1. Introduction
Chronic kidney disease (CKD) affects between 10 and 15% of the world’s population—a percentage that is rising along with comorbidities such as hypertension, obesity, and diabetes [
1]. CKD may have developmental origins from toxic environmental exposures that occur in early childhood [
2]. In particular, early-life exposure to common nephrotoxic elements, such as arsenic (As), cadmium (Cd), and lead (Pb), may disrupt nephrogenesis and primary renal developmental processes that are vital for nutrientwaste homeostasis [
3]. As, Cd, and Pb are established nephrotoxicants, pervasive in the environment [
4,
5,
6], with substantial evidence of glomerular or tubular toxicity [
7,
8,
9] as well as nephrotoxic effects in children [
10,
11,
12,
13,
14,
15,
16]. However, their joint impact on renal development as a mixture is less well known. In this study, we selected As, Cd, and Pb
a priori based on these reasons and measured levels in blood and urine in a population of healthy children in Mexico City—a population with elevated environmental Pb exposure [
17].
Because early-life exposure to nephrotoxicants may lead to kidney function decline, examining exposure to multiple toxicants individually and as a mixture is increasingly important in the context of real-life exposures. For example, blood Pb and Cd exposure have been individually associated with a reduced estimated glomerular filtration rate (eGFR) and co-exposure to both toxicants increases CKD risk [
6,
18]. Pb and Cd have also been shown to act non-additively, resulting in worse kidney function decline in adults [
6,
19,
20,
21]. Additionally, studies in mice show that As and Cd co-exposure exacerbates tubular injury [
22]. Despite these consistent results in adults and animal models, the effects of mixed exposures to known nephrotoxicants, such as As, Cd, and Pb, are understudied in the context of early-life exposure and both later childhood as well as adolescent kidney function.
In nearly all human studies, the true internal dose of a chemical, such as a single element, is unknown and must be estimated—most commonly by using exposure biomarker measurements of a compound or its metabolite in a biological medium (e.g., blood or urine) [
23]. However, each metal has unique toxicokinetics; i.e., the process by which a chemical enters the body, is metabolized, and excreted. Thus, different metals are distributed differently across different tissues and exposures assessed in a single biological media may not accurately capture exposure to multiple metals simultaneously [
24]. This can lead to misleading and uninformative exposure estimates for some chemicals in a mixture when a single exposure media (e.g., blood or urine) is used as a proxy of exposure, thus leading to inaccurate estimates for the effects of the mixture as a whole [
25,
26]. We have previously reported on a potential method to address this issue, namely, quantifying exposure through multi-media biomarkers (MMBs), estimates of exposure derived using environmental mixture methods across different biological media [
27,
28,
29]. MMBs have proven useful in uncovering the effects of metal mixtures on neurodevelopment [
27,
28,
29]; however, to our knowledge they have not been applied to the role of metal mixtures on kidney function.
In this study, we set out to examine whether early-life exposure to nephrotoxic metal mixtures (NMM) was associated with poorer kidney function, including altered tubular and glomerular parameters. We measured the As, Cd, and Pb levels in the blood and urine from 300 children participating in the Programming Research in Obesity, Growth, Environment, and Social Stressors (PROGRESS) cohort in Mexico City, Mexico. We estimated combined As–Cd–Pb effects, using weighted quantile sum regression (WQS), on renal health, quantified using serum cystatin C and cystatin-C-based eGFR. We compare the mixture effects, estimated using the two media of urine or blood individually, as well as with combined effects when the level of exposure is estimated using MMBs. We propose that MMBs may better highlight the effects of NMM on kidney parameters than any single medium—blood or urine.
4. Discussion
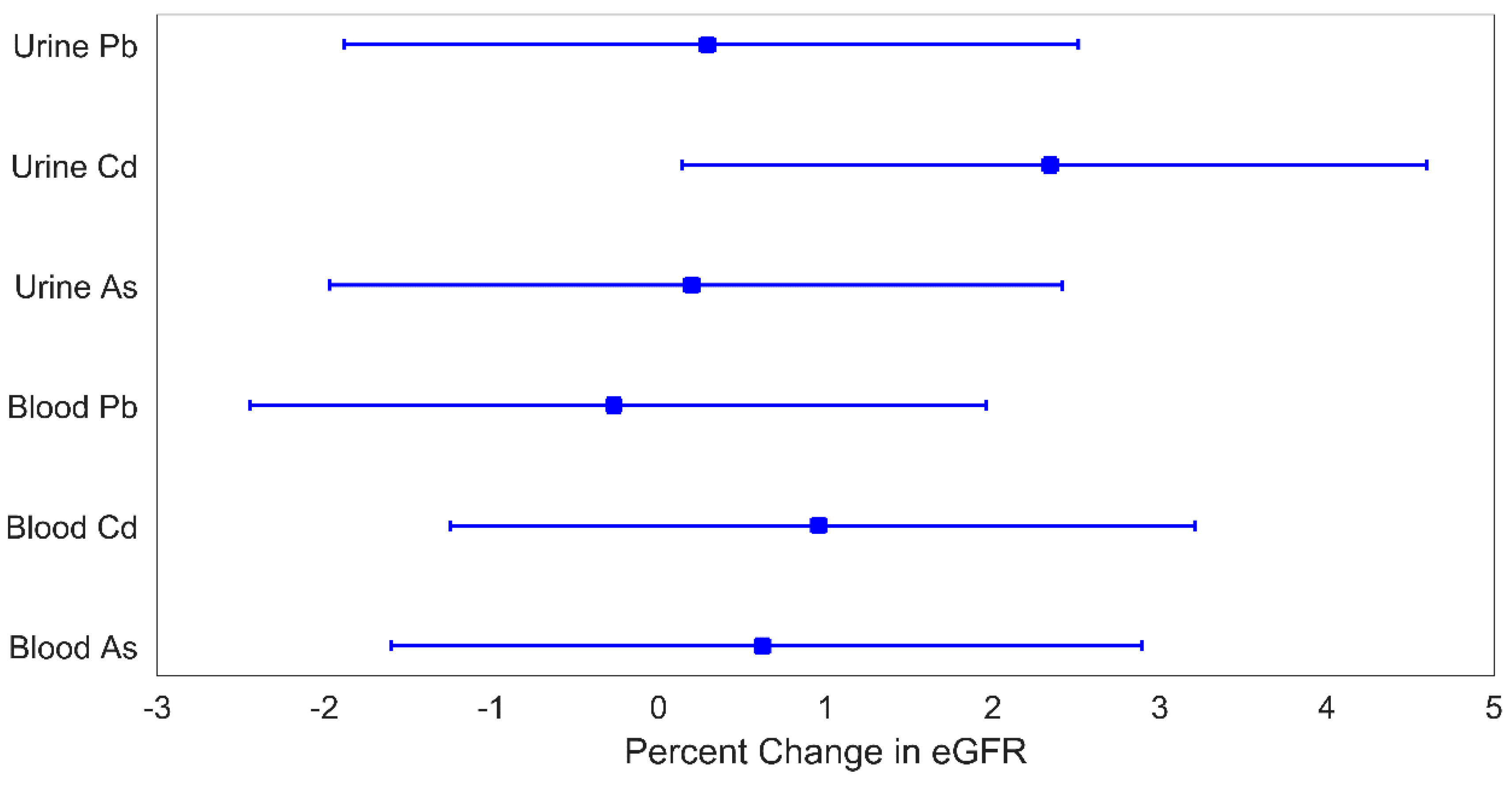
In this study, we examined the mixture effect of As, Cd, and Pb on markers of kidney function using WQS and MMBs. Overall, we found that the mixture effects differed depending on the medium used to measure exposure and that the MMB approach as well as the omnibus mixture consistently uncovered stronger mixture effects than those found using the biomarkers individually. This highlights the advantages of incorporating exposure information from multiple biomarkers, reducing errors in the estimation of exposure. In this work, we report modest increases in the metal mixture associated eGFR (~3% increases), largely driven by Cd. While these findings are not clinically of concern, early increases in filtration leading to hyperfiltration are beginning stages of eventual eGFR decline in some CKD [
38,
39]. Therefore, long-term follow-up among at risk groups with early-life elevated eGFR may be prudent.
In models for U
mix, the mixture of MMBs, and the omnibus mixture, urine Cd was consistently identified as the strongest contributor (73–84%) to the relationship with a higher subsequent eGFR. A prior study of As, Cd, Pb, and mercury (Hg) mixtures among US adolescents in NHANES similarly identified a positive association between a similar mixture of urinary metals and serum creatinine-derived eGFR wherein the top weighted metals contributing to the association were Hg (61%), Cd (17%), and As (13%) [
12]. While we did not assess Hg in this study, the relative contributions for the metals that overlap across the two studies, namely As, Cd, and Pb, is identical across studies. Although generally in alignment with our results, differences between the reported findings could be due to study design, source population exposure sources and characteristics, younger age of participants in the PROGRESS cohort, and differences in available metals and kidney function measures between the two studies. A study of metal mixtures assessed in blood from a large sample of US adults reported significant associations with multiple measures of worse kidney function, including reduced eGFR [
40]. The strongest contributors to those associations included blood Pb and Cd. In our study, we observed no significant associations with B
mix, similar to findings in the prior study of adolescents in the US [
12]. The differences between the reported findings for blood metal mixtures may be due to the age difference between the subjects or the differences in the mixture methods used. The present study, and the one reporting no associations for the blood metals and renal health, studied children and adolescents, and both used WQS [
12], while there were associations observed for adults detected using Bayesian kernel machine regression (BKMR) [
40]. The inconsistencies of the metal mixture effects on eGFR depending on the exposure biomarker has been reported previously [
41].
While there have been relatively few papers exploring the impact of metal mixtures on kidney function, especially in adolescents, there is a larger literature on these metals individually and kidney disease or dysfunction. For example, in two cross-sectional studies of adults and adolescents, increased levels of urine Cd were associated with increased eGFR [
42,
43]. Additionally, in a cross-sectional study of 1253 adolescents/young adults in the US, higher total urinary As was associated with higher eGFR [
44]. Among studies that examined individual metal levels in blood and kidney health, two reported associations between blood Pb increased eGFR [
45,
46]. A potential mechanism to explain these results is that increased exposure to metals leads to subclinical glomerular damage, resulting in overcompensation observed as increased filtration (hyperfiltration) [
41,
43,
44,
47,
48]. The cross-sectional design of the vast majority of prior studies makes temporal inference difficult. For example, chronic exposure to one or more toxicant metals can lead to decreased kidney filtration capacity due to reduced capacity for toxicant excretion, thus resulting in renal accumulation of multiple toxicants and further explaining mixture effects [
12]. In this work, we considered a relatively simple mixture of three established nephrotoxic metals selected a priori; however, issues of bias in exposure estimation become increasingly strong as the number of elements in the mixture increases. The reason for this is that a single biomarker is less likely to be an accurate measure of exposure for every element in a mixture with a large number of elements than in a simple mixture.
Our study has multiple strengths. The participants in the PROGRESS cohort are free of clinical renal disease. We measured metal exposure when the participants were 4–6 years of age and kidney outcomes when the participants were 8–10 years of age, thus enabling us to examine temporal relationships between exposure and outcome. Our use of multiple exposure biomarkers, uncommon in epidemiological studies, enabled us to explore how the association between metal mixtures and kidney function parameters differs depending on the choice of exposure media. Using MMBs allows us to minimize the potential bias that can be introduced through the selection of a single biological media to measure the mixture effects of As, Cd, and Pb. We used four complementary modeling techniques to explore associations between metals and renal parameters: (i) individual metal regressions, (ii) media-specific mixture analyses (Umix and Bmix); (iii) MMB mixture analyses, and (iv) an omnibus mixture of metals and biomarkers. We found strong consistency across these different modeling methods, increasing our confidence in our results. We used an established mixtures method, WQS, which assesses the joint effect of multiple metals on kidney function while being robust to exposure collinearly. Additionally, our use of WQS facilitates the estimation of weights highlighting the relative contributions of different metals to the mixture effect as well as the relative contributions of different exposure biomarkers to detect these associations. Although Cd dominated the weighted indices, there was a contribution from other metals, supporting the concept of a nephrotoxic mixture effect.
Our study also has some limitations. We assessed only eGFR at a single time point; follow-up studies in mid-adolescence will provide additional information about the eGFR trajectory. The PROGRESS cohort did not collect information about the hour of the day of urine collection as well as whether urine samples analyzed for metals were the first urine of the day, and so we were unable to account for this in our analyses. However, the samples were collected in the morning of each visit and, thus, constitute either the first or second urine of the day. Subsequent PROGRESS study visits are collecting information on time of urine collection, enabling its incorporation into analyses moving forward. We used spot urine samples for our analyses and do not have information about 24-h creatinine clearance. We were not able to measure iohexol clearance, considered the “gold standard” of measured GFR, as this may not be practical in large population-based studies. In our WQS analyses, we did not split our data into separate training and testing datasets; therefore, our results could suffer from a lack of generalization. As with all observational epidemiological studies, replication in other populations is needed to validate our results. Finally, as with all studies of metals and renal health, reverse causality is a major concern. As kidney function declines, the changes in metal excretion rates can lead to measured increases in urinary metal levels [
49]. However, the probability of this may be minimal in this study as the participants do not have any clinically-diagnosed kidney disease or dysfunction.