In vivo Efficacy and Safety Evaluation of Lactosyl-β-cyclodextrin as a Therapeutic Agent for Hepatomegaly in Niemann-Pick Type C Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Synthesis of HF647-Labeled β-CyDs
2.3. Cell Culture
2.4. Mice
2.5. In Vivo Fluorescence Imaging
2.6. Treatment of NPC Model Mice with CyDs
2.7. Biochemical and Histological Analysisβ
2.8. In Vivo Safety Evaluation of CyDs in WT Mice
2.9. Cellular Uptake Analysis
2.10. Free Cholesterol Levels in Endolysosomes
2.11. Analysis of Cholesterol Solubilization by CyDs
2.12. Hemolytic Activity of CyDs in Rabbit Red Blood Cells
2.13. Cytotoxicity of CyDs
2.14. Data Analysis
3. Results
3.1. In Vivo Fluorescence Imaging of HF647-Lac-β-CyD
3.2. Therapeutic Effect of Lac-β-CyD in NPC Mice
3.3. In Vivo Safety Evaluation of Lac-β-CyD in WT Mice
3.4. Intracellular Distribution of HF647-Lac-β-CyD in NPC-like Liver Cells
3.5. Free Cholesterol Lowering Effect of Lac-β-CyD in Endolysosomes
3.6. In Vitro Safety Evaluation of Lac-β-CyD
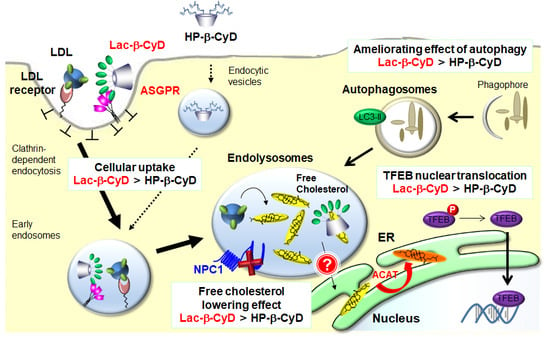
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Platt, F.M.; Boland, B.; van der Spoel, A.C. The cell biology of disease: Lysosomal storage disorders: The cellular impact of lysosomal dysfunction. J. Cell. Biol. 2012, 199, 723–734. [Google Scholar] [CrossRef]
- Futerman, A.H.; van Meer, G. The cell biology of lysosomal storage disorders. Nat. Rev. Mol. Cell. Biol. 2004, 5, 554–565. [Google Scholar] [CrossRef]
- Vellodi, A. Lysosomal storage disorders. Br. J. Haematol. 2005, 128, 413–431. [Google Scholar] [CrossRef]
- Winchester, B.; Vellodi, A.; Young, E. The molecular basis of lysosomal storage diseases and their treatment. Biochem. Soc. Trans. 2000, 28, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Vanier, M.T. Niemann-Pick disease type C. Orphanet J. Rare Dis. 2010, 5, 16. [Google Scholar] [CrossRef] [PubMed]
- Vanier, M.T. Pediatric Neurology Part III: Chapter 176. Niemann–Pick diseases. Handb. Clin. Neurol. 2013, 113, 1717–1721. [Google Scholar]
- Lyseng-Williamson, K.A. Miglustat: A review of its use in Niemann-Pick disease type C. Drugs 2014, 74, 61–74. [Google Scholar] [CrossRef] [PubMed]
- Helquist, P.; Maxfield, F.R.; Wiech, N.L.; Wiest, O. Treatment of Niemann-pick type C disease by histone deacetylase inhibitors. Neurotherapeutics 2013, 10, 688–697. [Google Scholar] [CrossRef]
- Lloyd-Evans, E.; Morgan, A.J.; He, X.; Smith, D.A.; Elliot-Smith, E.; Sillence, D.J.; Churchill, G.C.; Schuchman, E.H.; Galione, A.; Platt, F.M. Niemann-Pick disease type C1 is a sphingosine storage disease that causes deregulation of lysosomal calcium. Nat. Med. 2008, 14, 1247–1255. [Google Scholar] [CrossRef]
- Davidson, C.D.; Ali, N.F.; Micsenyi, M.C.; Stephney, G.; Renault, S.; Dobrenis, K.; Ory, D.S.; Vanier, M.T.; Walkley, S.U. Chronic cyclodextrin treatment of murine Niemann-Pick C disease ameliorates neuronal cholesterol and glycosphingolipid storage and disease progression. PLoS ONE 2009, 4, e6951. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Turley, S.D.; Burns, D.K.; Miller, A.M.; Repa, J.J.; Dietschy, J.M. Reversal of defective lysosomal transport in NPC disease ameliorates liver dysfunction and neurodegeneration in the npc1−/− mouse. Proc. Natl. Acad. Sci. USA 2009, 106, 2377–2382. [Google Scholar] [CrossRef] [PubMed]
- Darblade, B.; Caillaud, D.; Poirot, M.; Fouque, M.; Thiers, J.C.; Rami, J.; Bayard, F.; Arnal, J.F. Alteration of plasmalemmal caveolae mimics endothelial dysfunction observed in atheromatous rabbit aorta. Cardiovasc. Res. 2001, 50, 566–576. [Google Scholar] [CrossRef]
- Parpal, S.; Karlsson, M.; Thorn, H.; Stralfors, P. Cholesterol depletion disrupts caveolae and insulin receptor signaling for metabolic control via insulin receptor substrate-1, but not for mitogen-activated protein kinase control. J. Biol. Chem. 2001, 276, 9670–9678. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, M.; Shraishi, K.; Wada, K.; Ishitsuka, Y.; Doi, H.; Maeda, M.; Mizoguchi, T.; Eto, J.; Mochinaga, S.; Arima, H.; Irie, T. Effects of intracerebroventricular administration of 2-hydroxypropyl-b-cyclodextrin in a patient with Niemann-Pick Type C disease. Mol. Genet. Metab. Rep. 2014, 1, 391–400. [Google Scholar] [CrossRef]
- Rigopoulou, E.I.; Roggenbuck, D.; Smyk, D.S.; Liaskos, C.; Mytilinaiou, M.G.; Feist, E.; Conrad, K.; Bogdanos, D.P. Asialoglycoprotein receptor (ASGPR) as target autoantigen in liver autoimmunity: Lost and found. Autoimmun. Rev. 2012, 12, 260–269. [Google Scholar] [CrossRef]
- Guo, H.; Zhang, D.; Li, T.; Li, C.; Guo, Y.; Liu, G.; Hao, L.; Shen, J.; Qi, L.; Liu, X.; Luan, J.; Zhang, Q. In vitro and in vivo study of Gal-OS self-assembled nanoparticles for liver-targeting delivery of doxorubicin. J. Pharm. Sci. 2014, 103, 987–993. [Google Scholar] [CrossRef]
- Motoyama, K.; Hirai, Y.; Nishiyama, R.; Maeda, Y.; Higashi, T.; Ishitsuka, Y.; Kondo, Y.; Irie, T.; Era, T.; Arima, H. Cholesterol lowering effects of mono-lactose-appended b-cyclodextrin in Niemann-Pick type C disease-like HepG2 cells. Beilstein J. Org. Chem. 2015, 11, 2079–2086. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.C.; Lee, R.T. Carbohydrate-protein interactions: Basis of glycobiology. Acc. Chem. Res. 1995, 28, 321–327. [Google Scholar] [CrossRef]
- Rice, K.G.; Lee, Y.C. Modification of triantennary glycopeptide into probes for the asialoglycoprotein receptor of hepatocytes. J. Biol. Chem. 1990, 265, 18423–18428. [Google Scholar]
- Motoyama, K.; Nishiyama, R.; Maeda, Y.; Higashi, T.; Ishitsuka, Y.; Kondo, Y.; Irie, T.; Era, T.; Arima, H. Synthesis of multi-lactose-appended b-cyclodextrin and its cholesterol-lowering effects in Niemann-Pick type C disease-like HepG2 cells. Beilstein J. Org. Chem. 2017, 13, 10–18. [Google Scholar] [CrossRef]
- Irie, T.; Otagiri, M.; Sunada, M.; Uekama, K.; Ohtani, Y.; Yamada, Y.; Sugiyama, Y. Cyclodextrin-induced hemolysis and shape changes of human erythrocytes in vitro. J. Pharmacobiodyn. 1982, 5, 741–744. [Google Scholar] [CrossRef] [PubMed]
- Uekama, K.; Irie, T.; Sunada, M.; Otagiri, M.; Tsubaki, K. Protective effects of cyclodextrins on drug-induced hemolysis in vitro. J. Pharmacobiodyn. 1981, 4, 142–144. [Google Scholar] [CrossRef]
- Liu, B.; Ramirez, C.M.; Miller, A.M.; Repa, J.J.; Turley, S.D.; Dietschy, J.M. Cyclodextrin overcomes the transport defect in nearly every organ of NPC1 mice leading to excretion of sequestered cholesterol as bile acid. J. Lipid Res. 2010, 51, 933–944. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, C.M.; Liu, B.; Aqul, A.; Taylor, A.M.; Repa, J.J.; Turley, S.D.; Dietschy, J.M. Quantitative role of LAL, NPC2, and NPC1 in lysosomal cholesterol processing defined by genetic and pharmacological manipulations. J. Lipid Res. 2011, 52, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, C.M.; Liu, B.; Taylor, A.M.; Repa, J.J.; Burns, D.K.; Weinberg, A.G.; Turley, S.D.; Dietschy, J.M. Weekly cyclodextrin administration normalizes cholesterol metabolism in nearly every organ of the Niemann-Pick type C1 mouse and markedly prolongs life. Pediatr. Res. 2010, 68, 309–315. [Google Scholar] [CrossRef]
- Tanaka, Y.; Yamada, Y.; Ishitsuka, Y.; Matsuo, M.; Shiraishi, K.; Wada, K.; Uchio, Y.; Kondo, Y.; Takeo, T.; Nakagata, N.; et al. Efficacy of 2-Hydroxypropyl-b-cyclodextrin in Niemann-Pick disease type C model mice and its pharmacokinetic analysis in a patient with the disease. Biol. Pharm. Bull. 2015, 38, 844–851. [Google Scholar] [CrossRef] [PubMed]
- Vite, C.H.; Bagel, J.H.; Swain, G.P.; Prociuk, M.; Sikora, T.U.; Stein, V.M.; O’Donnell, P.; Ruane, T.; Ward, S.; Crooks, A.; et al. Intracisternal cyclodextrin prevents cerebellar dysfunction and Purkinje cell death in feline Niemann-Pick type C1 disease. Sci. Transl. Med. 2015, 7, 276ra226. [Google Scholar] [CrossRef] [PubMed]
- Chien, Y.H.; Shieh, Y.D.; Yang, C.Y.; Lee, N.C.; Hwu, W.L. Lung toxicity of hydroxypropyl-b-cyclodextrin infusion. Mol. Genet. Metab. 2013, 109, 231–232. [Google Scholar] [CrossRef]
- Gould, S.; Scott, R.C. 2-Hydroxypropyl-b-cyclodextrin (HP-b-CD): A toxicology review. Food Chem. Toxicol. 2005, 43, 1451–1459. [Google Scholar] [CrossRef]
- Okada, Y.; Ueda, E.; Kondo, Y.; Ishitsuka, Y.; Irie, T.; Higashi, T.; Motoyama, K.; Arima, H.; Matuso, M.; Higaki, K.; et al. Role of 6-O-a-maltosyl-b-cyclodextrin in lysosomal cholesterol deprivation in Npc1-deficient Chinese hamster ovary cells. Carbohydr. Res. 2018, 455, 54–61. [Google Scholar] [CrossRef]
- Sugimoto, Y.; Ninomiya, H.; Ohsaki, Y.; Higaki, K.; Davies, J.P.; Ioannou, Y.A.; Ohno, K. Accumulation of cholera toxin and GM1 ganglioside in the early endosome of Niemann-Pick C1-deficient cells. Proc. Natl. Acad. Sci. USA 2001, 98, 12391–12396. [Google Scholar] [CrossRef]
- Frijlink, H.W.; Visser, J.; Hefting, N.R.; Oosting, R.; Meijer, D.K.; Lerk, C.F. The pharmacokinetics of b-cyclodextrin and hydroxypropyl-b-cyclodextrin in the rat. Pharm. Res. 1990, 7, 1248–1252. [Google Scholar] [CrossRef]
- Andersen, C.B.; Moestrup, S.K. How calcium makes endocytic receptors attractive. Trends Biochem. Sci. 2014, 39, 82–90. [Google Scholar] [CrossRef] [PubMed]
- Weigel, P.H.; Yik, J.H. Glycans as endocytosis signals: The cases of the asialoglycoprotein and hyaluronan/chondroitin sulfate receptors. Biochim. Biophys. Acta 2002, 1572, 341–363. [Google Scholar] [CrossRef]
- van der Kant, R.; Neefjes, J. Small regulators, major consequences-Ca(2)(+) and cholesterol at the endosome-ER interface. J. Cell Sci. 2014, 127, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Abi-Mosleh, L.; Infante, R.E.; Radhakrishnan, A.; Goldstein, J.L.; Brown, M.S. Cyclodextrin overcomes deficient lysosome-to-endoplasmic reticulum transport of cholesterol in Niemann-Pick type C cells. Proc. Natl. Acad. Sci. USA 2009, 106, 19316–19321. [Google Scholar] [CrossRef] [PubMed]
- Blanchette-Mackie, E.J. Intracellular cholesterol trafficking: Role of the NPC1 protein. Biochim. Biophys. Acta 2000, 1486, 171–183. [Google Scholar] [CrossRef]










© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Maeda, Y.; Motoyama, K.; Nishiyama, R.; Higashi, T.; Onodera, R.; Nakamura, H.; Takeo, T.; Nakagata, N.; Yamada, Y.; Ishitsuka, Y.; et al. In vivo Efficacy and Safety Evaluation of Lactosyl-β-cyclodextrin as a Therapeutic Agent for Hepatomegaly in Niemann-Pick Type C Disease. Nanomaterials 2019, 9, 802. https://doi.org/10.3390/nano9050802
Maeda Y, Motoyama K, Nishiyama R, Higashi T, Onodera R, Nakamura H, Takeo T, Nakagata N, Yamada Y, Ishitsuka Y, et al. In vivo Efficacy and Safety Evaluation of Lactosyl-β-cyclodextrin as a Therapeutic Agent for Hepatomegaly in Niemann-Pick Type C Disease. Nanomaterials. 2019; 9(5):802. https://doi.org/10.3390/nano9050802
Chicago/Turabian StyleMaeda, Yuki, Keiichi Motoyama, Rena Nishiyama, Taishi Higashi, Risako Onodera, Hideaki Nakamura, Toru Takeo, Naomi Nakagata, Yusei Yamada, Yoichi Ishitsuka, and et al. 2019. "In vivo Efficacy and Safety Evaluation of Lactosyl-β-cyclodextrin as a Therapeutic Agent for Hepatomegaly in Niemann-Pick Type C Disease" Nanomaterials 9, no. 5: 802. https://doi.org/10.3390/nano9050802
APA StyleMaeda, Y., Motoyama, K., Nishiyama, R., Higashi, T., Onodera, R., Nakamura, H., Takeo, T., Nakagata, N., Yamada, Y., Ishitsuka, Y., Kondo, Y., Irie, T., Era, T., & Arima, H. (2019). In vivo Efficacy and Safety Evaluation of Lactosyl-β-cyclodextrin as a Therapeutic Agent for Hepatomegaly in Niemann-Pick Type C Disease. Nanomaterials, 9(5), 802. https://doi.org/10.3390/nano9050802