CoNiO2/Co3O4 Nanosheets on Boron Doped Diamond for Supercapacitor Electrodes
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis of BDD
2.2. Synthesis of CoNiO2/Co3O4/BDD
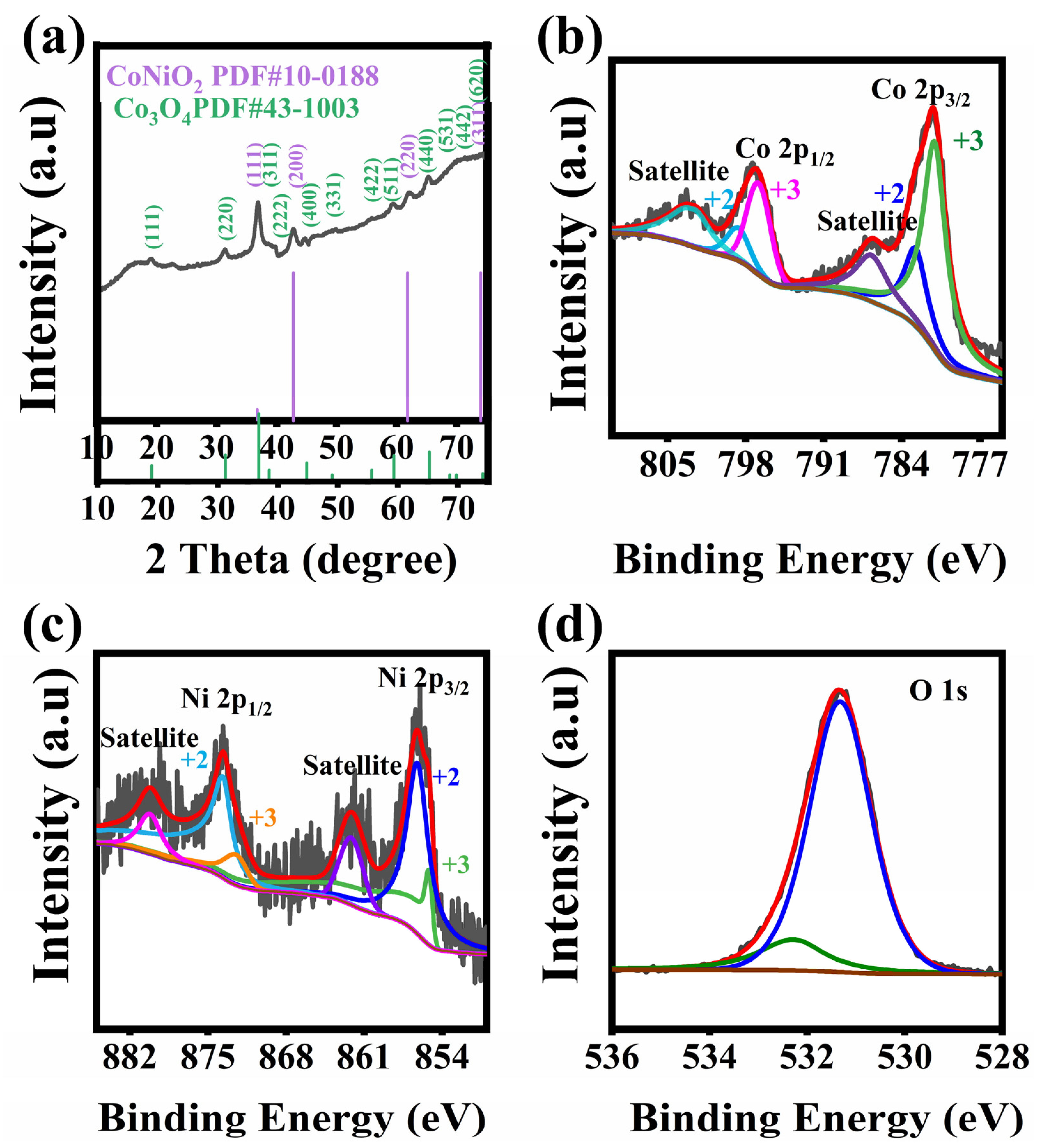
2.3. Materials Characterization
2.4. Negative Electrode Preparation and Assembly of Asymmetrical Supercapacitor
3. Results
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Lin, T.; Chen, I.W.; Liu, F.; Yang, C.; Bi, H.; Xu, F.; Huang, F. Nitrogen-doped mesoporous carbon of extraordinary capacitance for electrochemical energy storage. Science 2015, 350, 1508. [Google Scholar] [CrossRef]
- Huang, J.; Xie, Y.; You, Y.; Yuan, J.; Xu, Q.; Xie, H.; Chen, Y. Rational Design of Electrode Materials for Advanced Supercapacitors: From Lab Research to Commercialization. Adv. Funct. Mater. 2023, 33, 2213095. [Google Scholar] [CrossRef]
- Zhong, M.; Zhang, M.; Li, X. Carbon nanomaterials and their composites for supercapacitors. Carbon Energy 2022, 4, 950–985. [Google Scholar] [CrossRef]
- Chen, S.; Qiu, L.; Cheng, H.-M. Carbon-Based Fibers for Advanced Electrochemical Energy Storage Devices. Chem. Rev. 2020, 120, 2811–2878. [Google Scholar] [CrossRef]
- Wang, F.; Wu, X.; Yuan, X.; Liu, Z.; Zhang, Y.; Fu, L.; Zhu, Y.; Zhou, Q.; Wu, Y.; Huang, W. Latest advances in supercapacitors: From new electrode materials to novel device designs. Chem. Soc. Rev. 2017, 46, 6816–6854. [Google Scholar] [CrossRef]
- Yan, J.; Li, S.; Lan, B.; Wu, Y.; Lee, P.S. Rational Design of Nanostructured Electrode Materials toward Multifunctional Supercapacitors. Adv. Funct. Mater. 2019, 30, 1902564. [Google Scholar] [CrossRef]
- Zhai, Z.; Zhang, L.; Du, T.; Ren, B.; Xu, Y.; Wang, S.; Miao, J.; Liu, Z. A review of carbon materials for supercapacitors. Mater. Des. 2022, 221, 111017. [Google Scholar] [CrossRef]
- Kang, J.; Zhang, S.; Zhang, Z. Three-Dimensional Binder-Free Nanoarchitectures for Advanced Pseudocapacitors. Adv. Mater. 2017, 29, 1700515. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Wang, N.; Yang, C.; Hu, W. Sol–gel synthesis of nanoporous NiCo2O4 thin films on ITO glass as high-performance supercapacitor electrodes. Ceram. Int. 2016, 42, 11411–11416. [Google Scholar] [CrossRef]
- Senthilkumar, B.; Vijaya Sankar, K.; Kalai Selvan, R.; Danielle, M.; Manickam, M. Nano α-NiMoO4as a new electrode for electrochemical supercapacitors. RSC Adv. 2013, 3, 352–357. [Google Scholar] [CrossRef]
- Liang, R.; Du, Y.; Xiao, P.; Cheng, J.; Yuan, S.; Chen, Y.; Yuan, J.; Chen, J. Transition Metal Oxide Electrode Materials for Supercapacitors: A Review of Recent Developments. Nanomaterials 2021, 11, 1248. [Google Scholar] [CrossRef]
- Liu, R.; Zhou, A.; Zhang, X.; Mu, J.; Che, H.; Wang, Y.; Wang, T.-T.; Zhang, Z.; Kou, Z. Fundamentals, advances and challenges of transition metal compounds-based supercapacitors. Chem. Eng. J. 2021, 412, 128611. [Google Scholar] [CrossRef]
- Li, P.; Li, J.; Zhao, Z.; Fang, Z.; Yang, M.; Yuan, Z.; Zhang, Y.; Zhang, Q.; Hong, W.; Chen, X.; et al. A General Electrode Design Strategy for Flexible Fiber Micro-Pseudocapacitors Combining Ultrahigh Energy and Power Delivery. Adv. Sci. 2017, 4, 1700003. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, Z.; Wang, Y.; Li, H. Morphology-controllable synthesis of 3D CoNiO2 nano-networks as a high-performance positive electrode material for supercapacitors. Energy 2016, 113, 943–948. [Google Scholar] [CrossRef]
- Li, Y.; Han, X.; Yi, T.; He, Y.; Li, X. Review and prospect of NiCo2O4-based composite materials for supercapacitor electrodes. J. Energy Chem. 2019, 31, 54–78. [Google Scholar] [CrossRef]
- Pohlmann, S.; Balducci, A. A new conducting salt for high voltage propylene carbonate-based electrochemical double layer capacitors. Electrochim. Acta 2013, 110, 221–227. [Google Scholar] [CrossRef]
- Liu, G.; Kang, C.; Fang, J.; Fu, L.; Zhou, H.; Liu, Q. MnO2 nanosheet-coated Co3O4 complex for 1.4 V extra-high voltage supercapacitors electrode material. J. Power Sources 2019, 431, 48–54. [Google Scholar] [CrossRef]
- Yu, S.; Yang, N.; Liu, S.; Jiang, X. Diamond supercapacitors: Progress and perspectives. Curr. Opin. Solid State Mater. Sci. 2021, 25, 100922. [Google Scholar] [CrossRef]
- Banerjee, D.; Sankaran, K.J.; Deshmukh, S.; Ficek, M.; Yeh, C.-J.; Ryl, J.; Lin, I.N.; Bogdanowicz, R.; Kanjilal, A.; Haenen, K.; et al. Single-step grown boron doped nanocrystalline diamond-carbon nanograss hybrid as an efficient supercapacitor electrode. Nanoscale 2020, 12, 10117–10126. [Google Scholar] [CrossRef]
- Yu, S.; Yang, N.; Vogel, M.; Mandal, S.; Williams, O.A.; Jiang, S.; Schönherr, H.; Yang, B.; Jiang, X. Battery-like Supercapacitors from Vertically Aligned Carbon Nanofiber Coated Diamond: Design and Demonstrator. Adv. Energy Mater. 2018, 8, 1702947. [Google Scholar] [CrossRef]
- Garcia-Segura, S.; Vieira dos Santos, E.; Martínez-Huitle, C.A. Role of sp3/sp2 ratio on the electrocatalytic properties of boron-doped diamond electrodes: A mini review. Electrochem. Commun. 2015, 59, 52–55. [Google Scholar] [CrossRef]
- Mei, R.; Wei, Q.; Zhu, C.; Ye, W.; Zhou, B.; Ma, L.; Yu, Z.; Zhou, K. 3D macroporous boron-doped diamond electrode with interconnected liquid flow channels: A high-efficiency electrochemical degradation of RB-19 dye wastewater under low current. Appl. Catal. B 2019, 245, 420–427. [Google Scholar] [CrossRef]
- Yu, S.; Sankaran, K.J.; Korneychuk, S.; Verbeeck, J.; Haenen, K.; Jiang, X.; Yang, N. High-performance supercabatteries using graphite@diamond nano-needle capacitor electrodes and redox electrolytes. Nanoscale 2019, 11, 17939–17946. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Yang, N.; Zhuang, H.; Mandal, S.; Williams, O.A.; Yang, B.; Huang, N.; Jiang, X. Battery-like supercapacitors from diamond networks and water-soluble redox electrolytes. J. Mater. Chem. A 2017, 5, 1778–1785. [Google Scholar] [CrossRef]
- Li, X.; Ren, J.; Sridhar, D.; Xu, B.B.; Algadi, H.; El-Bahy, Z.M.; Ma, Y.; Li, T.; Guo, Z. Progress of layered double hydroxide-based materials for supercapacitors. Mater. Chem. Front. 2023, 7, 1520–1561. [Google Scholar] [CrossRef]
- Verma, S.; Arya, S.; Gupta, V.; Mahajan, S.; Furukawa, H.; Khosla, A. Performance analysis, challenges and future perspectives of nickel based nanostructured electrodes for electrochemical supercapacitors. J. Mater. Res. Technol. 2021, 11, 564–599. [Google Scholar] [CrossRef]
- Qi, D.; Liu, Y.; Liu, Z.; Zhang, L.; Chen, X. Design of Architectures and Materials in In-Plane Micro-supercapacitors: Current Status and Future Challenges. Adv. Mater. 2016, 29, 1602802. [Google Scholar] [CrossRef] [PubMed]
- Boryakov, A.V.; Surodin, S.I.; Kryukov, R.N.; Nikolichev, D.E.; Zubkov, S.Y. Spectral fit refinement in XPS analysis technique and its practical applications. J. Electron. Spectrosc. Relat. Phenom. 2018, 229, 132–140. [Google Scholar] [CrossRef]
- Biesinger, M.C.; Payne, B.P.; Grosvenor, A.P.; Lau, L.W.M.; Gerson, A.R.; Smart, R.S.C. Resolving surface chemical states in XPS analysis of first row transition metals, oxides and hydroxides: Cr, Mn, Fe, Co and Ni. Appl. Surf. Sci. 2011, 257, 2717–2730. [Google Scholar] [CrossRef]
- Jia, H.; Wang, Z.; Zheng, X.; Lin, J.; Liang, H.; Cai, Y.; Qi, J.; Cao, J.; Feng, J.; Fei, W. Interlaced Ni-Co LDH nanosheets wrapped Co9S8 nanotube with hierarchical structure toward high performance supercapacitors. Chem. Eng. J. 2018, 351, 348–355. [Google Scholar] [CrossRef]
- Fang, G.; Ji, X.; Shi, H.; Zhang, C.; Guo, Z.; Tang, T.; Liu, W. Constructing built-in electric field in 3D core-shell p-n heterostructure of NiFe-LDH nanosheets modified Cr-doped Ni3S2 nanofibers for efficient and stable oxygen evolution reaction. J. Alloys Compd. 2024, 970, 172583. [Google Scholar] [CrossRef]
- He, W.; Liang, Z.; Ji, K.; Sun, Q.; Zhai, T.; Xu, X. Hierarchical Ni-Co-S@Ni-W-O core–shell nanosheet arrays on nickel foam for high-performance asymmetric supercapacitors. Nano Res. 2018, 11, 1415–1425. [Google Scholar] [CrossRef]
- Chang, L.; Chen, S.; Fei, Y.; Stacchiola, D.J.; Hu, Y.H. Superstructured NiMoO4@CoMoO4 core-shell nanofibers for supercapacitors with ultrahigh areal capacitance. Proc. Natl. Acad. Sci. USA 2023, 120, e2219950120. [Google Scholar] [CrossRef]
- Minakshi, M.; Wickramaarachchi, K. Electrochemical aspects of supercapacitors in perspective: From electrochemical configurations to electrode materials processing. Prog. Solid State Chem. 2023, 69, 100390. [Google Scholar] [CrossRef]
- Wang, J.; He, Z.; Tan, X.; Wang, T.; Liu, L.; He, X.; Liu, X.D.; Zhang, L.; Du, K. High-performance 2.6 V aqueous symmetric supercapacitor based on porous boron-doped diamond via regrowth of diamond nanoparticles. Carbon 2020, 160, 71–79. [Google Scholar] [CrossRef]
- Gan, Z.; Yin, J.; Xu, X.; Cheng, Y.; Yu, T. Nanostructure and Advanced Energy Storage: Elaborate Material Designs Lead to High-Rate Pseudocapacitive Ion Storage. ACS Nano 2022, 16, 5131–5152. [Google Scholar] [CrossRef]
- Chen, Y.; Jiang, Y.; Liu, Z.; Yang, L.; Du, Q.; Zhuo, K. Hierarchical porous N-doped graphene aerogel with good wettability for high-performance ionic liquid-based supercapacitors. Electrochim. Acta 2021, 366, 137414. [Google Scholar] [CrossRef]
- Ray, P.K.; Mohanty, R.; Parida, K. Recent advancements of NiCo LDH and graphene based nanohybrids for supercapacitor application. J. Energy Storage 2023, 72, 108335. [Google Scholar] [CrossRef]
- Li, H.; Wang, J.; Chu, Q.; Wang, Z.; Zhang, F.; Wang, S. Theoretical and experimental specific capacitance of polyaniline in sulfuric acid. J. Power Sources 2009, 190, 578–586. [Google Scholar] [CrossRef]
- Fleischmann, S.; Mitchell, J.B.; Wang, R.; Zhan, C.; Jiang, D.-e.; Presser, V.; Augustyn, V. Pseudocapacitance: From Fundamental Understanding to High Power Energy Storage Materials. Chem. Rev. 2020, 120, 6738–6782. [Google Scholar] [CrossRef]
- Temesgen, T.; Bekele, E.T.; Gonfa, B.A.; Tufa, L.T.; Sabir, F.K.; Tadesse, S.; Dessie, Y. Advancements in biomass derived porous carbon materials and their surface influence effect on electrode electrochemical performance for sustainable supercapacitors: A review. J. Energy Storage 2023, 73, 109293. [Google Scholar] [CrossRef]
- Wang, J.; He, Z.; Tan, X.; Wang, T.; He, X.; Zhang, L.; Huang, J.; Chen, G.; Du, K. Hybrid supercapacitors from porous boron-doped diamond with watersoluble redox electrolyte. Surf. Coat. Technol. 2020, 398, 126103. [Google Scholar] [CrossRef]
- Pallavolu, M.R.; Kumar, Y.A.; Mani, G.; Nallapureddy, R.R.; Parvathala, A.; Albaqami, M.D.; Karami, A.M.; Joo, S.W. A novel hybridized needle-like Co3O4/N-CNO composite for superior energy storage asymmetric supercapacitors. J. Alloys Compd. 2022, 908, 164447. [Google Scholar] [CrossRef]
- Pan, X.; Chen, X.; Li, Y.; Yu, Z. Facile Synthesis of Co3O4 Nanosheets Electrode with Ultrahigh Specific Capacitance for Electrochemical Supercapacitors. Electrochim. Acta 2015, 182, 1101–1106. [Google Scholar] [CrossRef]
- Liu, S.; Kang, L.; Hu, J.; Jung, E.; Zhang, J.; Jun, S.C.; Yamauchi, Y. Unlocking the Potential of Oxygen-Deficient Copper-Doped Co3O4 Nanocrystals Confined in Carbon as an Advanced Electrode for Flexible Solid-State Supercapacitors. ACS Energy Lett. 2021, 6, 3011–3019. [Google Scholar] [CrossRef]
- Lai, C.; Guo, Y.; Zhao, H.; Song, H.; Qu, X.; Huang, M.; Hong, S.W.; Lee, K. High-performance double “ion-buffering reservoirs” of asymmetric supercapacitors enabled by battery-type hierarchical porous sandwich-like Co3O4 and 3D graphene aerogels. Adv. Compos. Hybrid Mater. 2022, 5, 2557–2574. [Google Scholar] [CrossRef]
- Umeshbabu, E.; Rajeshkhanna, G.; Ranga Rao, G. Effect of solvents on the morphology of NiCo2O4/graphene nanostructures for electrochemical pseudocapacitor application. J. Solid State Electrochem. 2015, 20, 1837–1844. [Google Scholar] [CrossRef]
- Yang, J.; Li, H.; He, S.; Du, H.; Liu, K.; Zhang, C.; Jiang, S. Facile Electrodeposition of NiCo2O4 Nanosheets on Porous Carbonized Wood for Wood-Derived Asymmetric Supercapacitors. Polymers 2022, 14, 2521. [Google Scholar] [CrossRef]
- Hu, X.; Wan, C.; Meng, X.; Tang, A.; Ju, X. Porous NiCoO2 nanospheres encapsulated in nitrogen-doped carbon shell achieving high energy storage for aqueous supercapacitors and zinc–ion batteries. Appl. Surf. Sci. 2022, 582, 152456. [Google Scholar] [CrossRef]
- Liu, B.; Hou, J.; Zhang, T.; Xu, C.; Liu, H. A three-dimensional multilevel nanoporous NiCoO2/Ni hybrid for highly reversible electrochemical energy storage. J. Mater. Chem. A 2019, 7, 16222–16230. [Google Scholar] [CrossRef]
- Wang, X.; Fang, Y.; Shi, B.; Huang, F.; Rong, F.; Que, R. Three-dimensional NiCo2O4@NiCo2O4 core–shell nanocones arrays for high-performance supercapacitors. Chem. Eng. J. 2018, 344, 311–319. [Google Scholar] [CrossRef]
- Wang, X.W.; Wang, X.E.; Liu, Y.P.; Kong, Y.Y.; Sun, L.Y.; Hu, Y.C.; Zhu, Q.Q. Hydrothermal process fabrication of NiO–NiCoO2–Co3O4 composites used as supercapacitor materials. J. Mater. Sci. Mater. Electron. 2017, 28, 14928–14934. [Google Scholar]
- Yu, S.; Yang, N.; Zhuang, H.; Meyer, J.; Mandal, S.; Williams, O.A.; Lilge, I.; Schönherr, H.; Jiang, X. Electrochemical Supercapacitors from Diamond. J. Phys. Chem. C 2015, 119, 18918–18926. [Google Scholar] [CrossRef]
- Gao, F.; Nebel, C.E. Diamond nanowire forest decorated with nickel hydroxide as a pseudocapacitive material for fast charging–discharging. Phys. Status Solidi (a) 2015, 212, 2533–2538. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, X.; Zhao, Z.-y.; Zhang, Z.; Li, J. Influence of pore size of Ti substrate on structural and capacitive properties of Ti/boron doped diamond electrode. J. Alloys Compd. 2019, 777, 84–93. [Google Scholar] [CrossRef]
- Lu, Z.; Huang, N.; Zhai, Z.; Chen, B.; Liu, L.; Song, H.; Yuan, Z.; Zhang, C.; Yang, B.; Jiang, X. Integration of 3D interconnected porous microstructure and high electrochemical property for boron-doped diamond by facile strategy. J. Mater. Sci. Technol. 2022, 105, 26–35. [Google Scholar]
- Shi, C.; Li, C.; Li, M.; Li, H.; Dai, W.; Wu, Y.; Yang, B. Fabrication of porous boron-doped diamond electrodes by catalytic etching under hydrogen–argon plasma. Appl. Surf. Sci. 2016, 360, 315–322. [Google Scholar] [CrossRef]
- Guo, T.; Yang, N.; Yang, B.; Schulte, A.; Jin, Q.; Koch, U.; Mandal, S.; Engelhard, C.; Williams, O.A.; Schönherr, H.; et al. Electrochemistry of nitrogen and boron Bi-element incorporated diamond films. Carbon 2021, 178, 19–25. [Google Scholar] [CrossRef]
- Xu, J.; Yang, N.; Heuser, S.; Yu, S.; Schulte, A.; Schönherr, H.; Jiang, X. Achieving Ultrahigh Energy Densities of Supercapacitors with Porous Titanium Carbide/Boron-Doped Diamond Composite Electrodes. Adv. Energy Mater. 2019, 9, 1803623. [Google Scholar] [CrossRef]
- Zhang, J.; Zhao, Z.; Zhang, Z.; Guo, L.; Xu, L.; Sun, P.; Wang, M.; Gao, M.; Li, Y.; Li, D.; et al. Construction of flexible fiber-shaped boron-doped diamond film and its supercapacitor application. J. Colloid Interface Sci. 2023, 629, 813–821. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, X.; Zhang, Z.-Q.; Zhao, Z.-Y. Preparation of boron-doped diamond foam film for supercapacitor applications. Appl. Surf. Sci. 2020, 506, 144645. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cui, Z.; Wang, T.; Geng, Z.; Wan, L.; Liu, Y.; Xu, S.; Gao, N.; Li, H.; Yang, M. CoNiO2/Co3O4 Nanosheets on Boron Doped Diamond for Supercapacitor Electrodes. Nanomaterials 2024, 14, 474. https://doi.org/10.3390/nano14050474
Cui Z, Wang T, Geng Z, Wan L, Liu Y, Xu S, Gao N, Li H, Yang M. CoNiO2/Co3O4 Nanosheets on Boron Doped Diamond for Supercapacitor Electrodes. Nanomaterials. 2024; 14(5):474. https://doi.org/10.3390/nano14050474
Chicago/Turabian StyleCui, Zheng, Tianyi Wang, Ziyi Geng, Linfeng Wan, Yaofeng Liu, Siyu Xu, Nan Gao, Hongdong Li, and Min Yang. 2024. "CoNiO2/Co3O4 Nanosheets on Boron Doped Diamond for Supercapacitor Electrodes" Nanomaterials 14, no. 5: 474. https://doi.org/10.3390/nano14050474
APA StyleCui, Z., Wang, T., Geng, Z., Wan, L., Liu, Y., Xu, S., Gao, N., Li, H., & Yang, M. (2024). CoNiO2/Co3O4 Nanosheets on Boron Doped Diamond for Supercapacitor Electrodes. Nanomaterials, 14(5), 474. https://doi.org/10.3390/nano14050474






