MPI Phantom Study with A High-Performing Multicore Tracer Made by Coprecipitation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Magnetic Particle Imaging (MPI) Tracers
2.3. Magnetic Nanoparticles (MNP) Characterization
2.4. MPI Phantom Studies
2.5. Image Reconstruction and Analysis
2.5.1. Reconstruction
2.5.2. Evaluation of SNR-Optimized Regularization Factor (Plotting SNR Against λ)
2.5.3. Methods for Volume and Iron Evaluation in The Single-Tube Phantom Series
3. Results and Discussion
3.1. MCP 3 Synthesis
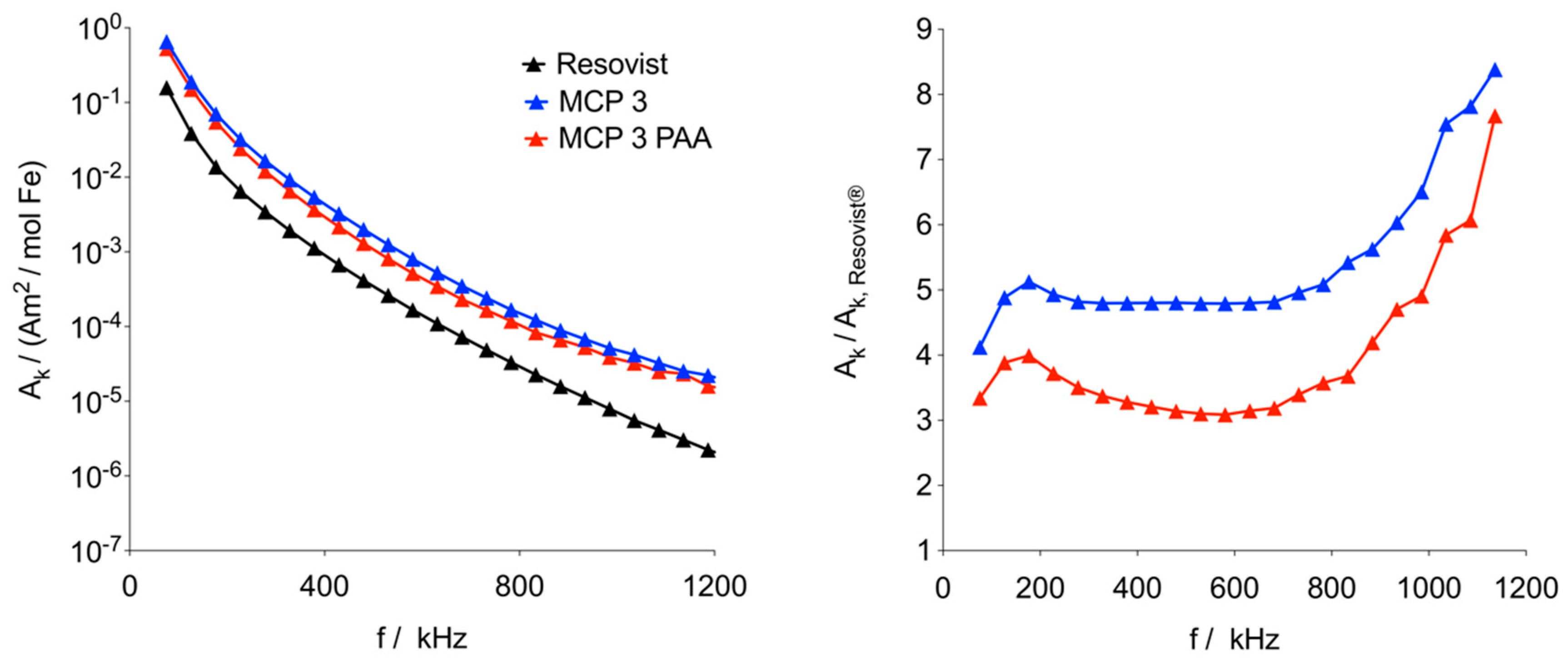
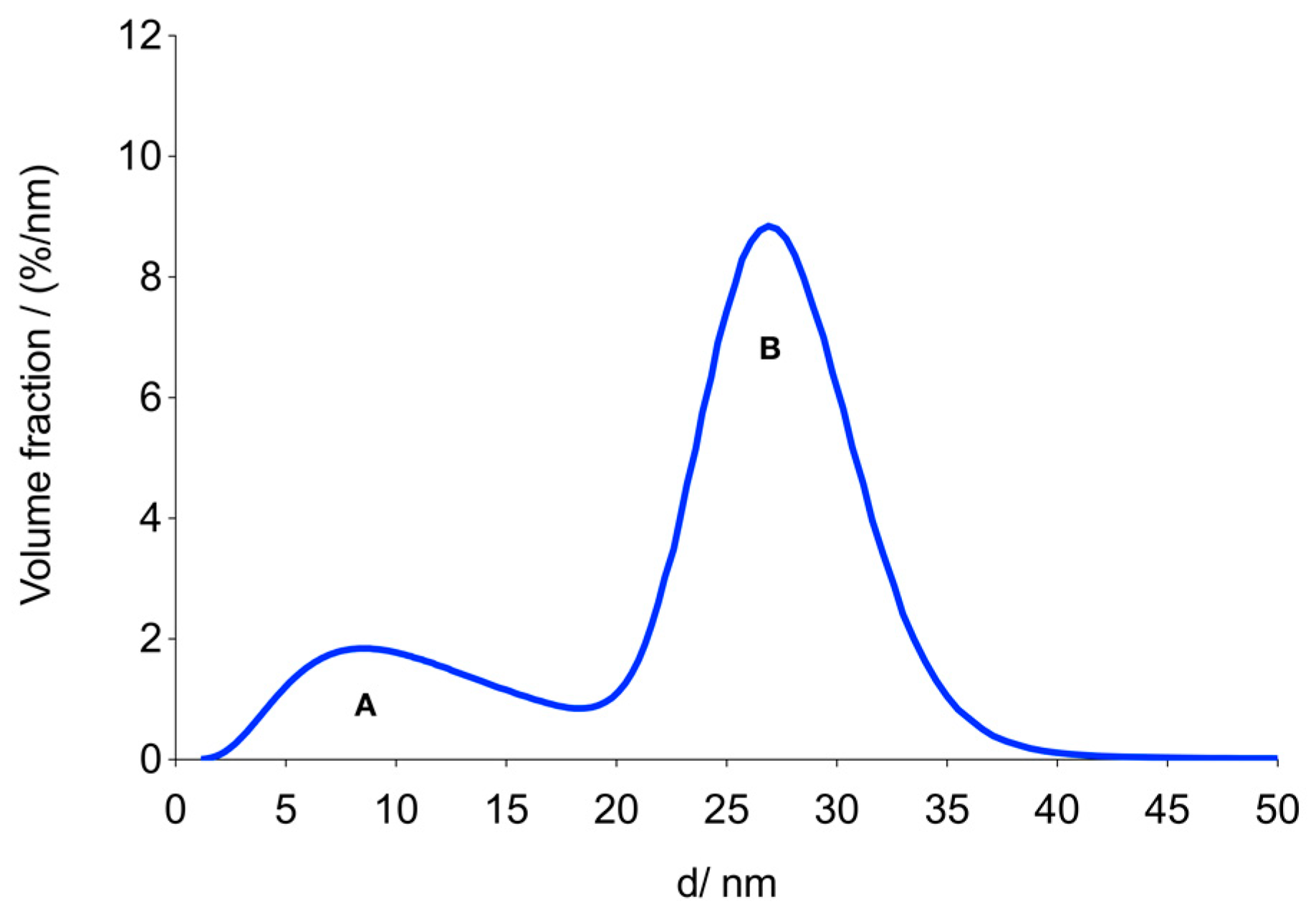
3.2. MNP Characterization
3.3. MPI Phantom Measurements/Image Reconstruction
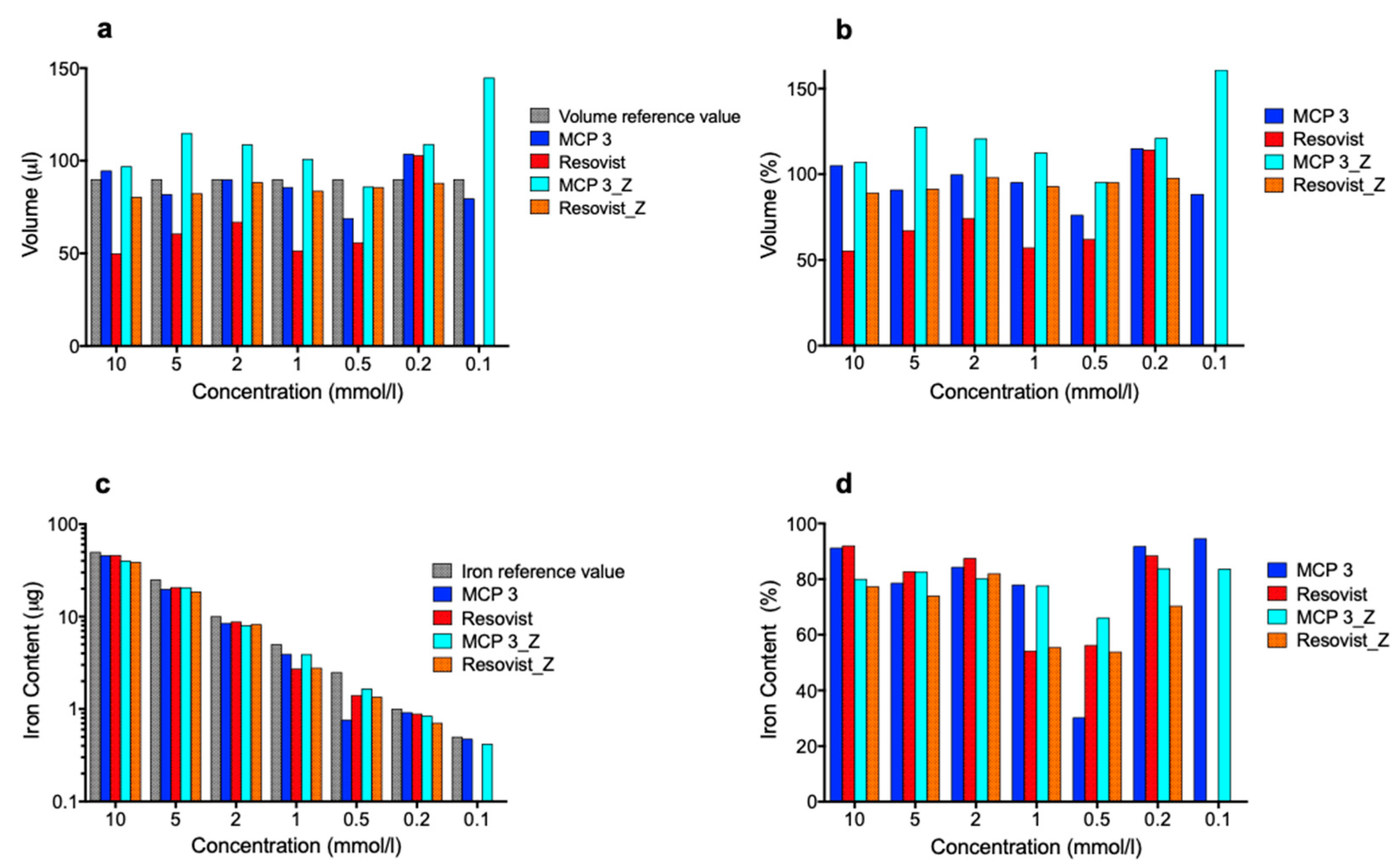
3.3.1. Phantom Dilution Series
3.3.2. Determination of Volumes and Iron Amounts in the Phantom Dilution Series
3.3.3. Dual-Tube Phantoms
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gleich, B.; Weizenecker, J. Tomographic imaging using the nonlinear response of magnetic particles. Nature 2005, 435, 1214–1217. [Google Scholar] [CrossRef] [PubMed]
- Haegele, J.; Duschka, R.L.; Graeser, M.; Schaecke, C.; Panagiotopoulos, N.; Ludtke-Buzug, K.; Buzug, T.M.; Barkhausen, J.; Vogt, F.M. Magnetic particle imaging: Kinetics of the intravascular signal in vivo. Int. J. Nanomed. 2014, 9, 4203–4209. [Google Scholar] [CrossRef] [PubMed]
- Weizenecker, J.; Gleich, B.; Rahmer, J.; Dahnke, H.; Borgert, J. Three-dimensional real-time in vivo magnetic particle imaging. Phys. Med. Biol. 2009, 54, L1–L10. [Google Scholar] [CrossRef] [PubMed]
- Borgert, J.; Schmidt, J.D.; Schmale, I.; Rahmer, J.; Bontus, C.; Gleich, B.; David, B.; Eckart, R.; Woywode, O.; Weizenecker, J.; et al. Fundamentals and applications of magnetic particle imaging. J. Cardiovasc. Comput. Tomogr. 2012, 6, 149–153. [Google Scholar] [CrossRef]
- Kratz, H.; Eberbeck, D.; Wagner, S.; Taupitz, M.; Schnorr, J. Synthetic routes to magnetic nanoparticles for MPI. Biomed. Tech. 2013, 58, 509–515. [Google Scholar] [CrossRef]
- Franke, J.; Heinen, U.; Lehr, H.; Weber, A.; Jaspard, F.; Ruhm, W.; Heidenreich, M.; Schulz, V. System characterization of a highly integrated preclinical hybrid MPI-MRI scanner. IEEE Trans. Med. Imaging 2016, 35, 1993–2004. [Google Scholar] [CrossRef]
- Rahmer, J.; Antonelli, A.; Sfara, C.; Tiemann, B.; Gleich, B.; Magnani, M.; Weizenecker, J.; Borgert, J. Nanoparticle encapsulation in red blood cells enables blood-pool magnetic particle imaging hours after injection. Phys. Med. Biol. 2013, 58, 3965–3977. [Google Scholar] [CrossRef] [Green Version]
- Kaul, M.G.; Weber, O.; Heinen, U.; Reitmeier, A.; Mummert, T.; Jung, C.; Raabe, N.; Knopp, T.; Ittrich, H.; Adam, G. Combined Preclinical Magnetic Particle Imaging and Magnetic Resonance Imaging: Initial Results in Mice. Rofo 2015, 187, 347–352. [Google Scholar] [CrossRef] [Green Version]
- Nishimoto, K.; Mimura, A.; Aoki, M.; Banura, N.; Murase, K. Application of Magnetic Particle Imaging to Pulmonary Imaging Using Nebulized Magnetic Nanoparticles. Open J. Med. Imaging 2015, 5, 49–55. [Google Scholar] [CrossRef] [Green Version]
- Zheng, B.; Vazin, T.; Goodwill, P.W.; Conway, A.; Verma, A.; Saritas, E.U.; Schaffer, D.; Conolly, S.M. Magnetic Particle Imaging tracks the long-term fate of in vivo neural cell implants with high image contrast. Sci. Rep. 2015, 5, 14055. [Google Scholar] [CrossRef]
- Ohki, A.; Tanoue, M.; Kobayashi, S.; Murase, K. Magnetic Particle Imaging for Quantitative Evaluation of Tumor Response to Magnetic Hyperthermia Treatment Combined with Chemotherapy Using Cisplatin. Therm. Med. 2017, 33, 39–51. [Google Scholar] [CrossRef] [Green Version]
- Kuboyabu, T.; Yabata, I.; Aoki, M.; Banura, N.; Nishimoto, K.; Mimura, A.; Murase, K. Magnetic Particle Imaging for Magnetic Hyperthermia Treatment: Visualization and Quantification of the Intratumoral Distribution and Temporal Change of Magnetic Nanoparticles in Vivo. Open J. Med. Imaging 2016, 6, 1–15. [Google Scholar] [CrossRef] [Green Version]
- Vogel, P.; Ruckert, M.A.; Klauer, P.; Kullmann, W.H.; Jakob, P.M.; Behr, V.C. First in vivo traveling wave magnetic particle imaging of a beating mouse heart. Phys. Med. Biol. 2016, 61, 6620–6634. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; von See, M.P.; Yu, E.; Gunel, B.; Lu, K.; Vazin, T.; Schaffer, D.V.; Goodwill, P.W.; Conolly, S.M. Quantitative Magnetic Particle Imaging Monitors the Transplantation, Biodistribution, and Clearance of Stem Cells In Vivo. Theranostics 2016, 6, 291–301. [Google Scholar] [CrossRef] [PubMed]
- Dieckhoff, J.; Kaul, M.G.; Mummert, T.; Jung, C.; Salamon, J.; Adam, G.; Knopp, T.; Ludwig, F.; Balceris, C.; Ittrich, H. In vivo liver visualizations with magnetic particle imaging based on the calibration measurement approach. Phys. Med. Biol. 2017, 62, 3470–3482. [Google Scholar] [CrossRef]
- Kaul, M.G.; Mummert, T.; Jung, C.; Salamon, J.; Khandhar, A.P.; Ferguson, R.M.; Kemp, S.J.; Ittrich, H.; Krishnan, K.M.; Adam, G.; et al. In vitro and in vivo comparison of a tailored magnetic particle imaging blood pool tracer with Resovist. Phys. Med. Biol. 2017, 62, 3454–3469. [Google Scholar] [CrossRef]
- Ohki, A.; Kuboyabu, T.; Aoki, M.; Yamawaki, M.; Murase, K. Quantitative Evaluation of Tumor Response to combination of Magnetic Hyperthermia Treatment and Radiation Therapy using Magnetic Particle Imaging. Int. J. Nanomed. Nanosurgery 2016, 2. [Google Scholar] [CrossRef]
- Khandhar, A.P.; Keselman, P.; Kemp, S.J.; Ferguson, R.M.; Goodwill, P.W.; Conolly, S.M.; Krishnan, K.M. Evaluation of PEG-coated iron oxide nanoparticles as blood pool tracers for preclinical magnetic particle imaging. Nanoscale 2017, 9, 1299–1306. [Google Scholar] [CrossRef]
- Starmans, L.W.; Burdinski, D.; Haex, N.P.; Moonen, R.P.; Strijkers, G.J.; Nicolay, K.; Grull, H. Iron oxide nanoparticle-micelles (ION-micelles) for sensitive (molecular) magnetic particle imaging and magnetic resonance imaging. PLoS ONE 2013, 8, e57335. [Google Scholar] [CrossRef]
- Ziemian, S.; Lowa, N.; Kosch, O.; Bajj, D.; Wiekhorst, F.; Schutz, G. Optimization of Iron Oxide Tracer Synthesis for Magnetic Particle Imaging. Nanomaterials 2018, 8, 180. [Google Scholar] [CrossRef]
- Eberbeck, D.; Dennis, C.L.; Huls, N.F.; Krycka, K.L.; Gruttner, C.; Westphal, F. Multicore Magnetic Nanoparticles for Magnetic Particle Imaging. IEEE Trans. Magn. 2013, 49, 269–274. [Google Scholar] [CrossRef]
- Tay, Z.W.; Chandrasekharan, P.; Zhou, X.Y.; Yu, E.; Zheng, B.; Conolly, S. In vivo tracking and quantification of inhaled aerosol using magnetic particle imaging towards inhaled therapeutic monitoring. Theranostics 2018, 8, 3676–3687. [Google Scholar] [CrossRef] [PubMed]
- Bender, P.; Fock, J.; Frandsen, C.; Hansen, M.F.; Balceris, C.; Ludwig, F.; Posth, O.; Wetterskog, E.; Bogart, L.K.; Southern, P.; et al. Relating Magnetic Properties and High Hyperthermia Performance of Iron Oxide Nanoflowers. J. Phys. Chem. C 2018, 122, 3068–3077. [Google Scholar] [CrossRef] [Green Version]
- Lu, A.H.; Salabas, E.L.; Schuth, F. Magnetic nanoparticles: Synthesis, protection, functionalization, and application. Angew. Chem. Int. Ed. 2007, 46, 1222–1244. [Google Scholar] [CrossRef] [PubMed]
- Cotin, G.; Kiefer, C.; Perton, F.; Ihiawakrim, D.; Blanco-Andujar, C.; Moldovan, S.; Lefevre, C.; Ersen, O.; Pichon, B.; Mertz, D.; et al. Unravelling the Thermal Decomposition Parameters for The Synthesis of Anisotropic Iron Oxide Nanoparticles. Nanomaterials 2018, 8, 881. [Google Scholar] [CrossRef] [PubMed]
- Kratz, H.; Taupitz, M.; Ariza de Schellenberger, A.; Kosch, O.; Eberbeck, D.; Wagner, S.; Trahms, L.; Hamm, B.; Schnorr, J. Novel magnetic multicore nanoparticles designed for MPI and other biomedical applications: From synthesis to first in vivo studies. PLoS ONE 2018, 13, e0190214. [Google Scholar] [CrossRef] [PubMed]
- Laurent, S.; Forge, D.; Port, M.; Roch, A.; Robic, C.; Vander Elst, L.; Muller, R.N. Magnetic iron oxide nanoparticles: Synthesis, stabilization, vectorization, physicochemical characterizations, and biological applications. Chem. Rev. 2008, 108, 2064–2110. [Google Scholar] [CrossRef]
- Eberbeck, D.; Wiekhorst, F.; Wagner, S.; Trahms, L. How the size distribution of magnetic nanoparticles determines their magnetic particle imaging performance. Appl. Phys. Lett. 2011, 98. [Google Scholar] [CrossRef]
- Ferguson, R.M.; Khandhar, A.P.; Arami, H.; Hua, L.; Hovorka, O.; Krishnan, K.M. Tailoring the magnetic and pharmacokinetic properties of iron oxide magnetic particle imaging tracers. Biomed. Tech. 2013, 58, 493–507. [Google Scholar] [CrossRef] [Green Version]
- Scharlach, C.; Müller, L.; Wagner, S.; Kobayashi, Y.; Kratz, H.; Ebert, M.; Jakubowski, N.; Schellenberger, E. LA-ICP-MS Allows Quantitative Microscopy of Europium-Doped Iron Oxide Nanoparticles and is a Possible Alternative to Ambiguous Prussian Blue Iron Staining. J. Biomed. Nanotechnol. 2016, 12, 1001–1010. [Google Scholar] [CrossRef] [PubMed]
- Wells, J.; Paysen, H.; Kosch, O.; Löwa, N.; Schmitzberger, F.; Makowksi, M.; Franke, J.; Trahms, L.; Wiekhorst, F. Characterizing a Preclinical Magnetic Particle Imaging System with Separate Pickup Coil. IEEE Trans. Magn. 2017, 53, 1–5. [Google Scholar] [CrossRef]
- Paysen, H.; Wells, J.; Kosch, O.; Steinhoff, U.; Franke, J.; Trahms, L.; Schaeffter, T.; Wiekhorst, F. Improved sensitivity and limit-of-detection using a receive-only coil in magnetic particle imaging. Phys. Med. Biol. 2018, 63, 13NT02. [Google Scholar] [CrossRef] [PubMed]
- Them, K.; Kaul, M.G.; Jung, C.; Hofmann, M.; Mummert, T.; Werner, F.; Knopp, T. Sensitivity Enhancement in Magnetic Particle Imaging by Background Subtraction. IEEE Trans. Med. Imaging 2016, 35, 893–900. [Google Scholar] [CrossRef] [PubMed]
- Hansen, P.C. Discrete Inverse Problems: Insight and Algorithms; Siam: Philadelphia, PA, USA, 2010. [Google Scholar]
- Hemalatha, R.J.; Thamizhvani, T.R.; Dhivya, A.J.A.; Joseph, J.E.; Babu, B.; Chandrasekaran, R. Active Contour Based Segmentation Techniques for Medical Image Analysis. In Medical and Biological Image Analysis; IntechOpen: London, UK, 2018. [Google Scholar] [Green Version]
- Reimer, P.; Balzer, T. Ferucarbotran (Resovist): A new clinically approved RES-specific contrast agent for contrast-enhanced MRI of the liver: Properties, clinical development, and applications. Eur. Radiol. 2003, 13, 1266–1276. [Google Scholar] [CrossRef]
- Wang, Y.X. Superparamagnetic iron oxide based MRI contrast agents: Current status of clinical application. Quant. Imaging Med. Surg. 2011, 1, 35–40. [Google Scholar] [CrossRef]
- Hyeon, T.; Lee, S.S.; Park, J.; Chung, Y.; Bin Na, H. Synthesis of highly crystalline and monodisperse maghemite nanocrystallites without a size-selection process. J. Am. Chem. Soc. 2001, 123, 12798–12801. [Google Scholar] [CrossRef]
- Roca, A.G.; Marco, J.F.; Morales, M.D.; Serna, C.J. Effect of nature and particle size on properties of uniform magnetite and maghemite nanoparticles. J. Phys. Chem. C 2007, 111, 18577–18584. [Google Scholar] [CrossRef]
- Kaiser, R.; Miskolczy, G. Magnetic properties of stable dispersions of subdomain magnetite particles. J. Appl. Phys. 1970, 41, 1064–1072. [Google Scholar] [CrossRef]
- Anthore, R.; Petipas, C.; Chandesris, D.; Martinet, A. X-ray and magnetization studies of the geometrical parameters of the grains of a ferrofluid. Le J. De Phys. Colloq. 1977, 38, C2–C203. [Google Scholar]
- Chantrell, R.; Popplewell, J.; Charles, S. Measurements of particle size distribution parameters in ferrofluids. IEEE Trans. Magn. 1978, 14, 975–977. [Google Scholar] [CrossRef]
- O’Grady, K.; Bradbury, A.; Popplewell, J.; Charles, S.; Chantrell, R. The effect of field induced texture on the properties of a fine particle system. J. Magn. Magn. Mater. 1985, 49, 106–116. [Google Scholar] [CrossRef]
- Van Leeuwen, D.A.; van Ruitenbeek, J.M.; de Jongh, L.J.; Ceriotti, A.; Pacchioni, G.; Haberlen, O.D.; Rosch, N. Quenching of magnetic moments by ligand-metal interactions in nanosized magnetic metal clusters. Phys. Rev. Lett. 1994, 73, 1432–1435. [Google Scholar] [CrossRef] [PubMed]
- Millan, A.; Urtizberea, A.; Silva, N.; Palacio, F.; Amaral, V.; Snoeck, E.; Serin, V. Surface effects in maghemite nanoparticles. J. Magn. Magn. Mater. 2007, 312, L5–L9. [Google Scholar] [CrossRef]
- Ahrens, T.J. Rock Physics & Phase Relations; Wiley Online Library: Hoboken, NJ, USA, 1995. [Google Scholar]
- Biederer, S.; Knopp, T.; Sattel, T.; Lüdtke-Buzug, K.; Gleich, B.; Weizenecker, J.; Borgert, J.; Buzug, T. Magnetization response spectroscopy of superparamagnetic nanoparticles for magnetic particle imaging. J. Phys. D Appl. Phys. 2009, 42, 205007. [Google Scholar] [CrossRef]
- Arami, H.; Khandhar, A.; Liggitt, D.; Krishnan, K.M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 2015, 44, 8576–8607. [Google Scholar] [CrossRef]
- Blyakhman, F.A.; Makarova, E.B.; Fadeyev, F.A.; Lugovets, D.V.; Safronov, A.P.; Shabadrov, P.A.; Shklyar, T.F.; Melnikov, G.Y.; Orue, I.; Kurlyandskaya, G.V. The Contribution of Magnetic Nanoparticles to Ferrogel Biophysical Properties. Nanomaterials 2019, 9, 232. [Google Scholar] [CrossRef] [PubMed]
- Arbab, A.S.; Wilson, L.B.; Ashari, P.; Jordan, E.K.; Lewis, B.K.; Frank, J.A. A model of lysosomal metabolism of dextran coated superparamagnetic iron oxide (SPIO) nanoparticles: Implications for cellular magnetic resonance imaging. NMR Biomed. 2005, 18, 383–389. [Google Scholar] [CrossRef]
- Wisotzki, E.I.; Eberbeck, D.; Kratz, H.; Mayr, S.G. Magnetic response of gelatin ferrogels across the sol-gel transition: The influence of high energy crosslinking on thermal stability. Soft Matter 2016, 12, 3908–3918. [Google Scholar] [CrossRef]
- Rahmer, J.; Weizenecker, J.; Gleich, B.; Borgert, J. Analysis of a 3-D system function measured for magnetic particle imaging. IEEE Trans. Med. Imaging 2012, 31, 1289–1299. [Google Scholar] [CrossRef]
- Vaalma, S.; Rahmer, J.; Panagiotopoulos, N.; Duschka, R.L.; Borgert, J.; Barkhausen, J.; Vogt, F.M.; Haegele, J. Magnetic Particle Imaging (MPI): Experimental Quantification of Vascular Stenosis Using Stationary Stenosis Phantoms. PLoS ONE 2017, 12, e0168902. [Google Scholar] [CrossRef]
- Rahmer, J.; Weizenecker, J.; Gleich, B.; Borgert, J. Signal encoding in magnetic particle imaging: properties of the system function. BMC Med. Imaging 2009, 9, 4. [Google Scholar] [CrossRef] [PubMed]
- Knopp, T.; Gdaniec, N.; Moddel, M. Magnetic particle imaging: from proof of principle to preclinical applications. Phys. Med. Biol. 2017, 62, R124–R178. [Google Scholar] [CrossRef] [PubMed]








| Mean Core Diameter (TEM) * [nm] | dV (DLS) [nm] by Volume | Z-Average (DLS) [nm] | PDI (DLS) | ζ-Potential [mV] |
|---|---|---|---|---|
| 31.72 ± 8.4 | 24.4–122.4 | 53.94 | 0.08 | −33.5 |
| Sample | β | dv2 (nm) | σ2 | Ms Am2/kg Fe | μ2 (aAm2) | β·μ2 (aAm2) | M3 Am2/mol(Fe) |
|---|---|---|---|---|---|---|---|
| MCP 3 | 0.73 ± 0.02 | 27 ± 0.5 | 0.13 ± 0.01 | 5.81 ± 0.25 | 3.9 ± 0.2 | 2.81 ± 0.16 | 0.65 |
| β | Volume fraction of the second mode (B) of the assumed bimodal size distribution obtained from M(H) data | ||||||
| dv2 | Mean volume diameter of the second mode (B) of the assumed bimodal size distribution obtained from M(H) data | ||||||
| σ2 | Geometric dispersion parameter of the second mode (B) of the assumed bimodal size distribution obtained from M(H) data | ||||||
| Ms | Saturation magnetization of the second mode (B) of the assumed bimodal size distribution obtained from M(H) data | ||||||
| μ2 | Mean magnetic moment of the second mode (B) of the assumed bimodal size distribution obtained from M(H) data | ||||||
| M3 | Third harmonic of measured MPS data (f = 25 kHz, B = 10 mT) | ||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kratz, H.; Mohtashamdolatshahi, A.; Eberbeck, D.; Kosch, O.; Hauptmann, R.; Wiekhorst, F.; Taupitz, M.; Hamm, B.; Schnorr, J. MPI Phantom Study with A High-Performing Multicore Tracer Made by Coprecipitation. Nanomaterials 2019, 9, 1466. https://doi.org/10.3390/nano9101466
Kratz H, Mohtashamdolatshahi A, Eberbeck D, Kosch O, Hauptmann R, Wiekhorst F, Taupitz M, Hamm B, Schnorr J. MPI Phantom Study with A High-Performing Multicore Tracer Made by Coprecipitation. Nanomaterials. 2019; 9(10):1466. https://doi.org/10.3390/nano9101466
Chicago/Turabian StyleKratz, Harald, Azadeh Mohtashamdolatshahi, Dietmar Eberbeck, Olaf Kosch, Ralf Hauptmann, Frank Wiekhorst, Matthias Taupitz, Bernd Hamm, and Jörg Schnorr. 2019. "MPI Phantom Study with A High-Performing Multicore Tracer Made by Coprecipitation" Nanomaterials 9, no. 10: 1466. https://doi.org/10.3390/nano9101466





