In Newborn Infants a New Intubation Method May Reduce the Number of Intubation Attempts: A Randomized Pilot Study
Abstract
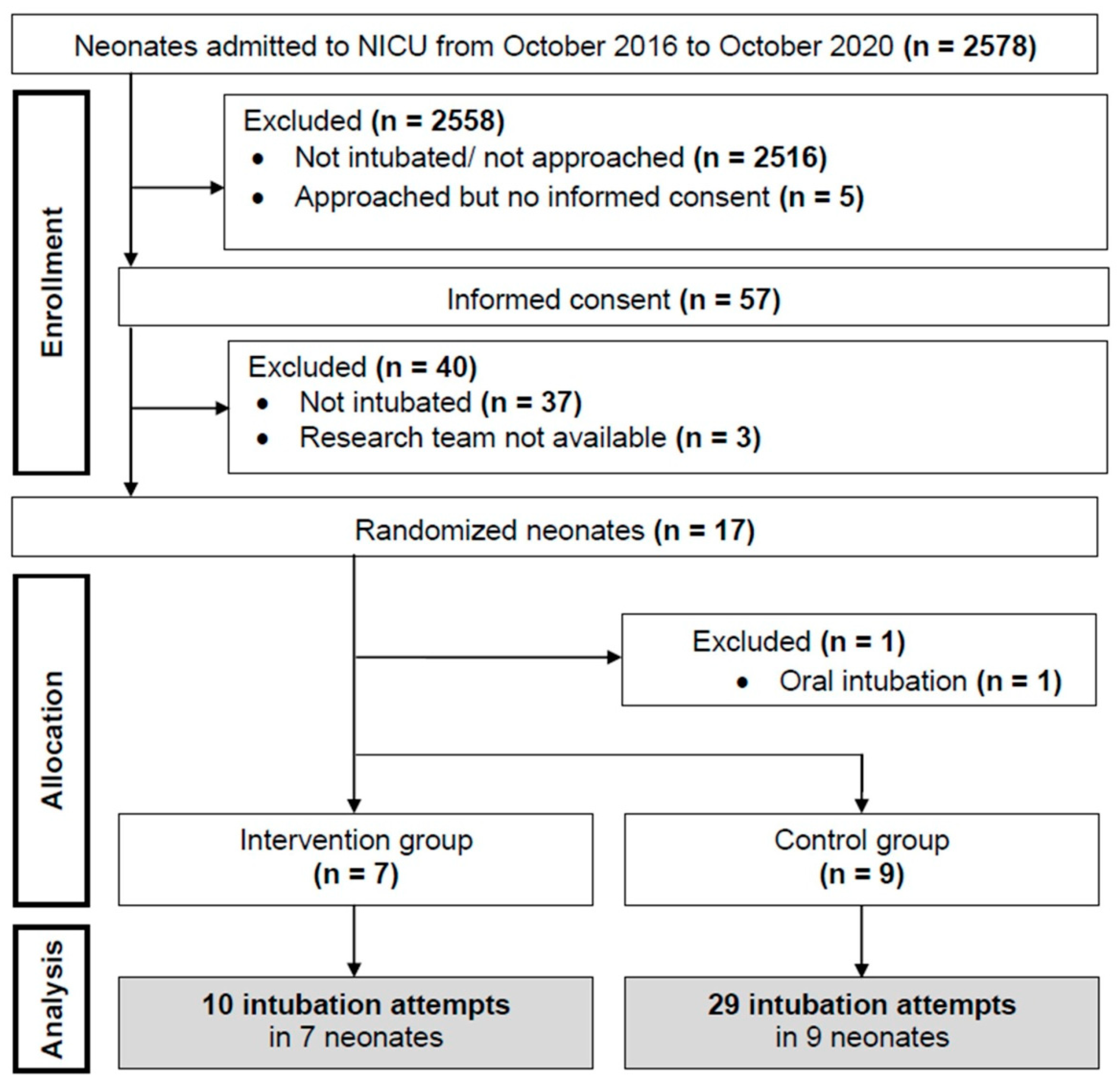
:1. Introduction
2. Materials and Methods
2.1. Study Poplulation
2.2. Randomization and Blinding
2.3. Sample Size
2.4. Interventions
2.5. Intubation Attempt Abortion Criteria
2.6. Data Collection and Statistical Analysis
3. Results
3.1. Intubation Attempts
3.2. Secondary Outcomes
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Foglia, E.E.; Ades, A.; Sawyer, T.; Glass, K.M.; Singh, N.; Jung, P.; Quek, B.H.; Johnston, L.C.; Barry, J.; Zenge, J.; et al. Neonatal Intubation Practice and Outcomes: An International Registry Study. Pediatrics 2019, 143, e20180902. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hatch, L.D.; Grubb, P.H.; Lea, A.S.; Walsh, W.F.; Markham, M.H.; Whitney, G.M.; Slaughter, J.C.; Stark, A.R.; Ely, E.W. Endotracheal Intubation in Neonates: A Prospective Study of Adverse Safety Events in 162 Infants. J. Pediatr. 2016, 168, 62–66. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fiadjoe, J.E.; Nishisaki, A.; Jagannathan, N.; Hunyady, A.I.; Greenberg, R.S.; Reynolds, P.I.; Matuszczak, M.E.; Rehman, M.A.; Polaner, D.M.; Szmuk, P.; et al. Airway management complications in children with difficult tracheal intubation from the Pediatric Difficult Intubation (PeDI) registry: A prospective cohort analysis. Lancet Respir. Med. 2016, 4, 37–48. [Google Scholar] [CrossRef]
- Soneru, C.N.; Hurt, H.F.; Petersen, T.R.; Davis, D.D.; Braude, D.A.; Falcon, R.J. Apneic nasal oxygenation prolongs safe apnea time during pediatric intubations by learners. Pediatr. Anesth. 2019, 29, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Latif, M.E.; Oei, J.; Lui, K. Propofol Compared with the Morphine, Atropine, and Suxamethonium Regimen as Induction Agents for Neonatal Endotracheal Intubation: A Randomized, Controlled Trial: In Reply. Pediatrics 2007, 120, 933. [Google Scholar] [CrossRef]
- Hodgson, K.A.; Owen, L.S.; Kamlin, C.O.; Roberts, C.T.; Donath, S.M.; Davis, P.G.; Manley, B.J. A multicentre, randomised trial of stabilisation with nasal high flow during neonatal endotracheal intubation (the SHINE trial): A study protocol. BMJ Open 2020, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]

| Study Group (n = 7) | Control Group (n = 9) | p-value | |
|---|---|---|---|
| Demographics | |||
| Gestational age (weeks) | 30 (26–31) | 32 (30–35) | 0.222 |
| Term infants (≥37 weeks gestation) | 0 (0) | 2 (22) | 0.475 |
| Female sex | 2 (29) | 5 (56) | 0.608 |
| Apgar 5 min | 9 (8–9) | 8 (8–9) | 0.834 |
| Postnatal age at intubation (hours) | 15 (17) | 36 (56) | 0.873 |
| Weight at intubation (grams) | 1487 (819) | 1857 (927) | 0.461 |
| Heart rate before intubation (beats/min) | 151 (13) | 145 (9) | 0.292 |
| Mean arterial blood pressure (mmHg) before intubation | 44 (9) | 42 (8) | 0.694 |
| Intubation | |||
| Number of intubation attempts | 1 (1–2) | 4 (2–5) | 0.056 |
| Success on first intubation attempt | 4 (57) | 2 (22) | 0.303 |
| Duration until successful intubation (seconds) | 204 (138–300) | 858 (330–924) | 0.114 |
| Aborted intubations due to desaturation and/or bradycardia (n) | 3 | 20 | 0.060 |
| FiO2 during intubation | 0.50 (0.40–0.75) | 0.55 (0.40–0.70) | 0.873 |
| Gas flow (liters/minute) | 6 (6–8) | 8 (6–8) | 0.289 |
| Intravenous Propofol dosage (mg/kg) | 1 (1–2) | 2 (2–3) | 0.072 |
| Parameters of hospital stay | |||
| Duration of invasive ventilation (days) | 6 (2–7) | 1 (0–3) | 0.459 |
| Duration of non-invasive ventilation (days) | 14 (10–61) | 6 (5–40) | 0.153 |
| Mortality (%) | 0 | 0 | 1.000 |
| Hospital stay (days) | 46 (39–106) | 30 (18–58) | 0.187 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bruckner, M.; Morris, N.M.; Pichler, G.; Wolfsberger, C.H.; Heschl, S.; Mileder, L.P.; Schwaberger, B.; Schmölzer, G.M.; Urlesberger, B. In Newborn Infants a New Intubation Method May Reduce the Number of Intubation Attempts: A Randomized Pilot Study. Children 2021, 8, 553. https://doi.org/10.3390/children8070553
Bruckner M, Morris NM, Pichler G, Wolfsberger CH, Heschl S, Mileder LP, Schwaberger B, Schmölzer GM, Urlesberger B. In Newborn Infants a New Intubation Method May Reduce the Number of Intubation Attempts: A Randomized Pilot Study. Children. 2021; 8(7):553. https://doi.org/10.3390/children8070553
Chicago/Turabian StyleBruckner, Marlies, Nicholas M. Morris, Gerhard Pichler, Christina H. Wolfsberger, Stefan Heschl, Lukas P. Mileder, Bernhard Schwaberger, Georg M. Schmölzer, and Berndt Urlesberger. 2021. "In Newborn Infants a New Intubation Method May Reduce the Number of Intubation Attempts: A Randomized Pilot Study" Children 8, no. 7: 553. https://doi.org/10.3390/children8070553
APA StyleBruckner, M., Morris, N. M., Pichler, G., Wolfsberger, C. H., Heschl, S., Mileder, L. P., Schwaberger, B., Schmölzer, G. M., & Urlesberger, B. (2021). In Newborn Infants a New Intubation Method May Reduce the Number of Intubation Attempts: A Randomized Pilot Study. Children, 8(7), 553. https://doi.org/10.3390/children8070553







