Assessment of Valuable and Critical Elements Recovery Potential in Ashes from Processes of Solid Municipal Waste and Sewage Sludge Thermal Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Sewage Sludge Incineration Technology
2.2. Municipal Waste Incineration Plant 1
2.3. Municipal Waste Incineration Plant 2
2.4. Analytical Methods
3. Results and Discussion
3.1. Grain Size Distribution and Mineral Composition
3.2. FA in the Rock Classification Scheme
3.3. Chemical Composition of FA in Comparison with Rocks And ores
3.4. Possible Applications of FA
4. Conclusions
- Chemical composition of FA exhibits strong variation related to the localisation of incinerators.
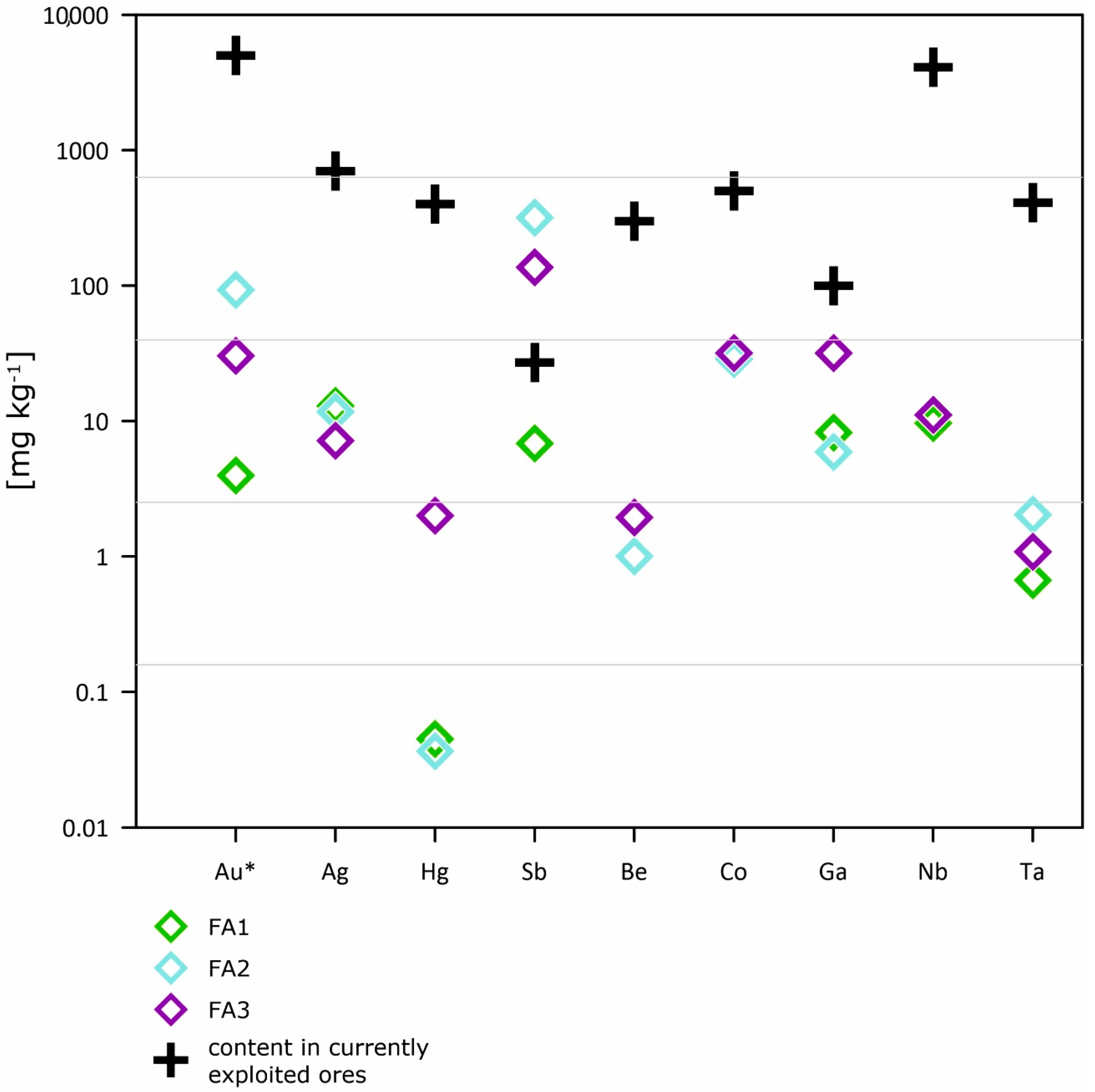
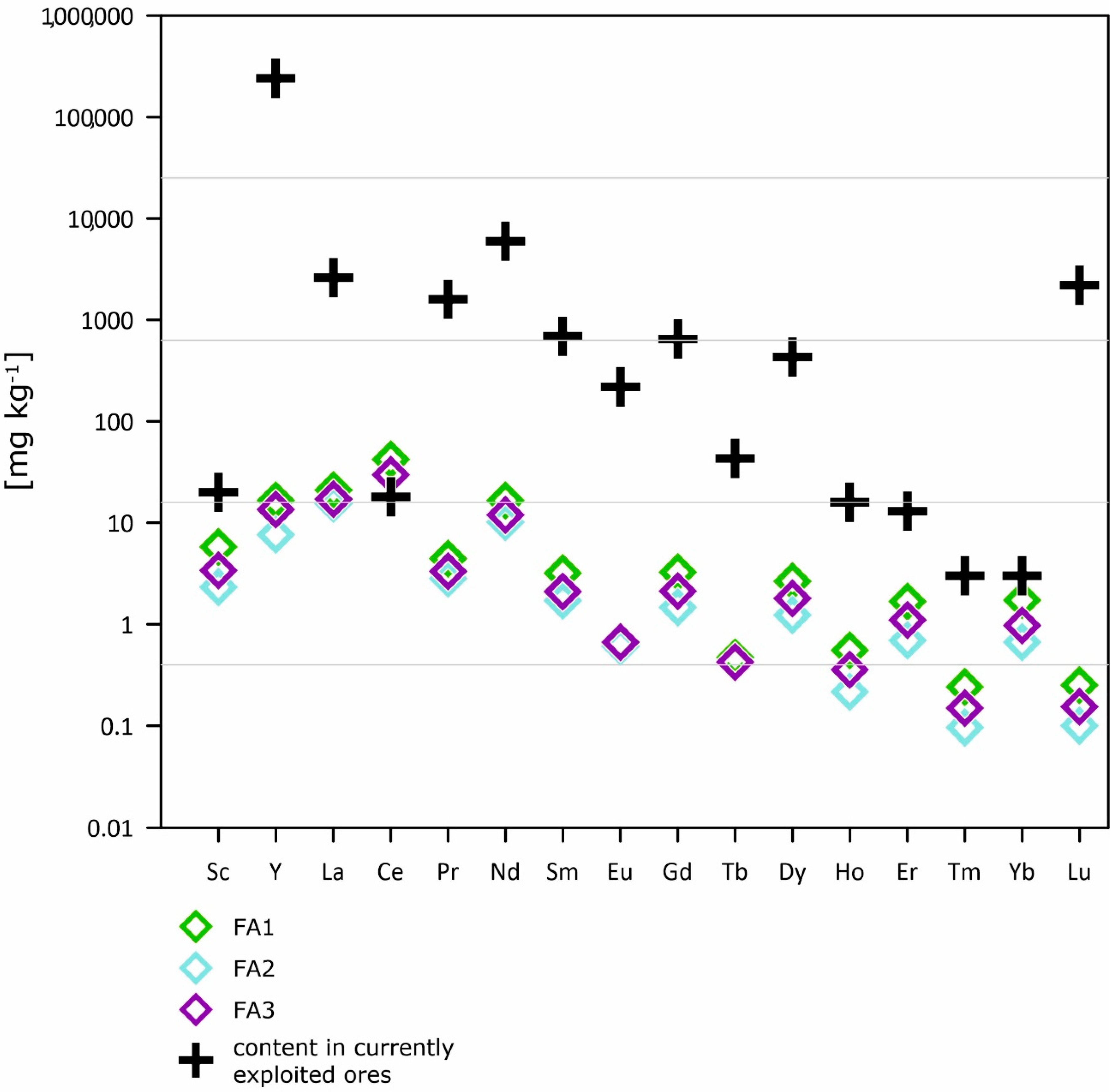
- FA contains more valuable and critical elements in comparison to the Earth materials, but less in comparison to the content of currently exploited ores; thus, the recovery of elements from FA seems to be unprofitable or unreasonable as direct extraction from unprocessed materials.
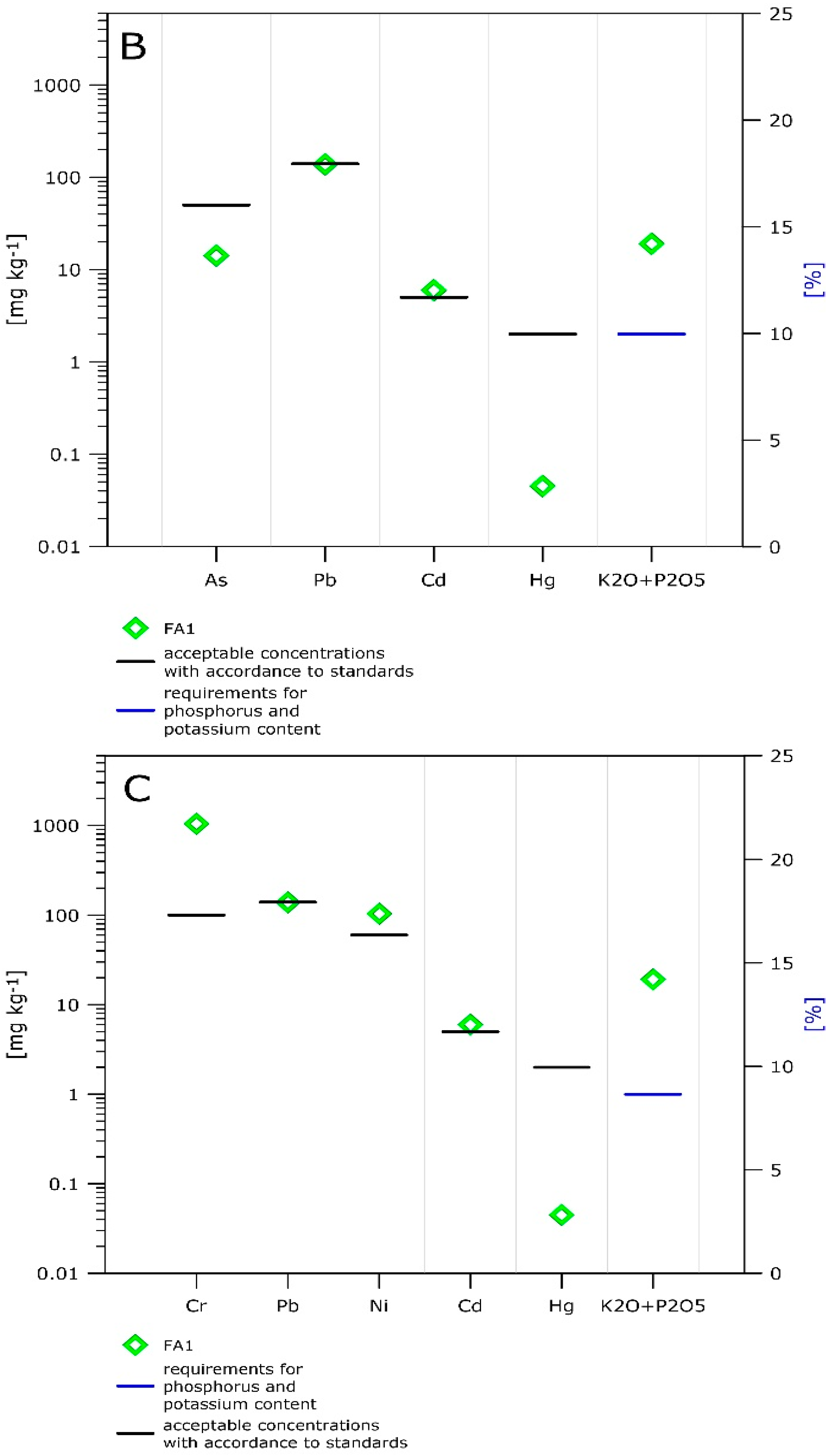
- The high phosphorus content in the sewage sludge FA (~17 wt% of P2O5), where the concentration is close to medium grade ores, enables us to consider this material as a source of this element.
- The decrease and exploitation of natural resources, mineral processing, and other technical costs such as building and maintaining infrastructure result in increasing environmental and social costs, whereas FA are easily accessible, cheap materials that do not require complicated processing methods due to the small fraction; therefore, FA can be considered as a future waste-based source of economically important elements (i.e., P, Zn, Sn, Cr, Pb, Au and Ag).
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Steffen, W.; Grinevald, J.; Crutzen, P.; McNeill, J. The Anthropocene: Conceptual and historical perspectives. Phil. Trans. R. Soc. A 2011, 369, 842–867. [Google Scholar] [CrossRef] [PubMed]
- Steffen, W.; Persson, Å.; Deutsch Zalasiewicz, J.; Williams, M.; Richardson, K.; Crumley, C.; Crutzen, P.; Folke, C.; Gordon, L.; Molina, M.; et al. The Anthropocene: From Global Change to Planetary Stewardship. Ambio 2011, 40, 739–761. [Google Scholar] [CrossRef] [PubMed]
- Blengini, G.A.; Blagoeva, D.; Dewulf, J.; Torres de Matos, C.; Nita, V.; Vidal-Legaz, B.; Latunussa, C.; Kayam, Y.; Talens Peirò, L.; Baranzelli, C.E.L. Assessment of the Methodology for Establishing the EU List of Critical Raw Materials. Background Report; Joint Research Centre (JRC); Publications Office of the European Union: Luxembourg, 2017; ISSN 1831-9424. [Google Scholar] [CrossRef]
- Morf, L.S.; Gloor, R.; Haag, O.; Haupt, M.; Skutan, S.; Di Lorenzo, F.; Böni, D. Precious metals and rare earth elements in municipal solid waste—Sources and fate in a Swiss incineration plant. Waste Manag. 2013, 33, 634–644. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Report on Critical Raw Materials for the EU 2014. In Report of the Ad-Hoc Working Group on Defining Critical Raw Materials; Raw Materials Supply Group: Brussels, Belgium, 2014. [Google Scholar]
- Communication from the Commission to the European Parliament, the Council, the European Economic and Social Committee and the Committee of the Regions on the 2017 list of Critical Raw Materials for the EU. Available online: https://ec.europa.eu/transparency/regdoc/rep/1/2017/EN/COM-2017-490-F1-EN-MAIN-PART-1.PDF (accessed on 13 September 2017).
- European Commission. Critical Raw Materials for the EU. 2010. In Report of the Ad-Hoc Working Group on Defining Critical Raw Materials; Raw Materials Supply Group: Brussels, Belgium, 2010. [Google Scholar]
- Achzet, B.; Helbig, C. How to evaluate raw material supply risks-an overview. Resour. Policy 2013, 38, 435–447. [Google Scholar] [CrossRef]
- Chapman, A.; Arendorf, J.; Castella, T.; Thompson, P.; Willis, P.; Tercero Espinoza, L.; Klug, S.; Wichmann, E. Study on Critical Raw Materials at EU Level. In Final Report Prepared by Oakdene Hollins and Fraunhofer ISI for EU COM DG Enterprise and Industry; Oakdene Hollins and Fraunhofer ISI: Aylesbury, UK, 2013. [Google Scholar]
- Glöser, S.; Tercero, L.; Gandenberger, C.M. Raw material criticality in the context of classical risk assessment. Resour. Policy 2015, 44, 35–46. [Google Scholar] [CrossRef]
- Helbig, C.; Wietschel, L.; Thorenz, A.; Tuma, A. How to Evaluate Raw Material Vulnerability—An Overview. Resour. Policy. 2016, 48, 13–24. [Google Scholar] [CrossRef]
- Zhang, F.-S.; Yamasaki, S.-I.; Nanzyo, M. Application of waste ashes to agricultural land—Effect of incineration temperature on chemical characteristics. Sci. Total Environ. 2001, 264, 205–214. [Google Scholar] [CrossRef]
- Binnemans, K.; Jones, P.T.; Blanpain, B.; Van Gerven, T.; Yang, Y.; Walton, A.; Buchert, M. Recycling of rare earths: A critical review. J. Clean Prod. 2013, 51, 1–22. [Google Scholar] [CrossRef]
- Mueller, S.R.; Wäger, P.A.; Widmer, R.; Williams, I.D. A geological reconnaissance of electrical and electronic waste as a source for rare earth metals. Waste Manag. 2015, 45, 226–234. [Google Scholar] [CrossRef]
- Brunner, P.H. Urban Mining A Contribution to Reindustrializing the City. J. Ind. Ecol. 2011, 15, 339–341. [Google Scholar] [CrossRef]
- Chandler, A.J.; Eighmy, T.T.; Hartlén, J.; Hjelmar, O.; Kosson, D.S.; Sawell, S.E.; van der Sloot, H.A.; Vehlow, J. Municipal Solid Waste Incineration Residues. In Studies in Environmental Science; Elsevier Science: Amsterdam, The Netherlands, 1997; Volume 67, ISBN 978-0-444-82563-6. [Google Scholar]
- Lynn, C.J.; Ghataora, G.S.; Dhir, O.B.E.R.K. Municipal incinerated bottom ash (MIBA) characteristics and potential for use in road pavements. Int. J. Pavement Res. Technol. 2017, 10, 185–201. [Google Scholar] [CrossRef]
- Kasina, M.; Wendorff-Belon, M.; Kowalski, P.R.; Michalik, M. Characterization of incineration residues from wastewater treatment plant in Polish city: A future waste based source of valuable elements? J. Mater. Cycles Waste Manag. 2019, 21, 885–896. [Google Scholar] [CrossRef]
- Cyr, M.; Coutand, M.; Clastres, P. Technological and environmental behaviour of sewage sludge ash (SSA) in cement-based materials. Cem. Concr. Res. 2007, 37, 1278–1289. [Google Scholar] [CrossRef]
- Alba, N.; Gasso, S.; Lacorte, T.; Baldasano, J.M. Characterization of municipal solid waste incineration residues from facilities with different air pollution control systems. J. Air Waste Manag. Assoc. 1997, 47, 1170–1179. [Google Scholar] [CrossRef]
- Shi, H.S.; Kan, L.L. Leaching behavior of heavy metals from municipal solid wastes incineration (MSWI) fly ash used in concrete. J. Hazard Mater. 2009, 164, 750–754. [Google Scholar] [CrossRef]
- Cheng, K.-L.; Hsu, S.-C.; Li, W.-M.; Ma, H.-W. Quantifying potential anthropogenic resources of buildings through hot spot analysis. Resour. Conserv. Recycl. 2018, 133, 10–20. [Google Scholar] [CrossRef]
- Wedepohl, K.H. The composition of the continental crust. Geochim. Cosmochim. Acta 1995, 59, 1217–1232. [Google Scholar] [CrossRef]
- Faure, G. Principles and Applications of Geochemistry; Prentice Hall: Pearson, NJ, USA, 1998; ISBN 0023364505. [Google Scholar]
- Rudnick, R.L.; Gao, S. The Composition of the Continental Crust; Holland, H.D., Turekian, K.K., Eds.; Elsevier-Pergamon: Oxford, UK, 2003; Volume 3, pp. 1–64. [Google Scholar]
- Laznicka, P. Giant Metallic Deposits; Springer-Verlag: Berlin Heidelberg, Germany, 2006; eBook; ISBN 978-3-540-33092-9. [Google Scholar] [CrossRef]
- Allegrini, E.; Maresca, A.; Olsson, M.E.; Holtze, M.S.; Boldrin, A.; Astrup, T.F. Quantification of the resource recovery potential of municipal solid waste incineration bottom ashes. Waste Manag. 2014, 34, 1627–1636. [Google Scholar] [CrossRef]
- Kowalski, P.R.; Kasina, M.; Michalik, M. Metallic Elements Fractionation in Municipal Solid Waste Incineration Residues. Energy Procedia 2016, 97, 31–36. [Google Scholar] [CrossRef]
- Hughes, C.J. Igneous Petrology, v. 7, 1st ed.; Elsevier: Amsterdam, The Netherlands, 2003; p. 96. ISBN1 9780444420114. ISBN2 9781483289694. [Google Scholar]
- Jaiswal, S.; Das, M.; Maiti, S.K. Environmental impact of flyash and its management: An overview. Indian J. Environ. Prot. 2006, 26, 68–275. [Google Scholar]
- Youcai, Z.; Lijie, S.; Guojian, L. Chemical stabilization of MSW incinerator fly ashes. J. Hazard Mater. 2002, 95, 47–63. [Google Scholar] [CrossRef]
- Colling, D.; Vasilos, T. Industrial Materials: Vol. 1, Metals and Alloys; Prentice Hall: Englewood Cliffs, NJ, USA, 1995. [Google Scholar]
- Preiss, M.K. Method Development to Recover Zinc from Fly Ash Originating from Municipal Solid Waste Incineration in Sweden. Master’s Thesis, University of Borås, Borås, Sweden, 2013. [Google Scholar]
- Funari, V.; Mäkinen, J.; Salminen, J.; Braga, R.; Dnelli, E.; Revitzer, H. Metal removal from Municipal Solid Waste Incineration fly ash: A comparison between chemical leaching and bioleaching. Waste Manag. 2017, 60, 397–406. [Google Scholar] [CrossRef]
- Ahmaruzzaman, M. A review on the utilization of fly ash. Prog. Energy Combust. Sci. 2010, 36, 327–363. [Google Scholar] [CrossRef]
- Van de Velden, M.; Dewil, R.; Baeyens, J.; Josson, L.; Lanssens, P. The distribution of heavy metals during fluidized bed combustion of sludge (FBSC). J Hazard Mater. 2008, 151, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Rozporządzenie Ministra Rolnictwa i Rozwoju Wsi z dnia 18 czerwca 2018 r. w sprawie wykonania niektórych przepisów ustawy o nawozach i nawożeniu. Dziennik Ustaw Rzeczypospolitej Polskiej nr 2008, 119, Poz. 765. (In Polih)
- Querol, X.; Moreno, N.; Umana, J.C.; Alastuey, A.; Hernandez, E.; Lopez-Soler, A.; Plana, F. Synthesis of zeolites from coal fly ash: An overview. Int. J. Coal Geol. 2002, 50, 413–423. [Google Scholar] [CrossRef]
- Bhattacharjee, U.; Kandpal, T.C. Potential of fly utilization in India. Energy 2002, 27, 151–166. [Google Scholar] [CrossRef]





| FA1 (n = 6) | FA2 (n = 4) | FA3 (n = 10) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Min. Content | Max. Content | Average | Min. Content | Max. Content | Average | Min. Content | Max. Content | Average | |
| % | |||||||||
| SiO2 | 35.04 | 40.56 | 37.65 | 15.99 | 20.75 | 17.99 | 29.42 | 32.73 | 31.64 |
| Al2O3 | 7.41 | 8.76 | 8.26 | 7.29 | 8.07 | 7.72 | 7.31 | 10.46 | 8.35 |
| Fe2O3 | 12.46 | 15.85 | 14.18 | 1.24 | 2.02 | 1.70 | 2.35 | 4.83 | 3.09 |
| MgO | 3.35 | 3.78 | 3.56 | 2.21 | 2.37 | 2.27 | 2.05 | 2.14 | 2.10 |
| CaO | 9.91 | 13.45 | 11.70 | 35.15 | 39.44 | 37.23 | 27.42 | 28.86 | 27.95 |
| Na2O | 0.63 | 0.74 | 0.69 | 3.21 | 4.47 | 3.93 | 2.51 | 3.31 | 2.86 |
| K2O | 1.72 | 1.93 | 1.86 | 2.54 | 3.91 | 3.38 | 2.40 | 2.81 | 2.56 |
| TiO2 | 0.85 | 0.98 | 0.94 | 1.80 | 1.97 | 1.92 | 1.34 | 1.85 | 1.57 |
| P2O5 | 16.10 | 17.92 | 17.20 | 1.39 | 1.50 | 1.45 | 1.23 | 1.58 | 1.39 |
| MnO | 0.10 | 0.12 | 0.11 | 0.08 | 0.10 | 0.08 | 0.08 | 0.11 | 0.10 |
| LOI | 2.30 | 6.00 | 3.28 | 5.00 | 10.80 | 7.93 | 9.20 | 17.08 | 14.87 |
| Ctot | 0.12 | 0.19 | 0.14 | 24.60 | 43.30 | 32.27 | 26.68 | 35.43 | 30.30 |
| Stot | 0.61 | 0.79 | 0.69 | 194.00 | 339.60 | 318.50 | 112.30 | 169.63 | 136.65 |
| mg kg−1 | |||||||||
| Ag | 12.00 | 14.40 | 12.97 | 846.00 | 1039.00 | 913.67 | 932.00 | 1108.00 | 1047.56 |
| As | 11.60 | 16.20 | 14.23 | 1.00 | 1.00 | 1.00 | 1.00 | 3.00 | 1.94 |
| Au* | 0.638 | 0.906 | 0.774 | 0.020 | 0.039 | 0.028 | 0.020 | 0.047 | 0.031 |
| Ba | 1015.00 | 1446.00 | 1186.83 | 1.20 | 2.70 | 2.00 | 2.50 | 2.70 | 2.65 |
| Be | 1.00 | 3.00 | 2.00 | 4.10 | 8.10 | 5.90 | 8.10 | 9.50 | 8.63 |
| Bi | 7.20 | 10.50 | 9.17 | 3.50 | 4.10 | 3.83 | 4.65 | 6.00 | 5.17 |
| Cd | 4.80 | 7.00 | 5.97 | 10.80 | 11.60 | 11.20 | 10.20 | 11.70 | 11.06 |
| Ce | 39.00 | 44.40 | 42.65 | 32.30 | 50.40 | 43.93 | 32.00 | 38.55 | 35.41 |
| Co | 26.00 | 41.50 | 30.40 | 398.00 | 595.00 | 515.33 | 169.00 | 266.75 | 226.31 |
| Cr | 770.00 | 1530.00 | 1040.00 | 570.00 | 700.00 | 626.67 | 56.50 | 69.00 | 62.88 |
| Cs | 4.10 | 6.90 | 5.15 | 466.30 | 550.70 | 498.57 | 349.00 | 422.18 | 393.54 |
| Cu | 570.30 | 666.60 | 632.58 | 1.70 | 2.70 | 2.03 | 0.90 | 1.23 | 1.08 |
| Dy | 2.45 | 2.97 | 2.74 | 3.70 | 4.50 | 4.13 | 4.60 | 4.85 | 4.73 |
| Er | 1.57 | 1.90 | 1.70 | 1.40 | 1.60 | 1.50 | 1.80 | 1.90 | 1.84 |
| Eu | 0.61 | 0.73 | 0.66 | 21.00 | 30.00 | 25.67 | 39.25 | 51.00 | 43.31 |
| Ga | 7.40 | 8.90 | 8.20 | 5.20 | 73.00 | 34.33 | 6.80 | 134.08 | 65.19 |
| Gd | 3.03 | 3.68 | 3.28 | 140.50 | 161.00 | 153.17 | 182.30 | 249.00 | 205.40 |
| Hf | 6.40 | 8.60 | 7.35 | 11.80 | 12.30 | 11.27 | 10.50 | 13.60 | 12.41 |
| Hg | 0.01 | 0.06 | 0.05 | 14.60 | 17.00 | 15.93 | 16.40 | 17.43 | 16.95 |
| Ho | 0.51 | 0.64 | 0.57 | 25.80 | 30.80 | 27.90 | 29.08 | 30.38 | 30.16 |
| La | 19.00 | 22.90 | 21.58 | 2.68 | 2.88 | 2.77 | 2.79 | 3.45 | 3.10 |
| Lu | 0.23 | 0.28 | 0.26 | 9.50 | 10.40 | 10.07 | 10.10 | 12.33 | 11.20 |
| Mo | 18.30 | 25.50 | 21.72 | 1.33 | 1.55 | 1.47 | 1.64 | 2.14 | 1.95 |
| Nb | 8.50 | 10.70 | 9.65 | 0.52 | 0.64 | 0.57 | 0.46 | 0.70 | 0.59 |
| Nd | 15.30 | 18.60 | 16.73 | 1.34 | 1.58 | 1.51 | 1.68 | 2.17 | 1.92 |
| Ni | 79.30 | 119.50 | 103.97 | 0.30 | 0.41 | 0.37 | 0.30 | 0.43 | 0.37 |
| Pb | 125.90 | 151.60 | 138.78 | 1.15 | 1.32 | 1.19 | 1.32 | 1.83 | 1.64 |
| Pr | 4.03 | 4.88 | 4.42 | 0.19 | 0.23 | 0.22 | 0.24 | 0.37 | 0.31 |
| Rb | 50.10 | 57.80 | 54.03 | 0.64 | 0.77 | 0.65 | 0.90 | 1.16 | 1.00 |
| Sb | 5.90 | 7.80 | 6.82 | 0.04 | 0.10 | 0.08 | 0.11 | 0.15 | 0.13 |
| Sc | 6.00 | 6.00 | 5.83 | 0.64 | 0.71 | 0.64 | 0.74 | 1.01 | 0.91 |
| Se | 2.40 | 8.50 | 5.62 | 0.05 | 0.11 | 0.08 | 0.11 | 0.16 | 0.13 |
| Sm | 2.83 | 3.51 | 3.21 | 0.22 | 1.38 | 0.78 | 1.65 | 2.57 | 2.14 |
| Sn | 147.00 | 473.00 | 333.00 | 5.16 | 6.47 | 5.75 | 2.16 | 3.93 | 2.93 |
| Sr | 491.70 | 548.10 | 519.82 | 11.30 | 14.40 | 11.67 | 9.80 | 12.80 | 10.71 |
| Ta | 0.60 | 0.70 | 0.67 | 258.70 | 300.90 | 284.20 | 362.98 | 407.10 | 388.72 |
| Tb | 0.45 | 0.51 | 0.48 | 119.70 | 461.60 | 227.73 | 404.13 | 1423.50 | 762.27 |
| Th | 5.70 | 6.60 | 6.10 | 5002.00 | 8067.00 | 5698.00 | 3729.25 | 4489.00 | 4198.06 |
| Tl | 0.70 | 1.10 | 0.88 | 36.90 | 58.20 | 45.20 | 47.90 | 62.83 | 54.76 |
| Tm | 0.22 | 0.28 | 0.25 | 7.90 | 19.30 | 10.80 | 8.80 | 10.65 | 9.74 |
| U | 7.70 | 9.40 | 8.63 | 13.60 | 21.10 | 16.73 | 7.20 | 9.65 | 8.02 |
| V | 62.00 | 89.00 | 71.67 | 10.00 | 13.60 | 11.67 | 5.30 | 8.60 | 7.16 |
| W | 7.50 | 137.20 | 50.98 | 64.10 | 134.60 | 93.27 | 11.00 | 45.05 | 30.44 |
| Y | 15.20 | 18.80 | 16.95 | 0.01 | 0.06 | 0.04 | 0.06 | 3.39 | 1.99 |
| Yb | 1.60 | 1.88 | 1.75 | 0.10 | 0.10 | 0.10 | 0.10 | 0.35 | 0.23 |
| Zn | 3550.00 | 4472.00 | 3975.83 | 1.50 | 4.20 | 2.60 | 1.90 | 3.05 | 2.59 |
| Zr | 246.60 | 344.70 | 290.87 | 2.00 | 3.00 | 2.00 | 3.00 | 3.75 | 3.19 |
| SiO2 | Al2O3 | Fe2O3 | MgO | CaO | Na2O | K2O | TiO2 | P2O5 | MnO | |
|---|---|---|---|---|---|---|---|---|---|---|
| FA 1 | 37.46 | 8.02 | 14.32 | 3.66 | 12.09 | 0.70 | 1.83 | 0.93 | 17.18 | 0.11 |
| FA 2 | 18.23 | 7.53 | 1.58 | 2.31 | 36.64 | 4.09 | 3.74 | 1.86 | 1.44 | 0.09 |
| FA 3 | 30.92 | 7.32 | 3.80 | 2.10 | 28.34 | 2.55 | 2.49 | 1.40 | 1.25 | 0.10 |
| Ultramafic rocks | 42.36 | 2.27 | 13.78 | 38.47 | 2.24 | 0.66 | 0.02 | 0.05 | 0.04 | 0.20 |
| Basalt | 50.27 | 15.65 | 12.30 | 7.54 | 10.07 | 2.52 | 1.00 | 1.90 | 0.26 | 0.23 |
| High-Ca granites | 67.17 | 15.49 | 4.23 | 1.56 | 3.54 | 3.83 | 3.04 | 0.57 | 0.21 | 0.07 |
| Low-Ca granites | 74.23 | 13.60 | 2.03 | 0.27 | 0.71 | 3.48 | 5.06 | 0.20 | 0.14 | 0.05 |
| Continental crust | 66.62 | 15.40 | 5.04 | 2.48 | 3.59 | 3.27 | 2.80 | 0.64 | 0.15 | 0.10 |
| Tonalites | 61.90 | 16.30 | 6.30 | 2.60 | 4.90 | 3.90 | 1.90 | 0.77 | 0.26 | 0.09 |
| Sandstone | 78.72 | 4.72 | 1.40 | 1.16 | 5.47 | 0.44 | 1.29 | 0.25 | 0.04 | 0.00 |
| Greywackes | 69.10 | 13.50 | 5.90 | 2.30 | 2.60 | 3.00 | 2.00 | 0.72 | 0.13 | 0.10 |
| Shale | 15.62 | 15.12 | 6.75 | 2.49 | 3.09 | 1.29 | 3.20 | 0.77 | 0.16 | 0.11 |
| Carbonate rocks | 5.13 | 0.79 | 0.47 | 7.79 | 42.30 | 0.05 | 0.33 | 0.07 | 0.09 | 0.14 |
| Deep-sea clay | 53.48 | 15.87 | 9.29 | 3.48 | 4.06 | 5.39 | 3.01 | 0.77 | 0.34 | 0.87 |
| Ag | As | Au | Ba | Be | Cd | Co | Cr | Cu | Hg | Ga | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| mg kg−1 | mg kg−1 | µg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mgkg−1 | mg kg−1 | |
| FA 1 | 12.85 | 13.98 | 737.58 | 1262.25 | 1.75 | 5.63 | 32.35 | 1200.00 | 639.30 | 0.03 | 8.05 |
| FA 2 | 12.87 | 14.60 | 74.27 | 978.00 | 28.27 | 38.50 | 1.00 | 910.00 | 286.23 | 0.04 | 6.23 |
| FA 3 | 8.26 | 10.24 | 38.93 | 1075.13 | 39.84 | 31.05 | 2.00 | 887.00 | 374.34 | 3.71 | 8.45 |
| Ultramafic | 0.05 | 0.80 | 6.00 | 0.70 | 0.20 | 0.05 | 175.00 | 1800.00 | 15.00 | 0.01 | 1.80 |
| Basalt | 0.11 | 2.20 | 4.00 | 315.00 | 0.70 | 0.21 | 47.00 | 185.00 | 94.00 | 0.09 | 18.00 |
| High-Ca granites | 0.05 | 1.90 | 4.00 | 420.00 | 2.00 | 0.13 | 7.00 | 22.00 | 30.00 | 0.08 | 17.00 |
| Low-Ca granites | 0.04 | 1.50 | 4.00 | 840.00 | 3.00 | 0.13 | 1.00 | 4.10 | 10.00 | 0.08 | 17.00 |
| Continental crust | 53.00 | 4.80 | 1.50 | 624.00 | 2.10 | 0.09 | 17.30 | 92.00 | 28.00 | 0.05 | 17.50 |
| Tonalites | n.d | n.d | n.d | 608.00 | n.d | n.d | 16.00 | 38.00 | 19.00 | n.d | 19.00 |
| Sandstone | 0.01 | 1.00 | n.d | 10.00 | n.d | n.d | 0.30 | 35.00 | n.d | 0.03 | 12.00 |
| Greywackes | n.d | n.d | 4.80 | 426.00 | n.d | n.d | 15.00 | 88.00 | 24.00 | n.d | 16.00 |
| Shale | 0.07 | 13.00 | n.d | 580.00 | 3.00 | 0.30 | 19.00 | 90.00 | 45.00 | 0.40 | 19.00 |
| Carbonate rocks | 0.01 | 1.00 | n.d | 10.00 | n.d | 0.04 | 0.10 | 11.00 | 4.00 | 0.04 | 4.00 |
| Deep-sea clay | 0.11 | 13.00 | n.d | 2300.00 | 2.60 | 0.42 | 74.00 | 90.00 | 250.00 | 0.10 | 20.00 |
| currently exploited ores | 700.00 | 1000.00 | 5000.00 | 590.00 | 300.00 | 1000.00 | 500.00 | 310.00 | 5000.00 | 400.00 | 100.00 |
| Mo | Nb | Ni | Pb | Sb | Se | Sn | Ta | Tl | V | Zn | |
| mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | |
| FA 1 | 21.48 | 7.93 | 97.00 | 135.73 | 6.93 | 5.68 | 340.75 | 0.73 | 0.80 | 74.50 | 3969.50 |
| FA 2 | 12.70 | 11.27 | 41.27 | 341.70 | 287.53 | 3.50 | 561.33 | 2.23 | 0.10 | 26.33 | 6669.67 |
| FA 3 | 11.58 | 11.18 | 58.96 | 472.24 | 151.10 | 4.38 | 255.63 | 1.16 | 0.38 | 45.13 | 4038.13 |
| Ultramafic | 0.30 | 9.00 | 2000.00 | 0.50 | 0.10 | 0.05 | 0.50 | 0.50 | 0.04 | 40.00 | 40.00 |
| Basalt | 1.50 | 20.00 | 145.00 | 7.00 | 0.60 | 0.05 | 1.50 | 0.80 | 0.21 | 225.00 | 118.00 |
| High-Ca granites | 1.00 | 20.00 | 15.00 | 15.00 | 0.20 | 0.05 | 1.50 | 3.60 | 0.72 | 88.00 | 60.00 |
| Low-Ca granites | 1.30 | 21.00 | 4.50 | 19.00 | 0.20 | 0.05 | 3.00 | 4.20 | 2.30 | 44.00 | 39.00 |
| Continental crust | 1.10 | 12.00 | 47.00 | 17.00 | 0.40 | 0.09 | 2.10 | 0.90 | 0.90 | 97.00 | 67.00 |
| Tonalites | n.d | 8.80 | 19.00 | 14.20 | n.d | n.d | 2.40 | 1.10 | n.d | 103.00 | 61.00 |
| Sandstone | 0.20 | n.d | 2.00 | 7.00 | 0.01 | 0.05 | 0.10 | 0.01 | 0.82 | 20.00 | 16.00 |
| Greywackes | n.d | 8.40 | 24.00 | 14.20 | n.d | n.d | n.d | n.d | n.d | 98.00 | 76.00 |
| Shale | 2.60 | 11.00 | 68.00 | 20.00 | 1.50 | 0.60 | 6.00 | 0.80 | 1.40 | 130.00 | 95.00 |
| Carbonate rocks | 0.40 | 0.30 | 20.00 | 9.00 | 0.20 | 0.08 | 0.10 | 0.01 | 0.01 | 20.00 | 20.00 |
| Deep-sea clay | 27.00 | 14.00 | 225.00 | 80.00 | 1.00 | 0.17 | 1.50 | 0.10 | 0.80 | 120.00 | 225.00 |
| currently exploited ores | 10,000.00 | 4100.00 | 15,000.00 | 300,000.00 | 27.00 | 20,000.00 | 4000.00 | 410.00 | 10,000.00 | 500.00 | 50,000.00 |
| Sc | Y | La | Ce | Pr | Nd | Sm | Eu | |
|---|---|---|---|---|---|---|---|---|
| mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | |
| FA 1 | 5.75 | 16.53 | 20.95 | 42.20 | 4.41 | 16.63 | 3.19 | 0.65 |
| FA 2 | 2.33 | 7.65 | 15.47 | 28.87 | 2.82 | 10.13 | 1.71 | 0.61 |
| FA 3 | 3.40 | 13.56 | 17.10 | 29.73 | 3.34 | 11.90 | 2.11 | 0.67 |
| Ultramafic | 10.00 | n.d | 1.30 | 3.50 | 0.49 | 1.90 | 0.42 | 0.14 |
| Basalt | 27.00 | 21.00 | 6.10 | 16.00 | 2.70 | 14.00 | 4.30 | 1.50 |
| High-Ca granites | 14.00 | 335.00 | 45.00 | 81.00 | 7.70 | 33.00 | 8.80 | 1.40 |
| Low-Ca granites | 7.00 | 40.00 | 55.00 | 92.00 | 8.80 | 37.00 | 10.00 | 1.60 |
| Continental crust | 14.00 | 21.00 | 31.00 | 63.00 | 7.10 | 27.00 | 4.70 | 1.00 |
| Tonalites | 11.00 | 22.00 | 23.00 | 53.00 | 8.50 | 25.00 | 4.90 | 1.40 |
| Sandstone | 1.00 | 40.00 | 30.00 | 92.00 | 8.80 | 37.00 | 10.00 | 1.60 |
| Greywackes | 16.00 | 26.00 | 34.00 | 58.00 | 6.10 | 25.00 | 4.60 | 1.20 |
| Shale | 13.00 | 26.00 | 92.00 | 59.00 | 5.60 | 24.00 | 6.40 | 1.00 |
| Carbonate rocks | 1.00 | 30.00 | 1.00 | 11.50 | 1.10 | 4.70 | 1.30 | 0.20 |
| Deep-sea clay | 19.00 | 90.00 | 115.00 | 345.00 | 33.00 | 140.00 | 38.00 | 6.00 |
| currently exploited ores | 20.00 | 240,000.00 | 2600.00 | 18.00 | 1600.00 | 6000.00 | 690.00 | 220.00 |
| Gd | Tb | Dy | Ho | Er | Tm | Yb | Lu | |
| mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | mg kg−1 | |
| FA 1 | 3.25 | 0.47 | 2.65 | 0.56 | 1.68 | 0.24 | 1.73 | 0.25 |
| FA 2 | 1.46 | 0.41 | 1.23 | 0.22 | 0.70 | 0.10 | 0.67 | 0.10 |
| FA 3 | 2.12 | 0.42 | 1.79 | 0.36 | 1.10 | 0.15 | 0.98 | 0.15 |
| Ultramafic rocks | 0.54 | 0.12 | 0.77 | 0.12 | 0.30 | 0.04 | 0.38 | 0.04 |
| Basalt | 6.20 | 1.10 | 5.90 | 1.40 | 3.60 | 0.60 | 3.20 | 0.55 |
| High-Ca granites | 8.80 | 1.40 | 6.30 | 1.80 | 3.50 | 0.30 | 3.50 | 1.10 |
| Low-Ca granites | 10.00 | 1.60 | 7.20 | 2.00 | 4.00 | 0.30 | 4.00 | 1.20 |
| Continental crust | 4.00 | 0.70 | 3.90 | 0.83 | 2.30 | 0.30 | 2.00 | 0.31 |
| Tonalites | 4.20 | 0.75 | 3.50 | 0.90 | 1.90 | n.d | 2.40 | 0.37 |
| Sandstone | 10.00 | 1.60 | 7.20 | 2.00 | 4.00 | 0.30 | 4.00 | 1.20 |
| Greywackes | 4.00 | 0.63 | 3.40 | 0.78 | 2.20 | n.d | 2.10 | 0.37 |
| Shale | 6.40 | 1.00 | 4.60 | 1.20 | 2.50 | 0.20 | 2.60 | 0.70 |
| Carbonate rocks | 1.30 | 0.20 | 0.90 | 0.30 | 0.50 | 0.04 | 0.50 | 0.20 |
| Deep-sea clay | 38.00 | 6.00 | 27.00 | 7.50 | 15.00 | 1.20 | 15.00 | 4.50 |
| currently exploited ores | 650.00 | 43.00 | 430.00 | 16.00 | 13.00 | 3.00 | 3.00 | 2200.00 |
| EU Dirrective [36] | Solid Mineral Fertilisers [37] | Solid Organic–Mineral Fertilisers [37] | |
|---|---|---|---|
| mg/kg Dry Mass | |||
| As | n.d | 50 | n.d |
| Cr | 800 | n.d | 100 |
| Cu | 800 | nd. | n.d |
| Pb | 500 | 50 | 5 |
| Ni | 200 | n.d | 60 |
| Zn | 2000 | n.d | n.d |
| Cd | 5 | 140 | 140 |
| Hg | 5 | 2 | 2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kasina, M.; Kowalski, P.R.; Kajdas, B.; Michalik, M. Assessment of Valuable and Critical Elements Recovery Potential in Ashes from Processes of Solid Municipal Waste and Sewage Sludge Thermal Treatment. Resources 2020, 9, 131. https://doi.org/10.3390/resources9110131
Kasina M, Kowalski PR, Kajdas B, Michalik M. Assessment of Valuable and Critical Elements Recovery Potential in Ashes from Processes of Solid Municipal Waste and Sewage Sludge Thermal Treatment. Resources. 2020; 9(11):131. https://doi.org/10.3390/resources9110131
Chicago/Turabian StyleKasina, Monika, Piotr Rafał Kowalski, Bartłomiej Kajdas, and Marek Michalik. 2020. "Assessment of Valuable and Critical Elements Recovery Potential in Ashes from Processes of Solid Municipal Waste and Sewage Sludge Thermal Treatment" Resources 9, no. 11: 131. https://doi.org/10.3390/resources9110131
APA StyleKasina, M., Kowalski, P. R., Kajdas, B., & Michalik, M. (2020). Assessment of Valuable and Critical Elements Recovery Potential in Ashes from Processes of Solid Municipal Waste and Sewage Sludge Thermal Treatment. Resources, 9(11), 131. https://doi.org/10.3390/resources9110131





