Antimicrobial Properties of Biofunctionalized Silver Nanoparticles on Clinical Isolates of Streptococcus mutans and Its Serotypes
Abstract
:1. Introduction
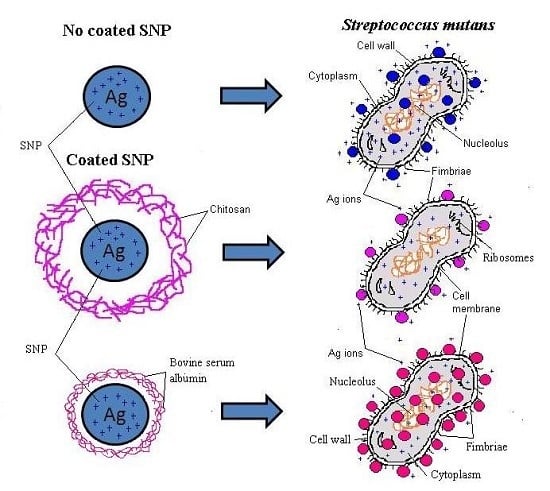
2. Results and Discussion
2.1. Characterization of BSA and CS Coated SNP
2.2. Antibacterial Test
2.3. Scanning Electron Microscopy
3. Materials and Methods
3.1. Materials and Reagents
3.2. Silver Nanoparticles Preparation
3.3. Preparation of BSA Coated Silver Nanoparticles
3.4. Preparation of CS Coated Silver Nanoparticles
3.5. Characterization
3.6. Identification of S. mutans Serotypes Using PCR
3.7. Antibacterial Assay
3.8. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| µg/mL | microgram per milliliter |
| BSA | Bovine serum albumin |
| CFU | colony forming units |
| CS | Chitosan |
| DLS | Dynamic light scattering |
| DSC | Differential scanning calorimetry |
| MIC | Minimum inhibitory concentration |
| S. mutans | Streptococcus mutans |
| SEM | Scanning electron microscopy |
| SNP | Silver nanoparticles |
| TEM | Transmission electron microscopy |
| TGA | Thermo-gravimetric analysis |
References
- Al-Darwish, M.; El Ansari, W.; Bener, A. Prevalence of dental caries among 12–14 year old children in Qatar. Saudi Dent. J. 2014, 26, 115–125. [Google Scholar] [CrossRef] [PubMed]
- Plonka, K.A.; Pukallus, M.L.; Barnett, A.G.; Walsh, L.J.; Holcombe, T.H.; Seow, W.K. Mutans streptococci and lactobacilli colonization in predentate children from the neonatal period to seven months of age. Caries Res. 2012, 46, 213–220. [Google Scholar] [CrossRef] [PubMed]
- Moreillon, P.; Que, Y.A. Infective endocarditis. Lancet 2004, 363, 139–149. [Google Scholar] [CrossRef]
- Nakano, K.; Ooshima, T. Serotype classification of Streptococcus mutans and its detection outside the oral cavity. Future Microbiol. 2009, 4, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Holbrook, W.P.; Beighton, D. Streptococcus mutans levels in saliva and distribution of serotypes among 9-year-old Icelandic children. Scand. J. Dent. Res. 1987, 95, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Hirasawa, M.; Takada, K. A new selective medium for Streptococcus mutans and the distribution of S. mutans and S. sobrinus and their serotypes in dental plaque. Caries Res. 2003, 37, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Nomura, R.; Nakagawa, I.; Hamada, S.; Ooshima, T. Demonstration of Streptococcus mutans with a cell wall polysaccharide specific to a new serotype, k, in the human oral cavity. J. Clin. Microbiol. 2004, 42, 198–202. [Google Scholar] [CrossRef] [PubMed]
- Nakano, K.; Tsuji, M.; Nishimura, K.; Nomura, R.; Ooshima, T. Contribution of cell surface protein antigen PAc of Streptococcus mutans to bacteremia. Microbes Infect. 2006, 8, 114–121. [Google Scholar] [CrossRef] [PubMed]
- Lapirattanakul, J.; Nomura, R.; Matsumoto-Nakano, M.; Srisatjaluk, R.; Ooshima, T.; Nakano, K. Variation of expression defects in cell surface 190-kDa protein antigen of Streptococcus mutans. Int. J. Med. Microbiol. 2015, 305, 383–391. [Google Scholar] [CrossRef] [PubMed]
- Nomura, R.; Otsugu, M.; Naka, S.; Teramoto, N.; Kojima, A.; Muranaka, Y.; Matsumoto-Nakano, M.; Ooshima, T.; Nakano, K. Contribution of the interaction of Streptococcus mutans serotype k strains with fibrinogen to the pathogenicity of infective endocarditis. Infect. Immun. 2014, 82, 5223–5234. [Google Scholar] [CrossRef] [PubMed]
- Naka, S.; Nomura, R.; Takashima, Y.; Okawa, R.; Ooshima, T.; Nakano, K. A specific Streptococcus mutans strain aggravates non-alcoholic fatty liver disease. Oral Dis. 2014, 20, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.H.; Seo, J.S.; Yoon, Y.J.; Kim, K.W.; Yoo, S.Y.; Kim, H.S.; Kook, J.K.; Lee, B.R.; Cha, J.H.; Park, J.Y. Effect of Leaf-Extract from Camellia sinensis and Seed-Extract from Casia tora on Viability of Mutans Streptococci isolated from the interface between orthodontic brackets and tooth surfaces. Korea J. Orthod. 2003, 33, 381–389. [Google Scholar]
- Yoo, S.Y.; Park, S.J.; Jeong, D.K.; Kim, K.W.; Lim, S.H.; Lee, S.H.; Choe, S.J.; Chang, Y.H.; Park, I.; Kook, J.K. Isolation and characterization of the mutans streptococci from the dental plaques in Koreans. J. Microbiol. 2007, 45, 246–255. [Google Scholar] [PubMed]
- Espinosa-Cristóbal, L.F.; Martínez-Castañón, G.A.; Martínez-Martínez, R.E.; Loyola-Rodríguez, J.P.; Patiño-Marín, N.; Reyes-Macías, J.F.; Ruiz, F. Antibacterial effect of silver nanoparticles against Streptococcus mutans. Mater. Lett. 2009, 63, 2603–2606. [Google Scholar] [CrossRef]
- Cury, J.A.; Tenuta, L.M. Evidence-based recommendation on toothpaste use. Braz. Oral Res. 2014, 28, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Walsh, T.; Worthington, H.V.; Glenny, A.M.; Appelbe, P.; Marinho, V.C.; Shi, X. Fluoride toothpastes of different concentrations for preventing dental caries in children and adolescents. Cochrane Database Syst. Rev. 2010, 20. [Google Scholar] [CrossRef]
- Aidara, A.W.; Bourgeois, D. Prevalence of dental caries: National pilot study comparing the severity of decay (CAO) vs. ICDAS index in Senegal. Odontostomatol. Trop. 2014, 37, 53–63. [Google Scholar] [PubMed]
- Bhardwaj, S.B.; Mehta, M.; Gauba, K. Nanotechnology: Role in dental biofilms. Indian J. Dent. Res. 2009, 20, 511–513. [Google Scholar] [PubMed]
- Feng, Q.L.; Wu, J.; Chen, G.Q.; Cui, F.Z.; Kim, T.N.; Kim, J.O. A mechanistic study of the antibacterial effect of silver ions on Escherichia coli and Staphylococcus aureus. J. Biomed. Mater. 2000, 52, 662–668. [Google Scholar] [CrossRef]
- Sraydich-Stolle, L.; Hussain, S.; Schlager, J.; Hofmann, M. In vitro cytotoxicity of nanoparticles in mammalian germline stem cells. Toxicol. Sci. 2005, 88, 412–419. [Google Scholar] [CrossRef] [PubMed]
- Ai, J.; Biazar, E.; Jafarpour, M.; Montazeri, M.; Majdi, A.; Aminifard, S.; Zafari, M.; Akbari, H.R.; Rad, H.G. Nanotoxicology and nanoparticle safety in biomedical designs. Int. J. Nanomed. 2011, 6, 1117–1127. [Google Scholar]
- Marini, M.; De Niederhausern, S.; Iseppi, R.; Bondi, M.; Sabia, C.; Toselli, M.; Pilati, F. Antibacterial activity of plastics coated with silver-doped organic-inorganic hybrid coatings prepared by sol-gel processes. Biomacromolecules 2007, 8, 1246–1254. [Google Scholar] [CrossRef] [PubMed]
- Ashoka, S.; Seetharamappa, J.; Kandagal, P.B.; Shaikh, S.M.T. Investigation of the interaction between trazodone hydrochloride and bovine serum albumin. J. Lumin. 2006, 121, 179–186. [Google Scholar] [CrossRef]
- Xia, W.; Liu, P.; Zhang, J.; Chen, J. Biological activities of chitosan and chitooligosaccharides. Food Hydrocoll. 2011, 25, 170–179. [Google Scholar] [CrossRef]
- Ravindran, A.; Singh, A.; Raichur, A.M.; Chandrasekaran, N.; Mukherjee, A. Studies on interaction of colloidal Ag nanoparticles with Bovine Serum Albumin (BSA). Colloids Surf. B 2010, 76, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Martins, A.M.; Alves, C.M.; Kasper, F.K.; Mikos, A.G.; Reis, R.L. Responsive and in situ-forming chitosan scaffolds for bone tissue engineering applications: An overview of the last decade. J. Mater. Chem. 2010, 20, 1638–1645. [Google Scholar] [CrossRef]
- Esmaeili, F.; Heuking, S.; Junginger, H.E.; Borchard, G.J. Progress in chitosan-based vaccine delivery systems. J. Drug Deliv. Sci. Technol. 2010, 20, 53–61. [Google Scholar] [CrossRef]
- Quang, D.V.; Lee, J.E.; Kim, J.K.; Kim, Y.N.; Shao, G.N.; Kim, H.T. A gentle method to graft thiol-functional groups onto silica gel for adsorption of silver ions and immobilization of silver nanoparticles. Powder Technol. 2013, 235, 221–227. [Google Scholar] [CrossRef]
- Reicha, F.M.; Sarhan, A.; Abdel-Hamid, M.I.; El-Sherbiny, I.M. Preparation of silver nanoparticles in the presence of chitosan by electrochemical method. Carbohydr. Polym. 2012, 89, 236–244. [Google Scholar] [CrossRef] [PubMed]
- Espinosa-Cristóbal, L.F.; Martínez-Castañón, G.A.; Loyola-Rodríguez, J.P.; Niño-Martínez, N.; Ruiz, F.; Zavala-Alonso, N.V.; Lara, R.H.; Reyes-López, S.Y. Bovine Serum Albumin and Chitosan Coated Silver Nanoparticles and Its Antimicrobial Activity against Oral and Nonoral Bacteria. J. Nanomater. 2015, 9. [Google Scholar] [CrossRef]
- Espinosa-Cristóbal, L.F.; Martínez-Castañon, G.A.; Martínez-Martínez, R.E.; Loyola-Rodriguez, J.P.; Patiño-Marin, N.; Reyes-Macias, J.F.; Ruiz, F. Antimicrobial sensibility of Streptococcus mutans serotypes to silver nanoparticles. Mater. Sci. Eng. C 2012, 32, 896–901. [Google Scholar] [CrossRef]
- Shibata, Y.; Ozaki, K.; Seki, M.; Kawato, T.; Tanaka, H.; Nakano, Y.; Yamashita, Y. Analysis of loci required for determination of serotype antigenicity in Streptococcus mutans and its clinical utilization. J. Clin. Microbiol. 2003, 41, 4107–4112. [Google Scholar] [CrossRef] [PubMed]
- Hoshino, T.; Kawaguchi, M.; Shimizu, N.; Hoshino, N.; Ooshima, T.; Fujiwara, T. PCR detection and identification of oral streptococci in saliva samples using GTF genes. Diagn. Microbiol. Infect. Dis 2004, 48, 195–199. [Google Scholar] [CrossRef] [PubMed]
- Gebregeorgis, A.; Bhan, C.; Wilson, O.; Raghavan, D.J. Characterization of Silver/Bovine Serum Albumin (Ag/BSA) nanoparticles structure: Morphological, compositional, and interaction studies. J. Colloid Interface Sci. 2013, 389, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.W.; Rajendran, S.; Joshi, M. Synthesis and characterization of chitosan and silver loaded chitosan nanoparticles for bioactive polyester. Carbohyd. Polym. 2011, 83, 438–446. [Google Scholar] [CrossRef]
- Lopez, F.; Merce, A.; Alguacil, F.; Lopez-Delgado, A. A kinetic study on the thermal behaviour of chitosan. J. Therm. Anal. Calorim. 2008, 91, 633–639. [Google Scholar] [CrossRef]
- An, J.; Ji, Z.; Wang, D.; Luo, Q.; Li, X. Preparation and characterization of uniform-sized chitosan/silver microspheres with antibacterial activities. Mater. Sci. Eng. C 2014, 36, 33–41. [Google Scholar] [CrossRef] [PubMed]
- Lu, Z.; Rong, K.; Li, J.; Yang, H.; Chen, R. Size-dependent antibacterial activities of silver nanoparticles against oral anaerobic pathogenic bacteria. J. Mater. Sci. Mater. Med. 2013, 24, 1465–1471. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Ma, L.; Shi, H.; Gao, C.; Han, C. Chitosan hyaluronic acid hybrid film as a novel wound dressing: In vitro and in vivo studies. Polym. Adv. Technol. 2007, 18, 869–875. [Google Scholar] [CrossRef]
- Treuel, L.; Brandholt, S.; Maffre, P.; Wiegele, S.; Shang, L.; Nienhaus, G.U. Impact of protein modification on the protein corona on nanoparticles and nanoparticle-cell interactions. ACS Nano 2014, 8, 503–513. [Google Scholar] [CrossRef] [PubMed]
- Lee, W.; Kim, K.J.; Lee, D.G. A novel mechanism for the antibacterial effect of silver nanoparticles on Escherichia coli. Biometals 2014, 27, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Mathew, T.V.; Kuriakose, S. Studies on the antimicrobial properties of colloidal silver nanoparticles stabilized by bovine serum albumin. Colloids Surf. B 2013, 101, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Alt, V.; Bechert, T.; Steinrücke, P.; Wagener, M.; Seidel, P.; Dingeldein, E.; Domann, E.; Schnettler, R. An in vitro assessment of the antibacterial properties and cytotoxicity of nanoparticulate silver bone cement. Biomaterials 2004, 25, 4383–4391. [Google Scholar] [CrossRef] [PubMed]
- Loyola-Rodriguez, J.P.; Martinez-Martinez, R.E.; Flores-Ferreyra, B.I.; Patiño-Marin, N.; Alpuche-Solis, A.G.; Reyes-Macias, J.F. Distribution of Streptococcus mutans and Streptococcus sobrinus in saliva of Mexican preschool caries-free and caries-active children by microbial and molecular (PCR) assays. J. Clin. Pediatr. Dent. 2008, 32, 121–126. [Google Scholar] [CrossRef] [PubMed]
- Slayton, R.L.; Cooper, M.E.; Marazita, M.L. Tuftelin, mutans streptococci, and dental caries susceptibility. J. Dent. Res. 2005, 84, 711–714. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Wang, L.; Chen, Q.; Chen, C. Cytotoxic potential of silver nanoparticles. Yonsei. Med. J. 2014, 55, 283–291. [Google Scholar] [CrossRef] [PubMed]






| SNP Sample (nm) | Diameter DLS (nm) | Zeta Potential (mV ± ZD) | Residues TGA (%) | Total Weight Loss TGA (%) |
|---|---|---|---|---|
| 7.1 | 7.1 | −48.4 ± 6.9 | 98.5 | 1.5 |
| 17.4 | 17.4 | −52.6 ± 8.5 | 98.2 | 1.7 |
| 87.6 | 87.6 | −55.7 ± 9.9 | 97.8 | 2.1 |
| BSA 16.5 | 16.5 | −13.5 ± 4.7 | 96.3 | 3.6 |
| BSA 23.3 | 23.3 | −44.0 ± 6.9 | 86.9 | 13.0 |
| BSA 115.2 | 115.2 | −32.7 ± 6.3 | 96.7 | 3.2 |
| CS 22.5 | 22.5 | 37.9 ± 13.0 | 67.3 | 32.6 |
| CS 44.1 | 44.1 | 48.3 ± 7.9 | 97.5 | 2.4 |
| CS 133.7 | 133.7 | 52.0 ± 5.4 | 95.9 | 4.1 |
© 2016 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martínez-Robles, Á.M.; Loyola-Rodríguez, J.P.; Zavala-Alonso, N.V.; Martinez-Martinez, R.E.; Ruiz, F.; Lara-Castro, R.H.; Donohué-Cornejo, A.; Reyes-López, S.Y.; Espinosa-Cristóbal, L.F. Antimicrobial Properties of Biofunctionalized Silver Nanoparticles on Clinical Isolates of Streptococcus mutans and Its Serotypes. Nanomaterials 2016, 6, 136. https://doi.org/10.3390/nano6070136
Martínez-Robles ÁM, Loyola-Rodríguez JP, Zavala-Alonso NV, Martinez-Martinez RE, Ruiz F, Lara-Castro RH, Donohué-Cornejo A, Reyes-López SY, Espinosa-Cristóbal LF. Antimicrobial Properties of Biofunctionalized Silver Nanoparticles on Clinical Isolates of Streptococcus mutans and Its Serotypes. Nanomaterials. 2016; 6(7):136. https://doi.org/10.3390/nano6070136
Chicago/Turabian StyleMartínez-Robles, Ángel Manuel, Juan Pablo Loyola-Rodríguez, Norma Verónica Zavala-Alonso, Rita Elizabeth Martinez-Martinez, Facundo Ruiz, René Homero Lara-Castro, Alejandro Donohué-Cornejo, Simón Yobanny Reyes-López, and León Francisco Espinosa-Cristóbal. 2016. "Antimicrobial Properties of Biofunctionalized Silver Nanoparticles on Clinical Isolates of Streptococcus mutans and Its Serotypes" Nanomaterials 6, no. 7: 136. https://doi.org/10.3390/nano6070136