Nanoarchitectonics of Fe-Doped Ni3S2 Arrays on Ni Foam from MOF Precursors for Promoted Oxygen Evolution Reaction Activity
Abstract
:1. Introduction
2. Experimental
2.1. Reagents and Materials
2.2. Synthesis of Electrocatalysts
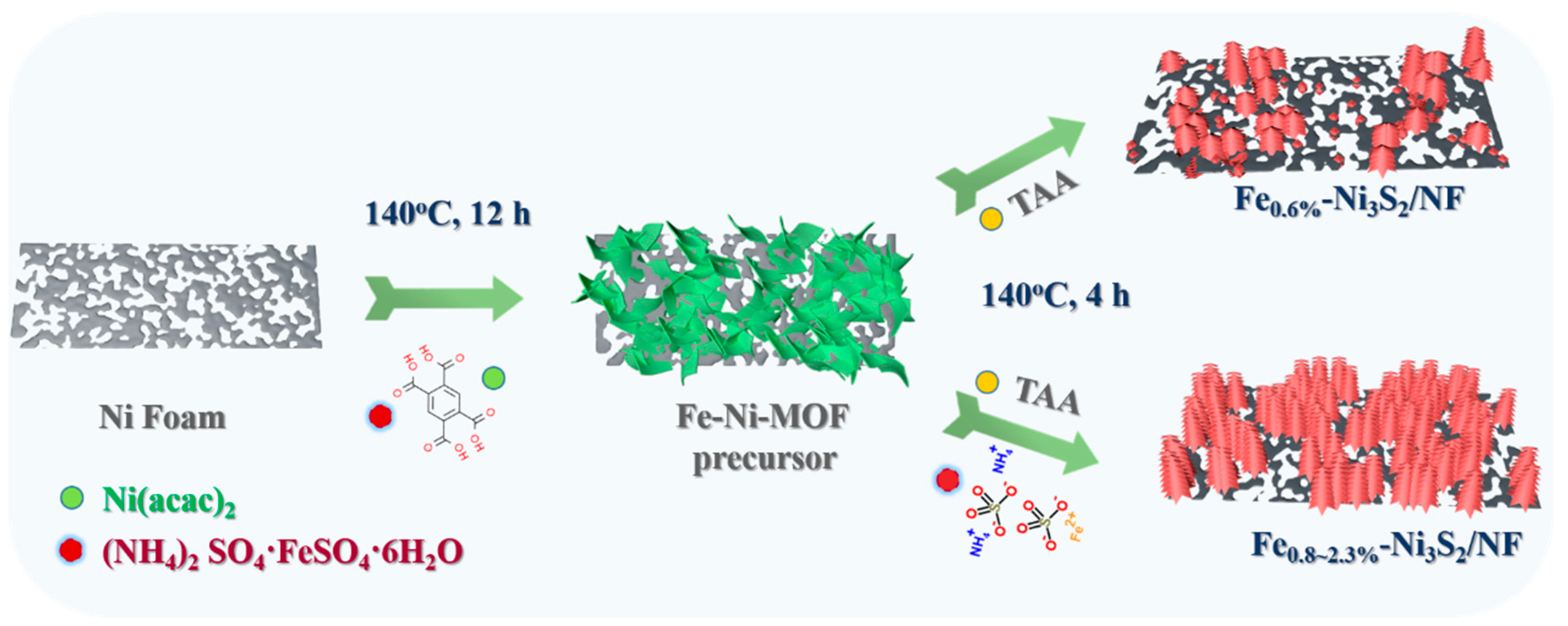
2.2.1. Synthesis of the FeNi-MOF Precursor
2.2.2. Synthesis of the Fex-Ni3S2/NF
2.3. Materials Characterization
2.4. Electrochemical Measurements
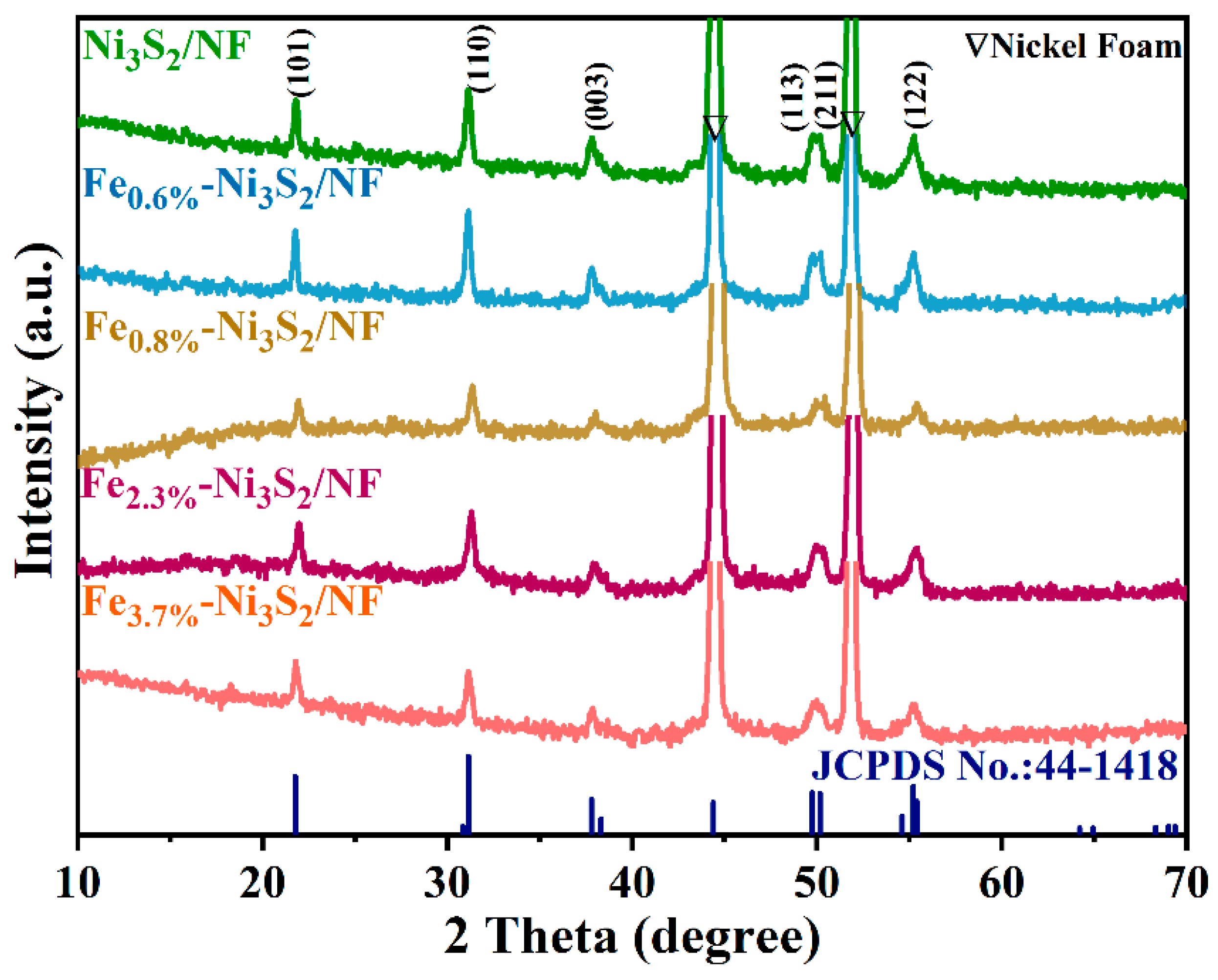
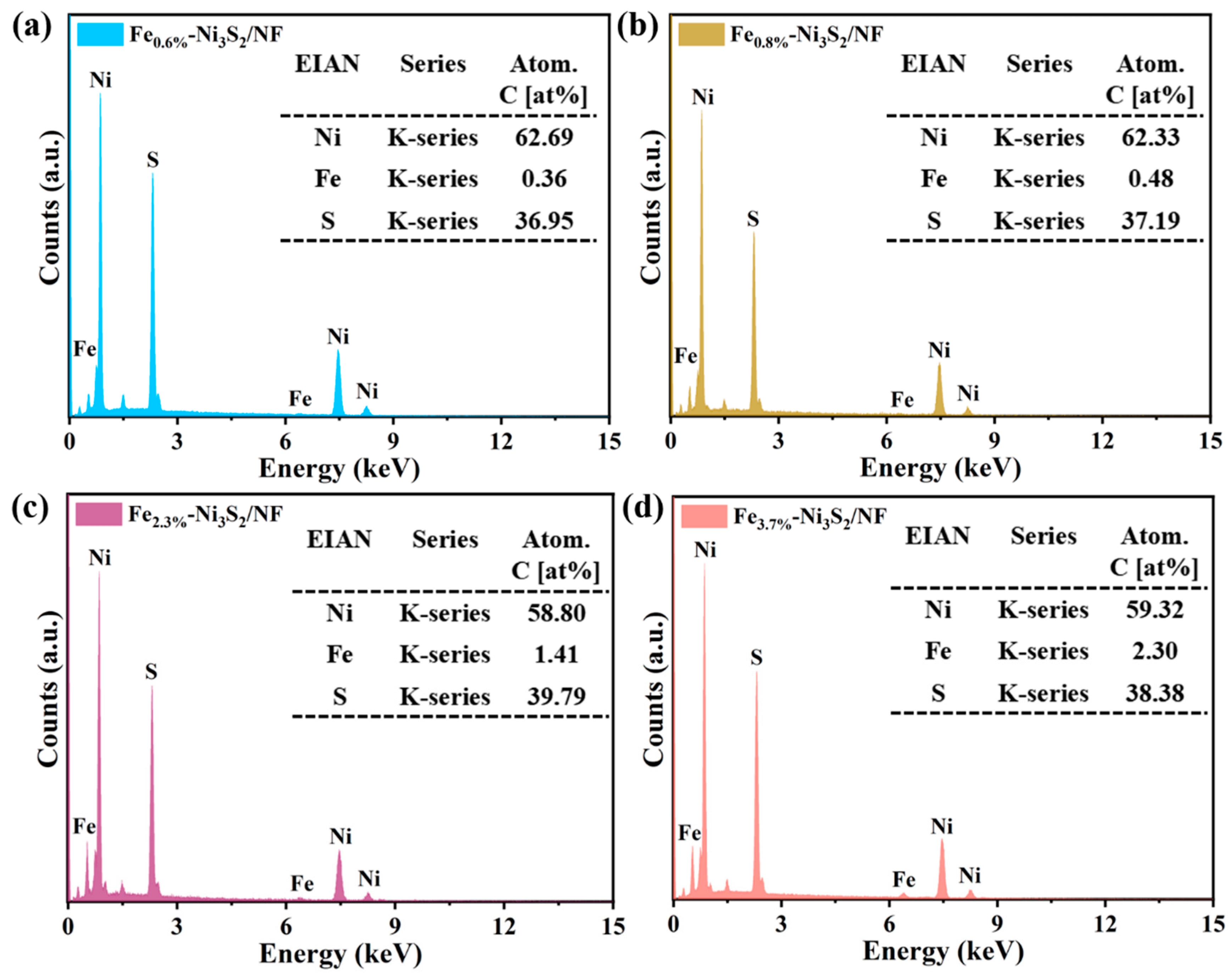
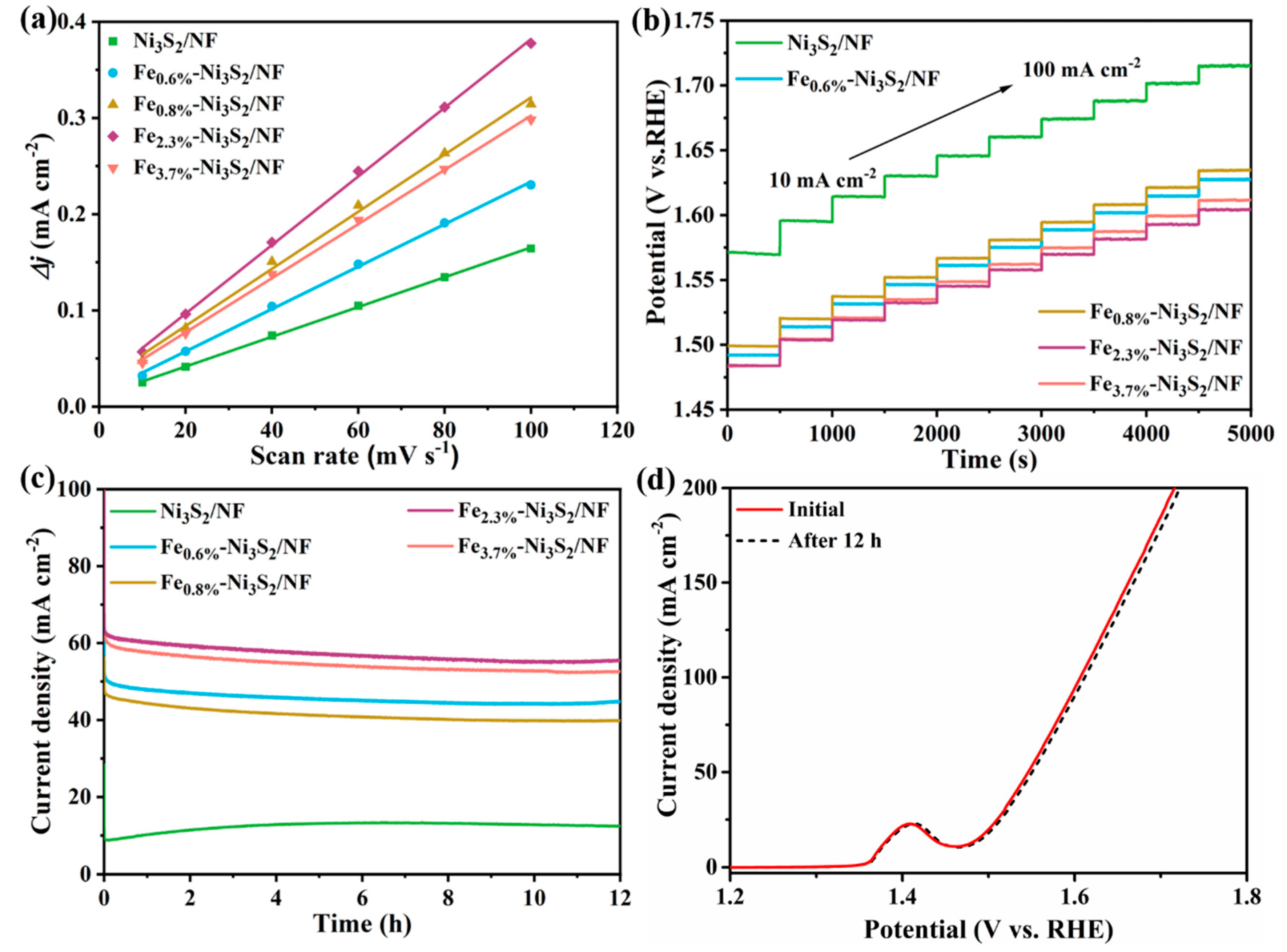
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhou, G.M.; Xu, L.; Hu, G.W.; Mai, L.Q.; Cui, Y. Nanowires for Electrochemical Energy Storage. Chem. Rev. 2019, 119, 11042–11109. [Google Scholar] [CrossRef]
- Zhang, K.; Han, X.P.; Hu, Z.; Zhang, X.L.; Tao, Z.L.; Chen, J. Nanostructured Mn-based oxides for electrochemical energy storage and conversion. Chem. Soc. Rev. 2015, 44, 699–728. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Liu, Q.; Bai, T.; Wang, W.G.; He, F.L.; Ye, M.D. Nickel and cobalt sulfide-based nanostructured materials for electrochemical energy storage devices. Chem. Eng. J. 2021, 409, 127237. [Google Scholar] [CrossRef]
- Yu, X.Y.; Lou, X.W. Mixed Metal Sulfides for Electrochemical Energy Storage and Conversion. Adv. Energy Mater. 2018, 8, 1701592. [Google Scholar] [CrossRef]
- Kulkarni, P.; Nataraj, S.K.; Balakrishna, R.G.; Nagarajua, D.H.; Reddy, M.V. Nanostructured binary and ternary metal sulfides: Synthesis methods and their application in energy conversion and storage devices. J. Mater. Chem. A 2017, 5, 22040–22094. [Google Scholar] [CrossRef]
- Yan, D.F.; Mebrahtu, C.; Wang, S.Y.; Palkovits, R. Innovative Electrochemical Strategies for Hydrogen Production: From Electricity Input to Electricity Output. Angew. Chem. Int. Ed. 2023, 62, e202214333. [Google Scholar] [CrossRef]
- Chapman, A.; Ertekin, E.; Kubota, M.; Nagao, A.; Bertsch, K.; Macadre, A.; Tsuchiyama, T.; Masamura, T.; Takaki, S.; Komoda, R.; et al. Achieving a Carbon Neutral Future through Advanced Functional Materials and Technologies. Bull. Chem. Soc. Jpn. 2022, 95, 73–103. [Google Scholar] [CrossRef]
- Ariga, K. Nanoarchitectonics: The method for everything in materials science. Bull. Chem. Soc. Jpn. 2024, 97, uoad001. [Google Scholar] [CrossRef]
- Kweon, D.H.; Jeon, I.Y.; Baek, J.-B. Electrochemical Catalysts for Green Hydrogen Energy. Adv. Energy Sustain. Res. 2021, 2, 2100019. [Google Scholar] [CrossRef]
- Mancera, J.J.C.; Manzano, F.S.; Andújar, J.M.; López, E.; Isorna, F. Sun, heat and electricity. A comprehensive study of nonpollutant alternatives to produce green hydrogen. Int. J. Energy Res. 2022, 46, 17999–18028. [Google Scholar] [CrossRef]
- Guo, Y.N.; Park, T.; Yi, J.W.; Henzie, J.; Kim, J.; Wang, Z.L.; Jiang, B.; Bando, Y.; Sugahara, Y.; Tang, J.; et al. Nanoarchitectonics for Transition-Metal-Sulfide-Based Electrocatalysts for Water Splitting. Adv. Mater. 2019, 31, 1807134. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Zhang, L.; He, Y.J.; Zhu, H.W. Recent advances in transition-metal-sulfide-based bifunctional electrocatalysts for overall water splitting. J. Mater. Chem. A 2021, 9, 5320–5363. [Google Scholar] [CrossRef]
- Jiang, W.J.; Tang, T.; Zhang, Y.; Hu, J.S. Synergistic Modulation of Non-Precious-Metal Electrocatalysts for Advanced Water Splitting. Acc. Chem. Res. 2020, 53, 1111–1123. [Google Scholar] [CrossRef] [PubMed]
- Suen, N.T.; Hung, S.F.; Quan, Q.; Zhang, N.; Xu, Y.J.; Chen, H.M. Electrocatalysis for the oxygen evolution reaction: Recent development and future perspectives. Chem. Soc. Rev. 2017, 46, 337–365. [Google Scholar] [CrossRef]
- Zhang, Y.C.; Han, C.D.; Gao, J.; Pan, L.; Wu, J.T.; Zhu, X.D.; Zou, J.J. NiCo-Based Electrocatalysts for the Alkaline Oxygen Evolution Reaction: A Review. ACS Catal. 2021, 11, 12485–12509. [Google Scholar] [CrossRef]
- Zhang, K.X.; Zou, R.Q. Advanced Transition Metal-Based OER Electrocatalysts: Current Status, Opportunities, and Challenges. Small 2021, 17, 2100129. [Google Scholar] [CrossRef]
- Ding, H.; Liu, H.F.; Chu, W.S.; Wu, C.Z.; Xie, Y. Structural Transformation of Heterogeneous Materials for Electrocatalytic Oxygen Evolution Reaction. Chem. Rev. 2021, 121, 13174–13212. [Google Scholar] [CrossRef]
- Yu, M.Q.; Budiyanto, E.; Tüysüz, H. Principles of Water Electrolysis and Recent Progress in Cobalt-, Nickel-, and Iron-Based Oxides for the Oxygen Evolution Reaction. Angew. Chem. Int. Ed. 2022, 61, e202103824. [Google Scholar] [CrossRef]
- Du, J.; Li, F.; Sun, L.C. Metal–organic frameworks and their derivatives as electrocatalysts for the oxygen evolution reaction. Chem. Soc. Rev. 2021, 50, 2663–2695. [Google Scholar] [CrossRef]
- Jiang, F.; Li, Y.C.; Pan, Y. Design Principles of Single-Atom Catalysts for Oxygen Evolution Reaction: From Targeted Structures to Active Sites. Adv. Mater. 2024, 36, 2306309. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhou, Q.; Zhu, J.X.; Yan, Q.Y.; Dou, S.X.; Sun, W.P. Nanostructured Metal Chalcogenides for Energy Storage and Electrocatalysis. Adv. Funct. Mater. 2017, 27, 1702317. [Google Scholar] [CrossRef]
- Zhao, Y.; You, J.H.; Wang, L.; Bao, W.T.; Yao, R.Y. Recent advances in Ni3S2-based electrocatalysts for oxygen evolution reaction. Int. J. Hydrogen Energy 2021, 46, 39146–39182. [Google Scholar] [CrossRef]
- Hai, Y.; Tao, K.Y.; Dan, H.M.; Liu, L.; Gong, Y. Cr-doped (Co, Ni)3S4/Co9S8/Ni3S2 nanowires/nanoparticles grown on Ni foam for hybrid supercapacitor. J. Alloys Compd. 2020, 835, 155254. [Google Scholar] [CrossRef]
- Yang, H.; Yuan, M.W.; Sun, Z.M.; Wang, D.; Lin, L.; Li, H.F.; Sun, G.B. In Situ Construction of a Mn2+-Doped Ni3S2 Electrode with Highly Enhanced Urea Oxidation Reaction Performance. ACS Sustain. Chem. Eng. 2020, 8, 8348–8355. [Google Scholar] [CrossRef]
- Du, H.T.; Kong, R.M.; Qu, F.L.; Lu, L.M. Enhanced electrocatalysis for alkaline hydrogen evolution by Mn doping in a Ni3S2 nanosheet array. Chem. Commun. 2018, 54, 10100–10103. [Google Scholar] [CrossRef] [PubMed]
- Yu, X.; Xu, S.R.; Liu, X.X.; Cheng, H.; Du, Y.S.; Wu, Q. Mn-doped NiCo2S4 nanosheet array as an efficient and durable electrocatalyst for oxygen evolution reaction. J. Alloys Compd. 2021, 878, 160388. [Google Scholar] [CrossRef]
- Li, G.F.; Cui, X.; Song, B.; Ouyang, H.Z.; Wang, K.L.; Sun, Y.M.; Wang, Y.Q. One-pot synthesis of Cu-doped Ni3S2 nano-sheet/rod nanoarray for high performance supercapacitors. Chem. Eng. J. 2020, 388, 124319. [Google Scholar] [CrossRef]
- Tong, X.; Li, Y.; Pang, N.; Qu, Y.H.; Yan, C.H.; Xiong, D.Y.; Xu, S.H.; Wang, L.W.; Chu, P.K. Co-doped Ni3S2 porous nanocones as high-performance bifunctional electrocatalysts in water splitting. Chem. Eng. J. 2021, 425, 130455. [Google Scholar] [CrossRef]
- Zhang, G.; Feng, Y.S.; Lu, W.T.; He, D.; Wang, C.Y.; Li, Y.K.; Wang, X.Y.; Cao, F.F. Enhanced Catalysis of Electrochemical Overall Water Splitting in Alkaline Media by Fe Doping in Ni3S2 Nanosheet Arrays. ACS Catal. 2018, 8, 5431–5441. [Google Scholar] [CrossRef]
- Lim, D.; Oh, E.; Lim, C.; Shim, S.E.; Baeck, S.-H. Fe-doped Ni3S2 nanoneedles directly grown on Ni foam as highly efficient bifunctional electrocatalysts for alkaline overall water splitting. Electrochim. Acta 2020, 361, 137080. [Google Scholar] [CrossRef]
- Xie, N.; Ma, D.D.; Wu, X.T.; Zhu, Q.L. Facile construction of self-supported Fe-doped Ni3S2 nanoparticle arrays for the ultralow-overpotential oxygen evolution reaction. Nanoscale 2021, 13, 1807–1812. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.X.; Jia, Q.; Liang, H.; Cui, L.; Wei, D.; Liu, J.Q. Iron doped Ni3S2 nanorods directly grown on FeNi3 foam as an efficient bifunctional catalyst for overall water splitting. Chem. Eng. J. 2020, 396, 125315. [Google Scholar] [CrossRef]
- Wu, C.R.; Liu, B.T.; Wang, J.; Su, Y.Y.; Yan, H.Q.; Ng, C.T.; Li, C.; Wei, J.M. 3D structured Mo-doped Ni3S2 nanosheets as efficient dual-electrocatalyst for overall water splitting. Appl. Surf. Sci. 2018, 441, 1024–1033. [Google Scholar] [CrossRef]
- Ma, L.Z.; Zhang, K.; Wang, S.; Gao, L.N.; Sun, Y.F.; Liu, Q.Y.; Guo, J.X.; Zhang, X. Vanadium doping over Ni3S2 nanosheet array for improved overall water splitting. Appl. Surf. Sci. 2019, 489, 815–823. [Google Scholar] [CrossRef]
- Jian, J.; Yuan, L.; Qi, H.; Sun, X.J.; Zhang, L.; Li, H.; Yuan, H.M.; Feng, S.H. Sn−Ni3S2 Ultrathin Nanosheets as Efficient Bifunctional Water-Splitting Catalysts with a Large Current Density and Low Overpotential. ACS Appl. Mater. Interfaces 2018, 10, 40568–40576. [Google Scholar] [CrossRef]
- Gao, W.; Ma, F.Y.; Wang, C.; Wen, D. Ce dopant significantly promotes the catalytic activity of Ni foam-supported Ni3S2 electrocatalyst for alkaline oxygen evolution reaction. J. Power Sources 2020, 450, 227654. [Google Scholar] [CrossRef]
- An, W.Y.; Lee, H.; Choi, S.R.; Choi, S.; Cho, H.-S.; Choi, M.; Park, J.-Y. Hierarchically porous Ni foam-supported Co and Sn doped Ni3S2 nanosheets for oxygen evolution reaction electrocatalysts. J. Mater. Chem. A 2023, 11, 5734–5745. [Google Scholar] [CrossRef]
- Li, J.Y.; Wang, X.D.; Yi, L.Y.; Fang, C.X.; Li, T.H.; Sun, W.; Hu, W.H. Plasma-assisted rhodium incorporation in nickel–iron sulfide nanosheets: Enhanced catalytic activity and the Janus mechanism for overall water splitting. Inorg. Chem. Front. 2022, 9, 6237–6247. [Google Scholar] [CrossRef]
- Liu, Q.; Xie, L.S.; Liu, Z.A.; Du, G.; Asiri, A.M.; Sun, X.P. A Zn-doped Ni3S2 nanosheet array as a high-performance electrochemical water oxidation catalyst in alkaline solution. Chem. Commun. 2017, 53, 12446–12449. [Google Scholar] [CrossRef]
- Lin, R.B.; Zhang, Z.J.; Chen, B.L. Achieving High Performance Metal−Organic Framework Materials through Pore Engineering. Acc. Chem. Res. 2021, 54, 3362–3376. [Google Scholar] [CrossRef]
- Cai, G.R.; Yan, P.; Zhang, L.L.; Zhou, H.C.; Jiang, H.L. Metal−Organic Framework-Based Hierarchically Porous Materials: Synthesis and Applications. Chem. Rev. 2021, 121, 12278–12326. [Google Scholar] [CrossRef]
- Wang, H.F.; Chen, L.Y.; Pang, H.; Kaskel, S.; Xu, Q. MOF-derived electrocatalysts for oxygen reduction, oxygen evolution and hydrogen evolution reactions. Chem. Soc. Rev. 2020, 49, 1414–1448. [Google Scholar] [CrossRef]
- da Silva, M.I.; Machado, ĺ.R.; Toma, H.E.; Araki, K.; Angnes, L.; Gonçalves, J.M. Recent progress in water-splitting and supercapacitor electrode materials based on MOF-derived sulfides. J. Mater. Chem. A 2022, 10, 430–474. [Google Scholar] [CrossRef]
- Liu, X.; Li, Y.Q.; Cao, Z.Y.; Yin, Z.H.; Ma, T.L.; Chen, S.R. Current progress of metal sulfides derived from metal–organic frameworks for advanced electrocatalysis: Potential electrocatalysts with diverse applications. J. Mater. Chem. A 2022, 10, 1617–1641. [Google Scholar] [CrossRef]
- Zhang, D.J.; Zhang, X.B.; Bu, Y.P.; Zhang, J.C.; Zhang, R.C. Copper Cobalt Sulfide Structures Derived from MOF Precursors with Enhanced Electrochemical Glucose Sensing Properties. Nanomaterials 2022, 12, 1394. [Google Scholar] [CrossRef]
- Sun, S.C.; Ma, F.X.; Li, Y.; Dong, L.W.; Liu, H.; Jiang, C.M.; Song, B.; Zhen, L.; Xu, C.Y. Sulfur Vacancies Promoting Fe-Doped Ni3S2 Nanopyramid Arrays as Efficient Bifunctional Electrocatalysts for Overall Water Splitting. Sustain. Energy Fuels 2020, 4, 3326–3333. [Google Scholar] [CrossRef]
- Manso, R.H.; Acharya, P.; Deng, S.Q.; Crane, C.C.; Reinhart, B.; Lee, S.; Tong, X.; Nykypanchuk, D.; Zhu, J.; Zhu, Y.M.; et al. Controlling the 3-D morphology of Ni–Fe-based nanocatalysts for the oxygen evolution reaction. Nanoscale 2019, 11, 8170–8184. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.C.; Chu, M.S.; Liu, M.M.; Zhang, J.H.; Jiang, H.; Li, C.Z. Fluorine-triggered surface reconstruction of Ni3S2 electrocatalysts towards enhanced water oxidation. Chem. Eng. J. 2021, 411, 128488. [Google Scholar] [CrossRef]
- Gao, W.K.; Qin, J.F.; Wang, K.; Yan, K.L.; Liu, Z.Z.; Lin, J.H.; Chai, Y.M.; Liu, C.G.; Dong, B. Facile synthesis of Fe-doped Co9S8 nano-microspheres grown on nickel foam for efficient oxygen evolution reaction. Appl. Surf. Sci. 2018, 454, 46–53. [Google Scholar] [CrossRef]
- Dong, W.X.; Zhou, H.B.; Mao, B.D.; Zhang, Z.Y.; Liu, Y.S.; Liu, Y.H.; Li, F.H.; Zhang, D.Q.; Zhang, D.X.; Shi, W.D. Efficient MOF-derived V-Ni3S2 nanosheet arrays for electrocatalytic overall water splitting in alkali. Int. J. Hydrogen Energy 2021, 46, 10773–10782. [Google Scholar] [CrossRef]
- Zhang, J.F.; Li, Y.; Zhu, T.Y.; Wang, Y.; Cui, J.W.; Wu, J.J.; Xu, H.; Shu, X.; Qin, Y.Q.; Zheng, H.M.; et al. 3D Coral-Like Ni3S2 on Ni Foam as a Bifunctional Electrocatalyst for Overall Water Splitting. ACS Appl. Mater. Interfaces 2018, 10, 31330–31339. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Yang, H.D.; Lan, K.; Iqbal, K.; Liu, Y.; Ma, P.; Zhao, Z.M.; Luo, S.; Luo, Y.T.; Ma, J.T. Optimization of iron-doped Ni3S2 nanosheets by disorder engineering for oxygen evolution reaction. Nanoscale 2019, 11, 2355–2365. [Google Scholar] [CrossRef] [PubMed]





| Catalysts | Overpotential (mV) (j = 10 mA cm−2) | Tafel Slope (mV dec−1) | References |
|---|---|---|---|
| Fe2.3%-Ni3S2/NF | 233 | 66 | This work |
| F-Ni3S2 | 239 | 36 | [48] |
| Fe-Co9S8 NM/NF | 270 | 70 | [49] |
| MOF-V-Ni3S2/NF | 268 | 99 | [50] |
| Mo-doped Ni3S2 | 260 | 85 | [33] |
| Ni3S2/NF-4 | 242 | 76 | [51] |
| Fe7.2%-Ni3S2/NF | 295 | 71 | [52] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, J.; Bu, Y.; Li, Z.; Yang, T.; Zhao, N.; Wu, G.; Zhao, F.; Zhang, R.; Zhang, D. Nanoarchitectonics of Fe-Doped Ni3S2 Arrays on Ni Foam from MOF Precursors for Promoted Oxygen Evolution Reaction Activity. Nanomaterials 2024, 14, 1445. https://doi.org/10.3390/nano14171445
Zhang J, Bu Y, Li Z, Yang T, Zhao N, Wu G, Zhao F, Zhang R, Zhang D. Nanoarchitectonics of Fe-Doped Ni3S2 Arrays on Ni Foam from MOF Precursors for Promoted Oxygen Evolution Reaction Activity. Nanomaterials. 2024; 14(17):1445. https://doi.org/10.3390/nano14171445
Chicago/Turabian StyleZhang, Jingchao, Yingping Bu, Zhuoyan Li, Ting Yang, Naihui Zhao, Guanghui Wu, Fujing Zhao, Renchun Zhang, and Daojun Zhang. 2024. "Nanoarchitectonics of Fe-Doped Ni3S2 Arrays on Ni Foam from MOF Precursors for Promoted Oxygen Evolution Reaction Activity" Nanomaterials 14, no. 17: 1445. https://doi.org/10.3390/nano14171445





