Time in Range in Children with Type 1 Diabetes before and during a Diabetes Camp—A Ceiling Effect?
Abstract
:1. Introduction
2. Materials and Methods
3. Results
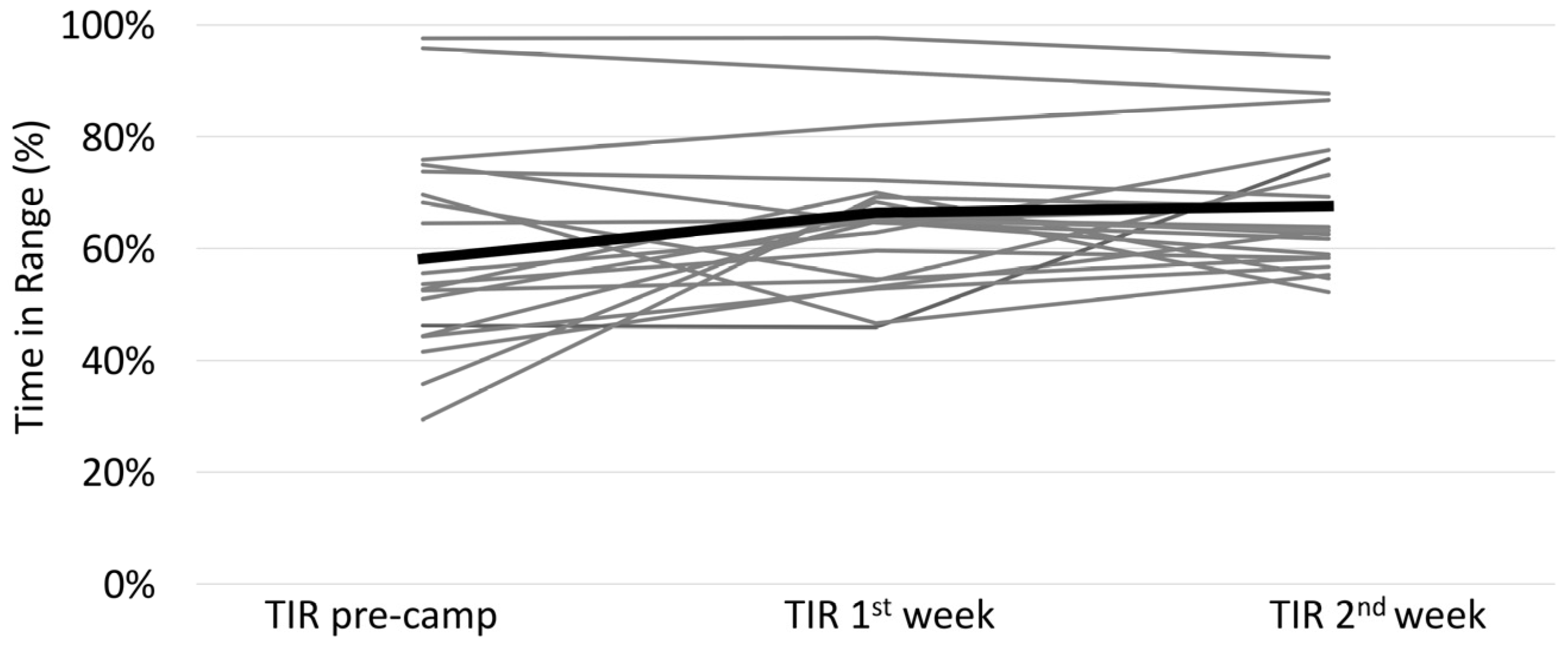
3.1. Comparison Pre-Camp to Camp
3.2. Comparison of Day- and Nighttime
3.3. Insulin Dose Adjustments
3.4. Comparison between Insulin Pen and Pump Users
3.5. Comparison between rtCGM and isCGM Users
3.6. Associations with Improvement of TIR
3.7. Targets and Safety
4. Discussion
4.1. Improvement of Glycemic Control
4.2. Day and Night
4.3. Comparison of isCGM vs. rtCGM
4.4. Therapy Modalities
4.5. Diabetes Camp Effect
4.6. Limitations
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Gandhi, G.Y.; Kovalaske, M.; Kudva, Y.; Walsh, K.; Elamin, M.B.; Beers, M.; Coyle, C.; Goalen, M.; Murad, M.S.H.; Erwin, P.J.; et al. Efficacy of continuous glucose monitoring in improving glycemic control and reducing hypoglycemia: A systematic review and meta-analysis of randomized trials. J. Diabetes Sci. Technol. 2011, 5, 952–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DeSalvo, D.J.; Miller, K.M.; Hermann, J.M.; Maahs, D.M.; Hofer, S.E.; Clements, M.A.; Lilienthal, E.; Sherr, J.L.; Tauschmann, M.; Holl, R.W. Continuous glucose monitoring and glycemic control among youth with type 1 diabetes: International comparison from the T1D Exchange and DPV Initiative. Pediatr. Diabetes 2018, 19, 1271–1275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tauschmann, M.; Hermann, J.M.; Freiberg, C.; Papsch, M.; Thon, A.; Heidtmann, B.; Placzeck, K.; Agena, D.; Kapellen, T.M.; Schenk, B.; et al. Reduction in diabetic ketoacidosis and severe hypoglycemia in pediatric type 1 diabetes during the first year of continuous glucose monitoring: A multicenter analysis of 3,553 subjects from the DPV registry. Diabetes Care 2020, 43, E40–E42. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Danne, T.; Nimri, R.; Battelino, T.; Bergenstal, R.M.; Close, K.L.; DeVries, J.H.; Garg, S.; Heinemann, L.; Hirsch, I.; Amiel, S.A.; et al. International consensus on use of continuous glucose monitoring. Diabetes Care 2017, 40, 1631–1640. [Google Scholar] [CrossRef] [Green Version]
- Sherr, J.L.; Tauschman, M.; Battelino, T.; de Bock, M.; Forlenza, G.; Roman, R.; Hood, K.K.; Maahs, D.M. ISPAD Clinical Practice Consensus Guidelines 2018 Diabetes Technologies. Pediatr. Diabetes 2018, 19, 302–325. [Google Scholar] [CrossRef]
- Battelino, T.; Danne, T.; Bergenstal, R.M.; Amiel, S.A.; Beck, R.; Biester, T.; Bosi, E.; Buckingham, B.A.; Cefalu, W.T.; Close, K.L.; et al. Clinical targets for continuous glucose monitoring data interpretation: Recommendations from the international consensus on time in range. Diabetes Care 2019, 42, 1593–1603. [Google Scholar] [CrossRef] [Green Version]
- Foster, N.C.; Miller, K.M.; Tamborlane, W.V.; Bergenstal, R.M.; Beck, R.W. Continuous Glucose Monitoring in Patients With Type 1 Diabetes Using Insulin Injections. Diabetes Care 2016, 39, e81–e82. [Google Scholar] [CrossRef] [Green Version]
- Franceschi, R.; Micheli, F.; Mozzillo, E.; Cauvin, V.; Liguori, A.; Soffiati, M.; Giani, E. Intermittently Scanned and Continuous Glucose Monitor Systems: A Systematic Review on Psychological Outcomes in Pediatric Patients. Front. Pediatr. 2021, 9, 660173. [Google Scholar] [CrossRef]
- Visser, M.M.; Charleer, S.; Fieuws, S.; De Block, C.; Hilbrands, R.; Van Huffel, L.; Maes, T.; Vanhaverbeke, G.; Dirinck, E.; Myngheer, N.; et al. Comparing real-time and intermittently scanned continuous glucose monitoring in adults with type 1 diabetes (ALERTT1): A 6-month, prospective, multicentre, randomised controlled trial. Lancet 2021, 397, 2275–2283. [Google Scholar] [CrossRef]
- Préau, Y.; Galie, S.; Schaepelynck, P.; Armand, M.; Raccah, D. Benefits of a switch from intermittently scanned continuous glucose monitoring (IsCGM) to real-time (rt) CGM in diabetes type 1 suboptimal controlled patients in real-life: A one-year prospective study. Sensors 2021, 21, 6131. [Google Scholar] [CrossRef]
- Reddy, M.; Jugnee, N.; Anantharaja, S.; Oliver, N. Switching from Flash Glucose Monitoring to Continuous Glucose Monitoring on Hypoglycemia in Adults with Type 1 Diabetes at High Hypoglycemia Risk: The Extension Phase of the i HART CGM Study. Diabetes Technol. Ther. 2018, 20, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Hásková, A.; Radovnická, L.; Petruželková, L.; Parkin, C.G.; Grunberger, G.; Horová, E.; Navrátilová, V.; Kádě, O.; Matoulek, M.; Prázný, M.; et al. Real-time cgm is superior to flash glucose monitoring for glucose control in type 1 diabetes: The corrida randomized controlled trial. Diabetes Care 2020, 43, 2744–2750. [Google Scholar] [CrossRef] [PubMed]
- Sandig, D.; Grimsmann, J.; Reinauer, C.; Melmer, A.; Zimny, S.; Müller-Korbsch, M.; Forestier, N.; Zeyfang, A.; Bramlage, P.; Danne, T.; et al. Continuous Glucose Monitoring in Adults with Type 1 Diabetes: Real-World Data from the German/Austrian Prospective Diabetes Follow-Up Registry. Diabetes Technol. Ther. 2020, 22, 602–612. [Google Scholar] [CrossRef] [PubMed]
- Massa, G.G.; Gys, I.; Bevilacqua, E.; Wijnands, A.; Zeevaert, R. Comparison of flash glucose monitoring with real time continuous glucose monitoring in children and adolescents with type 1 diabetes treated with continuous subcutaneous insulin infusion. Diabetes Res. Clin. Pract. 2019, 152, 111–118. [Google Scholar] [CrossRef]
- Acciaroli, G.; Vettoretti, M.; Facchinetti, A.; Sparacino, G. Calibration of Minimally Invasive Continuous Glucose Monitoring Sensors: State-of-The-Art and Current Perspectives. Biosensors 2018, 8, 24. [Google Scholar] [CrossRef] [Green Version]
- Worth, C.; Dunne, M.; Ghosh, A.; Harper, S.; Banerjee, I. Continuous glucose monitoring for hypoglycaemia in children: Perspectives in 2020. Pediatr. Diabetes 2020, 21, 697–706. [Google Scholar] [CrossRef]
- Nagl, K.; Berger, G.; Aberer, F.; Ziko, H.; Weimann, K.; Bozic, I.; Rami-Merhar, B.; Mader, J.K. Performance of three different continuous glucose monitoring systems in children with type 1 diabetes during a diabetes summer camp. Pediatr. Diabetes 2021, 22, 271–278. [Google Scholar] [CrossRef]
- Cole, T.J.; Green, P.J. Smoothing reference centile curves: The LMS method and penalized likelihood. Stat. Med. 1992, 11, 1305–1319. [Google Scholar] [CrossRef]
- De Onis, M.; Onyango, A.W.; Borghi, E.; Siyam, A.; Nishida, C.; Siekmann, J. Development of a WHO growth reference for school-aged children and adolescents. Bull. World Health Organ. 2007, 85, 660–667. [Google Scholar] [CrossRef]
- Hermann, J.M.; Miller, K.M.; Hofer, S.E.; Clements, M.A.; Karges, W.; Foster, N.C.; Fröhlich-Reiterer, E.; Rickels, M.R.; Rosenbauer, J.; DeSalvo, D.J.; et al. The Transatlantic HbA1c gap: Differences in glycaemic control across the lifespan between people included in the US T1D Exchange Registry and those included in the German/Austrian DPV registry. Diabet. Med. 2020, 37, 848–855. [Google Scholar] [CrossRef]
- Karges, B.; Schwandt, A.; Heidtmann, B.; Kordonouri, O.; Binder, E.; Schierloh, U.; Boettcher, C.; Kapellen, T.; Rosenbauer, J.; Holl, R.W. Association of Insulin Pump Therapy vs Insulin Injection Therapy With Severe Hypoglycemia, Ketoacidosis, and Glycemic Control Among Children, Adolescents, and Young Adults With Type 1 Diabetes. JAMA 2017, 318, 1358–1366. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.R.; Nebesio, T.D.; Dimeglio, L.A. Insulin dose changes in children attending a residential diabetes camp. Diabet. Med. 2011, 28, 480–486. [Google Scholar] [CrossRef] [PubMed]
- Gunasekera, H.; Ambler, G. Safety and efficacy of blood glucose management practices at a diabetes camp. J. Paediatr. Child Health 2006, 42, 643–648. [Google Scholar] [CrossRef] [PubMed]
- Duarte, D.B.; Fonseca, L.; Santos, T.; Silva, V.B.; Puga, F.M.; Saraiva, M.; Silva, I.L.; Teixeira, S.; Vilaverde, J.; Cardoso, M.H. Impact of intermittently scanned continuous glucose monitoring on quality of life and glycaemic control in persons with type 1 diabetes: A 12-month follow-up study in real life. Diabetes Metab. Syndr. Clin. Res. Rev. 2022, 16, 102509. [Google Scholar] [CrossRef]
- Franceschi, R.; Cauvin, V.; Stefani, L.; Berchielli, F.; Soffiati, M.; Maines, E. Early Initiation of Intermittently Scanned Continuous Glucose Monitoring in a Pediatric Population With Type 1 Diabetes: A Real World Study. Front. Endocrinol. 2022, 13, 1232. [Google Scholar] [CrossRef]
- Glocker, V.; Bachmann, S.; Hess, M.; Szinnai, G.; Burckhardt, M.A. Fear of hypoglycemia and quality of life in young people with type 1 diabetes and their parents in the era of sensor glucose monitoring. Front. Endocrinol. 2022, 13, 958671. [Google Scholar] [CrossRef]
- Urakami, T.; Terada, H.; Yoshida, K.; Kuwabara, R.; Mine, Y.; Aoki, M.; Shoji, Y.; Suzuki, J.; Morioka, I. Comparison of the clinical effects of intermittently scanned and real-time continuous glucose monitoring in children and adolescents with type 1 diabetes: A retrospective cohort study. J. Diabetes Investig. 2022, 13, 1745–1752. [Google Scholar] [CrossRef]
- Leiva-Gea, I.; Martos-Lirio, M.F.; Gómez-Perea, A.; Ariza-Jiménez, A.B.; Tapia-Ceballos, L.; Jiménez-Hinojosa, J.M.; Lopez-Siguero, J.P. Metabolic Control of the FreeStyle Libre System in the Pediatric Population with Type 1 Diabetes Dependent on Sensor Adherence. J. Clin. Med. 2022, 11, 286. [Google Scholar] [CrossRef]
- Miller, E.; Midyett, L.K. Just Because You Can, Doesn’t Mean You Should … Now. A Practical Approach to Counseling Persons with Diabetes on Use of Optional CGM Alarms. Diabetes Technol. Ther. 2021, 23, S66–S71. [Google Scholar] [CrossRef]
- Gupta, O.T.; MacKenzie, M.; Burris, A.; Jenkins, B.B.; Collins, N.; Shade, M.; Santa-Sosa, E.; Stewart, S.M.; White, P.C. Camp-based multi-component intervention for families of young children with type 1 diabetes: A pilot and feasibility study. Pediatr. Diabetes 2018, 19, 761–768. [Google Scholar] [CrossRef]
- Musolino, G.; Dovc, K.; Boughton, C.K.; Tauschmann, M.; Allen, J.M.; Nagl, K.; Fritsch, M.; Yong, J.; Metcalfe, E.; Schaeffer, D.; et al. Reduced burden of diabetes and improved quality of life: Experiences from unrestricted day-and-night hybrid closed-loop use in very young children with type 1 diabetes. Pediatr. Diabetes 2019, 20, 794–799. [Google Scholar] [CrossRef] [PubMed]
| All | Pen | Pump | p-Value | isCGM | rtCGM | p-Value | |
|---|---|---|---|---|---|---|---|
| N (%) | 26 | 10 (38.5%) | 16 (61.5%) | 13 (50%) | 13 (50%) | ||
| Age (years) | 11.0 ± 1.4 | 11.4 ± 1.1 | 10.7 ± 1.5 | 0.24 | 11.1 ± 1.2 | 10.8 ± 1.6 | 0.58 |
| female/male | 10/16 | 4/6 | 6/10 | 1.0 | 5/8 | 5/8 | 1.0 |
| Diabetes duration (years) | 4.1 ± 3.1 | 3.0 ± 3.1 | 5.0 ± 2.9 | 0.11 | 4.2 ± 2.5 | 4.2 ± 3.6 | 0.95 |
| Height (cm) | 145.9 ± 10.3 | 146.4 ± 9.8 | 145.7 ± 10.9 | 0.87 | 146.3 ± 8.1 | 145.5 ± 12.5 | 0.84 |
| Height-SDS | −0.83 ± 0.97 | −0.46 ± 0.83 | 0.15 ± 1.00 | 0.12 | −0.13 ± 1.16 | −0.03 ± 0.79 | 0.79 |
| Weight (kg) | 36.8 ± 7.63 | 35.4 ± 5.1 | 37.7 ± 8.9 | 0.41 | 38.5 ± 7.13 | 35.1 ± 8.0 | 0.265 |
| BMI-SDS | −0.13 ± 0.98 | −0.53 ± 0.65 | 0.13 ± 1.07 | 0.09 | 0.17 ± 0.92 | −0.42 ± 0.97 | 0.127 |
| HbA1c (%) | 7.3 ± 0.8 | 7.2 ± 1.1 | 7.3 ± 0.7 | 0.66 | 7.6 ± 0.8 | 7.0 ± 0.8 | 0.07 |
| HbA1c (mmol/mol) | 56.3 ± 8.7 | 55.2 ± 12.0 | 56.3 ± 7.7 | 59.6 ± 8.7 | 53.0 ± 8.7 |
| Difference between isCGM and rtCGM | Difference Pre-Camp to Camp | |||||||
|---|---|---|---|---|---|---|---|---|
| All | isCGM | rtCGM | p-Value | isCGM | rtCGM | |||
| N | 26 | p-Value | 13 | 13 | p-Value | p-Value | ||
| Time using CGM (%) | pre-camp | 95 [82;99] | 0.093 | 97 [71;100] | 94 [90;96] | 0.55 | 0.422 | 0.463 |
| camp | 96 [92;97] | 97 [96;98] | 92 [86;96] | 0.006 | ||||
| Time in range (TIR) | pre-camp | 58.2 ± 17.4 | 0.004 | 47.2 ± 12.2 | 69.1 ± 14.9 | <0.001 | 0.005 | 0.288 |
| (%) | camp | 67.0 ± 10.7 | 62.1 ± 6.3 | 71.9 ± 12.2 | 0.019 | |||
| Time below range | pre-camp | 3.2 ± 2.3 | <0.001 | 4.5 ± 2.4 | 1.8 ± 1.3 | 0.002 | 0.006 | 0.004 |
| (TBR) (%) | camp | 5.5 ± 3.0 | 7.1 ± 2.7 | 3.8 ± 2.3 | 0.002 | |||
| Time above range | pre-camp | 38.7 ± 16.9 | 0.001 | 48.3 ± 12.8 | 29.0 ± 15.0 | 0.002 | 0.002 | 0.109 |
| (TAR) (%) | camp | 27.5 ± 10.2 | 30.7 ± 7.1 | 24.3 ± 12.1 | 0.114 | |||
| Daytime TIR (%) | pre-camp | 55.5 ± 18.4 | 0.042 | 44.8 ± 12.9 | 66.2 ± 17.1 | 0.001 | 0.04 | 0.66 |
| camp | 62.5 ± 12.8 | 57.4 ± 9.5 | 67.6 ± 13.9 | 0.040 | ||||
| Nighttime TIR (%) | pre-camp | 62.5 ± 17.1 | <0.001 | 51.3 ± 13.4 | 73.8 ± 12.6 | <0.001 | <0.001 | 0.038 |
| camp | 74.1 ± 10.3 | 69.7 ± 7.8 | 78.5 ± 10.8 | 0.027 | ||||
| Daytime TBR (%) | pre-camp | 2.1 [1.0;4.0] | 0.038 | 2.3 [1.5;6.6] | 1.6 [0.8;2.5] | 0.86 | 0.507 | 0.009 |
| camp | 3.2 [2.3;5.7] | 4.2 [2.5;5.8] | 2.9 [1.8;5.3] | 0.34 | ||||
| Nighttime TBR (%) | pre-camp | 3.9 ± 3.2 | <0.001 | 5.9 ± 3.2 | 1.9 ± 1.5 | 0.001 | <0.001 | 0.017 |
| camp | 7.6 ± 5 | 11.3 ± 3.9 | 3.9 ± 2.7 | <0.001 | ||||
| Daytime TAR (%) | pre-camp | 41.7 ± 18.5 | 0.023 | 51.6 ± 14.2 | 31.9 ± 17.3 | 0.004 | 0.038 | 0.363 |
| camp | 33.4 ± 13 | 38.0 ± 10.1 | 28.7 ± 14.3 | 0.07 | ||||
| Nighttime TAR (%) | pre-camp | 33.6 ± 15.9 | <0.001 | 42.9 ± 13.5 | 24.3 ± 12.6 | 0.001 | <0.001 | 0.011 |
| camp | 18.3 ± 9 | 19.0 ± 8.1 | 17.6 ± 10.0 | 0.7 | ||||
| Total daily dose | pre-camp | 0.8 ± 0.2 | <0.001 | 0.85 ± 13.5 | 0.74 ± 18.1 | 0.08 | <0.001 | <0.001 |
| (TDD) (IU/kg/d) | camp | 0.6 ± 0.1 | 0.70 ± 0.12 | 0.59 ± 0.13 | 0.027 | |||
| Percentage of | pre-camp | 37.8 ± 9.7 | 0.040 | 40.4 ± 9.1 | 35.1 ± 9.9 | 0.16 | 0.526 | 0.011 |
| basal Insulin (%) | camp | 40.3 ± 8.3 | 41.8 ± 5.6 | 38.9 ± 10.4 | 0.40 | |||
| Daily intake of | pre-camp | 198.2 ± 30.9 | 0.003 | 212.7 ± 28.4 | 185.0 ± 28.02 | 0.022 | 0.07 | 0.026 |
| carbohydrates (g) | camp | 180.2 ± 32.5 | 197.5 ± 27.2 | 166.7 ± 16.5 | 0.012 | |||
| Scans per day | pre-camp | 13.1 ± 4.7 | 0.011 | . | . | . | . | . |
| with isCGM | camp | 17.7 ± 4.5 | . | . | . | . | . | |
| Difference TIR pre-camp to camp | (%) | 8.0 ± 14 | 15.0 ± 15.9 | 2.8 ± 9.0 | 0.025 | . | . | |
| Difference TBR pre-camp to camp | (%) | 2 ± 2 | 2.6 ± 2.8 | 1.9 ± 1.9 | 0.47 | . | . | |
| Difference TAR pre-camp to camp | (%) | −11 ± 15 | −17.6 ± 16.6 | −4.7 ± 9.8 | 0.024 | . | . | |
| Difference TDD pre-camp to camp | (%) | −19.2 ± 9.9 | −17.1 ± 12.4 | −19.6 ± 9.8 | 0.56 | . | . | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nagl, K.; Bozic, I.; Berger, G.; Tauschmann, M.; Blauensteiner, N.; Weimann, K.; Mader, J.K.; Rami-Merhar, B. Time in Range in Children with Type 1 Diabetes before and during a Diabetes Camp—A Ceiling Effect? Children 2022, 9, 1951. https://doi.org/10.3390/children9121951
Nagl K, Bozic I, Berger G, Tauschmann M, Blauensteiner N, Weimann K, Mader JK, Rami-Merhar B. Time in Range in Children with Type 1 Diabetes before and during a Diabetes Camp—A Ceiling Effect? Children. 2022; 9(12):1951. https://doi.org/10.3390/children9121951
Chicago/Turabian StyleNagl, Katrin, Ina Bozic, Gabriele Berger, Martin Tauschmann, Nicole Blauensteiner, Katharina Weimann, Julia K. Mader, and Birgit Rami-Merhar. 2022. "Time in Range in Children with Type 1 Diabetes before and during a Diabetes Camp—A Ceiling Effect?" Children 9, no. 12: 1951. https://doi.org/10.3390/children9121951





