Practical Liposomal Formulation for Taxanes with Polyethoxylated Castor Oil and Ethanol with Complete Encapsulation Efficiency and High Loading Efficiency
Abstract
:1. Introduction
2. Results
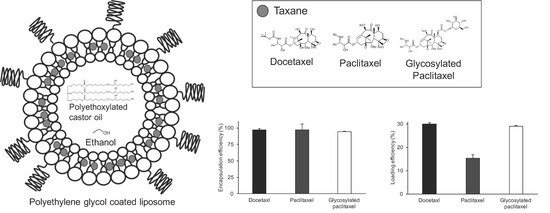
2.1. Liposomes Encapsulating Taxanes
2.2. Cytotoxicity of the Formulations for DTX
2.3. Internalization of Liposomes into the Cells
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Preparation of Liposomes Encapsulating Taxanes
4.3. Evaluation of the Leakage of Drugs from Liposomes
4.4. Preparation of EC1 Peptide Fused to Human IgG Fc Domain (EC-Fc)
4.5. Preparation of DTX-L Conjugated with EC1 Peptide (DTX-ECL)
4.6. Evaluation of Encapsulation Efficiency (EE) and Loading Efficiency (LE)
4.7. Characterization of Liposomes
4.8. Cell Culture
4.9. In Vitro Evaluation of Cellular Uptake of the Drugs
4.10. Cytotoxicity Assay
4.11. Confocal Microscopic Observation
4.12. Statistical Analysis
5. Conclusions
Supplementary Materials
Acknowledgements
Author Contributions
Conflicts of Interest
References
- Andresen, T.L.; Jensen, S.S.; Jørgensen, K. Advanced strategies in liposomal cancer therapy: Problems and prospects of active and tumor specific drug release. Prog. Lipid Res. 2005, 44, 68–97. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Kantoff, P.W.; Wooster, R.; Farokhzad, O.C. Cancer nanomedicine: Progress, challenges and opportunities. Nat. Rev. Cancer 2017, 17, 20–37. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, K. Intracellular targeting delivery of liposomal drugs to solid tumors based on epr effects. Adv. Drug Deliv. Rev. 2011, 63, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Li, S.D.; Huang, L. Nanoparticles evading the reticuloendothelial system: Role of the supported bilayer. Biochim. Biophys. Acta. 2009, 1788, 2259–2266. [Google Scholar] [CrossRef] [PubMed]
- Klibanov, A.L.; Maruyama, K.; Torchilin, V.P.; Huang, L. Amphipathic polyethyleneglycols effectively prolong the circulation time of liposomes. FEBS Lett. 1990, 268, 235–237. [Google Scholar] [CrossRef]
- Haran, G.; Cohen, R.; Bar, L.K.; Barenholz, Y. Transmembrane ammonium sulfate gradients in liposomes produce efficient and stable entrapment of amphipathic weak bases. Biochim. Biophys. Acta 1993, 1151, 201–215. [Google Scholar] [CrossRef]
- Hansen, C.B.; Kao, G.Y.; Moase, E.H.; Zalipsky, S.; Allen, T.M. Attachment of antibodies to sterically stabilized liposomes: Evaluation, comparison and optimization of coupling procedures. Biochim. Biophys. Acta 1995, 1239, 133–144. [Google Scholar] [CrossRef]
- Fenske, D.B.; Wong, K.F.; Maurer, E.; Maurer, N.; Leenhouts, J.M.; Boman, N.; Amankwa, L.; Cullis, P.R. Ionophore-mediated uptake of ciprofloxacin and vincristine into large unilamellar vesicles exhibiting transmembrane ion gradients. Biochim. Biophys. Acta 1998, 1414, 188–204. [Google Scholar] [CrossRef]
- Shmeeda, H.; Tzemach, D.; Mak, L.; Gabizon, A. HER2-targeted pegylated liposomal doxorubicin: Retention of target-specific binding and cytotoxicity after in vivo passage. J. Control. Release 2009, 136, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Heger, Z.; Polanska, H.; Merlos Rodrigo, M.A.; Guran, R.; Kulich, P.; Kopel, P.; Masarik, M.; Eckschlager, T.; Stiborova, M.; Kizek, R.; et al. Prostate tumor attenuation in the nu/nu murine model due to anti-sarcosine antibodies in folate-targeted liposomes. Sci. Rep. 2016, 6, 33379. [Google Scholar] [CrossRef] [PubMed]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal formulations in clinical use: An updated review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Gumulec, J.; Fojtu, M.; Raudenska, M.; Sztalmachova, M.; Skotakova, A.; Vlachova, J.; Skalickova, S.; Nejdl, L.; Kopel, P.; Knopfova, L.; et al. Modulation of induced cytotoxicity of doxorubicin by using apoferritin and liposomal cages. Int. J. Mol. Sci. 2014, 15, 22960–22977. [Google Scholar] [CrossRef] [PubMed]
- Shigehiro, T.; Kasai, T.; Murakami, M.; Sekhar, S.C.; Tominaga, Y.; Okada, M.; Kudoh, T.; Mizutani, A.; Murakami, H.; Salomon, D.S.; et al. Efficient drug delivery of paclitaxel glycoside: A novel solubility gradient encapsulation into liposomes coupled with immunoliposomes preparation. PLoS ONE 2014, 9, e107976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barenholz, Y. Doxil®—the first fda-approved nano-drug: Lessons learned. J. Control. Release 2012, 160, 117–134. [Google Scholar] [CrossRef] [PubMed]
- Hirai, M.; Minematsu, H.; Hiramatsu, Y.; Kitagawa, H.; Otani, T.; Iwashita, S.; Kudoh, T.; Chen, L.; Li, Y.; Okada, M.; et al. Novel and simple loading procedure of cisplatin into liposomes and targeting tumor endothelial cells. Int. J. Pharm. 2010, 391, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Pazdur, R.; Kudelka, A.P.; Kavanagh, J.J.; Cohen, P.R.; Raber, M.N. The taxoids: Paclitaxel (taxol) and docetaxel (taxotere). Cancer Treat. Rev. 1993, 19, 351–386. [Google Scholar] [CrossRef]
- Dykes, D.J.; Bissery, M.C.; Harrison, S.D.; Waud, W.R. Response of human tumor xenografts in athymic nude mice to docetaxel (RP 56976, Taxotere). Investig. New Drugs 1995, 13, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Hennenfent, K.L.; Govindan, R. Novel formulations of taxanes: A review. Old wine in a new bottle? Ann. Oncol. 2006, 17, 735–749. [Google Scholar] [CrossRef] [PubMed]
- Koudelka, S.; Turánek, J. Liposomal paclitaxel formulations. J. Control. Release 2012, 163, 322–334. [Google Scholar] [CrossRef] [PubMed]
- Crosasso, P.; Ceruti, M.; Brusa, P.; Arpicco, S.; Dosio, F.; Cattel, L. Preparation, characterization and properties of sterically stabilized paclitaxel-containing liposomes. J. Control. Release 2000, 63, 19–30. [Google Scholar] [CrossRef]
- Koudelka, S.; Turánek-Knötigová, P.; Masek, J.; Korvasová, Z.; Skrabalová, M.; Plocková, J.; Bartheldyová, E.; Turánek, J. Liposomes with high encapsulation capacity for paclitaxel: Preparation, characterisation and in vivo anticancer effect. J. Pharm. Sci. 2010, 99, 2309–2319. [Google Scholar] [CrossRef] [PubMed]
- Kan, P.; Tsao, C.W.; Wang, A.J.; Su, W.C.; Liang, H.F. A liposomal formulation able to incorporate a high content of paclitaxel and exert promising anticancer effect. J. Drug Deliv. 2011, 2011, 629234. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.S.; Kim, S.H.; Lim, S.J. Effects of triglycerides on the hydrophobic drug loading capacity of saturated phosphatidylcholine-based liposomes. Int. J. Pharm. 2015, 483, 142–150. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Cui, F.D.; Choi, M.K.; Cho, J.W.; Chung, S.J.; Shim, C.K.; Kim, D.D. Enhanced solubility and stability of pegylated liposomal paclitaxel: In vitro and in vivo evaluation. Int. J. Pharm. 2007, 338, 317–326. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Choi, M.K.; Cui, F.D.; Kim, J.S.; Chung, S.J.; Shim, C.K.; Kim, D.D. Preparation and evaluation of paclitaxel-loaded pegylated immunoliposome. J. Control. Release 2007, 120, 169–177. [Google Scholar] [CrossRef] [PubMed]
- Yang, T.; Choi, M.K.; Cui, F.D.; Lee, S.J.; Chung, S.J.; Shim, C.K.; Kim, D.D. Antitumor effect of paclitaxel-loaded pegylated immunoliposomes against human breast cancer cells. Pharm. Res. 2007, 24, 2402–2411. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Burt, H.M.; Mangold, G.; Dexter, D.; Von Hoff, D.; Mayer, L.; Hunter, W.L. Anti-tumor efficacy and biodistribution of intravenous polymeric micellar paclitaxel. Anticancer Drugs 1997, 8, 696–701. [Google Scholar] [CrossRef] [PubMed]
- Nicoletti, M.I.; Lucchini, V.; Massazza, G.; Abbott, B.J.; D'Incalci, M.; Giavazzi, R. Antitumor activity of taxol (NSC-125973) in human ovarian carcinomas growing in the peritoneal cavity of nude mice. Ann. Oncol. 1993, 4, 151–155. [Google Scholar] [CrossRef] [PubMed]
- Immordino, M.L.; Brusa, P.; Arpicco, S.; Stella, B.; Dosio, F.; Cattel, L. Preparation, characterization, cytotoxicity and pharmacokinetics of liposomes containing docetaxel. J. Control. Release 2003, 91, 417–429. [Google Scholar] [CrossRef]
- Wang, Q.W.; Lu, H.L.; Song, C.C.; Liu, H.; Xu, C.G. Radiosensitivity of human colon cancer cell enhanced by immunoliposomal docetaxel. World J. Gastroenterol 2005, 11, 4003–4007. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, Y.; Yoshida, M.; Sato, M.; Sato, K.; Kikuchi, S.; Sugishita, H.; Kuwabara, J.; Matsuno, Y.; Kojima, Y.; Morimoto, M.; et al. Feasibility of tailored, selective and effective anticancer chemotherapy by direct injection of docetaxel-loaded immunoliposomes into Her2/neu positive gastric tumor xenografts. Int. J. Oncol. 2011, 38, 33–39. [Google Scholar] [PubMed]
- Manjappa, A.S.; Goel, P.N.; Gude, R.P.; Ramachandra Murthy, R.S. Anti-neuropilin 1 antibody Fab' fragment conjugated liposomal docetaxel for active targeting of tumours. J. Drug Target. 2014, 22, 698–711. [Google Scholar] [CrossRef] [PubMed]
- Shigehiro, T.; Zhai, W.; Vaidyanath, A.; Masuda, J.; Mizutani, A.; Kasai, T.; Murakami, H.; Hamada, H.; Salomon, D.S.; Mikuni, K.; et al. Evaluation of glycosylated docetaxel-encapsulated liposomes prepared by remote loading under solubility gradient. J. Microencapsul. 2016, 33, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Pero, S.C.; Shukla, G.S.; Armstrong, A.L.; Peterson, D.; Fuller, S.P.; Godin, K.; Kingsley-Richards, S.L.; Weaver, D.L.; Bond, J.; Krag, D.N. Identification of a small peptide that inhibits the phosphorylation of ErbB2 and proliferation of ErbB2 overexpressing breast cancer cells. Int. J. Cancer 2004, 111, 951–960. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Yu, Y.; Marciniak, D.; Rishi, A.K.; Sarkar, F.H.; Kucuk, O.; Majumdar, A.P. Epidermal growth factor receptor (EGFR)-related protein inhibits multiple members of the EGFR family in colon and breast cancer cells. Mol. Cancer Ther. 2005, 4, 435–442. [Google Scholar] [PubMed]
- Rusnak, D.W.; Alligood, K.J.; Mullin, R.J.; Spehar, G.M.; Arenas-Elliott, C.; Martin, A.M.; Degenhardt, Y.; Rudolph, S.K.; Haws, T.F., Jr.; Hudson-Curtis, B.L.; et al. Assessment of epidermal growth factor receptor (EGFR, ErbB1) and HER2 (ErbB2) protein expression levels and response to lapatinib (Tykerb, GW572016) in an expanded panel of human normal and tumour cell lines. Cell Prolif. 2007, 40, 580–594. [Google Scholar] [CrossRef] [PubMed]
- Gelderblom, H.; Verweij, J.; Nooter, K.; Sparreboom, A. Cremophor EL: The drawbacks and advantages of vehicle selection for drug formulation. Eur. J. Cancer 2001, 37, 1590–1598. [Google Scholar] [CrossRef]
- Harris, L.; Batist, G.; Belt, R.; Rovira, D.; Navari, R.; Azarnia, N.; Welles, L.; Winer, E.; Group, T.D.-S. Liposome-encapsulated doxorubicin compared with conventional doxorubicin in a randomized multicenter trial as first-line therapy of metastatic breast carcinoma. Cancer 2002, 94, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Mandai, T.; Okumoto, H.; Oshitari, T. Synthesis and biological evaluation of water soluble taxoids bearing sugar moieties. Heterocycles 2001, 54, 561–566. [Google Scholar] [CrossRef]
- Vaidyanath, A.; Hashizume, T.; Nagaoka, T.; Takeyasu, N.; Satoh, H.; Chen, L.; Wang, J.; Kasai, T.; Kudoh, T.; Satoh, A.; et al. Enhanced internalization of ErbB2 in SK-BR-3 cells with multivalent forms of an artificial ligand. J. Cell Mol. Med. 2011, 15, 2525–2538. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, S.C.; Kasai, T.; Satoh, A.; Shigehiro, T.; Mizutani, A.; Murakami, H.; El-Aarag, B.Y.; Salomon, D.S.; Massaguer, A.; de Llorens, R.; et al. Identification of caveolin-1 as a potential causative factor in the generation of trastuzumab resistance in breast cancer cells. J. Cancer 2013, 4, 391–401. [Google Scholar] [CrossRef] [PubMed]





| Drug Formulation | EE (%) | LE (%) | Particle Size (nm) | Zeta Potential (mV) | PDI |
|---|---|---|---|---|---|
| DTX-L | 97.3 ± 2.0 | 29.9 ± 0.6 | 148.1 ± 16.1 | −4.65 ± 1.25 | 0.130 ± 0.030 |
| DTX-ECL | 69.3 ± 9.7 | 21.3 ± 3.0 | 186.7 ± 5.4 | −7.10 ± 2.80 | 0.154 ± 0.013 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shigehiro, T.; Masuda, J.; Saito, S.; Khayrani, A.C.; Jinno, K.; Seno, A.; Vaidyanath, A.; Mizutani, A.; Kasai, T.; Murakami, H.; et al. Practical Liposomal Formulation for Taxanes with Polyethoxylated Castor Oil and Ethanol with Complete Encapsulation Efficiency and High Loading Efficiency. Nanomaterials 2017, 7, 290. https://doi.org/10.3390/nano7100290
Shigehiro T, Masuda J, Saito S, Khayrani AC, Jinno K, Seno A, Vaidyanath A, Mizutani A, Kasai T, Murakami H, et al. Practical Liposomal Formulation for Taxanes with Polyethoxylated Castor Oil and Ethanol with Complete Encapsulation Efficiency and High Loading Efficiency. Nanomaterials. 2017; 7(10):290. https://doi.org/10.3390/nano7100290
Chicago/Turabian StyleShigehiro, Tsukasa, Junko Masuda, Shoki Saito, Apriliana C. Khayrani, Kazumasa Jinno, Akimasa Seno, Arun Vaidyanath, Akifumi Mizutani, Tomonari Kasai, Hiroshi Murakami, and et al. 2017. "Practical Liposomal Formulation for Taxanes with Polyethoxylated Castor Oil and Ethanol with Complete Encapsulation Efficiency and High Loading Efficiency" Nanomaterials 7, no. 10: 290. https://doi.org/10.3390/nano7100290