Determination of 27 Glucocorticoids in Urine by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Using UniSprayTM Source
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Sample Preparation
2.3. Instrumental Analysis
2.3.1. Liquid Chromatography Separation
2.3.2. Mass Spectrometry
3. Validation
3.1. Linearity
3.2. LOD, LOQ, and RRT
3.3. Extraction Recovery
3.4. Selectivity
3.5. Carryover
3.6. Accuracy and Intra-Day Precision
4. Results and Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Strehl, C.; Spies, C.M.; Buttgereit, F. Pharmacodynamics of glucocorticoids. Clin. Exp. Rheumatol. 2011, 29 (Suppl. 68), S13–S18. [Google Scholar] [PubMed]
- Ahi, S.; Beotra, A.; Dubey, S.; Upadhyay, A.; Jain, S. Simultaneous identification of prednisolone and its ten metabolites in human urine by high-performance liquid chromatography-tandem mass spectrometry. Drug Test. Anal. 2012, 4, 460–467. [Google Scholar] [CrossRef]
- Vegiopoulos, A.; Herzig, S. Glucocorticoids, metabolism and metabolic diseases. Mol. Cell. Endocrinol. 2007, 275, 43–61. [Google Scholar] [CrossRef] [PubMed]
- Mazzarino, M.; Piantadosi, C.; Comunita, F.; de la Torre, X.; Botre, F. Urinary excretion profile of prednisone and prednisolone after different administration routes. Drug Test. Anal. 2019, 11, 1601–1614. [Google Scholar] [CrossRef] [PubMed]
- Buckingham, J.C. Glucocorticoids: Exemplars of multi-tasking. Br. J. Pharmacol. 2006, 147, S258–S268. [Google Scholar] [CrossRef]
- Horst-Sikorska, W. Steroid therapy—Benefits and risks. Przew Lek 2008, 1, 133–136. [Google Scholar]
- Schäcke, H.; Döcke, W.D.; Asadullah, K. Mechanisms involved in the side effects of glucocorticoids. Pharmacol. Ther. 2002, 96, 23–43. [Google Scholar] [CrossRef]
- Kim, N.S.; Yoo, G.J.; Lee, J.H.; Park, H.J.; Cho, S.; Shin, D.W.; Kim, Y.; Baek, S.Y. Determination of 43 prohibited glucocorticoids in cosmetic products using a simultaneous LC-MS/MS method. Anal. Methods 2017, 9, 2104–2115. [Google Scholar] [CrossRef]
- Ganong, W.F. Physiology; Medical Publishing PZWL: Warsaw, Poland, 2007. [Google Scholar]
- Wrzeciono, U.; Zaprutko, L. Chemistry of Natural Compounds; Medical Academy Publishing of Karol Marcinkowski in Poznan: Poznan, Poland, 2001. [Google Scholar]
- Haneef, J.; Shaharyar, M.; Husain, A.; Rashid, M.; Mishra, R.; Parveen, S.; Ahmed, N.; Pal, M.; Kumar, D. Application of LC-MS/MS for quantitative analysis of glucocorticoids and stimulants in biological fluids. J. Pharm. Anal. 2013, 3, 341–348. [Google Scholar] [CrossRef] [PubMed]
- The World Anti-Doping Code 2021. Available online: https://www.wada-ama.org/en/resources/world-anti-doping-program/world-anti-doping-code (accessed on 16 January 2023).
- The World Anti-Doping Code. The 2004 Prohibited List International Standard, World Anti-Doping Agency, Montreal (Canada). 2004. Available online: https://www.wada-ama.org/sites/default/files/resources/files/WADA_Prohibited_List_2004_EN.pdf (accessed on 16 January 2023).
- The World Anti-Doping Code. The 2022 Prohibited List International Standard, World Anti-Doping Agency, Montreal (Canada). 2022. Available online: www.wada-ama.org (accessed on 16 January 2023).
- The World Anti-Doping Agency. Minimum Required performance level: TD2022MRPL. World Anti-Doping Agency, Montreal (Canada). 2022. Available online: www.wada-ama.org (accessed on 16 January 2023).
- Bajic, S. Atmospheric Pressure Ion Source by Interacting High Velocity Spray with a Target. U.S. Patent no. 8809777, 19 August 2014. [Google Scholar]
- Galani, J.H.Y.; Houbraken, M.; Van Hulle, M.; Spanoghe, P. Comparison of electrospray and UniSpray, a novel atmospheric pressure ionization interface, for LC-MS/MS analysis of 81 pesticide residues in food and water matrices. Anal. Bioanal. Chem. 2019, 411, 5099–5133. [Google Scholar] [CrossRef] [PubMed]
- Waters Corporation; Waters Application Note; Hammond, J.; Sanig, R.; Kirk, J.; Wrona, M. A Comparative Study of Electrospray and UniSpray Sources Using ACQUITY UPC2. May 2018. Available online: https://www.waters.com/content/dam/waters/en/app-notes/2018/720006286/720006286-de.pdf (accessed on 16 January 2023).
- The World Anti-Doping Agency (WADA). Minimum Criteria for Chromatographic-Mass Spectrometric Confirmation of The Identity of Analytes for Doping Control Purposes. Available online: https://www.wada-ama.org/en/resources/lab-documents/td2023idcr (accessed on 1 January 2023).
- Webster, T.E. A New Robust Technique for Testing of Glucocorticosteroids in Dogs and Horses; Retrospective Theses and Dissertations; Iowa State University: Ames, IA, USA, 2007; p. 15029. [Google Scholar]
- Scientific Working Group for Forensic Toxicology (SWGTOX). Standard Practices for Method Validation in Forensic Toxicology. J. Anal. Toxicol. 2013, 37, 452–474. [Google Scholar] [CrossRef] [PubMed]
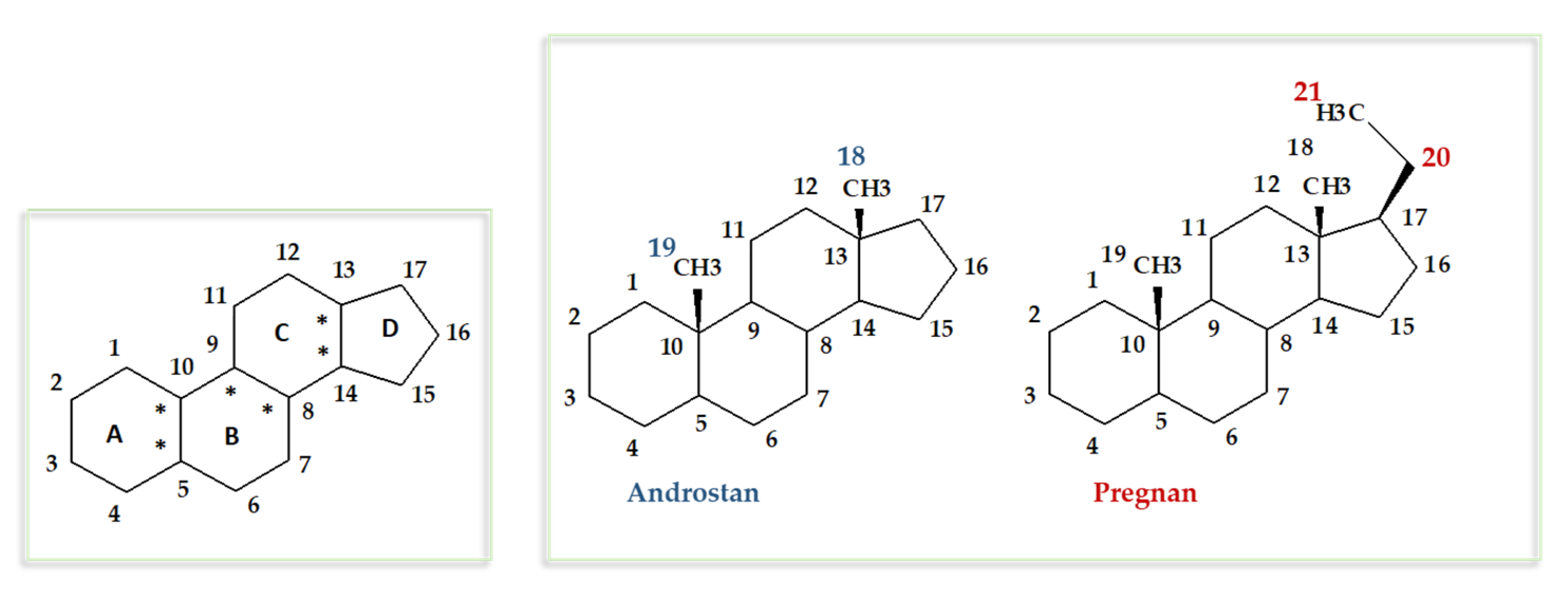
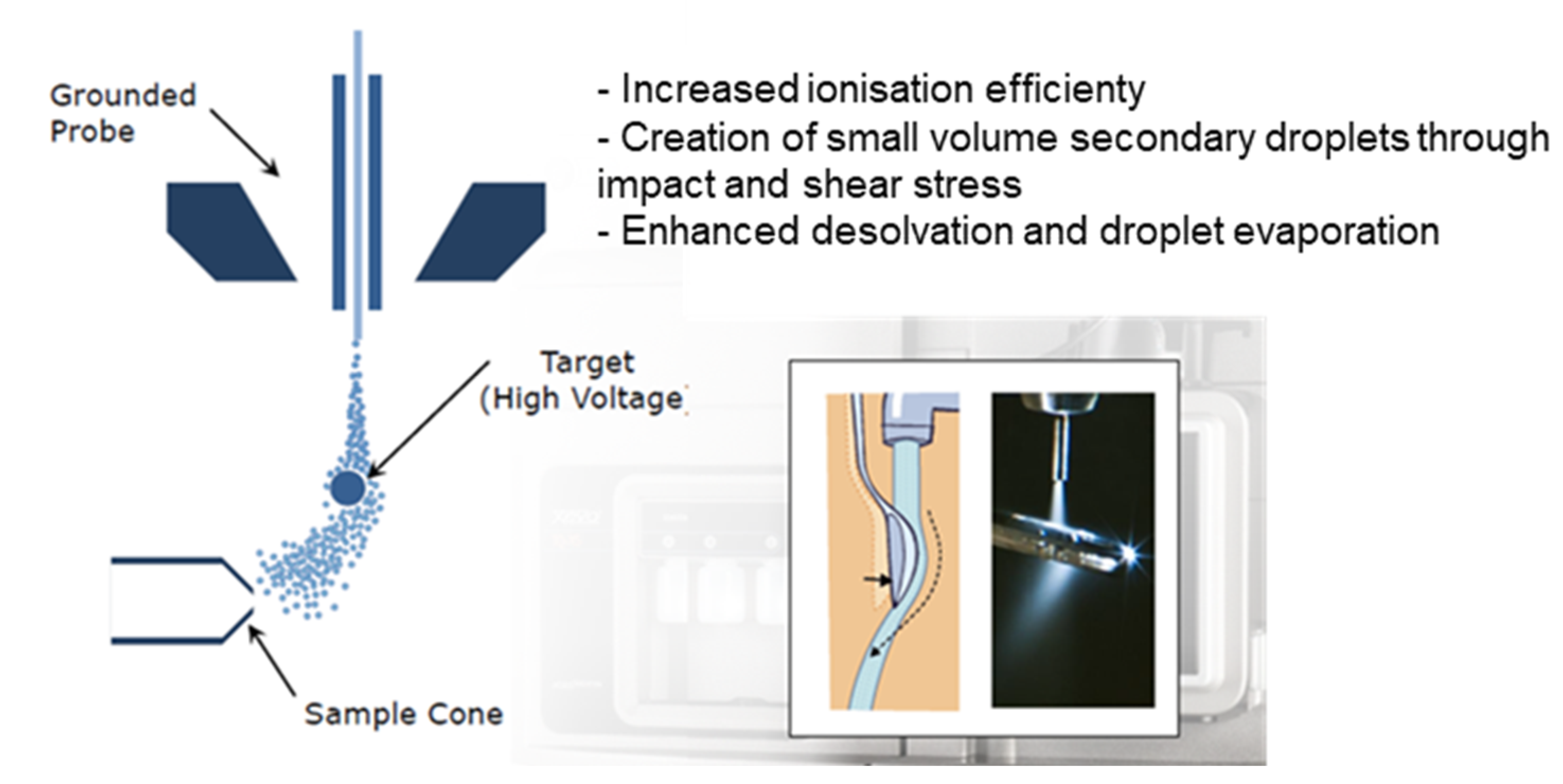




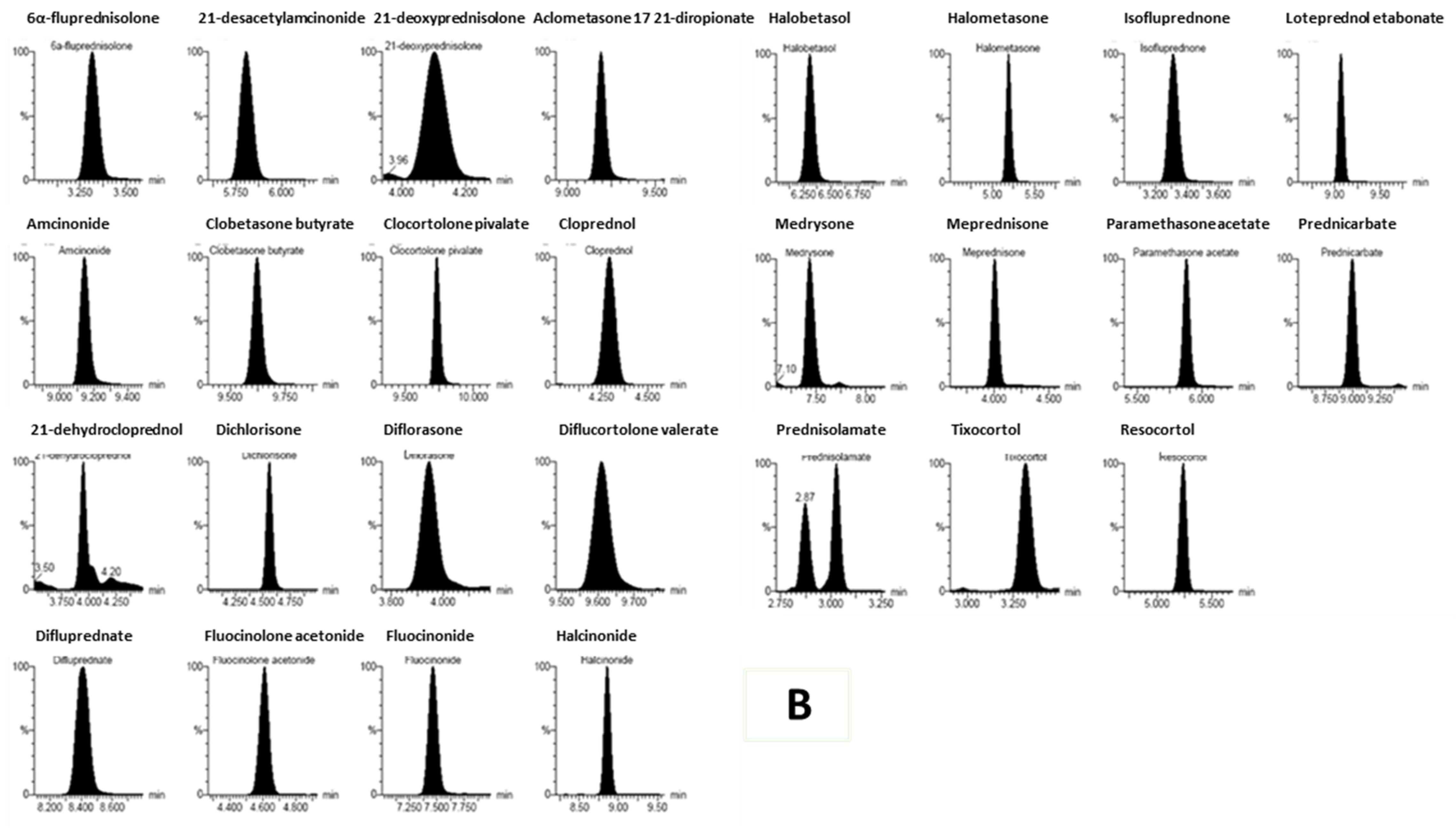
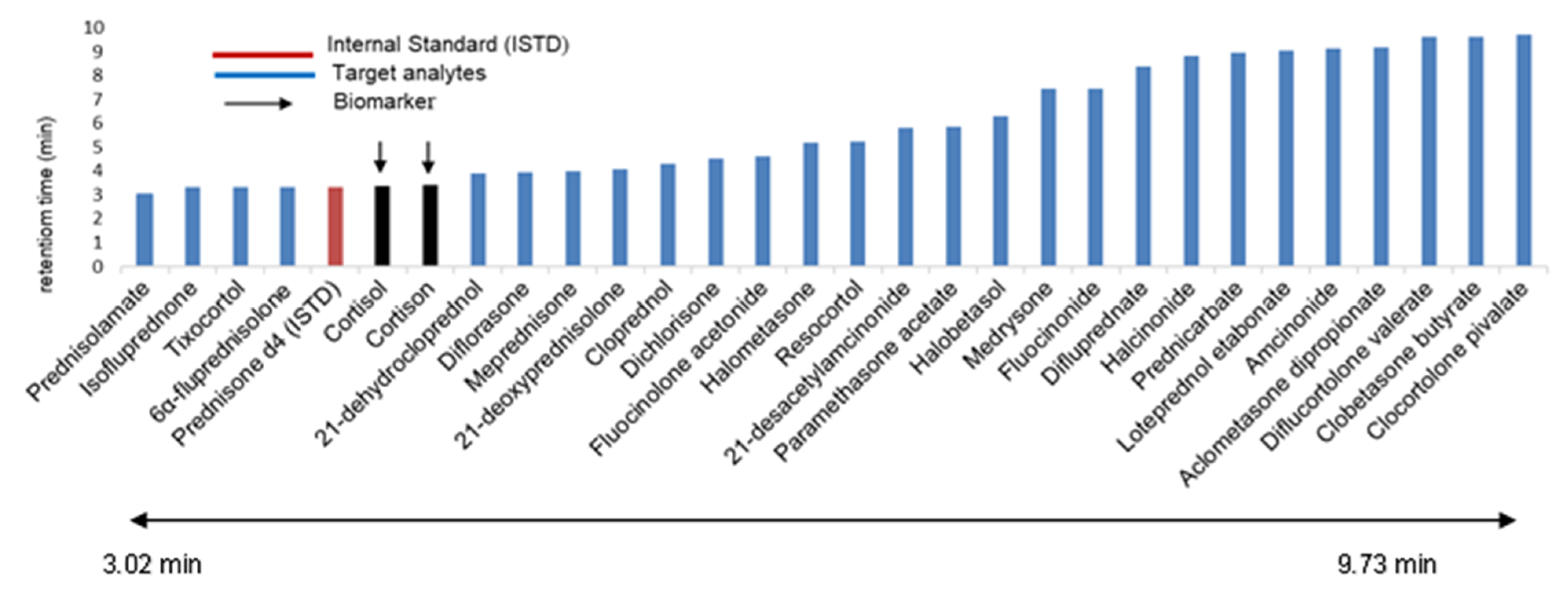
 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Synthetic | |||||||||||
| Type of Glucocorticoids | |||||||||||
| Δ1,2 | R6 | R9 | R11 | R16 | R17 | R21 | Progesterone | Hydrocortisone | Methasone (16-Methylated) | Acetonides and Related | |
| 6α-fluprednisolone | ═ | ─F | ─H | ─OH | ─H | ─OH | ─OH | ✓ | |||
| 21-desacetylamcinonide | ═ | ─H | ─F | ─OH | 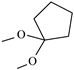 | ─OH | ✓ | ||||
| 21-deoxyprednisolone | ═ | ─H | ─H | ─OH | ─H | ─OH | ─CH3 | ✓ | |||
| Aclometasone17 21-dipropionate | ═ | ─H | ─H | ─OH | ─CH3 | ─OCOCH2CH3 | ─OCOCH2CH3 | ✓ | |||
| Amcinonide | ═ | ─H | ─F | ─OH |  | ─OCOCH3 | ✓ | ||||
| Clobetasone butyrate | ═ | ─H | ─F | ═O | ─CH3 | ─OCOCH2CH2CH3 | ─Cl | ✓ | |||
| Clocortolone pivalate | ═ | ─F | ─Cl | ─OH | ─CH3 | ─H | 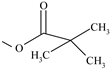 | ✓ | |||
| Cloprednol | ═ | ─Cl | ─H | ─OH | ─H | ─OH | ─OH | ✓ | |||
| 21-dehydrocloprednol | ═ | ─Cl | ─H | ─OH | ─H | ─OH | ═O | ✓ | |||
| Dichlorisone | ═ | ─H | ─Cl | ─Cl | ─H | ─OH | ─OH | ✓ | |||
| Diflorasone | ═ | ─F | ─F | ─OH | ─CH3 | ─OH | ─OH | ✓ | |||
| Diflucortolone valerate | ═ | ─F | ─F | ─OH | ─CH3 | ─H | ─OCOCH2CH2CH2CH3 | ✓ | |||
| Difluprednate | ═ | ─F | ─F | ─OH | ─H | ─OCOCH2CH2CH3 | ─OCOCH3 | ✓ | |||
| Fluocinolone acetonide | ═ | ─F | ─F | ─OH | 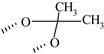 | ─OH | ✓ | ||||
| Fluocinonide | ═ | ─F | ─F | ─OH |  | ─OCOCH3 | ✓ | ||||
| Halcinonide | ─ | ─H | ─F | ─OH |  | ─Cl | ✓ | ||||
| Halobetasol | ═ | ─F | ─F | ─OH | ─CH3 | ─OH | ─Cl | ✓ | |||
| Halometasone | ═ | ─F | ─F | ─OH | ─CH3 | ─OH | ─OH | ✓ | |||
| Isofluprednone | ═ | ─H | ─F | ─OH | ─H | ─OH | ─OH | ||||
| Loteprednol etabonate | ═ | ─H | ─H | ─OH | ─H | ─OCOCOCH2CH3 | ─OCH2Cl | ✓ | |||
| Medrysone | ─ | ─CH3 | ─H | ─OH | ─H | ─H | ─CH3 | ✓ | |||
| Meprednisone | ═ | ─H | ─H | ═O | ─CH3 | ─OH | ─OH | ✓ | |||
| Paramethasone acetate | ═ | ─F | ─H | ─OH | ─CH3 | ─OH | ─OCOCH3 | ✓ | |||
| Prednicarbate | ═ | ─H | ─H | ─OH | ─H | 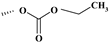 | ─OCOCH2CH3 | ✓ | |||
| Prednisolamate | ═ | ─H | ─H | ─OH | ─H | ─OH | 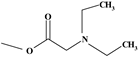 | ✓ | |||
| Resocortol | ─ | ─H | ─H | ─OH | ─H | ─OH | ─CH3 | ||||
| Tixocortol | ─ | ─H | ─H | ─OH | ─H | ─OH | ─SH | ✓ | |||
| Compound | Empirical Formula | Exact Mass | [M + H+] | RT ± SD (n = 168) | RRT (CV%) | Product Ions (m/z) /(Ion Ratio (%)) | Collision Energy (eV) |
|---|---|---|---|---|---|---|---|
| 6α-fluprednisolone | C21H27FO5 | 378.1843 | 379.1 | 3.30 ± 0.01 | 0.23 | 341.1 (100)/323.1 (68)/121 (66)/305.1 (60) | 10/10/35/10 |
| 21-desacetylamcinonide | C26H33FO6 | 460.2261 | 461.2 | 5.80 ± 0.01 | 0.12 | 357.1 (100)/339.1 (70)/213.1 (68)/441.1 (40) | 10/15/30/10 |
| 21-deoxyprednisolone | C21H28O4 | 344.1988 | 345.2 | 4.09 ± 0.01 | 0.14 | 327.1 (100)/147.1 (94)/309.1 (81)/171.1 (73) | 10/25/10/25 |
| Aclometasone17 21-dipropionate | C28H37ClO7 | 520.2228 | 521.2 | 9.18 ± 0.01 | 0.06 | 171.1 (100)/301.1 (89)/279.18 (76)/319.1(70) | 35/20/15/15 |
| Amcinonide | C28H35FO7 | 502.2367 | 503.2 | 9.14 ± 0.01 | 0.08 | 339.1 (100)/321.1 (72)/293.1 (51)/483.1 (32) | 15/20/20/10 |
| Clobetasone butyrate | C26H32ClFO5 | 478.1922 | 479.2 | 9.62 ± 0.00 | 0.03 | 343.1 (100)/279.1 (77)/371.1 (68)/266.2 (65) | 15/20/15/30 |
| Clocortolone pivalate | C27H36ClFO5 | 494.2235 | 495.2 | 9.73 ± 0.00 | 0.04 | 477.1 (100)/337.1 (97)/171.1 (82)/457.1 (57) | 10/15/25/15 |
| Cloprednol | C21H25ClO5 | 392.1391 | 393.1 | 4.28 ± 0.01 | 0.20 | 271.1 (100)/263.1 (97)/375.1 (46)/339.1 (15) | 20/20/10/15 |
| 21-dehydrocloprednol | C21H23ClO5 | 390.1234 | 391.1 | 3.92 ± 0.02 | 0.14 | 309.0 (100)/373 (92)/221.1 (55)/263.1 (37) | 15/10/30/30 |
| Dichlorisone | C21H26Cl2O4 | 412.1208 | 413.1 | 4.54 ± 0.02 | 0.17 | 237.1 (100)/121.1 (62)/377.1 (38)/135.1 (35) | 15/35/10/25 |
| Diflorasone | C22H28F2O5 | 410.1905 | 411.2 | 3.93 ± 0.01 | 0.12 | 121.1 (100)/253.1 (49)/135.1 (47)/371.1 (38) | 25/20/25/10 |
| Diflucortolone valerate | C27H36F2O5 | 478.2531 | 479.2 | 9.61 ± 0.00 | 0.04 | 121.1 (100)/355.1 (62)/439.1 (33)/375.1 (29) | 35/15/10/15 |
| Difluprednate | C27H34F2O7 | 508.2273 | 509.2 | 8.39 ± 0.01 | 0.14 | 303.1 (100)/279.1 (97)/261.1 (71)/321.1 (46) | 15/15/25/15 |
| Fluocinolone acetonide | C24H30F2O6 | 452.2011 | 453.2 | 4.60 ± 0.01 | 0.13 | 121.1 (100)/413.1 (48)/253.1 (40)/337.1 (35) | 35/10/15/15 |
| Fluocinonide | C26H32F2O37 | 494.2116 | 495.2 | 7.45 ± 0.01 | 0.16 | 121.1 (100)/337.1 (69)/319.1 (54)/291.4 (51) | 35/15/15/20 |
| Halcinonide | C24H32ClFO5 | 454.1922 | 455.2 | 8.85 ± 0.01 | 0.12 | 227.1 (100)/359.1 (66)/377.1 (48)/341.1 (46) | 25/25/25/25 |
| Halobetasol | C22H27ClF2O4 | 428.1566 | 429.1 | 6.30 ± 0.01 | 0.14 | 121.1 (100)/253.1 (70)/389 (66)/409 (25) | 30/20/10/10 |
| Halometasone | C22H27ClF2O5 | 444.1515 | 445.1 | 5.19 ± 0.01 | 0.15 | 155 (100)/287 (23)/369 (15)/307 (13) | 35/15/10/15 |
| Isofluprednone | C21H27FO5 | 378.1843 | 379.1 | 3.30 ± 0.01 | 0.20 | 341.1 (100)/147.1 (86)/237.1 (65)/265.1 (46) | 10/25/15/20 |
| Loteprednol etabonate | C24H31ClO7 | 466.1758 | 467.1 | 9.06 ± 0.01 | 0.08 | 265.1 (100)/359 (86)/147.1 (60)/171.1 (58) | 20/10/35/30 |
| Medrysone | C22H32O3 | 344.2352 | 345.2 | 7.43 ± 0.01 | 0.12 | 135.1 (100)/327.1 (77)/309.1 (50)/267.2 (15) | 20/15/15/20 |
| Meprednisone | C12H21NO4 | 243.1471 | 373.2 | 3.99 ± 0.00 | 0.11 | 147.1 (100)/171.1 (55)/355.1 (54)/337.1 (47) | 25/30/10/10 |
| Paramethasone acetate | C24H31FO6 | 434.2105 | 435.2 | 5.87 ± 0.01 | 0.17 | 319.1 (100)/171.1 (76)/337.1 (65)/121.1 (62) | 10/25/10/25 |
| Prednicarbate | C27H36O8 | 488.2410 | 489.2 | 8.99 ± 0.01 | 0.09 | 381.1 (100)/289.1 (90)/307.1 (61)/471.1 (35) | 10/15/15/10 |
| Prednisolamate | C27H39NO6 | 473.2777 | 474.2 | 3.02 ± 0.01 | 0.22 | 86.1 (100)/132.1 (30) | 35/30 |
| Resocortol | C22H32O4 | 360.2301 | 361.2 | 5.22 ± 0.01 | 0.13 | 325.1 (100)/269.1 (88)/121.1 (81)/287.2 (52) | 15/15/25/15 |
| Tixocortol | C21H30O4S | 378.1865 | 379.1 | 3.31 ± 0.01 | 0.15 | 147.2 (100)/121.1 (71) | 25/25 |
| Cortisol (Biomarker) | C21H30O5 | 362.2093 | 363 | 3.38 ± 0.01 | - | 121.0 | 20 |
| Cortisone (Biomarker) | C21H28O5 | 360.1937 | 361.5 | 3.41 ± 0.02 | - | 163.1 | 26 |
| Prednisone d4 (ISTD) | C21H22D4O5 | 362.2031 | 363.1 | 3.37 ± 0.02 | - | 269.1 | 15 |
| Substance | LOD (ng/mL) | LOQ (ng/mL) | Linearity (ng/mL) | R2 ± SD (n = 12) | RE (%)±SD [Mean] |
|---|---|---|---|---|---|
| 6α-fluprednisolone | 0.09 | 0.75 | 0.75–30 | 0.9723 ± 0.01 | 66.00 ± 1.79 |
| 21-desacetylamcinonide | 0.07 | 0.75 | 0.75–30 | 0.9704 ± 0.02 | 80.00 ± 3.50 |
| 21-deoxyprednisolone | 0.11 | 0.75 | 0.75–30 | 0.9724 ± 0.01 | 88.70 ± 4.16 |
| Aclometasone17 21-dipropionate | 0.14 | 0.75 | 0.75–30 | 0.9453 ± 0.04 | 80.30 ± 1.90 |
| Amcinonide | 0.15 | 0.75 | 0.75–30 | 0.9513 ± 0.02 | 93.70 ± 2.77 |
| Clobetasone butyrate | 0.12 | 0.75 | 0.75–30 | 0.9583 ± 0.01 | 97.30 ± 2.41 |
| Clocortolone pivalate | 0.14 | 0.75 | 0.75–30 | 0.9656 ± 0.00 | 99.80 ± 10.85 |
| Cloprednol | 0.04 | 0.75 | 0.75–30 | 0.9879 ± 0.01 | 65.40 ± 1.21 |
| 21-dehydrocloprednol | 0.12 | 0.75 | 0.75–30 | 0.9567 ± 0.03 | 78.70 ± 4.52 |
| Dichlorisone | 0.09 | 0.75 | 0.75–30 | 0.9691 ± 0.02 | 71.80 ± 1.25 |
| Diflorasone | 0.06 | 0.75 | 0.75–30 | 0.9697 ± 0.03 | 75.90 ± 1.03 |
| Diflucortolone valerate | 0.12 | 0.75 | 0.75–30 | 0.9571 ± 0.03 | 97.70 ± 4.59 |
| Difluprednate | 0.11 | 0.75 | 0.75–30 | 0.9476 ± 0.05 | 103.90 ± 1.50 |
| Fluocinolone acetonide | 0.07 | 0.75 | 0.75–30 | 0.9704 ± 0.02 | 82.50 ± 0.68 |
| Fluocinonide | 0.12 | 0.75 | 0.75–30 | 0.9648 ± 0.02 | 96.00 ± 2.33 |
| Halcinonide | 0.09 | 0.75 | 0.75–30 | 0.9737 ± 0.01 | 95.60 ± 1.40 |
| Halobetasol | 0.08 | 0.75 | 0.75–30 | 0.9729 ± 0.02 | 97.40 ± 3.42 |
| Halometasone | 0.11 | 0.75 | 0.75–30 | 0.9663 ± 0.02 | 78.60 ± 2.50 |
| Isofluprednone | 0.09 | 0.75 | 0.75–30 | 0.9641 ± 0.03 | 66.10 ± 3.80 |
| Loteprednol etabonate | 0.14 | 0.75 | 0.75–30 | 0.9536 ± 0.03 | 97.30 ± 0.56 |
| Medrysone | 0.09 | 0.75 | 0.75–30 | 0.9764 ± 0.02 | 99.90 ± 2.46 |
| Meprednisone | 0.06 | 0.75 | 0.75–30 | 0.9701 ± 0.02 | 77.50 ± 1.75 |
| Paramethasone acetate | 0.09 | 0.75 | 0.75–30 | 0.9743 ± 0.02 | 85.90 ± 1.61 |
| Prednicarbate | 0.12 | 0.75 | 0.75–30 | 0.9528 ± 0.03 | 87.30 ± 2.38 |
| Prednisolamate | 0.08 | 0.75 | 0.75–30 | 0.9859 ± 0.01 | 28.30 ± 1.04 |
| Resocortol | 0.09 | 0.75 | 0.75–30 | 0.9777 ± 0.01 | 87.00 ± 1.46 |
| Tixocortol | 0.09 | 0.75 | 0.75–30 | 0.9654 ± 0.03 | 77.40 ± 2.79 |
| Intra-Day | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Concentration (ng/mL) | |||||||||
| 3 | 7.5 | 15 | 22.5 | 30 | ||||||
| Precision (CV%) | Accuracy (%) | Precision (CV%) | Accuracy (%) | Precision (CV%) | Accuracy (%) | Precision (CV%) | Accuracy (%) | Precision (CV%) | Accuracy (%) | |
| 6α-fluprednisolone | 6.90 | 3.33 | 5.99 | 4.00 | 6.20 | 3.67 | 4.93 | 1.39 | 7.26 | 5.83 |
| 21-desacetylamcinonide | 10.54 | 0.001 | 11.14 | 3.67 | 4.80 | 0.08 | 5.92 | 5.83 | 6.01 | 3.54 |
| 21-deoxyprednisolone | 7.98 | 0.83 | 5.01 | 0.50 | 7.86 | 0.17 | 6.57 | 3.67 | 5.16 | 8.04 |
| Aclometasone17 21-dipropionate | 11.25 | 5.00 | 4.97 | 1.00 | 7.80 | 6.42 | 6.46 | 3.22 | 4.33 | 7.83 |
| Amcinonide | 8.75 | 6.67 | 9.68 | 8.17 | 4.43 | 0.58 | 6.36 | 4.89 | 4.75 | 9.58 |
| Clobetasone butyrate | 7.30 | 3.54 | 10.52 | 5.67 | 5.48 | 1.42 | 3.70 | 2.06 | 9.90 | 3.08 |
| Clocortolone pivalate | 9.96 | 2.08 | 11.96 | 0.33 | 6.95 | 1.58 | 5.52 | 1.33 | 9.62 | 4.63 |
| Cloprednol | 3.32 | 0.42 | 12.39 | 6.00 | 3.52 | 2.58 | 5.15 | 0.67 | 3.84 | 0.58 |
| 21-dehydrocloprednol | 6.78 | 5.42 | 8.05 | 2.33 | 4.86 | 0.17 | 2.17 | 1.67 | 5.11 | 4.96 |
| Dichlorisone | 7.51 | 5.83 | 6.31 | 3.50 | 2.66 | 0.83 | 5.57 | 2.78 | 7.06 | 3.50 |
| Diflorasone | 7.51 | 5.83 | 4.79 | 1.33 | 2.43 | 1.75 | 4.80 | 3.72 | 3.66 | 6.54 |
| Diflucortolone valerate | 6.53 | 2.50 | 3.78 | 2.00 | 4.96 | 2.17 | 6.65 | 2.67 | 6.58 | 0.96 |
| Difluprednate | 10.22 | 4.58 | 5.07 | 2.67 | 7.96 | 0.75 | 6.07 | 4.83 | 7.63 | 5.75 |
| Fluocinolone acetonide | 3.66 | 5.83 | 2.72 | 2.22 | 2.95 | 2.00 | 3.24 | 0.22 | 5.18 | 2.25 |
| Fluocinonide | 10.20 | 1.25 | 7.18 | 1.83 | 5.48 | 0.58 | 2.55 | 0.89 | 6.22 | 5.17 |
| Halcinonide | 7.73 | 3.75 | 2.51 | 1.83 | 7.47 | 2.42 | 2.61 | 1.11 | 3.01 | 0.75 |
| Halobetasol | 2.51 | 1.25 | 2.17 | 1.83 | 2.19 | 1.92 | 1.45 | 2.33 | 5.59 | 6.50 |
| Halometasone | 6.55 | 2.08 | 2.14 | 0.50 | 3.40 | 0.83 | 2.87 | 2.39 | 4.73 | 1.63 |
| Isofluprednone | 7.69 | 4.58 | 2.33 | 1.17 | 2.25 | 0.58 | 3.81 | 0.50 | 4.69 | 1.67 |
| Loteprednol etabonate | 2.22 | 3.75 | 3.22 | 1.67 | 4.51 | 1.67 | 2.35 | 1.72 | 4.75 | 3.71 |
| Medrysone | 8.45 | 1.25 | 10.02 | 4.33 | 5.83 | 2.08 | 6.06 | 3.28 | 6.92 | 6.92 |
| Meprednisone | 7.26 | 5.00 | 4.42 | 2.33 | 6.50 | 1.42 | 3.05 | 2.11 | 6.83 | 1.29 |
| Paramethasone acetate | 4.19 | 5.00 | 3.27 | 1.50 | 3.87 | 0.33 | 3.71 | 1.11 | 3.66 | 4.33 |
| Prednicarbate | 6.16 | 0.83 | 4.78 | 2.83 | 4.89 | 0.67 | 3.65 | 1.28 | 3.70 | 2.67 |
| Prednisolamate | 5.89 | 4.58 | 7.05 | 0.50 | 1.91 | 0.58 | 3.04 | 0.67 | 5.26 | 6.63 |
| Resocortol | 6.51 | 5.42 | 5.09 | 2.50 | 2.87 | 1.25 | 1.58 | 3.06 | 6.41 | 6.17 |
| Tixocortol | 5.61 | 0.83 | 1.86 | 2.83 | 3.85 | 1.83 | 2.99 | 1.83 | 3.51 | 0.54 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wicka, M.; Grucza, K.; Drapała, A.; Konarski, P.; Kwiatkowska, D. Determination of 27 Glucocorticoids in Urine by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Using UniSprayTM Source. Separations 2023, 10, 155. https://doi.org/10.3390/separations10030155
Wicka M, Grucza K, Drapała A, Konarski P, Kwiatkowska D. Determination of 27 Glucocorticoids in Urine by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Using UniSprayTM Source. Separations. 2023; 10(3):155. https://doi.org/10.3390/separations10030155
Chicago/Turabian StyleWicka, Mariola, Krzysztof Grucza, Aleksandra Drapała, Patryk Konarski, and Dorota Kwiatkowska. 2023. "Determination of 27 Glucocorticoids in Urine by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Using UniSprayTM Source" Separations 10, no. 3: 155. https://doi.org/10.3390/separations10030155
APA StyleWicka, M., Grucza, K., Drapała, A., Konarski, P., & Kwiatkowska, D. (2023). Determination of 27 Glucocorticoids in Urine by Ultra-High-Performance Liquid Chromatography-Tandem Mass Spectrometry Using UniSprayTM Source. Separations, 10(3), 155. https://doi.org/10.3390/separations10030155






