Methadone for Analgesia in Children with Life-Limiting Illness: Experience from a Tertiary Children’s Health Service
Abstract
:1. Introduction
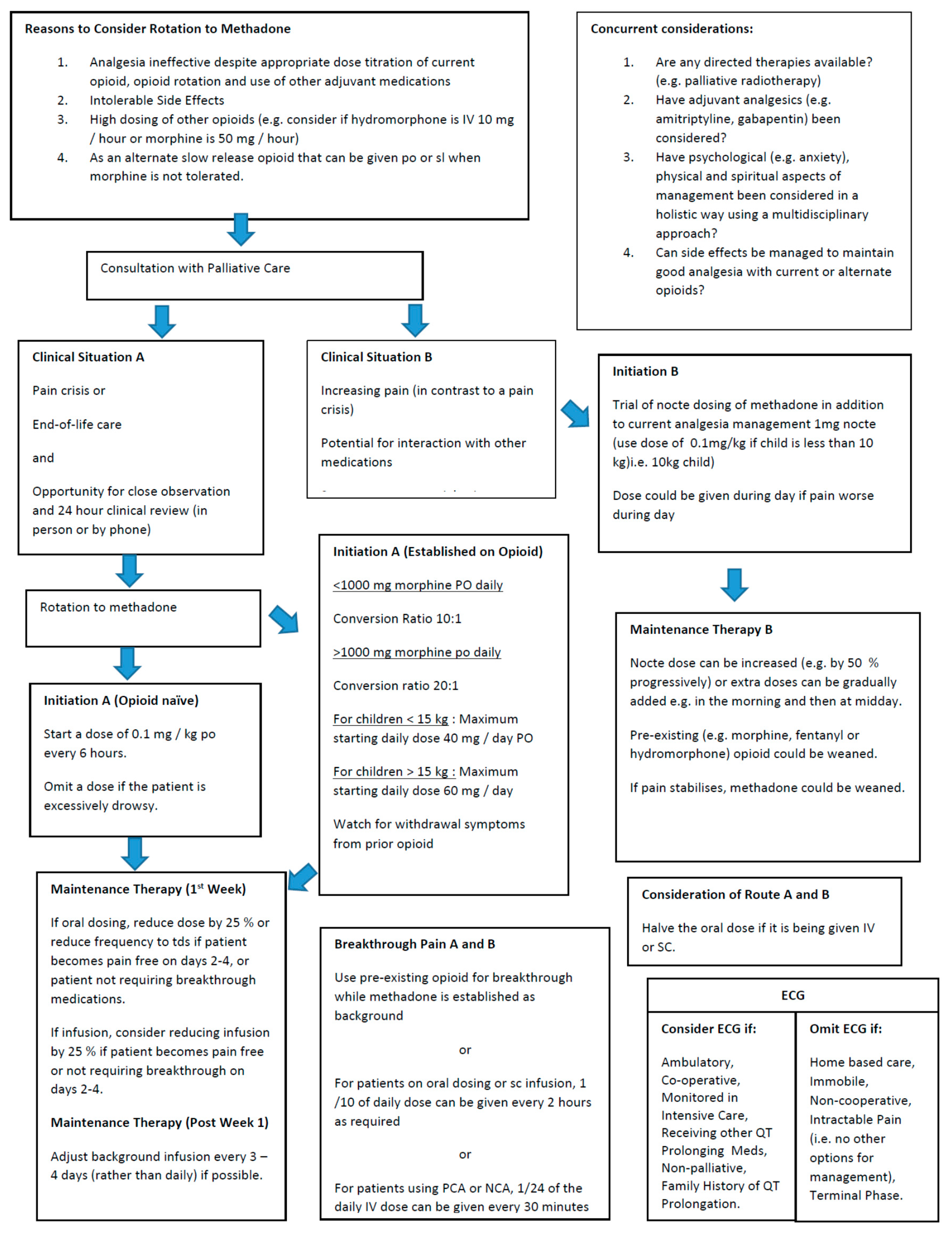
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ashby, M.; Fleming, B.; Wood, M.; Somoqyi, A. Plasma morphine and glucuronide (M3G and M6G) concentrations in hospice inpatients. J. Pain Symptom Manag. 1997, 14, 157–167. [Google Scholar] [CrossRef]
- Chhabra, S.; Bull, J. Methadone. Am. J. Hosp. Palliat. Care 2008, 25, 146–150. [Google Scholar] [CrossRef] [PubMed]
- Williams, P.I.; Sarginson, R.E.; Ratcliffe, J.M. Use of methadone in the morphine-tolerant burned paediatric patient. Br. J. Anaesth. 1998, 80, 92–95. [Google Scholar] [CrossRef] [PubMed]
- Mao, J. NMDA and opioid receptors: Their interactions in antinociception, tolerance and neuroplasticity. Brain Res. Brain Res. Rev. 1999, 30, 289–304. [Google Scholar] [CrossRef]
- Moulin, D.E.; Clark, A.J.; Gilron, I.; Ware, M.A.; Watson, C.P.; Sessle, B.J.; Coderre, T.; Morley-Forster, P.K.; Stinson, J.; Boulanger, A.; et al. Pharmacological Management of Chronic Neuropathic Pain: Consensus Statement and Guidelines from the Canadian Pain Society. Pain Res. Manag. 2007, 12, 13–21. [Google Scholar] [CrossRef] [PubMed]
- Barbosa Neto, J.O.; Garcia, M.A.; Garcia, J.B.S. Revisiting methadone: Pharmacokinetics, pharmacodynamics and clinical indication. Rev. Dor 2015, 16, 60–66. [Google Scholar] [CrossRef]
- Ward, R.M.; Drover, D.R.; Hammer, G.B.; Stemland, C.J.; Kern, S.; Tristani-Firouzi, M.; Lugo, R.A.; Satterfield, K.; Anderson, B.J. The pharmacokinetics of methadone and its metabolites in neonates, infants, and children. Paediatr. Anaesth. 2014, 24, 591–601. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharma, A.; Tallchief, D.; Blood, J.; Kim, T.; London, A.; Kharasch, E.D. Perioperative pharmacokinetics of methadone in adolescents. Anesthesiology 2011, 115, 1153–1161. [Google Scholar] [CrossRef] [PubMed]
- Horst, J.; Frei-Jones, M.; Deych, E.; Shannon, W.; Kharasch, E.D. Pharmacokinetics and analgesic effects of methadone in children and adults with sickle cell disease. Pediatr. Blood Cancer 2016, 63, 2123–2130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Verebely, K.; Volavka, J.; Mulé, S.; Resnick, R. Methadone in man: Pharmacokinetic and excretion studies in acute and chronic treatment. Clin. Pharmacol. Ther. 1975, 18, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, V.F.; Lundberg, V.; Jespersen, T.W.; Hasle, H. Extreme doses of intravenous methadone for severe pain in two children with cancer. Pediatr. Blood Cancer 2015, 62, 1087–1090. [Google Scholar] [CrossRef] [PubMed]
- Davies, D.; DeVlaming, D.; Haines, C. Methadone analgesia for children with advanced cancer. Pediatr. Blood Cancer 2008, 51, 393–397. [Google Scholar] [CrossRef] [PubMed]
- McCaffery, M.; Pasero, C. The merits of methadone. Am. J. Nurs. 2000, 100, 22–23. [Google Scholar] [CrossRef] [PubMed]
- Sirkiä, K.; Hovi, L.; Pouttu, J.; Saarinen-Pihkala, U. Pain Medication During Terminal Care of Children with Cancer. J. Pain Symptom Manag. 1998, 15, 220–226. [Google Scholar] [CrossRef]
- Shir, Y.; Shenkman, Z.; Shavelson, V.; Davidson, E.M.; Rosen, G. Oral methadone for the treatment of severe pain in hospitalized children: A report of five cases. Clin. J. Pain 1998, 14, 350–353. [Google Scholar] [CrossRef] [PubMed]
- Anghelescu, D.L.; Faughnan, L.G.; Hankins, G.M.; Ward, D.A.; Oakes, L.L. Methadone use in children and young adults at a cancer center: A retrospective study. J. Opioid Manag. 2011, 7, 353–361. [Google Scholar] [CrossRef] [PubMed]
- Hamunen, K. Ventilatory effects of morphine, pethidine and methadone in children. Br. J. Anaesth. 1993, 70, 414–418. [Google Scholar] [CrossRef] [PubMed]
- Berde, C.B.; Beyer, J.E.; Bournaki, M.C.; Levin, C.R.; Sethna, N.F. Comparison of morphine and methadone for prevention of postoperative pain in 3- to 7-year-old children. J. Pediatr. 1991, 119, 136–141. [Google Scholar] [CrossRef]
- Toombs, J.D.; Kral, L.A. Methadone treatment for pain states. Am. Fam. Phys. 2005, 71, 1353–1358. [Google Scholar] [PubMed]
- Sabatowski, R.; Kasper, S.M.; Radbruch, L. Patient-controlled analgesia with intravenous L-methadone in a child with cancer pain refractory to high-dose morphine. J. Pain Symptom Manag. 2002, 23, 3–5. [Google Scholar] [CrossRef]
- Hagen, N.A.; Wasylenko, E. Methadone: Outpatient titration and monitoring strategies in cancer patients. J. Pain Symptom Manag. 1999, 18, 369–375. [Google Scholar] [CrossRef]
- McLean, S.; Twomey, F. Methods of Rotation from Another Strong Opioid to Methadone for the Management of Cancer Pain: A Systematic Review of the Available Evidence. J. Pain Symptom Manag. 2015, 50, 248–259. [Google Scholar] [CrossRef] [PubMed]
- Weschules, D.J.; Bain, K.T. A systematic review of opioid conversion ratios used with methadone for the treatment of pain. Pain Med. 2008, 9, 595–612. [Google Scholar] [CrossRef] [PubMed]
- Amos, L.B.; D’Andrea, L.A. Severe central sleep apnea in a child with leukemia on chronic methadone therapy. Pediatr. Pulmonol. 2013, 48, 85–87. [Google Scholar] [CrossRef] [PubMed]
- Anghelescu, D.L.; Patel, R.M.; Mahoney, D.P.; Trujillo, L.; Faughnan, L.G.; Steen, B.D.; Baker, J.N.; Pei, D. Methadone prolongs cardiac conduction in young patients with cancer-related pain. J. Opioid Manag. 2016, 12, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Kornick, C.A.; Kilborn, M.J.; Santiago-Palma, J.; Schulman, G.; Thaler, H.T.; Keefe, D.L.; Katchman, A.N.; Pezzullo, J.C.; Ebert, S.N.; Woosley, R.L.; et al. QTc interval prolongation associated with intravenous methadone. Pain 2003, 105, 499–506. [Google Scholar] [CrossRef]
- Boyer, E.W.; McCance-Katz, E.F.; Marcus, S. Methadone and buprenorphine toxicity in children. Am. J. Addict. 2010, 19, 89–95. [Google Scholar] [CrossRef] [PubMed]
- Irving, H.; Liebke, K.; Lockwood, L.; Noyes, M.; Pfingst, D.; Rogers, T. A Practical Guide to Palliative Care in Paediatrics; Children’s Health Queensland Hospital and Health Service: Brisbane, Australia, 2014; pp. 44–55. ISBN 978-0-9875207-0-8. Available online: https://www.caresearch.com.au/QuoCCA/Portals/6/Documents/A-Practical-guide-to-Palliative-Care-in-Paediatrics.pdf (accessed on 17 June 2018).
- Eastern Metropolitan Region Palliative Care Consortium, Opioid Conversion Ratios Guide to Palliative Care Practice: Rangeview, Australia, 2016. Available online: http://www.emrpcc.org.au/wp-content/uploads/2016/05/Opioid-Conversions-May-3-2016-final.pdf (accessed on 17 June 2018).
- Collins, J.J.; Holcombe, E.; Hannah, C.; Charles, B. Control of severe pain in children with terminal malignancy. J. Pediatr. 1995, 126, 653–657. [Google Scholar] [CrossRef]
- Yap, Y.G.; Camm, A.J. Drug induced QT prolongation and torsades de pointes. Heart 2003, 89, 1363–1372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siddappa, R.; Fletcher, J.E.; Heard, A.M.; Kielma, D.; Cimino, M.; Heard, C.M. Methadone dosage for prevention of opioid withdrawal in children. Paediatr. Anaesth. 2003, 13, 805–810. [Google Scholar] [CrossRef] [PubMed]
- Kuhlen, M.; Hoell, J.I.; Gagnon, G.; Balzer, S.; Oommen, P.T.; Borkhardt, A.; Janßen, G. Effective treatment of spasticity using dronabinol in pediatric palliative care. Eur. J. Paediatr. Neurol. 2016, 20, 898–903. [Google Scholar] [CrossRef] [PubMed]
- Fife, A.; Postier, A.; Flood, A.; Friedrichsdorf, S.J. Methadone conversion in infants and children: Retrospective cohort study of 199 pediatric inpatients. J. Opioid Manag. 2016, 12, 123–130. [Google Scholar] [CrossRef] [PubMed]
- Good, P.D.; Ravenscroft, P.J.; Cavenagh, J. Effects of opioids and sedatives on survival in an Australian inpatient palliative care population. Intern. Med. J. 2005, 35, 512–517. [Google Scholar] [CrossRef] [PubMed]
- Sathornviriyapong, A.; Nagaviroj, K.; Anothaisintawee, T. The association between different opioid doses and the survival of advanced cancer patients receiving palliative care. BMC Palliat. Care 2016, 15, 95. [Google Scholar] [CrossRef] [PubMed]
- Sykes, N.; Thorns, A. The use of opioids and sedatives at the end of life. Lancet Oncol. 2003, 4, 312–318. [Google Scholar] [CrossRef]
| Morphine | Fentanyl | Hydromorphone | |
|---|---|---|---|
| Relative Potency | 1 | 40 | 5 |
| Example Doses | 4 mg | 100 mcg | 800 mcg |
| Patient | Age | Sex | Weight (kg) | Diagnosis | Prior Opioid/Analgesia | Care Location | Goals of Care | ECG |
|---|---|---|---|---|---|---|---|---|
| 1 | 4 years | F | 19.0 | Relapsed biliary embryonal rhabdomyosarcoma | Hydromorphone | Hospital (ward) | Palliative Care | Yes |
| 2 | 2 years | M | 12.7 | Haemophagocytic lymphohistiocytosis | Hydromorphone, ketamine | Hospital (ward) | Pain Management | No |
| 3 | 8 years | F | 27.0 | Relapsed acute lymphoblastic leukaemia | Hydromorphone, ketamine | Hospital (intensive care) | Palliative Care | Yes |
| 4 | 3 years | M | 14.5 | Metastatic hepatoblastoma | Hydromorphone | Hospital (ward) | Pain Management | No |
| 5 | 5 years | M | 21.0 | Relapsed acute lymphoblastic leukaemia | Hydromorphone | Home | Palliative Care | No |
| 6 | 16 months | M | 11.0 | Meningococcal septicaemia with brain injury and four limb amputation | Morphine | Hospital (ward) | Pain Management | No |
| 7 | 12 years | M | 34.6 | Relapsed pelvic alveolar rhabdomyosarcoma | Hydromorphone | Home | Palliative Care | No |
| 8 | 9 years | F | 45.9 | Frontotemporal pleomorphic xanthoastrocytoma | Hydromorphone | Hospital (ward) | Pain Management | No |
| 9 | 16 years | M | 46.0 | Refractory graft versus host disease post bone marrow transplant for acute myeloid leukaemia | Hydromorphone | Hospital (ward) | Palliative Care | No |
| 10 | 16 years | M | 38.6 | Cerebral palsy with spastic quadriplegia, parental nutrition | Fentanyl patch | Hospital (ward) | Palliative Care | No |
| 11 | 6 years | F | 20.0 | Low grade sarcoma | Hydromorphone | Hospital (ward) | Pain Management | No |
| 12 | 5 years | F | 18.0 | Juvenile myelomonocytic leukaemia, graft versus host disease | Hydromorphone, ketamine | Hospital (intensive care) | Pain Management | Yes |
| 13 | 7 years | F | 25.8 | Epileptic encephalopathy | Oxycodone | Home | Pain Management | No |
| 14 | 7 years | F | 22.0 | Pulmonary atresia with ventricular septal defect and major aortopulmonary collateral arteries | Morphine | Home | Palliative Care | No |
| 15 | 15 years | F | 45.0 | Severe veno-occlusive disease and renal impairment post bone marrow transplant for acute lymphoblastic leukaemia | Hydromorphone | Hospital (intensive care) | Pain Management | Yes |
| 16 | 23 months | M | 15.0 | Bi-lineage leukaemia requiring bone marrow transplant | Hydromorphone | Hospital (intensive care) | Pain Management | Yes |
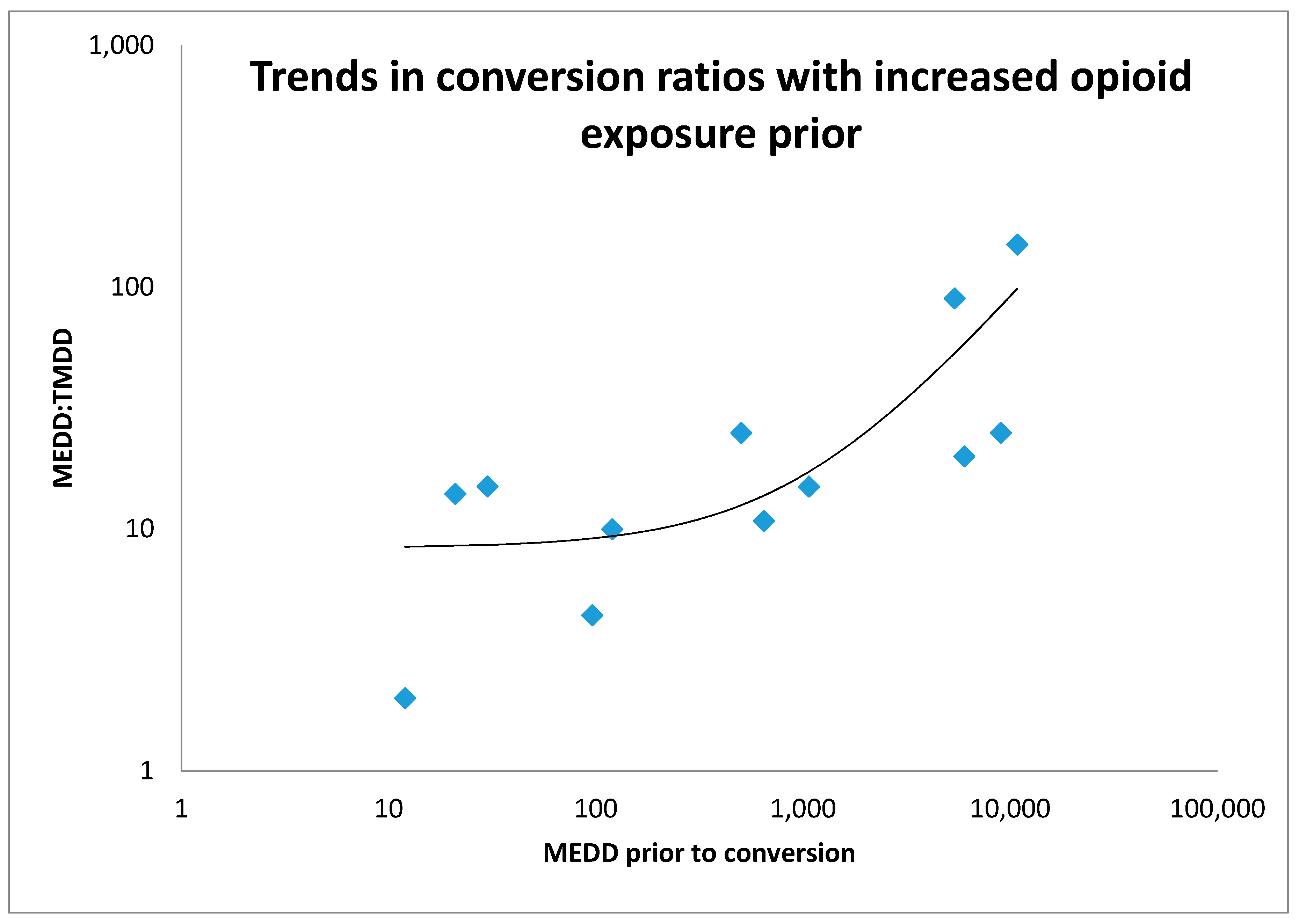
| Pt | Approx. MEDD (PO) | Methadone Starting Dose and Route | Maximal Methadone Dose and Route | Rapid Conversion or Adjuvant | Reason for Rotation to Methadone | Conversion Ratio (PO Morph: PO Meth) | Breakthrough Analgesia | Side Effects or Issues Noted | Outcome |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 504 mg | 5 mg QID PO | 15 mg PO QID. | Rapid conversion | Increasing Drowsiness and Inadequate Analgesia. | 25:1 | Hydromorph PCA IV. | Less drowsy on methadone. Able to start walking again. Withdrawal symptoms. | Died 6 months after commencing methadone. |
| Changed to 30 mg IV infusion over 24 h | Changed to meth PCA IV subsequently | ||||||||
| 2 | 1080 mg | 1 mg nocte NG | 2 mg QID SL | Adjuvant | Loss of central venous access and unable to maintain SC route. | N/A | Hydromorph PCA and ketamine IV | Drowsiness and low respiratory rate requiring doses to be skipped. | Died 3 months after commencing methadone. |
| 3 | 5400 mg | 15 mg QID PO | 240 mg IV infusion over 24 h | Rapid conversion | Inadequate Analgesia. | 90:1 | Hydromorph PCA IV.Eventually converted to meth PCA IV | Improved Analgesia. | Died one month after commencing methadone. |
| 4 | 12 mg | 1.5 mg QID PO | 3 mg TDS PO. Changed to Fentanyl patch | Rapid conversion | Side effects with morphine—inadequate analgesia and itch. Methadone only alternative slow release opioid that comes as elixir. | 2:1 | Hydromorph NCA | Unsteadiness attributed to methadone. | Died 3 months after ceasing methadone. |
| 5 | 6000 mg | 150 mg SC infusion over 24 h | 150 mg SC infusion over 24 h | Rapid conversion | Inadequate Analgesia. | 20:1 | Meth SC | Improved analgesia. | Died 7 days after commencing methadone. |
| 6 | 96 mg | 5.5 mg QID NG | 5.5 mg QID NG | Rapid conversion | Irritability. | 4.4:1 | Morph NCA IV | Withdrawal symptoms. | Weaned off methadone. Alive. |
| 7 | 9000 mg | 180 mg SC infusion over 24 h | 324 mg daily SC infusion | Rapid conversion | Inadequate Analgesia. | 25:1 | Meth SC | Improved analgesia. | Died 30 h after methadone rotation. |
| 8 | 648 mg | 15 mg QID PO | 15 mg QID PO | Rapid conversion | Inadequate Analgesia. | 10.8:1 | Hydromorph PCA IV | No significant improvement in analgesia. | Weaned off methadone. Alive. |
| 9 | 10,800 mg | 36 mg IV infusion over 24 h | 600 mg IV infusion over 24 h | Rapid conversion | Myoclonus. | 150:1 | Meth PCA IV | Less mycolonus. | Died 4 days after methadone rotation. |
| 10 | 288 mg | 1 mg IV NOCTE | 10 mg QID SL | Adjuvant. Gradual conversion to methadone | Inadequate analgesia. | N/A | NCA fentanyl IV | Improved analgesia. | Weaning Methadone. Alive. |
| 11 | 30 mg | 0.5 mg QIDPO | 2.5 mg TDS PO | Rapid conversion | Inadequate analgesia. Methadone syrup available as elixir. | 15:1 | PCA hydromorph IV | Improved analgesia. Episode of drowsiness and reduced RR (did not require naloxone). | Able to wean methadone. Alive. |
| 12 | 1080 mg | 1 mg IV NOCTE | 10 mg QID SL | Adjuvant for one week, then total conversion to methadone | Inadequate analgesia. Seizures (neurotoxicity) possibly due to hydromorphone. | N/A | NCA hydromorph IV | Improved analgesia. No further seizures. | Weaned off methadone. Alive. |
| 13 | Not on regular opioid, but having PRN codeine | 2 mg BD PO | 4 mg QID PO | Commenced as primary pain management (de novo) at 0.1 mg/kg bd | Complex pain. Neuropathic pain. | Dose based on weight | PRN oxycodone PO | No improvement. | Weaned off methadone. Alive. |
| 14 | 120 mg | 1 mg at night for 1 day and then 3 mg QID SL | 10 mg QID SL | Initially adjuvant and then rapid conversion | Neurotoxicity. Severe dysponea. Chest pain. Required elixir. | 10:1 | PRN morph PO | Less confusion. Improved analgesia and less dyspnoea. | Alive. |
| 15 | 1068 mg | 36 mg IV infusion over 24 h | 72 mg IV infusion over 24 h | Rapid conversion | Inadequate Analgesia. Agitation. | 15:1 | Meth NCA IV | Improved analgesia and sedation. | Died 16 days after methadone rotation. |
| 16 | 21 mg | 0.5 mg TDS PO | 0.5 mg TDS PO | Rapid conversion | Available as elixir. Experienced pruritis with morphine. | 14:1 | PRN meth PO | Stable analgesia. | Weaned methadone. Died 3 weeks after methadone ceased. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mott, C.; Sarpal, A.; Moss, K.; Herbert, A. Methadone for Analgesia in Children with Life-Limiting Illness: Experience from a Tertiary Children’s Health Service. Children 2018, 5, 86. https://doi.org/10.3390/children5070086
Mott C, Sarpal A, Moss K, Herbert A. Methadone for Analgesia in Children with Life-Limiting Illness: Experience from a Tertiary Children’s Health Service. Children. 2018; 5(7):86. https://doi.org/10.3390/children5070086
Chicago/Turabian StyleMott, Christine, Amrita Sarpal, Krista Moss, and Anthony Herbert. 2018. "Methadone for Analgesia in Children with Life-Limiting Illness: Experience from a Tertiary Children’s Health Service" Children 5, no. 7: 86. https://doi.org/10.3390/children5070086






