Biodegradable Microrobots and Their Biomedical Applications: A Review
Abstract
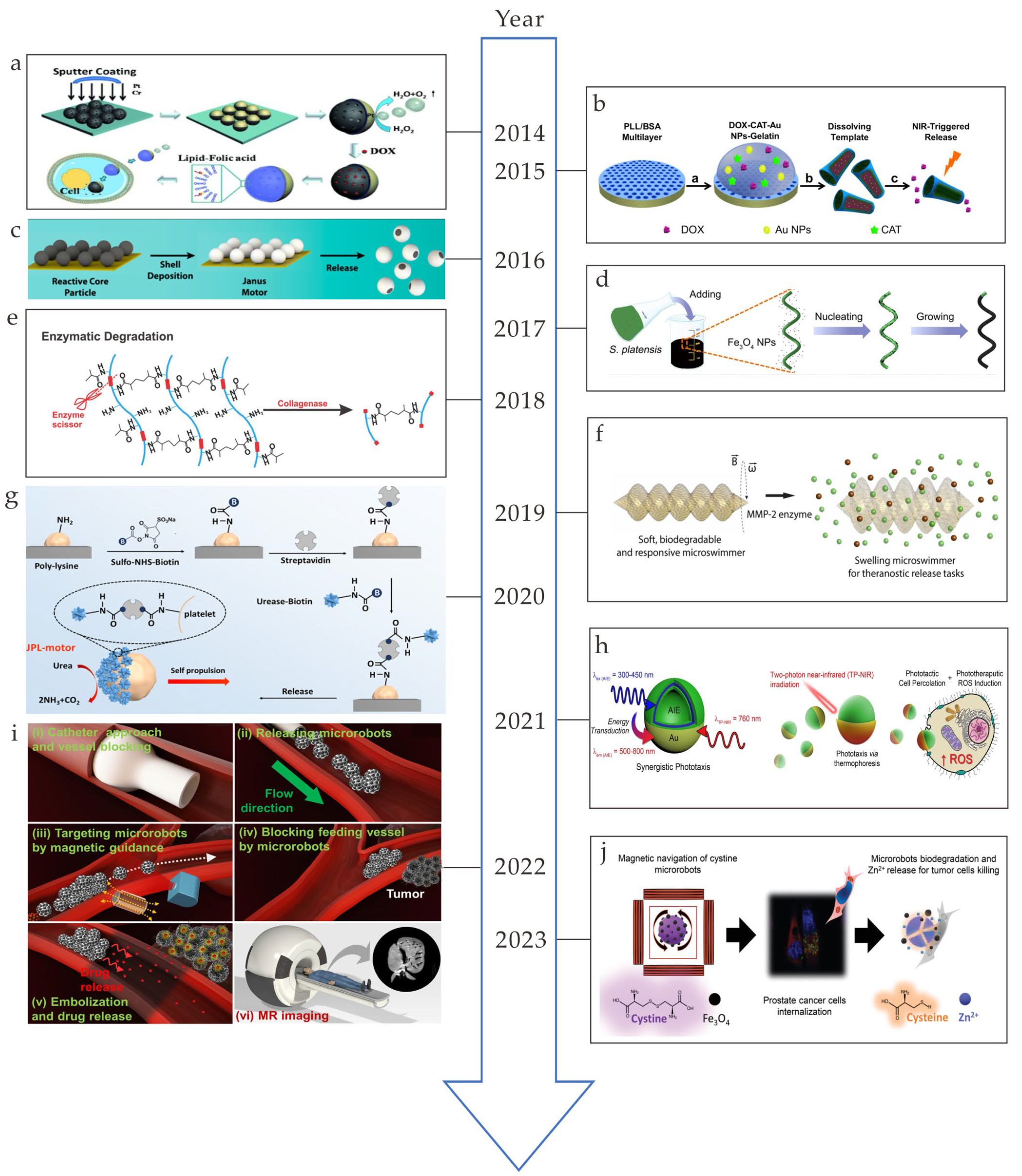
:1. Introduction
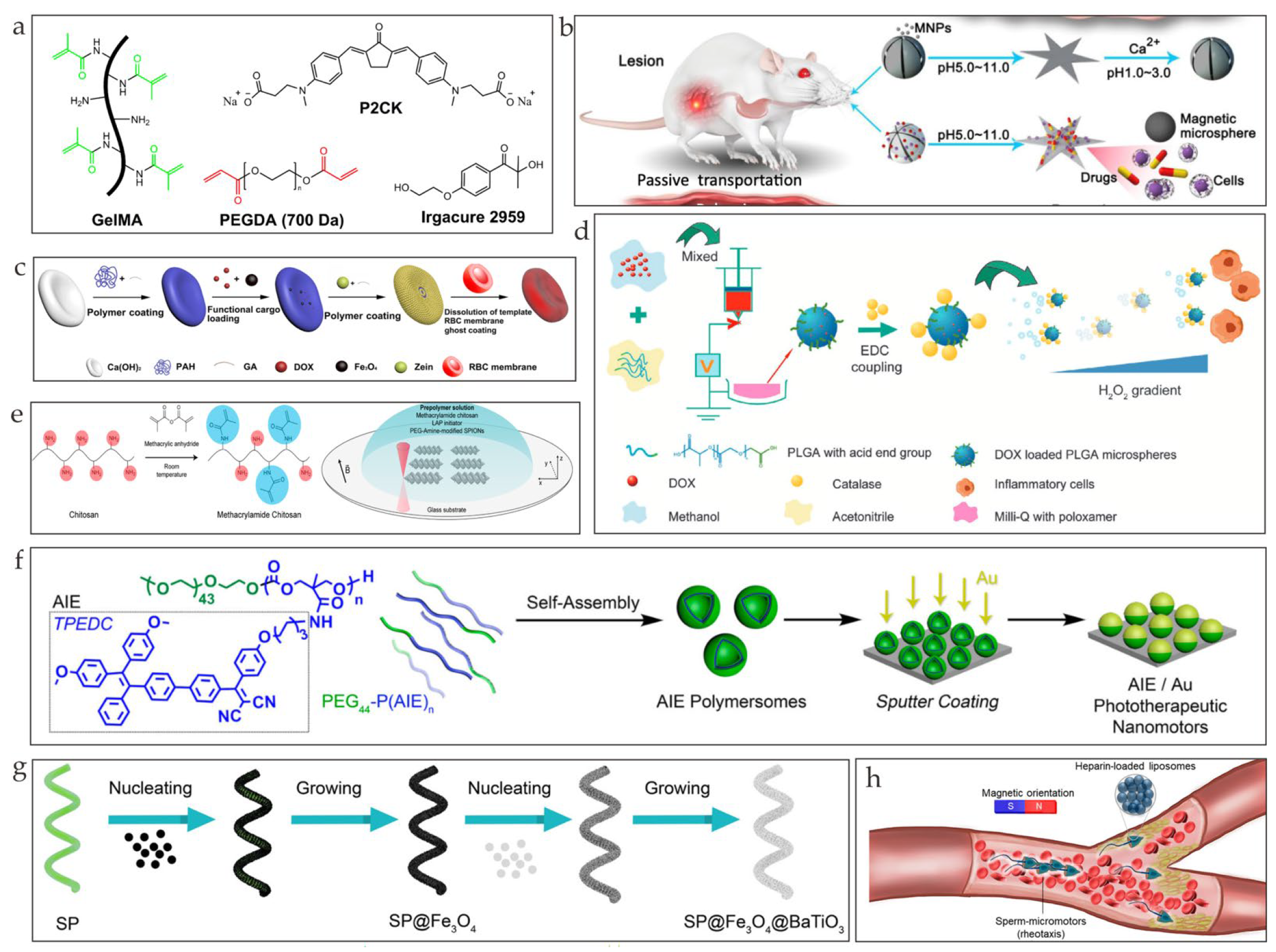
2. Biodegradable Materials
2.1. Metal-Based Microrobots
2.2. Polymer-Based Microrobots
2.3. Biohybrid Microrobots
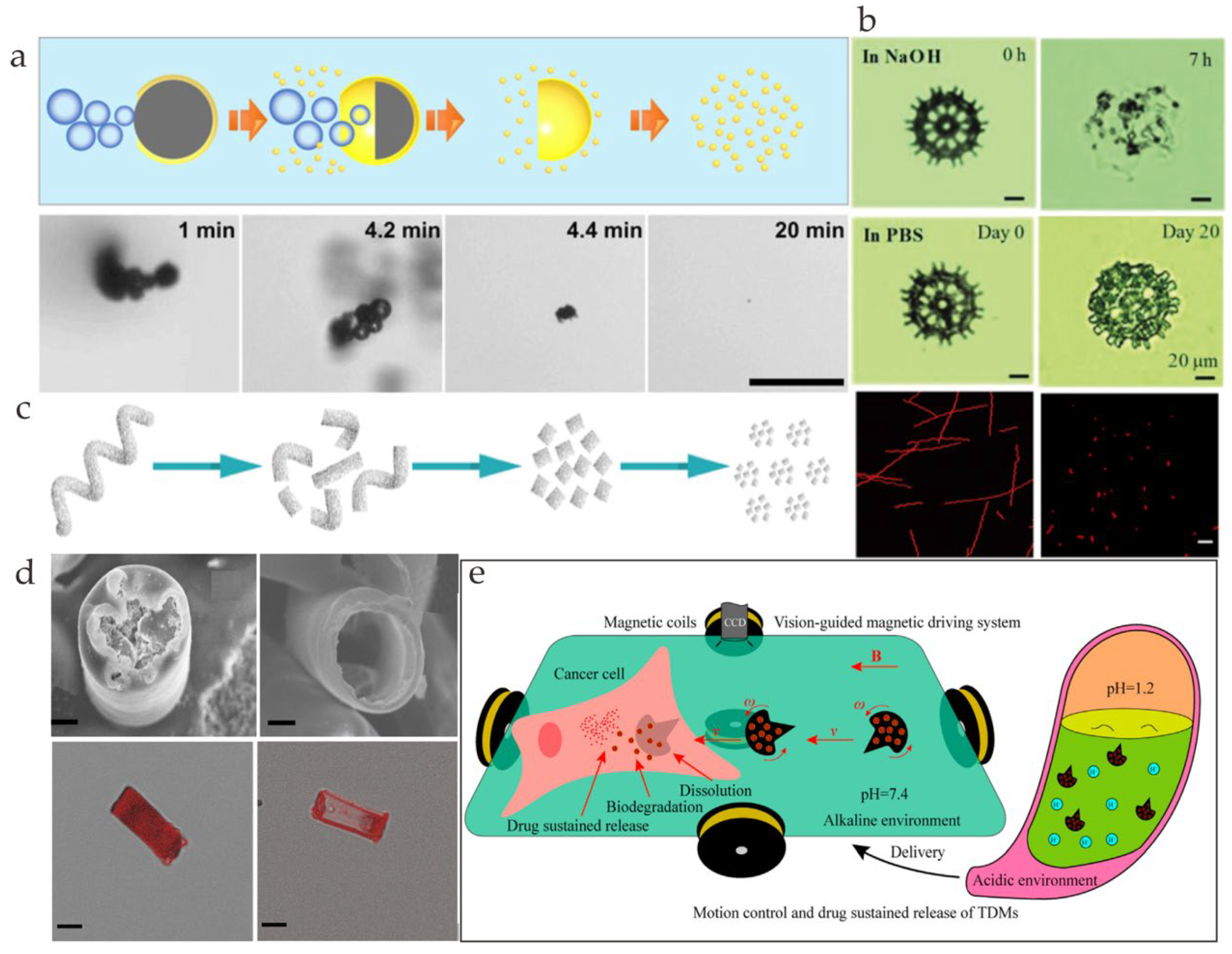
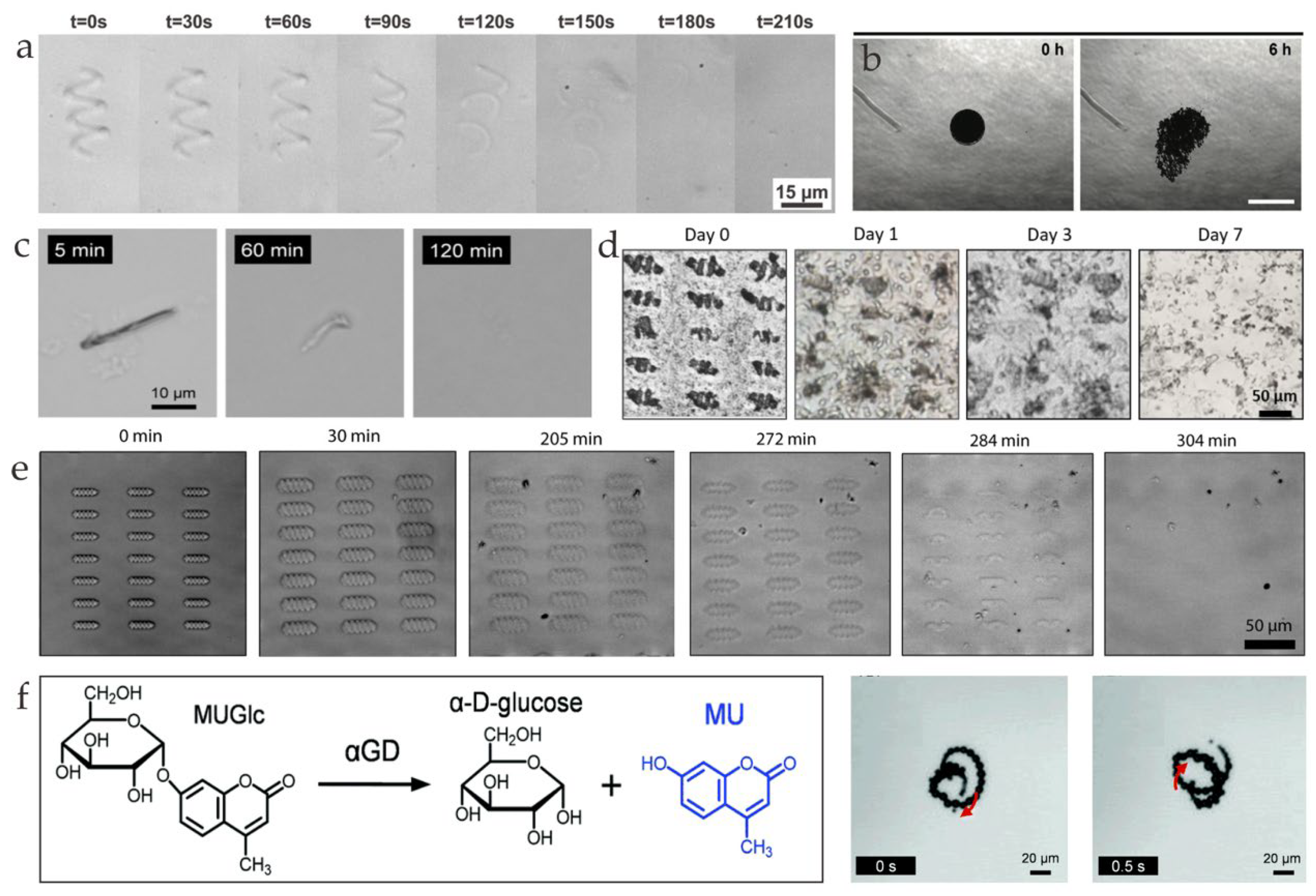
3. Degradation Mechanism
3.1. pH Degradation
3.2. Protease
3.3. Lipase
4. Biomedical Application
4.1. Drug Delivery
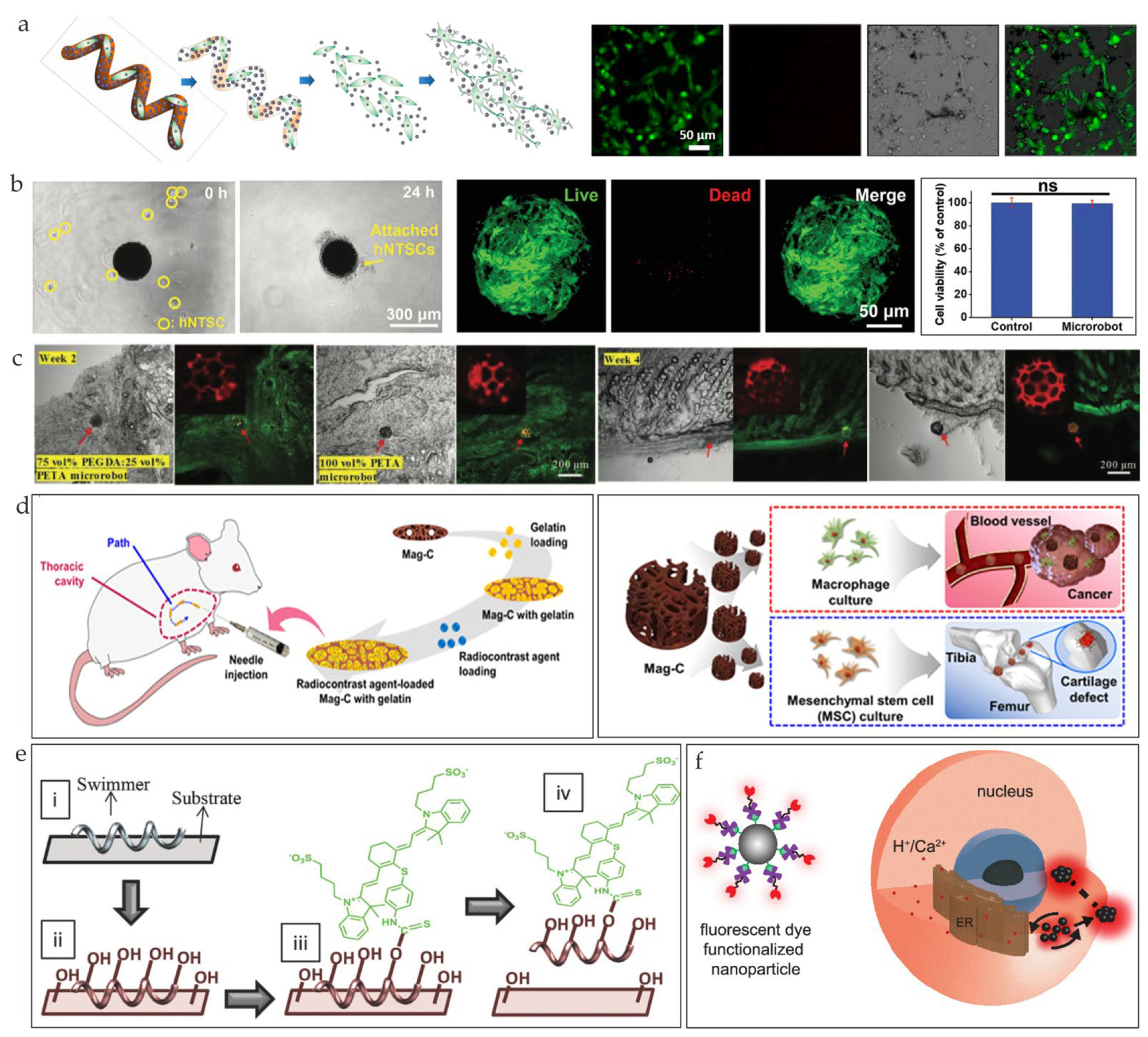
4.2. Cell Delivery
4.3. Bioimaging
5. Conclusions and Outlook
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Nelson, B.J.; Kaliakatsos, I.K.; Abbott, J.J. Microrobots for Minimally Invasive Medicine. Annu. Rev. Biomed. Eng. 2010, 12, 55–85. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Yang, Z.; Ferreira, A.; Zhang, L. Control and Autonomy of Microrobots: Recent Progress and Perspective. Adv. Intell. Syst. 2022, 4, 2100279. [Google Scholar] [CrossRef]
- Law, J.; Chen, H.; Wang, Y.; Yu, J.; Sun, Y. Gravity-resisting colloidal collectives. Sci. Adv. 2022, 8, eade3161. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Yang, L.; Du, X.; Chen, H.; Xu, T.; Zhang, L. Adaptive Pattern and Motion Control of Magnetic Microrobotic Swarms. IEEE Trans. Robot. 2022, 38, 1552–1570. [Google Scholar] [CrossRef]
- Zhou, H.; Mayorga-Martinez, C.C.; Pané, S.; Zhang, L.; Pumera, M. Magnetically Driven Micro and Nanorobots. Chem. Rev. 2021, 121, 4999–5041. [Google Scholar] [CrossRef] [PubMed]
- Nocentini, S.; Parmeggiani, C.; Martella, D.; Wiersma, D.S. Optically Driven Soft Micro Robotics. Adv. Opt. Mater. 2018, 6, 1800207. [Google Scholar] [CrossRef]
- Liu, D.K.-C.; Friend, J.; Yeo, L. A brief review of actuation at the micro-scale using electrostatics, electromagnetics and piezoelectric ultrasonics. Acoust. Sci. Technol. 2010, 31, 115–123. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, H.; Xu, T.; Yu, J. An Overview of Micronanoswarms for Biomedical Applications. ACS Nano 2021, 15, 15625–15644. [Google Scholar] [CrossRef] [PubMed]
- Law, J.; Wang, X.; Luo, M.; Xin, L.; Du, X.; Dou, W.; Wang, T.; Shan, G.; Wang, Y.; Song, P.; et al. Microrobotic swarms for selective embolization. Sci. Adv. 2022, 8, eabm5752. [Google Scholar] [CrossRef]
- Sitti, M.; Ceylan, H.; Hu, W.; Giltinan, J.; Turan, M.; Yim, S.; Diller, E. Biomedical Applications of Untethered Mobile Milli/Microrobots. Proc. IEEE 2015, 103, 205–224. [Google Scholar] [CrossRef]
- Schmidt, C.K.; Medina-Sánchez, M.; Edmondson, R.J.; Schmidt, O.G. Engineering microrobots for targeted cancer therapies from a medical perspective. Nat. Commun. 2020, 11, 56. [Google Scholar] [CrossRef] [PubMed]
- Qualliotine, J.R.; Bolat, G.; Beltrán-Gastélum, M.; de Ávila, B.E.; Wang, J.; Califano, J.A. Acoustic Nanomotors for Detection of Human Papillomavirus–Associated Head and Neck Cancer. Otolaryngol. Neck Surg. 2019, 161, 814–822. [Google Scholar] [CrossRef]
- Liu, M.; Chen, L.; Zhao, Z.; Liu, M.; Zhao, T.; Ma, Y.; Zhou, Q.; Ibrahim, Y.S.; Elzatahry, A.A.; Li, X.; et al. Enzyme-Based Mesoporous Nanomotors with Near-Infrared Optical Brakes. J. Am. Chem. Soc. 2022, 144, 3892–3901. [Google Scholar] [CrossRef] [PubMed]
- Dong, R.; Zhang, Q.; Gao, W.; Pei, A.; Ren, B. Highly Efficient Light-Driven TiO2–Au Janus Micromotors. ACS Nano 2016, 10, 839–844. [Google Scholar] [CrossRef] [PubMed]
- Cao, H.; Xing, L.; Mo, H.; Li, D.; Sun, D. Image-Guided Corridor-Based Motion Planning and Magnetic Control of Microrotor in Dynamic Environments. IEEE/ASME Trans. Mechatron. 2022, 27, 5415–5426. [Google Scholar] [CrossRef]
- de Ávila, B.E.-F.; Angsantikul, P.; Li, J.; Lopez-Ramirez, M.A.; Ramírez-Herrera, D.E.; Thamphiwatana, S.; Chen, C.; Delezuk, J.; Samakapiruk, R.; Ramez, V.; et al. Micromotor-enabled active drug delivery for in vivo treatment of stomach infection. Nat. Commun. 2017, 8, 272. [Google Scholar] [CrossRef]
- Peng, X.; Urso, M.; Ussia, M.; Pumera, M. Shape-Controlled Self-Assembly of Light-Powered Microrobots into Ordered Microchains for Cells Transport and Water Remediation. ACS Nano 2022, 16, 7615–7625. [Google Scholar] [CrossRef]
- Dong, R.; Hu, Y.; Wu, Y.; Gao, W.; Ren, B.; Wang, Q.; Cai, Y. Visible-Light-Driven BiOI-Based Janus Micromotor in Pure Water. J. Am. Chem. Soc. 2017, 139, 1722–1725. [Google Scholar] [CrossRef]
- Gao, W.; Pei, A.; Wang, J. Water-Driven Micromotors. ACS Nano 2012, 6, 8432–8438. [Google Scholar] [CrossRef]
- Wang, B.; Kostarelos, K.; Nelson, B.J.; Zhang, L. Trends in Micro-/Nanorobotics: Materials Development, Actuation, Localization, and System Integration for Biomedical Applications. Adv. Mater. 2021, 33, e2002047. [Google Scholar] [CrossRef]
- Peyer, K.E.; Zhang, L.; Nelson, B.J. Bio-inspired magnetic swimming microrobots for biomedical applications. Nanoscale 2013, 5, 1259–1272. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; de Ávila, B.E.-F.; Gao, W.; Zhang, L.; Wang, J. Micro/nanorobots for biomedicine: Delivery, surgery, sensing, and detoxification. Sci. Robot. 2017, 2, eaam6431. [Google Scholar] [CrossRef]
- Gao, C.; Wang, Y.; Ye, Z.; Lin, Z.; Ma, X.; He, Q. Biomedical Micro-/Nanomotors: From Overcoming Biological Barriers to In Vivo Imaging. Adv. Mater. 2021, 33, e2000512. [Google Scholar] [CrossRef] [PubMed]
- Aziz, A.; Pane, S.; Iacovacci, V.; Koukourakis, N.; Czarske, J.; Menciassi, A.; Medina-Sánchez, M.; Schmidt, O.G. Medical Imaging of Microrobots: Toward In Vivo Applications. ACS Nano 2020, 14, 10865–10893. [Google Scholar] [CrossRef]
- Xuan, M.; Shao, J.; Lin, X.; Dai, L.; He, Q. Self-Propelled Janus Mesoporous Silica Nanomotors with Sub-100 nm Diameters for Drug Encapsulation and Delivery. ChemPhysChem 2014, 15, 2255–2260. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Lin, X.; Zou, X.; Sun, J.; He, Q. Biodegradable Protein-Based Rockets for Drug Transportation and Light-Triggered Release. ACS Appl. Mater. Interfaces 2015, 7, 250–255. [Google Scholar] [CrossRef]
- Chen, C.; Karshalev, E.; Li, J.; Soto, F.; Castillo, R.; Campos, I.; Mou, F.; Guan, J.; Wang, J. Transient Micromotors That Disappear When No Longer Needed. ACS Nano 2016, 10, 10389–10396. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Zhou, Q.; Vincent, M.; Deng, Y.; Yu, J.; Xu, J.; Xu, T.; Tang, T.; Bian, L.; Wang, Y.-X.J.; et al. Multifunctional biohybrid magnetite microrobots for imaging-guided therapy. Sci. Robot. 2017, 2, eaaq1155. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Qin, X.-H.; Hu, C.; Terzopoulou, A.; Chen, X.; Huang, T.-Y.; Maniura, K.; Pané, S.; Nelson, B.J. 3D Printed Enzymatically Biodegradable Soft Helical Microswimmers. Adv. Funct. Mater. 2018, 28, 1804107. [Google Scholar] [CrossRef]
- Ceylan, H.; Yasa, I.C.; Yasa, O.; Tabak, A.F.; Giltinan, J.; Sitti, M. 3D-Printed Biodegradable Microswimmer for Theranostic Cargo Delivery and Release. ACS Nano 2019, 13, 3353–3362. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.; Zhang, F.; Gong, H.; Wei, F.; Zhuang, J.; Karshalev, E.; de Ávila, B.E.-F.; Huang, C.; Zhou, Z.; Li, Z.; et al. Enzyme-powered Janus platelet cell robots for active and targeted drug delivery. Sci. Robot. 2020, 5, eaba6137. [Google Scholar] [CrossRef]
- Cao, S.; Shao, J.; Wu, H.; Song, S.; De Martino, M.T.; Pijpers, I.A.B.; Friedrich, H.; Abdelmohsen, L.K.E.A.; Williams, D.S.; van Hest, J.C.M. Photoactivated nanomotors via aggregation induced emission for enhanced phototherapy. Nat. Commun. 2021, 12, 2077. [Google Scholar] [CrossRef] [PubMed]
- Go, G.; Yoo, A.; Nguyen, K.T.; Nan, M.; Darmawan, B.A.; Zheng, S.; Kang, B.; Kim, C.-S.; Bang, D.; Lee, S.; et al. Multifunctional microrobot with real-time visualization and magnetic resonance imaging for chemoembolization therapy of liver cancer. Sci. Adv. 2022, 8, eabq8545. [Google Scholar] [CrossRef] [PubMed]
- Ussia, M.; Urso, M.; Kratochvilova, M.; Navratil, J.; Balvan, J.; Mayorga-Martinez, C.C.; Vyskocil, J.; Masarik, M.; Pumera, M. Magnetically Driven Self-Degrading Zinc-Containing Cystine Microrobots for Treatment of Prostate Cancer. Small 2023, 19, 2208259. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Xu, J.; Zhou, Q.; Geng, P.; Wang, B.; Zhou, Y.; Liu, K.; Peng, F.; Tu, Y. Biodegradability of Micro/Nanomotors: Challenges and Opportunities. Adv. Healthc. Mater. 2021, 10, e2100335. [Google Scholar] [CrossRef]
- Zhao, S.; Sun, D.; Zhang, J.; Lu, H.; Wang, Y.; Xiong, R.; Grattan, K. Actuation and biomedical development of micro-/nanorobots—A review. Mater. Today Nano 2022, 18, 100223. [Google Scholar] [CrossRef]
- Feng, Y.; An, M.; Liu, Y.; Sarwar, M.T.; Yang, H. Advances in Chemically Powered Micro/Nanorobots for Biological Applications: A Review. Adv. Funct. Mater. 2022, 33, 2209883. [Google Scholar] [CrossRef]
- Bente, K.; Codutti, A.; Bachmann, F.; Faivre, D. Biohybrid and Bioinspired Magnetic Microswimmers. Small 2018, 14, e1704374. [Google Scholar] [CrossRef]
- Palagi, S.; Fischer, P. Bioinspired microrobots. Nat. Rev. Mater. 2018, 3, 113–124. [Google Scholar] [CrossRef]
- Wang, L.; Meng, Z.; Chen, Y.; Zheng, Y. Engineering Magnetic Micro/Nanorobots for Versatile Biomedical Applications. Adv. Intell. Syst. 2021, 3, 2000267. [Google Scholar] [CrossRef]
- Carlotti, M.; Mattoli, V. Functional Materials for Two-Photon Polymerization in Microfabrication. Small 2019, 15, e1902687. [Google Scholar] [CrossRef]
- Tan, L.; Davis, A.C.; Cappelleri, D.J. Smart Polymers for Microscale Machines. Adv. Funct. Mater. 2021, 31, 2007125. [Google Scholar] [CrossRef]
- Mei, Y.; Solovev, A.A.; Sanchez, S.; Schmidt, O.G. Rolled-up nanotech on polymers: From basic perception to self-propelled catalytic microengines. Chem. Soc. Rev. 2011, 40, 2109–2119. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Pumera, M. 3D printing of functional microrobots. Chem. Soc. Rev. 2021, 50, 2794–2838. [Google Scholar] [CrossRef] [PubMed]
- Spiegel, C.A.; Hippler, M.; Münchinger, A.; Bastmeyer, M.; Barner-Kowollik, C.; Wegener, M.; Blasco, E. 4D Printing at the Microscale. Adv. Funct. Mater. 2020, 30, 1907615. [Google Scholar] [CrossRef]
- Lin, Z.; Gao, C.; Wang, D.; He, Q. Bubble-Propelled Janus Gallium/Zinc Micromotors for the Active Treatment of Bacterial Infections. Angew. Chem. Int. Ed. 2021, 60, 8750–8754. [Google Scholar] [CrossRef]
- Xu, C.; Wang, S.; Wang, H.; Liu, K.; Zhang, S.; Chen, B.; Liu, H.; Tong, F.; Peng, F.; Tu, Y.; et al. Magnesium-Based Micromotors as Hydrogen Generators for Precise Rheumatoid Arthritis Therapy. Nano Lett. 2021, 21, 1982–1991. [Google Scholar] [CrossRef]
- Li, J.; Angsantikul, P.; Liu, W.; De Ávila, B.E.-F.; Thamphiwatana, S.; Xu, M.; Sandraz, E.; Wang, X.; Delezuk, J.; Gao, W.; et al. Micromotors Spontaneously Neutralize Gastric Acid for pH-Responsive Payload Release. Angew. Chem. Int. Ed. 2017, 56, 2156–2161. [Google Scholar] [CrossRef]
- Zhou, M.; Hou, T.; Li, J.; Yu, S.; Xu, Z.; Yin, M.; Wang, J.; Wang, X. Self-Propelled and Targeted Drug Delivery of Poly(aspartic acid)/Iron–Zinc Microrocket in the Stomach. ACS Nano 2019, 13, 1324–1332. [Google Scholar] [CrossRef]
- Jose, A.; Jagatheeswari, P. Neutroscopic data synthesis of biodegradable polymer for industrial bio robots. Cogn. Syst. Res. 2019, 56, 72–81. [Google Scholar] [CrossRef]
- Hu, N.; Wang, L.; Zhai, W.; Sun, M.; Xie, H.; Wu, Z.; He, Q. Magnetically Actuated Rolling of Star-Shaped Hydrogel Microswimmer. Macromol. Chem. Phys. 2018, 219, 1700540. [Google Scholar] [CrossRef]
- Bhushan, V.; Heitz, M.P.; Baker, G.A.; Pandey, S. Ionic Liquid-Controlled Shape Transformation of Spherical to Nonspherical Polymersomes via Hierarchical Self-Assembly of a Diblock Copolymer. Langmuir 2021, 37, 5081–5088. [Google Scholar] [CrossRef] [PubMed]
- Toebes, B.J.; Cao, F.; Wilson, D.A. Spatial control over catalyst positioning on biodegradable polymeric nanomotors. Nat. Commun. 2019, 10, 5308. [Google Scholar] [CrossRef] [PubMed]
- Bagheri, A.; Jin, J. Photopolymerization in 3D Printing. ACS Appl. Polym. Mater. 2019, 1, 593–611. [Google Scholar] [CrossRef]
- Layani, M.; Wang, X.; Magdassi, S. Novel Materials for 3D Printing by Photopolymerization. Adv. Mater. 2018, 30, e1706344. [Google Scholar] [CrossRef]
- Li, J.; Wu, C.; Chu, P.K.; Gelinsky, M. 3D printing of hydrogels: Rational design strategies and emerging biomedical applications. Mater. Sci. Eng. R Rep. 2020, 140, 100543. [Google Scholar] [CrossRef]
- Xing, J.-F.; Zheng, M.-L.; Duan, X.-M. Two-photon polymerization microfabrication of hydrogels: An advanced 3D printing technology for tissue engineering and drug delivery. Chem. Soc. Rev. 2015, 44, 5031–5039. [Google Scholar] [CrossRef]
- Kotz, F.; Quick, A.S.; Risch, P.; Martin, T.; Hoose, T.; Thiel, M.; Helmer, D.; Rapp, B.E. Two-Photon Polymerization of Nanocomposites for the Fabrication of Transparent Fused Silica Glass Microstructures. Adv. Mater. 2021, 33, e2006341. [Google Scholar] [CrossRef]
- Louis, F.; Piantino, M.; Liu, H.; Kang, D.-H.; Sowa, Y.; Kitano, S.; Matsusaki, M. Bioprinted Vascularized Mature Adipose Tissue with Collagen Microfibers for Soft Tissue Regeneration. Cyborg Bionic Syst. 2021, 2021, 1412542. [Google Scholar] [CrossRef]
- Hou, K.; Zhang, Y.; Bao, M.; Xin, C.; Wei, Z.; Lin, G.; Wang, Z. A Multifunctional Magnetic Red Blood Cell-Mimetic Micromotor for Drug Delivery and Image-Guided Therapy. ACS Appl. Mater. Interfaces 2022, 14, 3825–3837. [Google Scholar] [CrossRef]
- Zheng, Z.; Wang, H.; Dong, L.; Shi, Q.; Li, J.; Sun, T.; Huang, Q.; Fukuda, T. Ionic shape-morphing microrobotic end-effectors for environmentally adaptive targeting, releasing, and sampling. Nat. Commun. 2021, 12, 411. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Toebes, B.J.; Plachokova, A.S.; Liu, Q.; Deng, D.; Jansen, J.A.; Yang, F.; Wilson, D.A. Self-Propelled PLGA Micromotor with Chemotactic Response to Inflammation. Adv. Healthc. Mater. 2020, 9, e1901710. [Google Scholar] [CrossRef] [PubMed]
- Bozuyuk, U.; Yasa, O.; Yasa, I.C.; Ceylan, H.; Kizilel, S.; Sitti, M. Light-Triggered Drug Release from 3D-Printed Magnetic Chitosan Microswimmers. ACS Nano 2018, 12, 9617–9625. [Google Scholar] [CrossRef] [PubMed]
- Quashie, D.; Benhal, P.; Chen, Z.; Wang, Z.; Mu, X.; Song, X.; Jiang, T.; Zhong, Y.; Cheang, U.K.; Ali, J. Magnetic bio-hybrid micro actuators. Nanoscale 2022, 14, 4364–4379. [Google Scholar] [CrossRef]
- Xie, L.; Pang, X.; Yan, X.; Dai, Q.; Lin, H.; Ye, J.; Cheng, Y.; Zhao, Q.; Ma, X.; Zhang, X.; et al. Photoacoustic Imaging-Trackable Magnetic Microswimmers for Pathogenic Bacterial Infection Treatment. ACS Nano 2020, 14, 2880–2893. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Wu, J.; Wang, S.; Kun, L.; Gao, J.; Chen, B.; Ye, Y.; Wang, F.; Tong, F.; Jiang, J.; et al. Control the Neural Stem Cell Fate with Biohybrid Piezoelectrical Magnetite Micromotors. Nano Lett. 2021, 21, 3518–3526. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Xu, J.; Zhou, Q.; Jin, D.; Vong, C.I.; Feng, Q.; Ng, D.H.; Bian, L.; Zhang, L. Molecular cargo delivery using multicellular magnetic microswimmers. Appl. Mater. Today 2019, 15, 242–251. [Google Scholar] [CrossRef]
- Magdanz, V.; Khalil, I.S.M.; Simmchen, J.; Furtado, G.P.; Mohanty, S.; Gebauer, J.; Xu, H.; Klingner, A.; Aziz, A.; Medina-Sánchez, M.; et al. IRONSperm: Sperm-templated soft magnetic microrobots. Sci. Adv. 2020, 6, eaba5855. [Google Scholar] [CrossRef]
- Xu, H.; Medina-Sánchez, M.; Maitz, M.F.; Werner, C.; Schmidt, O.G. Sperm Micromotors for Cargo Delivery through Flowing Blood. ACS Nano 2020, 14, 2982–2993. [Google Scholar] [CrossRef]
- Go, G.; Jeong, S.-G.; Yoo, A.; Han, J.; Kang, B.; Kim, S.; Nguyen, K.T.; Jin, Z.; Kim, C.-S.; Seo, Y.R.; et al. Human adipose–derived mesenchymal stem cell–based medical microrobot system for knee cartilage regeneration in vivo. Sci. Robot. 2020, 5, eaay6626. [Google Scholar] [CrossRef]
- Zhang, H.; Li, Z.; Gao, C.; Fan, X.; Pang, Y.; Li, T.; Wu, Z.; Xie, H.; He, Q. Dual-responsive biohybrid neutrobots for active target delivery. Sci. Robot. 2021, 6, eaaz9519. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Cao, Z.; Zhang, Q.; Xu, J.; Yun, S.L.J.; Liang, K.; Gu, Z. Chemotaxis-Driven 2D Nanosheet for Directional Drug Delivery toward the Tumor Microenvironment. Small 2020, 16, e2002732. [Google Scholar] [CrossRef] [PubMed]
- Wei, T.; Liu, J.; Li, D.; Chen, S.; Zhang, Y.; Li, J.; Fan, L.; Guan, Z.; Lo, C.; Wang, L.; et al. Development of Magnet-Driven and Image-Guided Degradable Microrobots for the Precise Delivery of Engineered Stem Cells for Cancer Therapy. Small 2020, 16, 1906908. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Sun, M.; Fan, X.; Xie, H. Magnetic/pH-sensitive double-layer microrobots for drug delivery and sustained release. Appl. Mater. Today 2020, 19, 100583. [Google Scholar] [CrossRef]
- Noh, S.; Jeon, S.; Kim, E.; Oh, U.; Park, D.; Park, S.H.; Kim, S.W.; Pané, S.; Nelson, B.J.; Kim, J.; et al. A Biodegradable Magnetic Microrobot Based on Gelatin Methacrylate for Precise Delivery of Stem Cells with Mass Production Capability. Small 2022, 18, 2107888. [Google Scholar] [CrossRef]
- Dong, M.; Wang, X.; Chen, X.; Mushtaq, F.; Deng, S.; Zhu, C.; Torlakcik, H.; Terzopoulou, A.; Qin, X.; Xiao, X.; et al. 3D-Printed Soft Magnetoelectric Microswimmers for Delivery and Differentiation of Neuron-Like Cells. Adv. Funct. Mater. 2020, 30, 1910323. [Google Scholar] [CrossRef]
- Sugai, N.; Morita, Y.; Komatsu, T. Nonbubble-Propelled Biodegradable Microtube Motors Consisting Only of Protein. Chem. Asian J. 2019, 14, 2953–2957. [Google Scholar] [CrossRef]
- Umebara, M.; Sugai, N.; Murayama, K.; Sugawara, T.; Akashi, Y.; Morita, Y.; Kato, R.; Komatsu, T. Catalase-driven protein microtube motors with different exterior surfaces as ultrasmall biotools. Mater. Adv. 2021, 2, 6428–6438. [Google Scholar] [CrossRef]
- Goudu, S.R.; Yasa, I.C.; Hu, X.; Ceylan, H.; Hu, W.; Sitti, M. Biodegradable Untethered Magnetic Hydrogel Milli-Grippers. Adv. Funct. Mater. 2020, 30, 2004975. [Google Scholar] [CrossRef]
- Liu, M.; Sun, Y.; Wang, T.; Ye, Z.; Zhang, H.; Dong, B.; Li, C.Y. A biodegradable, all-polymer micromotor for gas sensing applications. J. Mater. Chem. C 2016, 4, 5945–5952. [Google Scholar] [CrossRef]
- Kroupa, T.; Hermanová, S.; Mayorga-Martinez, C.C.; Novotný, F.; Sofer, Z.; Pumera, M. Micromotors as “Motherships”: A Concept for the Transport, Delivery, and Enzymatic Release of Molecular Cargo via Nanoparticles. Langmuir 2019, 35, 10618–10624. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.; Peng, F.; André, A.A.M.; Men, Y.; Srinivas, M.; Wilson, D.A. Biodegradable Hybrid Stomatocyte Nanomotors for Drug Delivery. ACS Nano 2017, 11, 1957–1963. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, M.; Mayorga-Martinez, C.C.; Viktorova, J.; Ruml, T.; Escarpa, A.; Pumera, M. Microrobotic carrier with enzymatically encoded drug release in the presence of pancreatic cancer cells via programmed self-destruction. Appl. Mater. Today 2022, 27, 101494. [Google Scholar] [CrossRef]
- Yan, M.; Ma, D.; Qiu, B.; Liu, T.; Xie, L.; Zeng, J.; Liang, K.; Xin, H.; Lian, Z.; Jiang, L.; et al. Superassembled Hierarchical Asymmetric Magnetic Mesoporous Nanorobots Driven by Smart Confined Catalytic Degradation. Chem. A Eur. J. 2022, 28, e202200307. [Google Scholar] [CrossRef]
- Toebes, B.J.; Abdelmohsen, L.K.E.A.; Wilson, D.A. Enzyme-driven biodegradable nanomotor based on tubular-shaped polymeric vesicles. Polym. Chem. 2018, 9, 3190–3194. [Google Scholar] [CrossRef]
- Pena-Francesch, A.; Giltinan, J.; Sitti, M. Multifunctional and biodegradable self-propelled protein motors. Nat. Commun. 2019, 10, 3188. [Google Scholar] [CrossRef]
- Feng, A.; Huang, X.; Cheng, X.; Chu, M.; Wang, S.; Yan, X. Programmable degrading engine powered photoactivated organic colloidal motors. Chem. Eng. J. 2022, 440, 135838. [Google Scholar] [CrossRef]
- Kim, D.-I.; Lee, H.; Kwon, S.-H.; Choi, H.; Park, S. Magnetic nano-particles retrievable biodegradable hydrogel microrobot. Sens. Actuators B Chem. 2019, 289, 65–77. [Google Scholar] [CrossRef]
- Wang, H.; Kan, J.; Zhang, X.; Gu, C.; Yang, Z. Pt/CNT Micro-Nanorobots Driven by Glucose Catalytic Decomposition. Cyborg Bionic Syst. 2021, 2021, 9876064. [Google Scholar] [CrossRef]
- Singh, A.V. Targeted Drug Delivery and Imaging Using Mobile Milli/Microrobots: A Promising Future Towards Theranostic Pharmaceutical Design. Curr. Pharm. Des. 2016, 22, 1418–1428. [Google Scholar] [CrossRef]
- Manzari, M.T.; Shamay, Y.; Kiguchi, H.; Rosen, N.; Scaltriti, M.; Heller, D.A. Targeted drug delivery strategies for precision medicines. Nat. Rev. Mater. 2021, 6, 351–370. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, K.T.; Go, G.; Jin, Z.; Darmawan, B.A.; Yoo, A.; Kim, S.; Nan, M.; Lee, S.B.; Kang, B.; Kim, C.; et al. A Magnetically Guided Self-Rolled Microrobot for Targeted Drug Delivery, Real-Time X-Ray Imaging, and Microrobot Retrieval. Adv. Healthc. Mater. 2021, 10, e2001681. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Choi, H.; Lee, M.; Park, S. Preliminary study on alginate/NIPAM hydrogel-based soft microrobot for controlled drug delivery using electromagnetic actuation and near-infrared stimulus. Biomed. Microdevices 2018, 20, 103. [Google Scholar] [CrossRef] [PubMed]
- Shao, J.; Abdelghani, M.; Shen, G.; Cao, S.; Williams, D.S.; van Hest, J.C.M. Erythrocyte Membrane Modified Janus Polymeric Motors for Thrombus Therapy. ACS Nano 2018, 12, 4877–4885. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; Zhong, Y.; Jiang, T.; Cheang, U.K. Effect of solvation on the synthesis of MOF-based microrobots and their targeted-therapy applications. Mater. Adv. 2021, 2, 3871–3880. [Google Scholar] [CrossRef]
- Mair, L.O.; Chowdhury, S.; Paredes-Juarez, G.A.; Guix, M.; Bi, C.; Johnson, B.; English, B.W.; Jafari, S.; Baker-McKee, J.; Watson-Daniels, J.; et al. Magnetically Aligned Nanorods in Alginate Capsules (MANiACs): Soft Matter Tumbling Robots for Manipulation and Drug Delivery. Micromachines 2019, 10, 230. [Google Scholar] [CrossRef]
- Terzopoulou, A.; Wang, X.; Chen, X.; Palacios-Corella, M.; Pujante, C.; Herrero-Martín, J.; Qin, X.; Sort, J.; Demello, A.J.; Nelson, B.J.; et al. Biodegradable Metal–Organic Framework-Based Microrobots (MOFBOTs). Adv. Healthc. Mater. 2020, 9, 2001031. [Google Scholar] [CrossRef]
- Wu, Z.; Wu, Y.; He, W.; Lin, X.; Sun, J.; He, Q. Self-Propelled Polymer-Based Multilayer Nanorockets for Transportation and Drug Release. Angew. Chem. Int. Ed. 2013, 52, 7000–7003. [Google Scholar] [CrossRef]
- Wu, Z.; Li, L.; Yang, Y.; Hu, P.; Li, Y.; Yang, S.-Y.; Wang, L.V.; Gao, W. A microrobotic system guided by photoacoustic computed tomography for targeted navigation in intestines in vivo. Sci. Robot. 2019, 4, eaax0613. [Google Scholar] [CrossRef]
- Akiyama, Y. Design of Temperature-Responsive Cell Culture Surfaces for Cell Sheet Engineering. Cyborg Bionic Syst. 2021, 2021, 5738457. [Google Scholar] [CrossRef]
- Go, G.; Yoo, A.; Song, H.-W.; Min, H.-K.; Zheng, S.; Nguyen, K.T.; Kim, S.; Kang, B.; Hong, A.; Kim, C.-S.; et al. Multifunctional Biodegradable Microrobot with Programmable Morphology for Biomedical Applications. ACS Nano 2021, 15, 1059–1076. [Google Scholar] [CrossRef]
- Than, T.D.; Alici, G.; Zhou, H.; Li, W. A Review of Localization Systems for Robotic Endoscopic Capsules. IEEE Trans. Biomed. Eng. 2012, 59, 2387–2399. [Google Scholar] [CrossRef] [PubMed]
- Pané, S.; Puigmartí-Luis, J.; Bergeles, C.; Chen, X.; Pellicer, E.; Sort, J.; Počepcová, V.; Ferreira, A.; Nelson, B.J. Imaging Technologies for Biomedical Micro- and Nanoswimmers. Adv. Mater. Technol. 2019, 4, 1800575. [Google Scholar] [CrossRef]
- Luo, Y.; Chen, Z.; Wen, S.; Han, Q.; Fu, L.; Yan, L.; Jin, D.; Bünzli, J.-C.G.; Bao, G. Magnetic regulation of the luminescence of hybrid lanthanide-doped nanoparticles. Co-Ord. Chem. Rev. 2022, 469, 214653. [Google Scholar] [CrossRef]
- Servant, A.; Qiu, F.; Mazza, M.; Kostarelos, K.; Nelson, B.J. Controlled In Vivo Swimming of a Swarm of Bacteria-Like Microrobotic Flagella. Adv. Mater. 2015, 27, 2981–2988. [Google Scholar] [CrossRef]
- Wang, X.; Wang, T.; Chen, X.; Law, J.; Shan, G.; Tang, W.; Gong, Z.; Pan, P.; Liu, X.; Yu, J.; et al. Microrobotic Swarms for Intracellular Measurement with Enhanced Signal-to-Noise Ratio. ACS Nano 2022, 16, 10824–10839. [Google Scholar] [CrossRef]
- Wrede, P.; Degtyaruk, O.; Kalva, S.K.; Deán-Ben, X.L.; Bozuyuk, U.; Aghakhani, A.; Akolpoglu, B.; Sitti, M.; Razansky, D. Real-time 3D optoacoustic tracking of cell-sized magnetic microrobots circulating in the mouse brain vasculature. Sci. Adv. 2022, 8, eabm9132. [Google Scholar] [CrossRef]
- Cheng, X.; Sun, R.; Yin, L.; Chai, Z.; Shi, H.; Gao, M. Light-Triggered Assembly of Gold Nanoparticles for Photothermal Therapy and Photoacoustic Imaging of Tumors In Vivo. Adv. Mater. 2017, 29, 1604894. [Google Scholar] [CrossRef]
- Ou, J.; Tian, H.; Wu, J.; Gao, J.; Jiang, J.; Liu, K.; Wang, S.; Wang, F.; Tong, F.; Ye, Y.; et al. MnO2-Based Nanomotors with Active Fenton-like Mn2+ Delivery for Enhanced Chemodynamic Therapy. ACS Appl. Mater. Interfaces 2021, 13, 38050–38060. [Google Scholar] [CrossRef]
- Torlakcik, H.; Sarica, C.; Bayer, P.; Yamamoto, K.; Iorio-Morin, C.; Hodaie, M.; Kalia, S.K.; Neimat, J.S.; Hernesniemi, J.; Bhatia, A.; et al. Magnetically Guided Catheters, Micro- and Nanorobots for Spinal Cord Stimulation. Front. Neurorobot. 2021, 15, 749024. [Google Scholar] [CrossRef]
- Wang, K.; Wang, W.; Pan, S.; Fu, Y.; Dong, B.; Wang, H. Fluorescent self-propelled covalent organic framework as a microsensor for nitro explosive detection. Appl. Mater. Today 2020, 19, 100550. [Google Scholar] [CrossRef]
- Wang, S.; Liu, K.; Zhou, Q.; Xu, C.; Gao, J.; Wang, Z.; Wang, F.; Chen, B.; Ye, Y.; Ou, J.; et al. Hydrogen-Powered Microswimmers for Precise and Active Hydrogen Therapy Towards Acute Ischemic Stroke. Adv. Funct. Mater. 2021, 31, 2009475. [Google Scholar] [CrossRef]
- Park, J.; Jin, C.; Lee, S.; Kim, J.; Choi, H. Magnetically Actuated Degradable Microrobots for Actively Controlled Drug Release and Hyperthermia Therapy. Adv. Healthc. Mater. 2019, 8, e1900213. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Wang, H.; Zhong, S.; Qiu, Y.; Shi, Q.; Sun, T.; Huang, Q.; Fukuda, T. Design and Control of a Surface-Dimple-Optimized Helical Microdrill for Motions in High-Viscosity Fluids. IEEE/ASME Trans. Mechatron. 2022, 28, 429–439. [Google Scholar] [CrossRef]
- Singh, A.V.; Ansari, M.H.D.; Mahajan, M.; Srivastava, S.; Kashyap, S.; Dwivedi, P.; Pandit, V.; Katha, U. Sperm Cell Driven Microrobots—Emerging Opportunities and Challenges for Biologically Inspired Robotic Design. Micromachines 2020, 11, 448. [Google Scholar] [CrossRef] [PubMed]
- Hashizume, M. Perspective for Future Medicine: Multidisciplinary Computational Anatomy-Based Medicine with Artificial Intelligence. Cyborg Bionic Syst. 2021, 2021, 9160478. [Google Scholar] [CrossRef]
- Yang, W.; Wang, X.; Wang, Z.; Liang, W.; Ge, Z. Light-powered microrobots: Recent progress and future challenges. Opt. Lasers Eng. 2023, 161, 107380. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, L.; Chan, T.K.F.; Chen, Z.; Ip, M.; Chan, P.K.S.; Sung, J.J.Y.; Zhang, L. Micro-/Nanorobots in Antimicrobial Applications: Recent Progress, Challenges, and Opportunities. Adv. Healthc. Mater. 2022, 11, 2101991. [Google Scholar] [CrossRef]
- Wang, H.; Pumera, M. Fabrication of Micro/Nanoscale Motors. Chem. Rev. 2015, 115, 8704–8735. [Google Scholar] [CrossRef]
- Zhang, H.; Hutmacher, D.W.; Chollet, F.; Poo, A.N.; Burdet, E. Microrobotics and MEMS-Based Fabrication Techniques for Scaffold-Based Tissue Engineering. Macromol. Biosci. 2005, 5, 477–489. [Google Scholar] [CrossRef]
- Aghakhani, A.; Pena-Francesch, A.; Bozuyuk, U.; Cetin, H.; Wrede, P.; Sitti, M. High shear rate propulsion of acoustic microrobots in complex biological fluids. Sci. Adv. 2022, 8, eabm5126. [Google Scholar] [CrossRef]
- Wu, Z.; Chen, Y.; Mukasa, D.; Pak, O.S.; Gao, W. Medical micro/nanorobots in complex media. Chem. Soc. Rev. 2020, 49, 8088–8112. [Google Scholar] [CrossRef]
- Li, G.; Lauga, E.; Ardekani, A.M. Microswimming in viscoelastic fluids. J. Non-Newton. Fluid Mech. 2021, 297, 104655. [Google Scholar] [CrossRef]
- Spagnolie, S.E.; Underhill, P.T. Swimming in Complex Fluids. Annu. Rev. Condens. Matter Phys. 2023, 14, 381–415. [Google Scholar] [CrossRef]
- Chen, Y.; Demir, E.; Gao, W.; Young, Y.-N.; Pak, O.S. Wall-induced translation of a rotating particle in a shear-thinning fluid. J. Fluid Mech. 2021, 927, R2. [Google Scholar] [CrossRef]
- Zou, Z.; Liu, Y.; Young, Y.-N.; Pak, O.S.; Tsang, A.C.H. Gait switching and targeted navigation of microswimmers via deep reinforcement learning. Commun. Phys. 2022, 5, 1–9. [Google Scholar] [CrossRef]
- Hartl, B.; Hübl, M.; Kahl, G.; Zöttl, A. Microswimmers learning chemotaxis with genetic algorithms. Proc. Natl. Acad. Sci. USA 2021, 118, e2019683118. [Google Scholar] [CrossRef] [PubMed]
- Nasiri, M.; Löwen, H.; Liebchen, B. Optimal active particle navigation meets machine learning (a). Europhys. Lett. 2023, 142, 17001. [Google Scholar] [CrossRef]
- Schneider, E.; Stark, H. Optimal steering of a smart active particle. Europhys. Lett. 2019, 127, 64003. [Google Scholar] [CrossRef]
- Qin, K.; Pak, O.S. Purcell’s swimmer in a shear-thinning fluid. Phys. Rev. Fluids 2023, 8, 033301. [Google Scholar] [CrossRef]
- Muiños-Landin, S.; Fischer, A.; Holubec, V.; Cichos, F. Reinforcement learning with artificial microswimmers. Sci. Robot. 2021, 6, eabd9285. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Wang, Y.; Liu, Y.; Zou, Q.; Yu, J. Sensing of Fluidic Features Using Colloidal Microswarms. ACS Nano 2022, 16, 16281–16291. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.; Du, X.; Liu, Y.; Chen, H.; Wang, Y.; Yu, J. Dynamic Path Planning and Motion Control of Microrobotic Swarms for Mobile Target Tracking. IEEE Trans. Autom. Sci. Eng. 2022, 1–15. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Yu, J. Biodegradable Microrobots and Their Biomedical Applications: A Review. Nanomaterials 2023, 13, 1590. https://doi.org/10.3390/nano13101590
Li J, Yu J. Biodegradable Microrobots and Their Biomedical Applications: A Review. Nanomaterials. 2023; 13(10):1590. https://doi.org/10.3390/nano13101590
Chicago/Turabian StyleLi, Jinxin, and Jiangfan Yu. 2023. "Biodegradable Microrobots and Their Biomedical Applications: A Review" Nanomaterials 13, no. 10: 1590. https://doi.org/10.3390/nano13101590
APA StyleLi, J., & Yu, J. (2023). Biodegradable Microrobots and Their Biomedical Applications: A Review. Nanomaterials, 13(10), 1590. https://doi.org/10.3390/nano13101590




