Polybenzimidazole Confined in Semi-Interpenetrating Networks of Crosslinked Poly (Arylene Ether Ketone) for High Temperature Proton Exchange Membrane
Abstract
:1. Introduction
2. Experimental
2.1. Materials
2.2. Methods
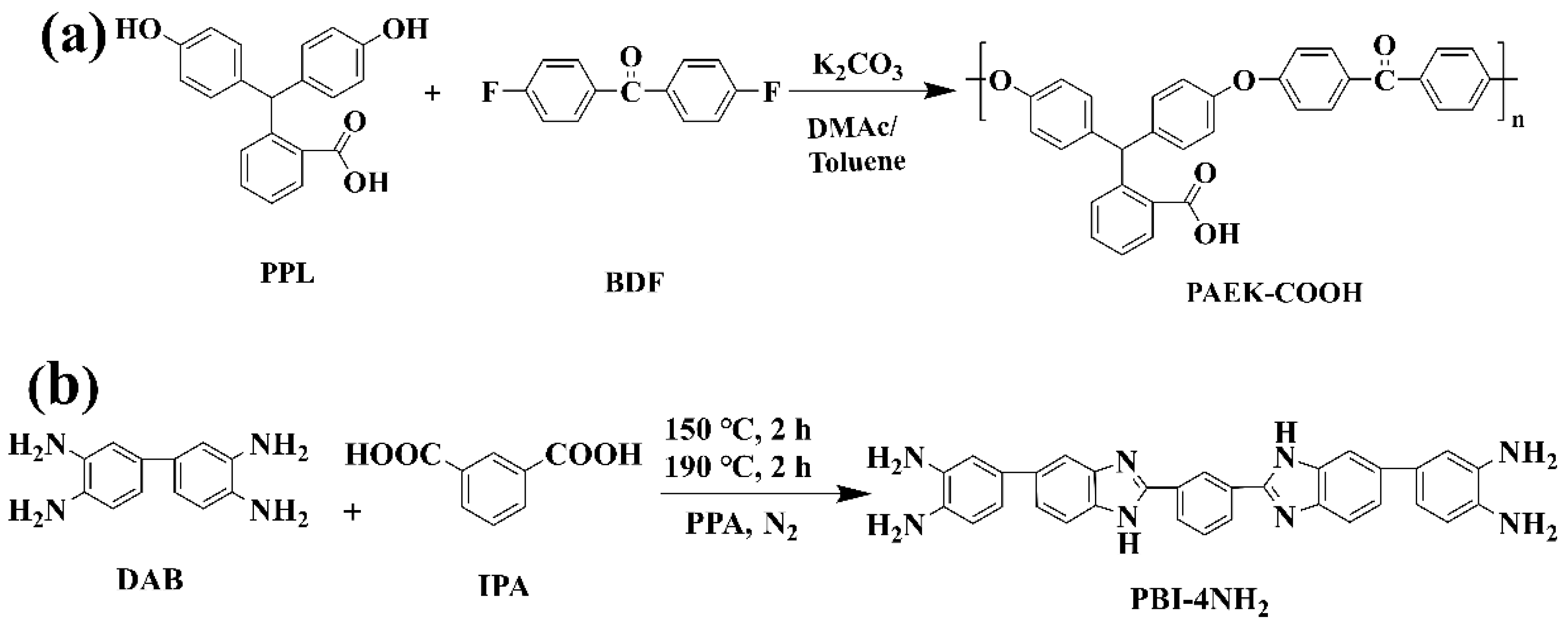
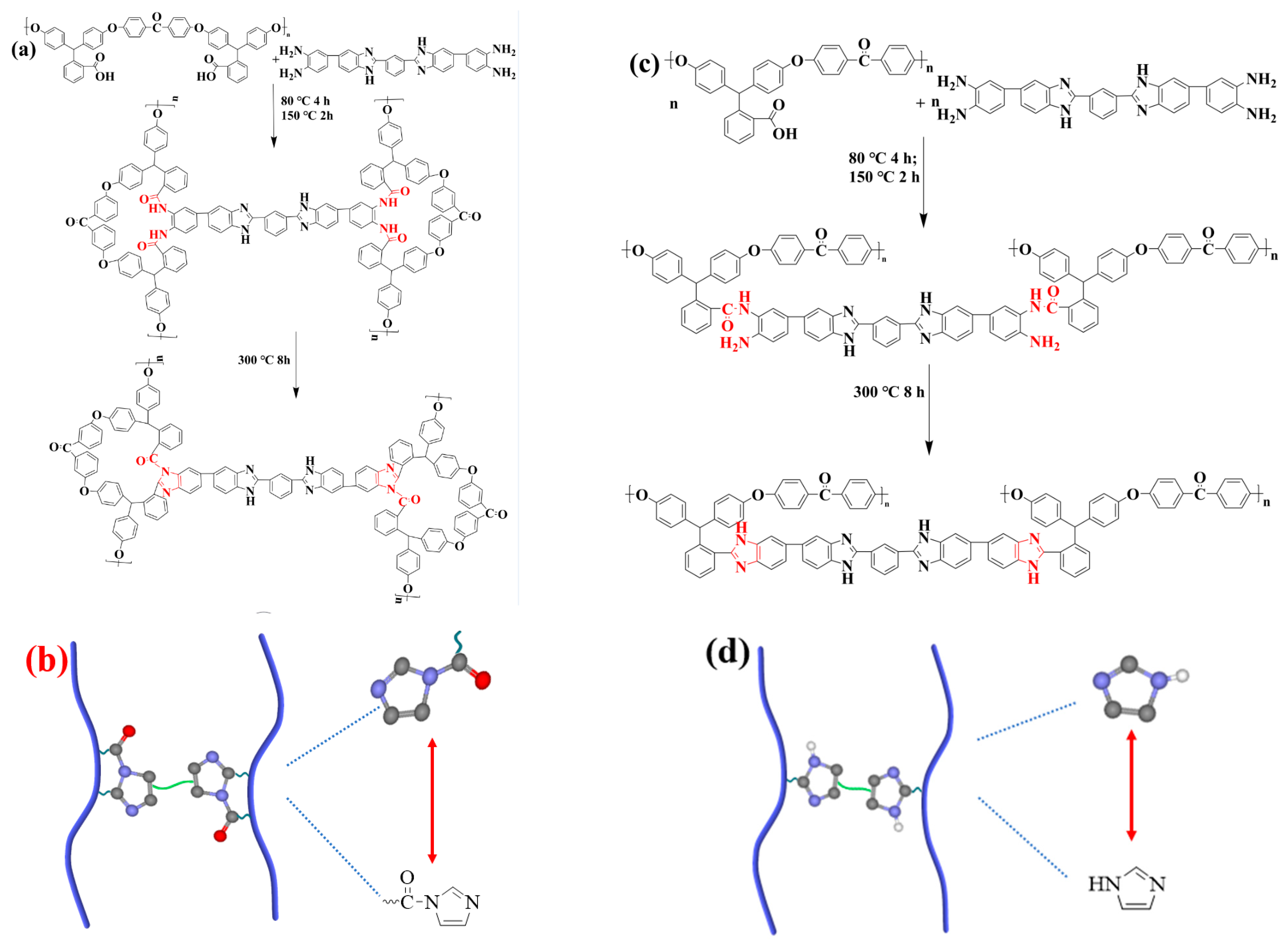
2.2.1. Preparation of Poly (Arylene Ether Ketone) with Pendant Carboxyl Groups (PAEK-COOH)
2.2.2. Preparation of Amino-Ended Crosslinker (PBI-4NH2)
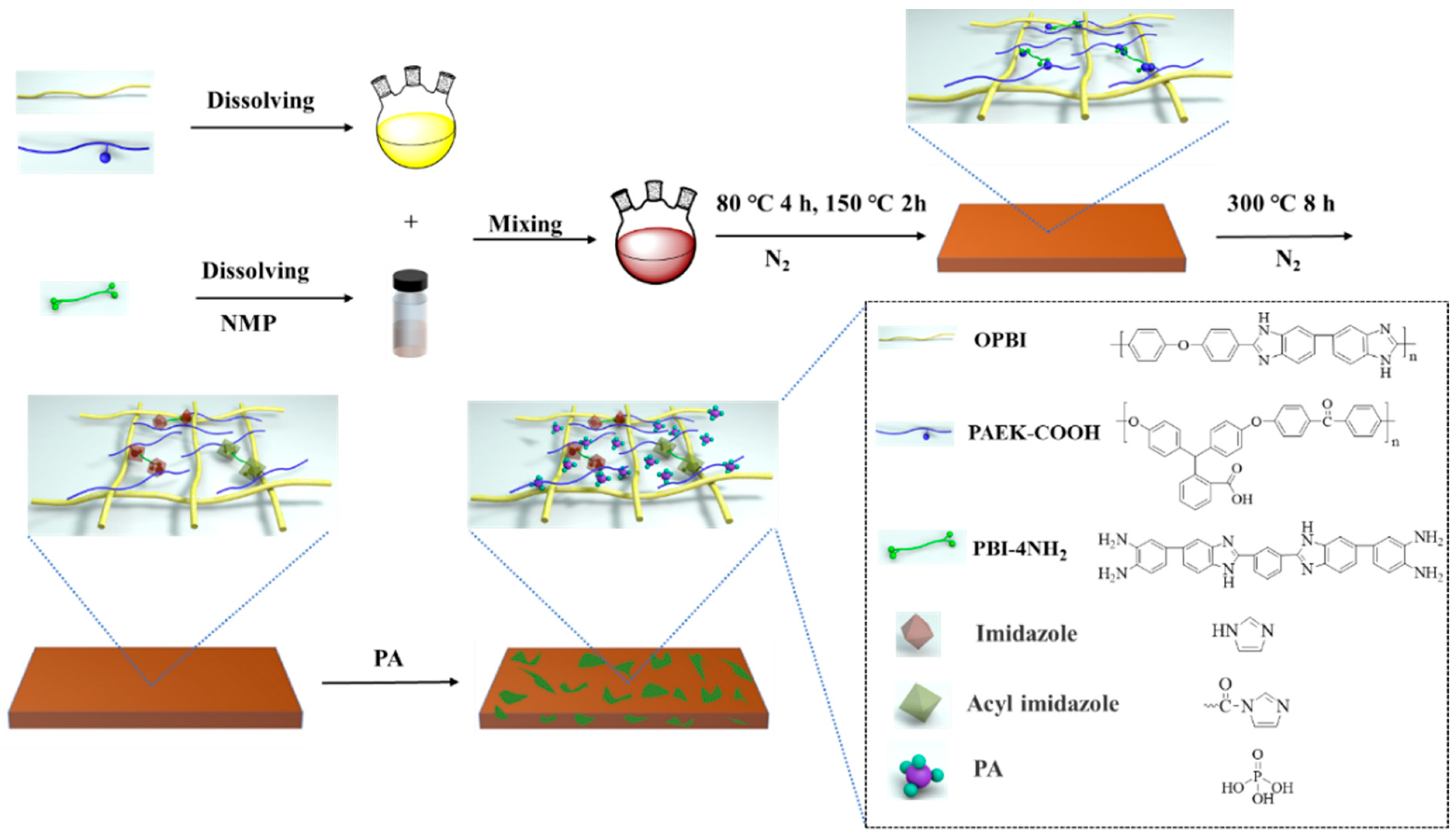
2.2.3. Preparation of Semi-IPN/xPBI Membranes
2.3. Characterization and Measurements
2.3.1. Characterization of Chemical Structure
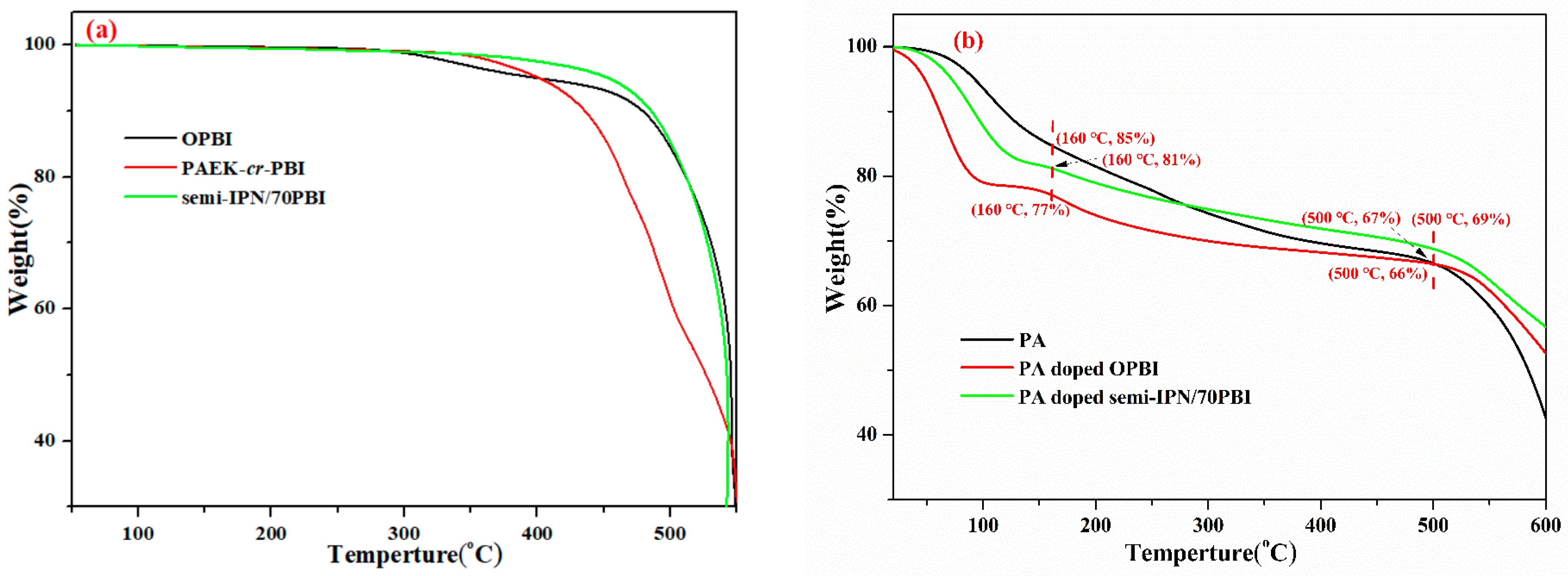
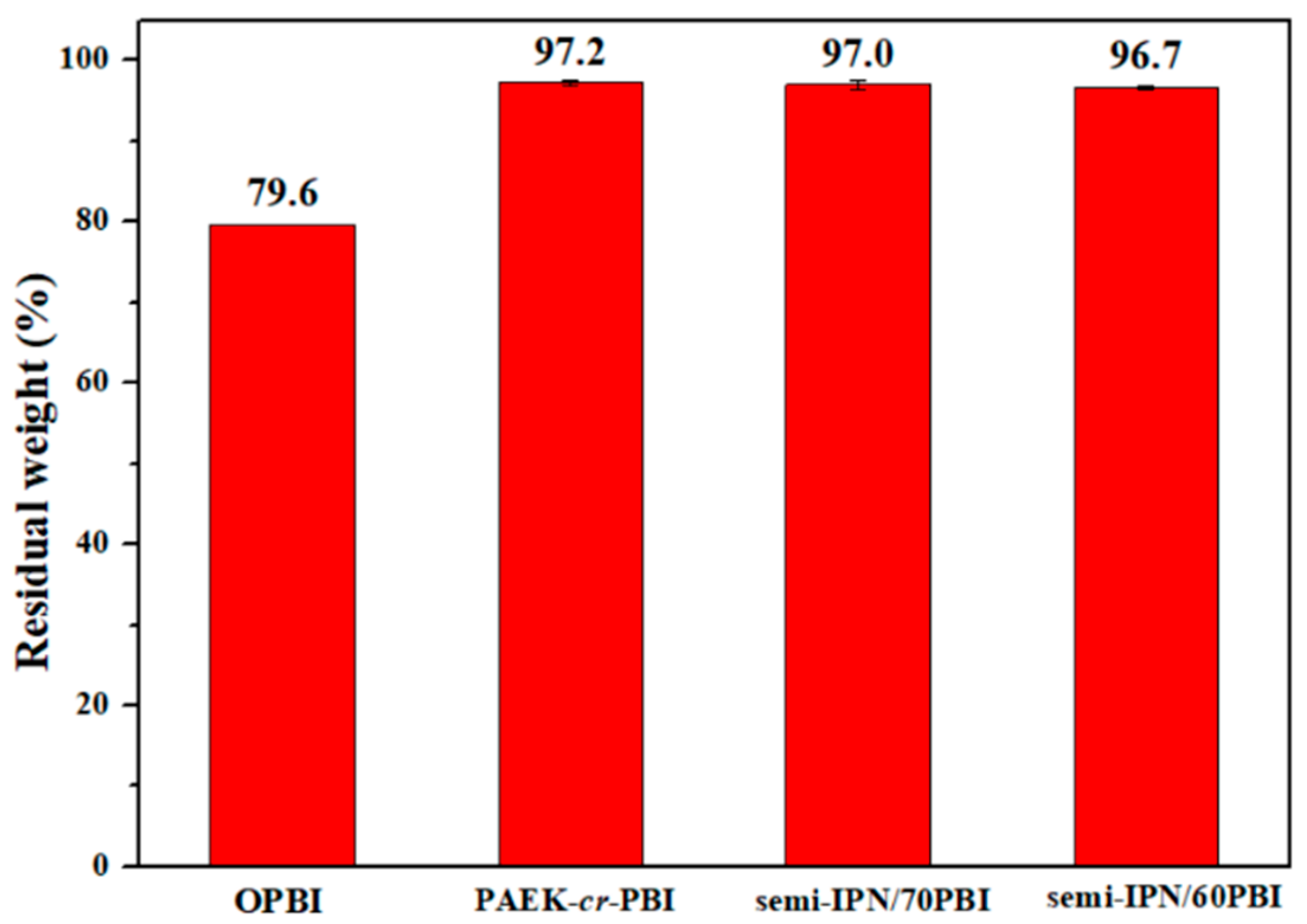
2.3.2. Thermal Stability
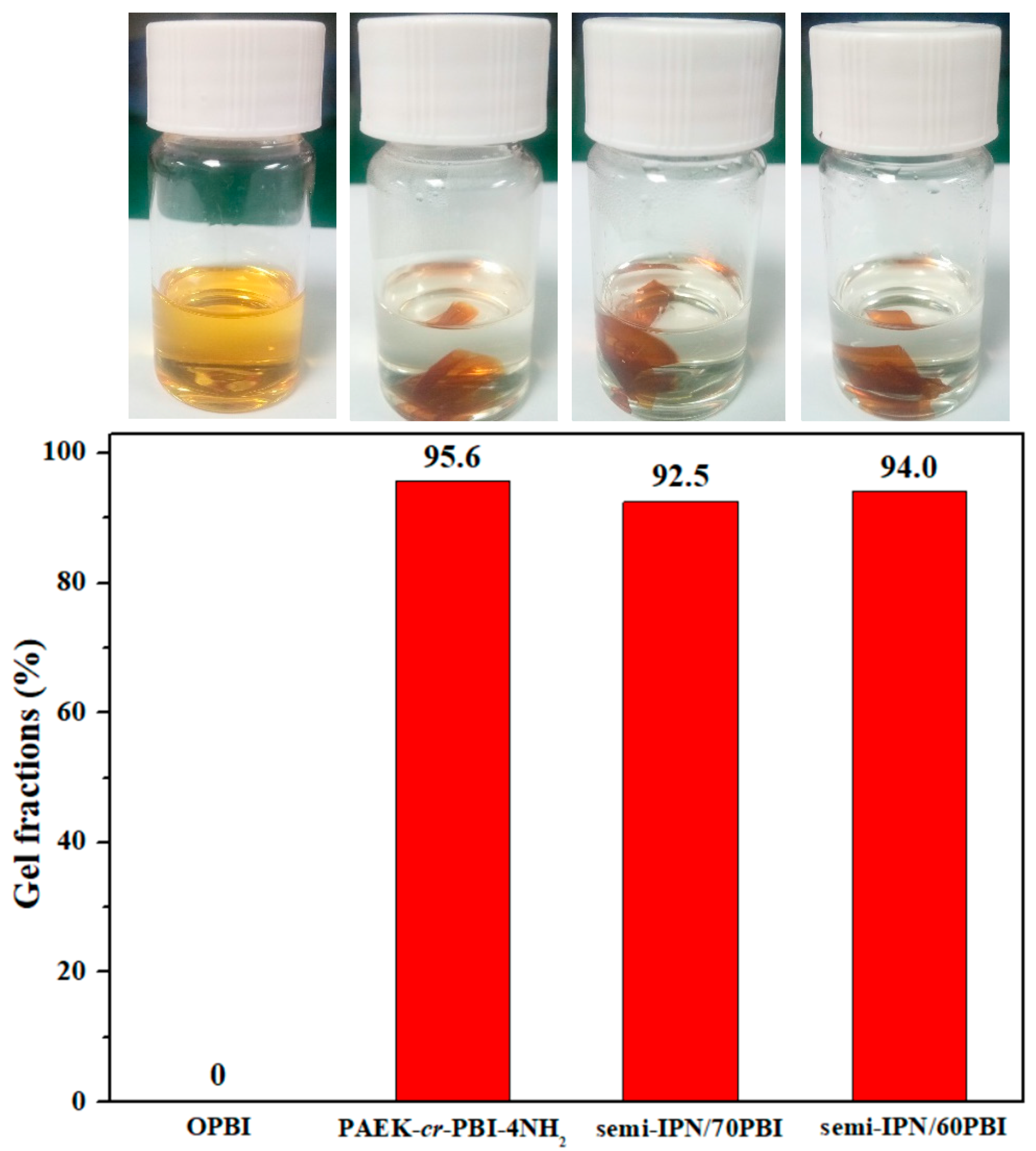
2.3.3. Gel Fraction Test
2.3.4. PA Uptake and Volumetric Swelling Ratio
2.3.5. Evaluation of Oxidative Stability
2.3.6. Mechanical Properties
2.3.7. Proton Conductivity
2.3.8. Single Cell Test
3. Results and Discussion
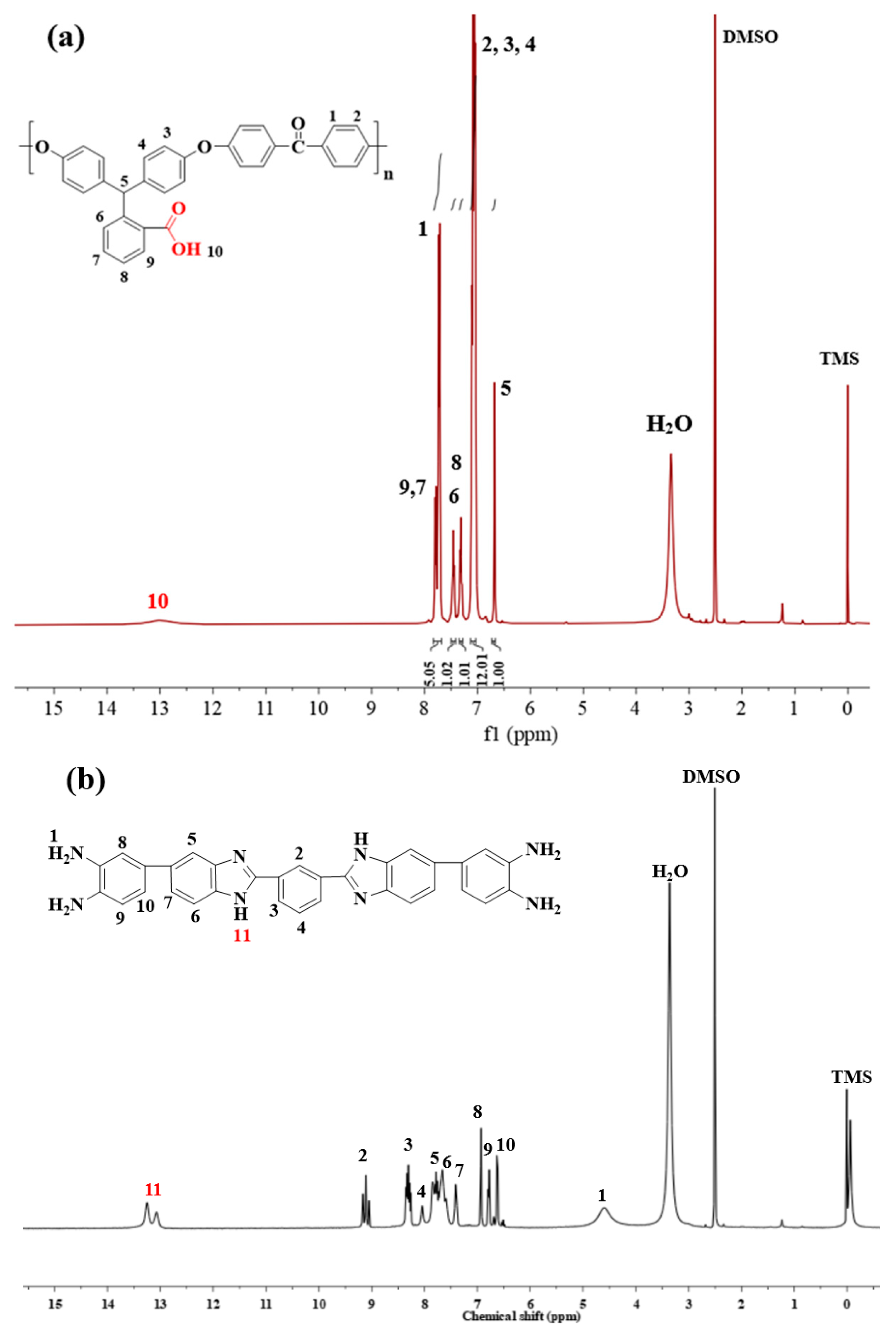
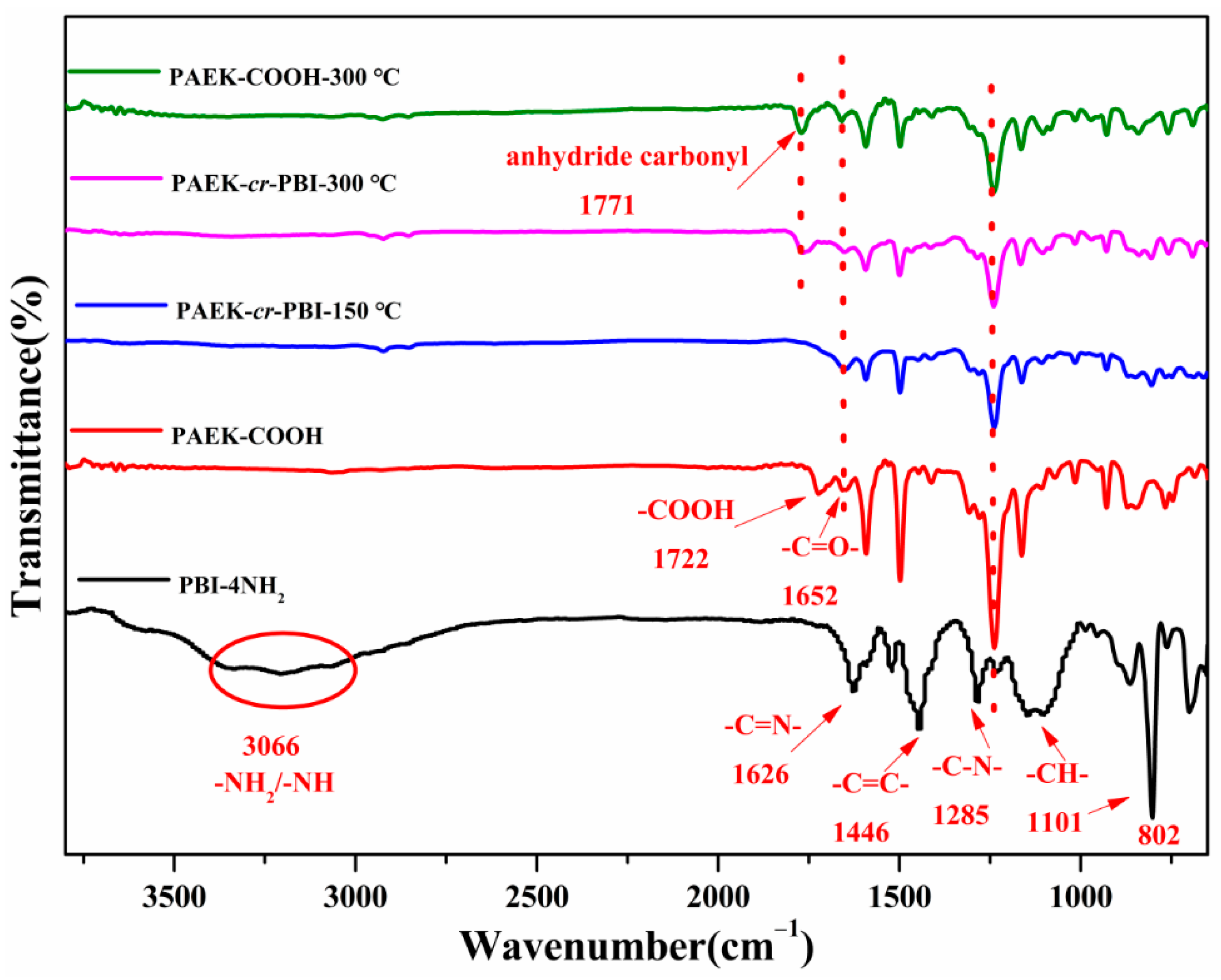
3.1. Chemical Structure Identification
3.2. Thermal and Oxidative Stability
3.3. PA Doping Level, Volumetric Swelling Ratio, and Mechanical Properties
3.4. Proton Conductivity
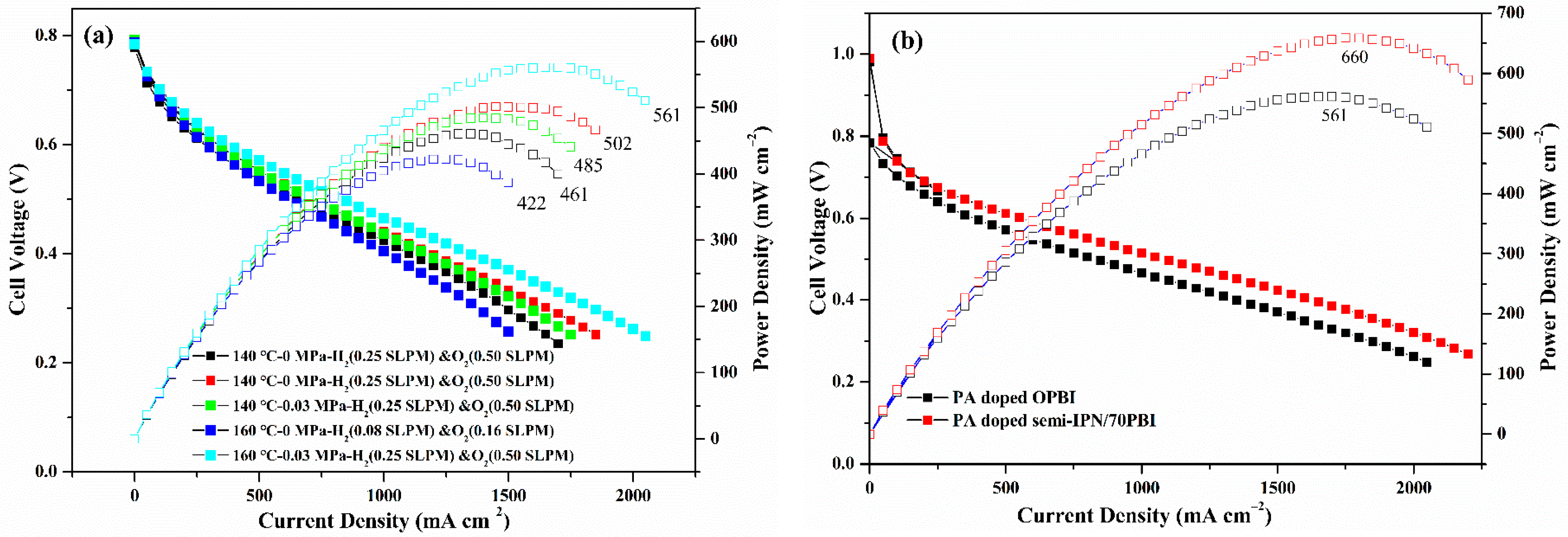
3.5. Single Fuel Cell Performance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Guo, Z.; Xu, X.; Xiang, Y.; Lu, S.; Jiang, S.P. New anhydrous proton exchange membranes for high-temperature fuel cells based on PVDF–PVP blended polymers. J. Mater. Chem. A 2014, 3, 148–155. [Google Scholar] [CrossRef]
- Kim, S.-K.; Kim, K.-H.; Park, J.O.; Ko, T.; Choi, S.-W.; Pak, C.; Chang, H.; Lee, J.-C. Highly durable polymer electrolyte membranes at elevated temperature: Cross-linked copolymer structure consisting of poly(benzoxazine) and poly(benzimidazole). J. Power Source 2013, 226, 346–353. [Google Scholar] [CrossRef]
- Li, Q.; Aili, D.; Hjuler, H.A.; Jensen, J.O. High Temperature Polymer Electrolyte Membrane Fuel Cells: Approaches, Status, and Perspectives; Springer International AG Switzerland: Cham, Switzerland, 2016. [Google Scholar]
- Rahman, S.; Masdar, M.S.; Rosli, M.; Majlan, E.H.; Husaini, T.; Kamarudin, S.; Daud, W. Overview biohydrogen technologies and application in fuel cell technology. Renew. Sustain. Energy Rev. 2016, 66, 137–162. [Google Scholar] [CrossRef]
- Tsai, T.-H.; Ertem, S.P.; Maes, A.M.; Seifert, S.; Herring, A.M.; Coughlin, E.B. Thermally Cross-Linked Anion Exchange Membranes from Solvent Processable Isoprene Containing Ionomers. Macromolecules 2015, 48, 655–662. [Google Scholar] [CrossRef]
- Bose, S.; Kuila, T.; Nguyen, T.X.H.; Kim, N.H.; Lau, K.-T.; Lee, J.H. Polymer membranes for high temperature proton exchange membrane fuel cell: Recent advances and challenges. Prog. Polym. Sci. 2011, 36, 813–843. [Google Scholar] [CrossRef]
- Mamlouk, M.; Scott, K. A boron phosphate-phosphoric acid composite membrane for medium temperature proton exchange membrane fuel cells. J. Power Source 2015, 286, 290–298. [Google Scholar] [CrossRef] [Green Version]
- Tian, X.; Wang, S.; Li, J.; Liu, F.; Wang, X.; Chen, H.; Wang, D.; Ni, H.; Wang, Z. Benzimidazole grafted polybenzimidazole cross-linked membranes with excellent PA stability for high-temperature proton exchange membrane applications. Appl. Surf. Sci. 2019, 465, 332–339. [Google Scholar] [CrossRef]
- Kallem, P.; Drobek, M.; Julbe, A.; Vriezekolk, E.J.; Mallada, R.; Pina, M.P. Hierarchical Porous Polybenzimidazole Microsieves: An Efficient Architecture for Anhydrous Proton Transport via Polyionic Liquids. ACS Appl. Mater. Interfaces 2017, 9, 14844–14857. [Google Scholar] [CrossRef] [Green Version]
- Rosli, R.E.; Sulong, A.B.; Daud, W.R.W.; Zulkifley, M.A.; Husaini, T.; Rosli, M.I.; Majlan, E.H.; Haque, M.A. A review of high-temperature proton exchange membrane fuel cell (HT-PEMFC) system. Int. J. Hydrogen Energy 2017, 42, 9293–9314. [Google Scholar] [CrossRef]
- He, R.; Li, Q.; Xiao, G.; Bjerrum, N.J. Proton conductivity of phosphoric acid doped polybenzimidazole and its composites with inorganic proton conductors. J. Membr. Sci. 2003, 226, 169–184. [Google Scholar] [CrossRef]
- Hu, M.; Ni, J.; Zhang, B.; Neelakandan, S.; Wang, L. Crosslinked polybenzimidazoles containing branching structure as membrane materials with excellent cell performance and durability for fuel cell applications. J. Power Source 2018, 389, 222–229. [Google Scholar] [CrossRef]
- Kallem, P.; Eguizabal, A.; Mallada, R.; Pina, M.P. Constructing Straight Polyionic Liquid Microchannels for Fast Anhydrous Proton Transport. ACS Appl. Mater. Interfaces 2016, 8, 35377–35389. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.S.; Choe, Y.-K.; Matanovic, I.; Kim, Y.S. The energetics of phosphoric acid interactions reveals a new acid loss mechanism. J. Mater. Chem. A 2019, 7, 9867–9876. [Google Scholar] [CrossRef]
- Wainright, J.S.; Wang, J.T.; Weng, D.; Savinell, R.F.; Litt, M. Acid-Doped Polybenzimidazoles: A New Polymer Electrolyte. J. Electrochem. Soc. 1994, 142, L121–L123. [Google Scholar] [CrossRef]
- Li, X.; Ma, H.; Wang, P.; Liu, Z.; Peng, J.; Hu, W.; Jiang, Z.; Liu, B. Construction of High-Performance, High-Temperature Proton Exchange Membranes through Incorporating SiO2 Nanoparticles into Novel Cross-linked Polybenzimidazole Networks. ACS Appl. Mater. Interfaces 2019, 11, 30735–30746. [Google Scholar] [CrossRef]
- Liu, F.; Wang, S.; Li, J.; Tian, X.; Wang, X.; Chen, H.; Wang, Z. Polybenzimidazole/ionic-liquid-functional silica composite membranes with improved proton conductivity for high temperature proton exchange membrane fuel cells. J. Membr. Sci. 2017, 541, 492–499. [Google Scholar] [CrossRef]
- Mader, J.A.; Benicewicz, B.C. Sulfonated Polybenzimidazoles for High Temperature PEM Fuel Cells. Macromolecules 2010, 43, 6706–6715. [Google Scholar] [CrossRef]
- Quartarone, E.; Mustarelli, P. Polymer fuel cells based on polybenzimidazole/H3PO4. Energy Environ. Sci. 2012, 5, 6436–6444. [Google Scholar] [CrossRef]
- Chandan, A.; Hattenberger, M.; El-Kharouf, A.; Du, S.; Dhir, A.; Self, V.; Pollet, B.G.; Ingram, A.; Bujalski, W. High temperature (HT) polymer electrolyte membrane fuel cells (PEMFC)—A review. J. Power Source 2013, 231, 264–278. [Google Scholar] [CrossRef]
- Tian, D.; Gu, T.; Yellamilli, S.N.; Bae, C. Phosphoric Acid-Doped Ion-Pair Coordinated PEMs with Broad Relative Humidity Tolerance. Energies 2020, 13, 1924. [Google Scholar] [CrossRef] [Green Version]
- Won, S.; Oh, K.; Ju, H. Numerical degradation studies of high-temperature proton exchange membrane fuel cells with phosphoric acid-doped PBI membranes. Int. J. Hydrogen Energy 2016, 41, 8296–8306. [Google Scholar] [CrossRef]
- Hu, M.; Li, T.; Neelakandan, S.; Wang, L.; Chen, Y. Cross-linked polybenzimidazoles containing hyperbranched cross-linkers and quaternary ammoniums as high-temperature proton exchange membranes: Enhanced stability and conductivity. J. Membr. Sci. 2019, 593, 117435. [Google Scholar] [CrossRef]
- Lee, H.-S.; Roy, A.; Lane, O.; McGrath, J.E. Synthesis and characterization of poly(arylene ether sulfone)-b-polybenzimidazole copolymers for high temperature low humidity proton exchange membrane fuel cells. Polymer 2008, 49, 5387–5396. [Google Scholar] [CrossRef]
- Zuo, Z.; Fu, Y.; Manthiram, A. Novel Blend Membranes Based on Acid-Base Interactions for Fuel Cells. Polymers 2012, 4, 1627–1644. [Google Scholar] [CrossRef]
- Suryani; Liu, Y.-L. Preparation and properties of nanocomposite membranes of polybenzimidazole/sulfonated silica nanoparticles for proton exchange membranes. J. Membr. Sci. 2009, 332, 121–128. [Google Scholar] [CrossRef]
- Bai, H.; Wang, H.; Zhang, J.; Wu, C.; Zhang, J.; Xiang, Y.; Lu, S. Simultaneously enhancing ionic conduction and mechanical strength of poly(ether sulfones)-poly(vinyl pyrrolidone) membrane by introducing graphitic carbon nitride nanosheets for high temperature proton exchange membrane fuel cell application. J. Membr. Sci. 2018, 558, 26–33. [Google Scholar] [CrossRef]
- Joseph, D.; Krishnan, N.N.; Henkensmeier, D.; Jang, J.H.; Choi, S.H.; Kim, H.-J.; Han, J.; Nam, S.W. Thermal crosslinking of PBI/sulfonated polysulfone based blend membranes. J. Mater. Chem. A 2017, 1, 409–417. [Google Scholar] [CrossRef]
- Ossiander, T.; Perchthaler, M.; Heinzl, C.; Schönberger, F.; Völk, P.; Welsch, M.; Chromik, A.; Hacker, V.; Scheu, C. Influence of membrane type and molecular weight distribution on the degradation of PBI-based HTPEM fuel cells. J. Membr. Sci. 2016, 509, 27–35. [Google Scholar] [CrossRef]
- Zkan, N.; Devrim, Y. Fabrication and Characterization of Cross-linked Polybenzimidazole Based Membranes for High Temperature PEM Fuel Cells. Electrochim. Acta 2017, 245, 1–13. [Google Scholar]
- Søndergaard, T.; Cleemann, L.N.; Becker, H.; Aili, D.; Steenberg, T.; Hjuler, H.A.; Seerup, L.; Li, Q.; Jensen, J.O. Long-term durability of HT-PEM fuel cells based on thermally cross-linked polybenzimidazole. J. Power Source 2017, 342, 570–578. [Google Scholar] [CrossRef] [Green Version]
- Yang, J.; Gao, L.; Wang, J.; Xu, Y.; Liu, C.; He, R. Strengthening Phosphoric Acid Doped Polybenzimidazole Membranes with Siloxane Networks for Using as High Temperature Proton Exchange Membranes. Macromol. Chem. Phys. 2017, 218. [Google Scholar] [CrossRef]
- Yang, J.; Li, Q.; Cleemann, L.N.; Jensen, J.O.; Pan, C.; Bjerrum, N.J.; He, R. Crosslinked Hexafluoropropylidene Polybenzimidazole Membranes with Chloromethyl Polysulfone for Fuel Cell Applications. Adv. Energy Mater. 2013, 3, 622–630. [Google Scholar] [CrossRef]
- Li, Q.; Pan, C.; Jensen, J.O.; Noyé, A.P.; Bjerrum, N.J. Cross-Linked Polybenzimidazole Membranes for Fuel Cells. Chem. Mater. 2007, 19, 350–352. [Google Scholar] [CrossRef]
- Kim, S.-K.; Choi, S.-W.; Jeon, W.S.; Park, J.O.; Ko, T.; Chang, H.; Lee, J.-C. Cross-Linked Benzoxazine–Benzimidazole Copolymer Electrolyte Membranes for Fuel Cells at Elevated Temperature. Macromolecules 2012, 45, 1438–1446. [Google Scholar] [CrossRef]
- Wang, S.; Zhao, C.; Ma, W.; Zhang, N.; Zhang, Y.; Zhang, G.; Liu, Z.; Na, H. Silane-cross-linked polybenzimidazole with improved conductivity for high temperature proton exchange membrane fuel cells. J. Mater. Chem. A Mater. Energy Sustain. 2013, 1, 621–629. [Google Scholar] [CrossRef]
- Thomas, O.D.; Peckham, T.J.; Thanganathan, U.; Yang, Y.; Holdcroft, S. Sulfonated polybenzimidazoles: Proton conduction and acid-base crosslinking. J. Polym. Sci. Part A Polym. Chem. 2010, 48, 3640–3650. [Google Scholar] [CrossRef]
- Nasef, M.M. Radiation-Grafted Membranes for Polymer Electrolyte Fuel Cells: Current Trends and Future Directions. Chem. Rev. 2014, 114, 12278–12329. [Google Scholar] [CrossRef]
- Valtcheva, I.B.; Kumbharkar, S.C.; Kim, J.F.; Bhole, Y.; Livingston, A. Beyond polyimide: Crosslinked polybenzimidazole membranes for organic solvent nanofiltration (OSN) in harsh environments. J. Membr. Sci. 2014, 457, 62–72. [Google Scholar] [CrossRef]
- Wang, S.; Zhang, G.; Han, M.; Li, H.; Ni, J.; Ma, W.; Li, M.; Wang, J.; Liu, Z.; Zhang, L.; et al. Novel epoxy-based cross-linked polybenzimidazole for high temperature proton exchange membrane fuel cells. Int. J. Hydrogen Energy 2011, 21, 2187–2193. [Google Scholar] [CrossRef]
- An, D.; Wu, B.; Zhang, G.; Zhang, W.; Wang, Y. Gradiently crosslinked polymer electrolyte membranes in fuel cells. J. Power Source 2016, 301, 204–209. [Google Scholar] [CrossRef]
- Anderson, L.J.; Yuan, X.; Fahs, G.B.; Moore, R.B. Blocky Ionomers via Sulfonation of Poly(ether ether ketone) in the Semicrystalline Gel State. Macromolecules 2018, 51, 6226–6237. [Google Scholar] [CrossRef]
- Kumar, G.G.; Manthiram, A. Sulfonated polyether ether ketone/strontium zirconite@TiO2 nanocomposite membranes for direct methanol fuel cells. J. Mater. Chem. A 2017, 5, 20497–20504. [Google Scholar] [CrossRef]
- Jiang, J.; Qu, E.; Xiao, M.; Han, D.; Wang, S.; Meng, Y. 3D Network Structural Poly (Aryl Ether Ketone)-Polybenzimidazole Polymer for High-Temperature Proton Exchange Membrane Fuel Cells. Adv. Polym. Technol. 2020, 2020, 1–13. [Google Scholar] [CrossRef]
- Ma, W.; Zhao, C.; Lin, H.; Zhang, G.; Ni, J.; Wang, J.; Wang, S.; Na, H. High-temperature water-free proton conducting membranes based on poly(arylene ether ketone) containing pendant quaternary ammonium groups with enhanced proton transport. J. Power Source 2011, 196, 9331–9338. [Google Scholar] [CrossRef]


| Sample | OPBI | PAEK-cr-PBI | Semi-IPN/70PBI |
|---|---|---|---|
| T–5%/°C | 399 | 401 | 453 |
| Membrane | PA Uptake (%) | Svolume (%) | Tensile Strength (MPa) | Elongation (%) |
|---|---|---|---|---|
| OPBI | 219 ± 3 | 128 ± 12 | 5.1 ± 0.8 | 22.1 ± 2.2 |
| PAEK-cr-PBI | 121 ± 2 | 46 ± 3 | 20.4 ± 1.2 | 4.6 ± 1.1 |
| Semi-IPN/70PBI | 191 ± 1 | 78 ± 3 | 11.8 ± 0.3 | 20.3 ± 4.8 |
| Semi-IPN/60PBI | 179 ± 5 | 72 ± 3 | 12.8 ± 0.2 | 18.6 ± 1.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Qu, E.; Jiang, J.; Xiao, M.; Han, D.; Huang, S.; Huang, Z.; Wang, S.; Meng, Y. Polybenzimidazole Confined in Semi-Interpenetrating Networks of Crosslinked Poly (Arylene Ether Ketone) for High Temperature Proton Exchange Membrane. Nanomaterials 2022, 12, 773. https://doi.org/10.3390/nano12050773
Qu E, Jiang J, Xiao M, Han D, Huang S, Huang Z, Wang S, Meng Y. Polybenzimidazole Confined in Semi-Interpenetrating Networks of Crosslinked Poly (Arylene Ether Ketone) for High Temperature Proton Exchange Membrane. Nanomaterials. 2022; 12(5):773. https://doi.org/10.3390/nano12050773
Chicago/Turabian StyleQu, Erli, Junqiao Jiang, Min Xiao, Dongmei Han, Sheng Huang, Zhiheng Huang, Shuanjin Wang, and Yuezhong Meng. 2022. "Polybenzimidazole Confined in Semi-Interpenetrating Networks of Crosslinked Poly (Arylene Ether Ketone) for High Temperature Proton Exchange Membrane" Nanomaterials 12, no. 5: 773. https://doi.org/10.3390/nano12050773








