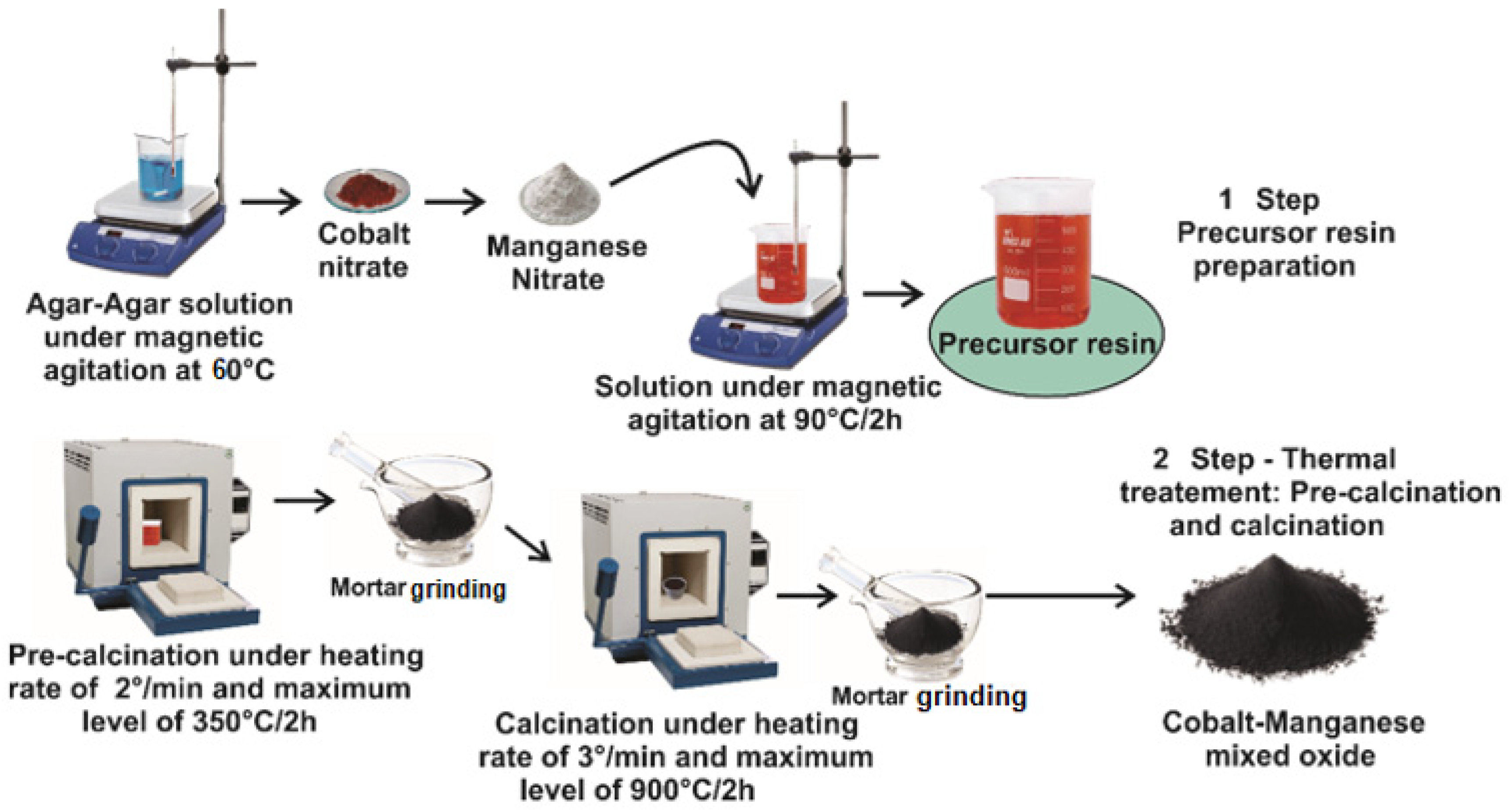
3.1. X-ray Diffraction (XRD)
The refined X-ray diffraction patterns of the Mn
XCo
3-XO
4 (0 ≤ X ≤ 1) are shown in
Figure 3a. As noted, all peaks are characteristic of the cubic phases of Co
3O
4 (structure of Spinel#MgAl
2O
4 type, with lattice parameter
a = b = 8.072(3) Å, ICSD n° 36256, space group Fd-3mS (227)) [
56] and MnCo
2O
4 (structure of Spinel#MgAl
2O
4 type, with lattice parameter
a = b = 8.28(2) Å, ICSD n° 18544, space group Fd-3mZ (227)) [
57]. No secondary phases corresponding to impurities were detected. The ICSD n° 36,256 was used to fit the Mn
XCo
3-XO
4 samples (X < 1), while the 18,544 ICSD file was applied to refine the sample with composition X = 1. The observed patterns are similar to those reported previously for pure and doped cobaltites [
16,
20,
58]. All crystallographic parameters, including crystallite size and lattice parameter, as well as the agreement indices (
Rwp,
Rexp e χ
2) for samples of Mn
XCo
3-XO
4 (0 ≤ X ≤ 1) are gathered in
Table 1.
Figure 3b shows the magnification of the most intense diffraction peak (311), located between 36° and 37.5°. As shown, increasing Mn content shifts the (311) peak to lower angles, indicating a continuous increase in the lattice parameter from 8.0757(1) for x = 0, 8.0759(8) for x = 0.2, 8.1183(7) for x = 0.6, 8.1675(8) for x = 0.8 to 8.2381(6) for x = 1. The total width at half maximum intensity (FWHM) also increases gradually with the increase in Mn (0.1322 for x = 0, 0.1433 for x = 0.2, 0.1513 for x = 0.4, 0.1613 for x = 0.6, 0.1633 for x = 0.8 and 0.1692 for x = 1), signaling a progressive reduction in crystallite size and increase in strain (90.6 nm for x = 0, 82.5 nm for x = 0.2, 80 nm for x = 0.4, 71 nm for x = 0.6, 68.8 nm for x = 0.8, 66 nm for x = 1, with the only exception of the sample corresponding to X = 0.4 for which the 2θ angle is slightly higher than the one displayed by the sample with X = 0.2). The largest variations of FWHM occur in the 0 ≤ X ≤ 0.6 range. The Mn
+2 has a radius of 0.80 Å and it is larger than the radii of Co
+3 (0.63 Å) and Co
+2 (0.65 Å); thus, it causes distortion and strain in the Co
3O
4 lattice, resulting in a decrease in crystallite size. In addition, other manganese oxidation states may be present in the samples such as Mn
+3 and Mn
+4. Mn
+3 (3d
4) is responsible for the Jahn–Teller phenomenon, which also develops a distortion in the lattice and an intrinsic strain that leads to a decrease in the crystallite size [
20,
59,
60,
61]. The trends of lattice parameter and crystallite size are in agreement with previous reports [
16,
20]. The maximum values of the agreement factors
Rwp and
Rexp from the Rietveld analyses are 8.14% and 6.92%, respectively. The low values of fitting quality (χ
2 ≤ 1.30) indicate excellent agreement between the data and the refined models.
3.4. Fourier-Transform Infrared (FT-IR) Spectroscopy
The FT-IR technique shows the vibrational fingerprint of the sample, with absorption peaks that correspond to the frequencies of vibrations of the bonds among the atoms that make up the material [
62].
Figure 6 shows the spectra of the Mn
XCo
3-XO
4 samples (0 ≤ X ≤ 1) in the range from 400 to 4000 cm
−1, where two bands with the highest intensities are located at 552–570 and 643–663 cm
−1, which are related to the stretching vibrations of the metal–oxygen bond, which confirms the formation of the pure Co
3O
4 phase [
63]. The
v1 band at 552–570 cm
−1 is characteristic of the vibration of Co
3+ at the octahedral site, and the
ν2 band at 643–663 cm
−1 is related to the vibration of Co
2+ at the tetrahedral site, confirming the formation of the spinel-like oxide [
64], in agreement with the XRD study. The low-intensity band that appears at 1100 cm
−1 is due to C-O stretching vibrations. The band at 1383 cm
−1 is attributed to the symmetric deformations of C-N and CH
2 groups, originating from the residues of nitrate ions and agar-agar [
65,
66,
67]. The band at 1635 cm
−1 was attributed to the angular deformation of the adsorbed water molecules [
68]. The broad absorption band in the region of about 3440 cm
−1 is due to OH stretching of the water molecules adsorbed from the moisture during the storage process [
69]. Furthermore, it is observed that the bands at 554–643 cm
−1 for sample X = 0.8 and the bands at 552–643 cm
−1 for sample X = 1.0 are similar, which may be related to the oxidation states of manganese (Mn
+2, Mn
+3, and Mn
+4) or to the vibrational intensity between the manganese and oxygen bond. In general, the frequency of the peaks of the absorption bands at (552–570) and (643–663) cm
−1 (
Table 2) decreases with the replacement of cobalt with manganese ions, i.e., they shift to the right as the amount of manganese increases, and this is related to the increase in the metal–oxygen distance, as indicated by the increase in the lattice parameter of the unit cells (
Table 1), since Mn ions are larger than Co ions [
21].
3.6. Raman Spectroscopy
Figure 8 shows the Raman spectra of the Mn
XCo
3-XO
4 samples in the range from 100 to 1000 cm
−1. The observed bands in the intervals 193–688 cm
−1 for x = 0.0, 186–682 cm
−1 for x = 0.2, 186–675 cm
−1 for x = 0.4, 185–667 cm
−1 for x = 0.6, 183–661 cm
−1 for x = 0.8, and 182–660 cm
−1 for x = 1.0 correspond to the active Raman modes A1g+Eg+3F2g (
Table 3), confirming the formation of the pure phase of mixed-valence oxides of spinel-like structure [
79,
80,
81,
82,
83]. The most intense band at 688–660 cm
−1 is assigned to the octahedral site MO
6 related to the A1g mode of the O
7h spectroscopic symmetry, which corresponds to the stretching vibrational modes of these oxides M-O, where M = {Co, Mn}, thus substantiating the formation of MnCo
2O
4. The Raman bands with medium intensity in the intervals 468–488 cm
−1 and 508–518 cm
−1 are assigned to Eg and F2g, respectively; meanwhile, the Raman bands with lower intensities in the interval 603–617 cm
−1 are caused by the F2g mode. Moreover, the Raman bands with very low intensity at 182–193 cm
−1 are attributed to the F2g mode related to the tetrahedral sites of CoO
4 [
82,
83,
84]. In general, when comparing the positions of the peaks, it is observed that as the amount of manganese increases, the peaks shift to the left, analogously to what was noticed in the FT-IR spectra. This change may be due to the greater ionic radius of Mn
2+, in comparison to that of Co
2+/Co
3+ [
85,
86,
87], which when entering the structure of Co
3O
4, generates a large distortion in the crystalline structure and increases the distance between the metal and the oxygen, and consequently a weakening of bonds occurs. Another reason would be due to vibrations in the structure, where the Co
2+ and Co
3+ cations are located in tetrahedral and octahedral sites in the cubic crystal structure [
88].
3.7. X-ray Photoelectron Spectroscopy (XPS)
The surface oxidation states of the samples were analyzed by XPS.
Figure 9 shows the high-resolution Co 2p, Mn 2p, and O 1s spectra obtained from the analysis. All data were corrected for the carbon peak position. In the case of the Co 2p spectra (
Figure 9a), four peaks were deconvoluted, at lower binding energies, corresponding to Co
3+ and Co
2+, as well as two satellite peaks at higher binding energies. The binding energies obtained for Co
3+ were found to fall in the 779.64–780.19 eV range, while for Co
2+, in the 781.19–781.85 eV range, in agreement with previous work [
90]. We also found a Co
2+/Co
3+ ratio varying from 0.31 to 0.40 among the samples, for which the sample X = 0.8 was found to have the lowest value of Co
2+/Co
3+ = 0.31. Higher oxidation states can induce more bonded oxygen species, which may have a positive impact on oxidation reactions.
Conversely, in the case of the Mn 2p spectra (
Figure 9b), the data were deconvoluted into three peaks, which were ascribed to Mn
4+ (ranging from 644.154 eV to 645.146 eV), Mn
3+ (ranging from 642.790 eV to 643.125 eV), and Mn
2+ (641.223 eV to 641.459 eV), in agreement with previous reports [
90,
91]. The lowest oxidation state species, Mn
2+, was found to represent the largest fraction of the total species present at the surface, with a relative value varying from 0.42 to 0.51 among the samples. Conversely, the presence of Mn
4+ and Mn
3+ oxidation states is related to the relatively high calcination temperature used in this work, i.e., 900 °C, as found in previous literature [
92]. In this respect, we also noted a higher Mn
3+/Mn
4+ ratio for the tested samples, which correlates well with the previously discussed Jahn–Teller phenomenon, with a concurrent distortion of the crystal lattice.
Finally, the O 1s high-resolution spectra (
Figure 9c) display three deconvoluted peaks: O
1 (529.94–530.27 eV), O
2 (531.16–531.72 eV), and O
3 (532.55–533.19 eV). Based on the characteristic binding energies determined for these peaks, they are likely related to surface lattice oxygen (O
lat, O
2−), adsorbed oxygen species (O
ads, O
2−, O
22−, and O
−), and adsorbed water species (O
H2O), in agreement with earlier reports on similar compounds [
91,
93]. From the analysis of the O 1s high-resolution spectra (
Table 4), we determined higher O
2/O
1 ratios with increasing Mn content, with a maximum value obtained for the X = 1.0 sample. This suggests that the compounds with the highest Mn content possess increased catalytic activity, as a likely result of increased oxygen-ion vacancies in these samples.
3.8. Vibrating Sample Magnetometer (VSM)
Magnetization measurements were done to study the magnetic behavior of samples at room temperature and to determine the cation magnetic moment in an approximate manner. For all samples, the isothermal magnetization at T = 300 K showed a linear behavior with the magnetic field, and their magnetization at a given field increased with the Mn concentration, as shown in
Figure 10.
This trend was observed for all samples and it is typical of paramagnetic samples as shown in
Figure 10. From the classical theory of paramagnetism we know that the relationship between the magnetization (M) and magnetic field (H) is given by the Langevin function L(
a) = M/M
o = coth(
a)-1/
a, where M and M
o are the mass magnetizations per total amount of Mn and Co (without oxygen),
a = μH/K
BT, μ is the average magnetic moment per cation, K
B is the Boltzmann constant (1.3807 × 10
−16 cm
2gK/s
2), and T = 300 K. It is known that L(
a) tends to
a/3 when
a is less than about 0.5 [
94]. In the present case, if μ~4.51 × 10
−20 Erg/Oe (theoretical magnetic moment for Mn
3+ and Co
3+) and H = 15 × 10
3 Oe, then, one can get
a = 0.01633, which is smaller than 0.5. Thus, L(
a) = M/M
o ≈
a/3 and, therefore, M = [M
oμ/(3K
BT)]H. In a paramagnetic system M
o = Nμ/
A, where N is the Avogadro’s number and
A = (x*54.938 + (3-x)*58.933)/3 is the average atomic mass provided by Mn and Co in Mn
XCo
3-XO
4 (where X = {0.0, 0.2, 0.4, 0.6, 0.8, 1.0}). Thus, the DC susceptibility is χ = M/H= Nμ
2/(3
AK
BT) [
94]. One can study the M-H data by fitting the curve to a linear equation and comparing the slope to Nμ
2/(3
AK
BT). Then, to obtain μ in Bohr magnetons (μ
B), one has to calculate μ/0.9274 × 10
−20. The results provided an effective magnetic moment per cation of 2.204 2.294, 2.342, 2.344, 2.348, 2.378 μ
B for the samples prepared with x = 0.0, 0.2, 0.4, 0.6, 0.8, 1.0, respectively.
The magnetic moments expected for low spin configuration have a total spin of S(Mn2+) = 0.5, S(Mn3+) = 0, S(Mn4+) = 0.5, S(Co2+) = 0.5, S(Co3+) = 0, whose magnetic moments are, μ = μB, i.e., μ(Mn2+) = 1.732 μB, μ(Mn3+) = 0, μ(Mn4+) = 1.732 μB, μ(Co2+) = 1.732 μB, μ(Co3+) = 0. Furthermore, for high spin configuration S(Mn2+) = 2.5, S(Mn3+) = 2.0, S(Mn4+) = 1.5, S(Co2+) = 1.5, S(Co3+) = 2, whose magnetic moments are μ(Mn2+) = 5.916 μB, μ(Mn3+) = 4.899 μB, μ(Mn4+) = 3.873 μB, μ(Co2+) = 3.873 μB, μ(Co3+) = 4.899 μB. Therefore, the magnetic moments for Co and Mn seem to be mainly in the low spin configuration; however, we cannot rule out the presence of some moments in the high spin configuration.
3.9. Oxygen Evolution Reaction (OER)
The samples were also evaluated as electrocatalysts for the oxygen evolution reaction (OER). According to the results of the anodic polarization (
Figure 11a), the electrodes presented values of 515 (Ni foam), 342 (X = 0.0), 342 (X = 0.2), 339 (X = 0.4), 337 (X = 0.6), 323 (X = 0.8), 299 (X = 1.0) and 235 (RuO
2/Ni foam benchmark, extracted from reference [
95]) mV vs. RHE, respectively, to record a current density J = 10 mA cm
2. Among the investigated materials, the MnCo
2O
4 (X = 1.0) samples displayed the best catalytic activity for OER, i.e., the lowest overpotential because the incorporation of Mn into the structure enhanced the defect concentrations, thus increasing the amount of catalytically active sites, which facilitated the mass transfer process, favoring OER [
96]. Moreover, the crystalline size decreased with the increase of manganese content, which indicates that the Co
3O
4 sample (X = 0.0) has larger average crystal sizes than the other samples, especially MnCo
2O
4 (X = 1.0); thus, Co
3O
4 was the sample that had the highest overpotential. This indicates that the presence of Mn, has a suppressive effect on Co
3O
4 [
97]. The obtained values are in agreement with others reported in the literature for Mn
XCo
3-XO
4 nanostructures, as shown in
Table 5.
The electrocatalytic kinetics for OER was investigated by the Tafel plots extracted from the LSV (linear sweep voltammetry) curves (
Figure 11a), using the Tafel equation (η = a + b log j), where b is the Tafel slope, η is the overpotential, j is the current density, and a is a constant. The values of the Tafel slope (
Figure 11b) were 63 (X = 0.0), 73 (X = 0.2), 72 (X = 0.4), 69 (X = 0.6), 68 (X = 0.8), and 52 mV dec
−1 (X = 1.0). Therefore, the results did not follow exactly a sequence like the ղ
10 values (
Figure 11a), but it can be observed that the electrode based on the MnCo
2O
4 sample (X = 1.0) exhibited the best reaction kinetics for OER, as it showed the lowest Tafel slope, which demonstrates a higher efficiency for oxygen evolution. The Tafel slope of 63 mV dec
−1 for the Co
3O
4 sample (X = 0.0) corresponds to slightly slower kinetics, indicating limitation in charge and mass transfer processes compared to the x = 1.0 sample. The Co
3O
4 sample was also the one with the highest overpotential, with no distortion in the structure, which reduces defects and consequently the oxygen vacancies [
106,
107]. These results are consistent with the XPS values as well as the electrochemical impedance spectroscopy. The samples with X = 0.2, 0.4, 0.6, and 0.8 show values next to 70 mV dec
−1, which means much slower kinetics for OER.
All this evidence can be explained by the distortion of the lattice with the increase of the amount of Mn in the Co
3O
4 structure, which changes the electronic charge distribution and increases the disorder in the crystalline system [
106,
107,
108,
109]. Furthermore, with increasing Mn percentages, the availability of oxygen and flexibility in the lattice is greater, which in turn is related to the M-O bond length [
110]. In any case, the samples (X = 0.2), (X = 0.4), (X = 0.6) and (X = 0.8) show values of Tafel slope below 80 mV dec
−1, and these results indicate the adsorption of intermediate species as the rate-determining step (rds), based on the Krasil’shchikov reaction model for OER in alkaline medium [
111,
112].
The double-layer capacitance (C
DL) can be obtained from the relationship between the anode current density (i
a) and the scan rate (ʋ), according to (i
a = ʋ x C
DL) [
100].
Figure 11c shows the double-layer capacitance values obtained for the samples: 1.78 (X = 0.0), 2.75 (X = 0.2), 2.57 (X = 0.4), 2.41 (X = 0.6), 3.16 (X = 0.8), and 2.03 mF cm
−2 (X = 1.0). These results suggest that the largest number of active sites is organized on the electrode surfaces. Although among the samples of this series, the one with X = 0.8 had the second lowest performance for OER, it displays the highest C
DL value, which may be linked to the high amount of oxygen vacancies that improves the absorption of reactive species (like OH¯) [
113]. For the samples (X = 0.2), (X = 0.4), and (X = 0.6), the values of 2.75, 2.57, and 2.41 mF cm
−2, respectively, are consistent with the XPS data (
Figure 9b), where the species in the lowest oxidation state, Mn
2+, represented the largest fraction of the total species present on the surface, with a relative fraction ranging from 0.42–0.51 among the samples. However, even with a low C
DL value for the sample (X = 1.0), the presence of Mn ions in the structure is essential for superior electrocatalytic properties. This was proven by the best overpotential extracted from the LSV curves (
Figure 11a) and the XPS data (
Figure 9c), as the substitution of Mn in spinel oxide cobalt occurs selectively in the (Co
3+) lattices, and the energy required for Mn
2+ to substitute Co
3+ is lower than that of Co
2+, [
114]. Moreover, Mn
+2, Mn
+3, and Mn
+4 have ionic ratios of 0.80, 0.66, and 0.60 Å, respectively, and the ratios of Mn
+2 and Mn
+3 are larger than that of Co
3+ (0.63 Å). Therefore, Mn doping results in the expansion of the Co
3O
4 lattice, generating defects, which influences the mass diffusion and charge transfer properties, contributing to oxygen-ion vacancies, which are consistent with the O
2/O
1 ratio that was highest for the MnCo
2O
4 sample (X = 1.0), with a value of 0.940 [
108,
109,
115,
116].
Durability is another important indicator of catalytic performance. The stability of the electrocatalysts was evaluated by chronopotentiometry. Tests were performed at a current density of 10 mA cm
−2 for 15 h. According to the curves shown in
Figure 11d, it can be seen that the samples (X = 0) and (X = 0.6) exhibited a potential decrease until about 2 h, but then they remained stable, whereas for the samples (X = 0.8) and (X = 1.0), the potential was practically stable for the entire time period tested. In general, all samples showed satisfactory stability over 15 h of testing [
117].
3.10. Electrochemical Impedance Spectroscopy (EIS)
The electrocatalytic activity also was investigated by electrochemical impedance spectroscopy (EIS). The EIS spectra of all samples were collected at 1.4 V vs. RHE. As seen in the Bode plots (
Figure 12b), the OER is composed of complex processes involving electrosorption of intermediate species during the reaction progress. This suggests an equivalent circuit model (ECM) able to describe these processes [
118,
119] that is composed of R
S (uncompensated solution resistance), R
P (polarization resistance, which denotes the overall rate of the OER),
QDL (double-layer pseudo-capacitance), R
-ad (resistance associated with adsorption of intermediate species), and
Q-ad (pseudo-capacitance of these species throughout the reaction). A constant phase element (
Q) was used to model an imperfect capacitor, and its impedance was obtained by:
Then, the values were used to calculate true capacitance (C
DL or C
-ad) by:
In
Figure 12a, the impedance of the electrodes is composed of two incomplete semicircles. The first is attributed to the polarization process (charge transfer), and the second indicates limitations on mass transfer processes, related to the intermediate species adsorption process [
120]. For the electrodes, the obtained R
P values were consistent with the OER performance, i.e., the X = 1.0 sample showed the lowest value (6.02 Ω), followed by X = 0.8 (9.70 Ω). The other samples revealed R
P values very close, but the result was expected as their overpotential values were close. The C
DL values varied slightly (
Table 6) due to the oxidation peak shown in
Figure 11a. The R
adC
ad loop associated with relaxation, which was attributed to the adsorbed intermediate species, revealed the difficulty of these electrodes to work in the diffusive processes observed at low frequencies. The high R
-ad (>1100 Ω) values displayed by the electrodes in those low frequency (>1 Hz) confirm that the adsorption of intermediates should be a rate-limiting step as predicted by Tafel analysis (
Figure 11b) [
118,
121]. The values obtained from the fitting of the spectra are listed in
Table 6.