Filter Modified with Hydrophilic and Oleophobic Coating for Efficient and Affordable Oil/Water Separation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials and Equipment
2.2. Coated Filter Preparation
2.2.1. Refrigeration-Assisted Treatment
2.2.2. Microwave-Assisted Treatment
2.2.3. Sonication-Assisted Treatment
2.3. Porosity Ratio
2.4. Mass Distribution
2.5. Performance Testing
2.5.1. Pure Oil Filtration Test
2.5.2. Contact Angle Test
2.5.3. Photocatalytic Activity Assessment
2.6. Characterization
3. Results and Discussion
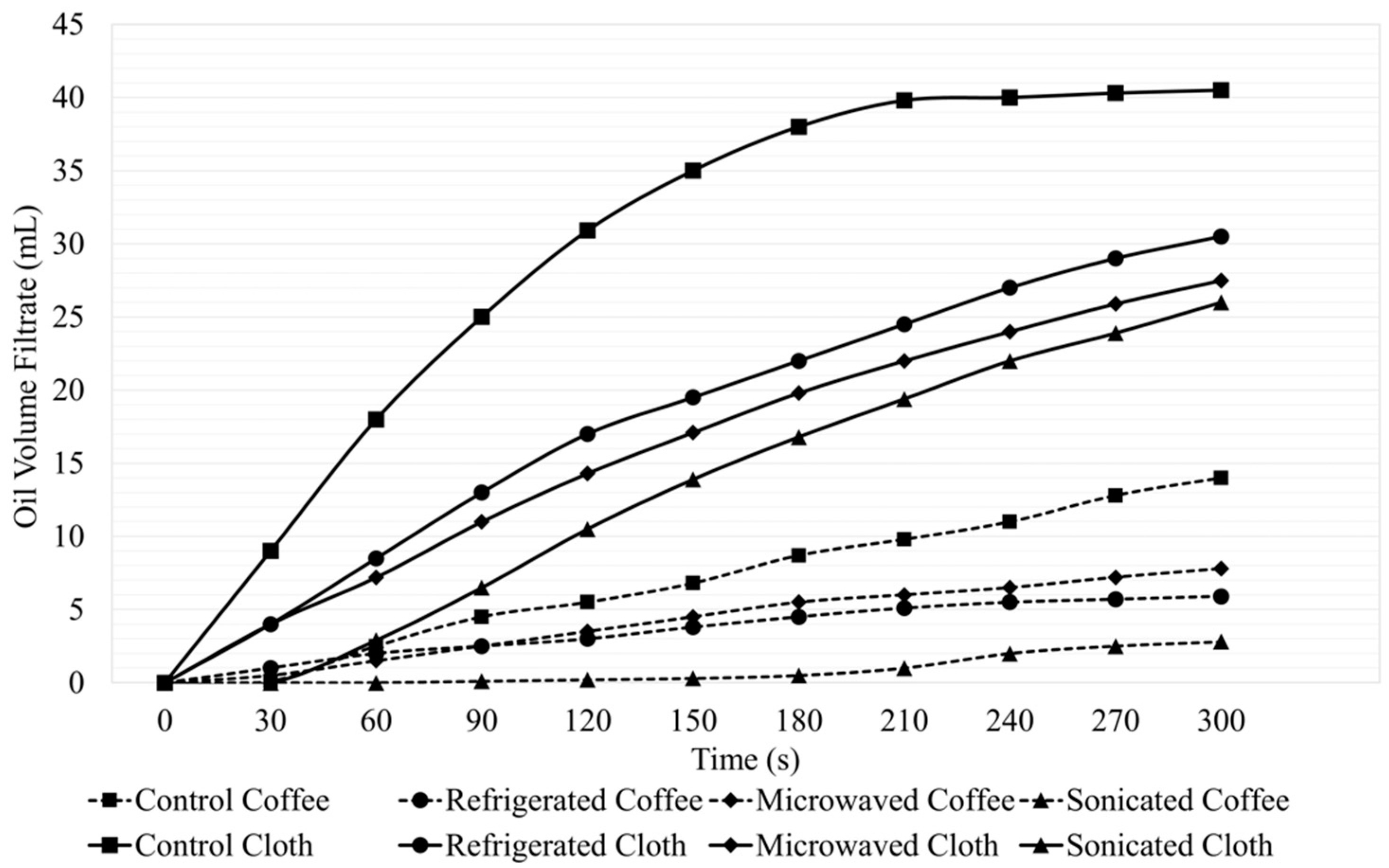
3.1. Oil Filtration and Oil–Water Separation Efficiency
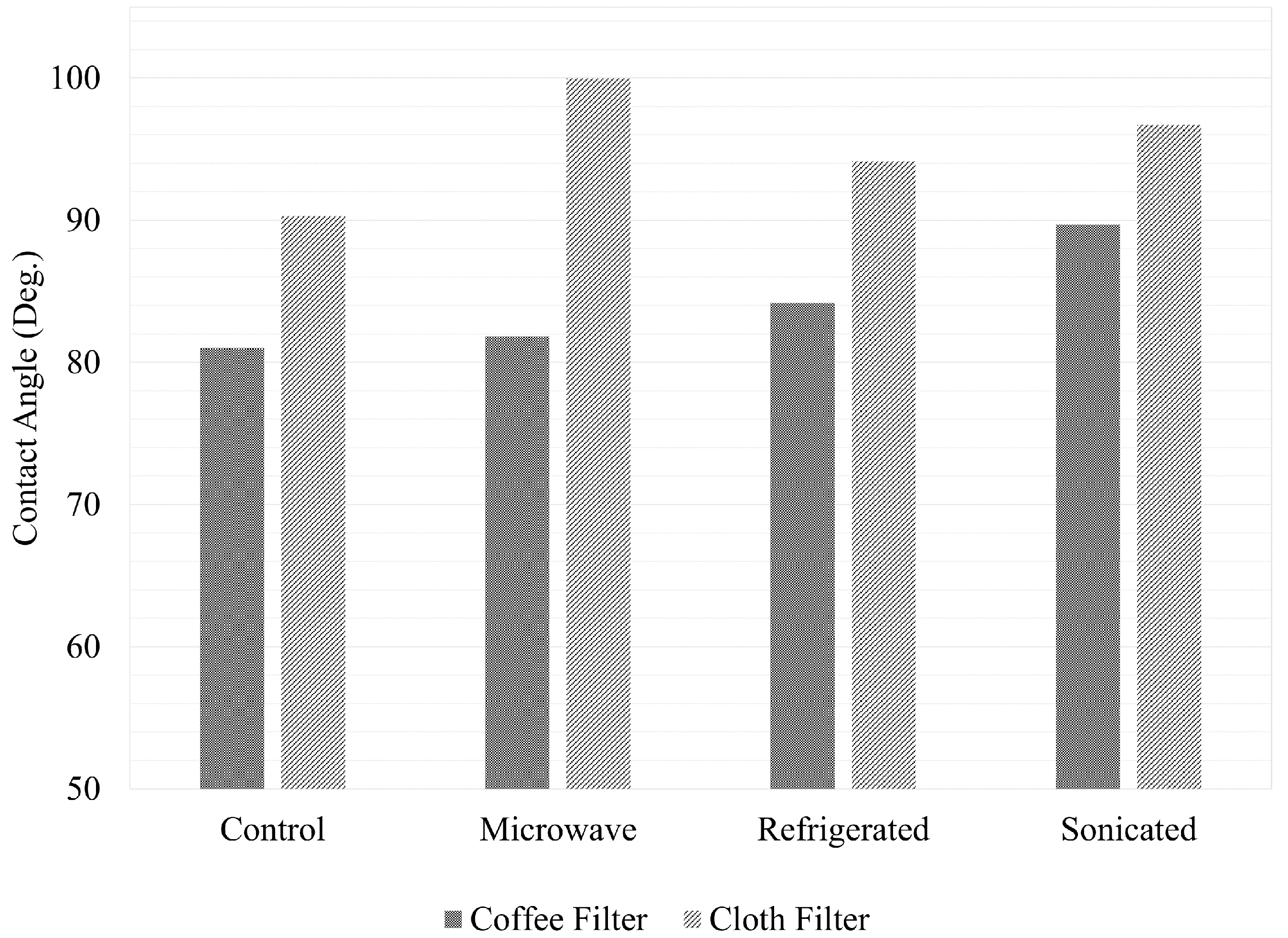
3.2. Wettability of Oil to Different Oil Filters
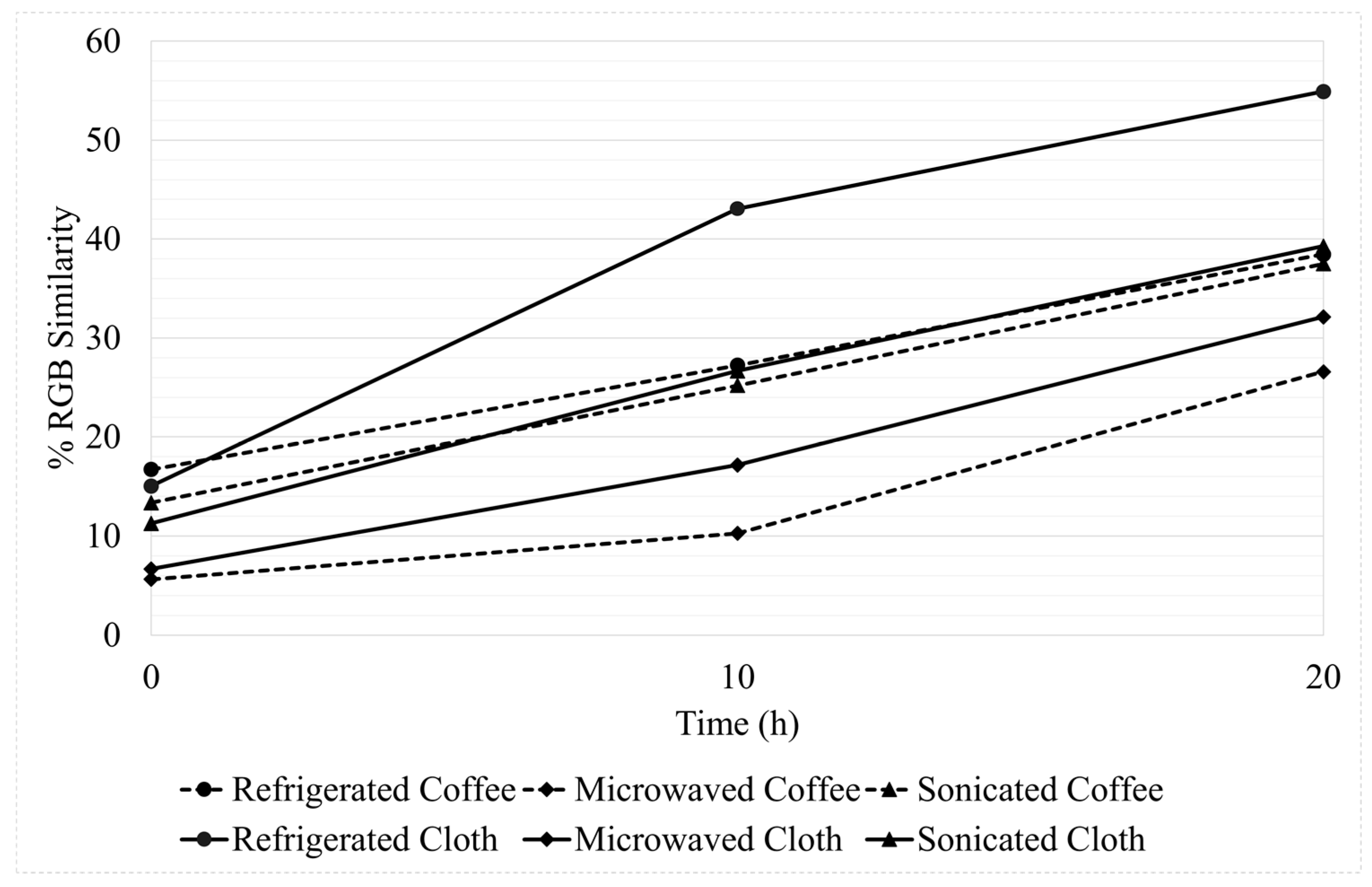
3.3. Photocatalytic Decomposition of Dye by the Coated Filters
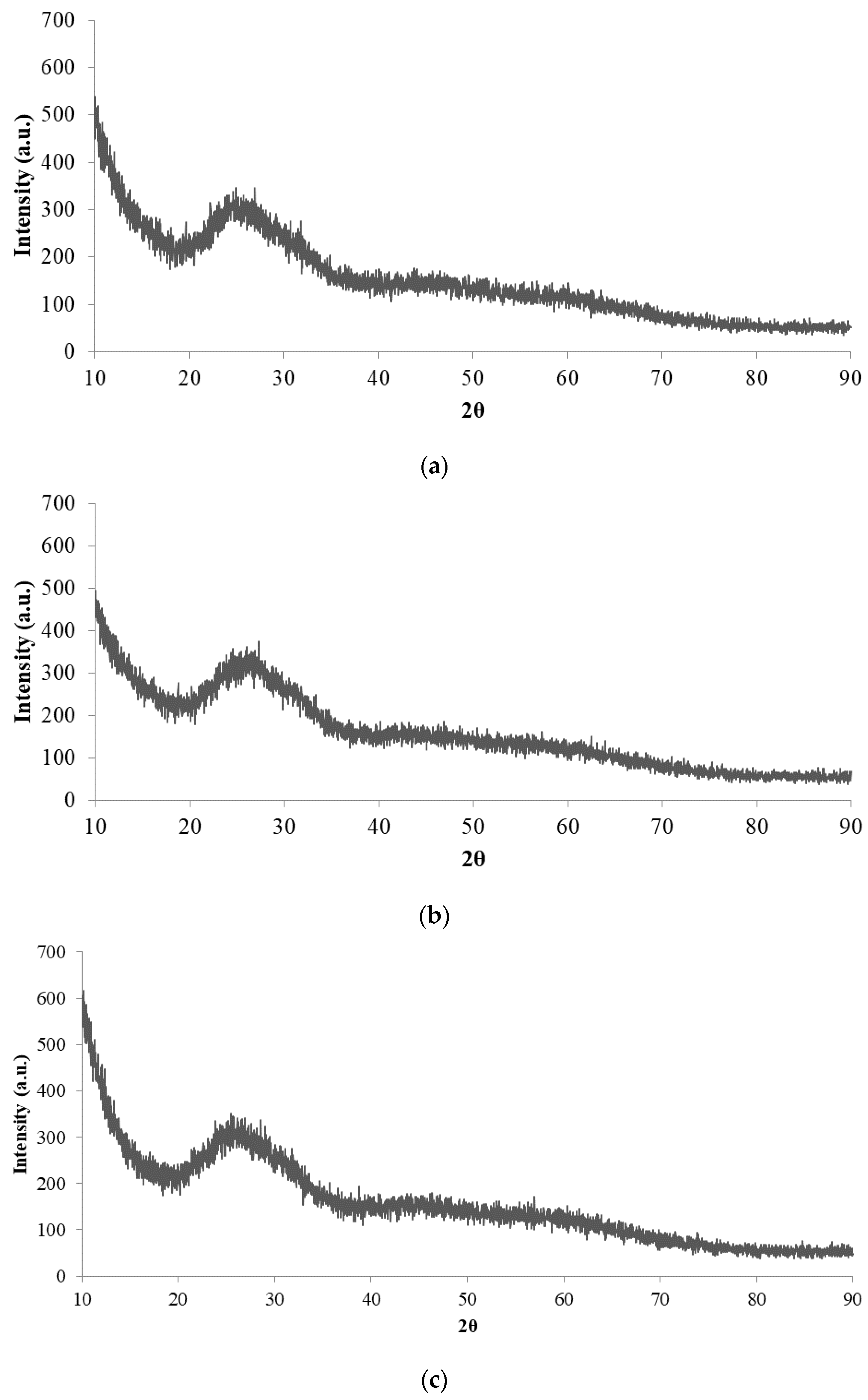
3.4. Structure of the Oxide Coating Determined by XRD
3.5. Reaction Mechanisms for Sol–Gel Coating Formation and Photocatalysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Huntington Beach 2022 Water Quality Report. City of Huntington Beach, California. Available online: https://huntingtonbeachca.gov/files/users/public_works/2022-Drinking-Water-Quality-Report.pdf (accessed on 16 August 2022).
- Office of Emergency and Remedial Response, Agency Response PB2000-963401 Oil Program Center Understanding Oil. United States Environmental Protection Agency. Available online: https://www.epa.gov/sites/default/files/2018-01/documents/ospguide99.pdf (accessed on 16 August 2022).
- Office of Response and Restoration. How Do Oil Spills out at Sea Typically Get Cleaned Up? National Oceanic and Atmospheric Administration. Available online: https://response.restoration.noaa.gov/about/media/how-do-oil-spills-out-sea-typically-get-cleaned.html (accessed on 15 August 2022).
- Nakajima, A.; Koizumi, S.I.; Watanabe, T.; Hashimoto, K. Photoinduced amphiphilic surface on polycrystalline anatase TiO2 thin films. Langmuir 2000, 16, 7048–7050. [Google Scholar] [CrossRef]
- Wang, Y.; Gong, X. Special oleophobic and hydrophilic surfaces: Approaches, mechanisms, and applications. J. Mater. Chem. A 2017, 5, 3759–3773. [Google Scholar] [CrossRef]
- Li, S.; Huang, J.; Ge, M.; Cao, C.; Deng, S.; Zhang, S.; Chen, G.; Zhang, K.; Al-Salem, S.; Lai, Y. Robust flower-like TiO2@Cotton fabrics with special wettability for effective self-cleaning and versatile oil/water separation. Adv. Mater. Interfaces 2015, 2, 1500220. [Google Scholar] [CrossRef]
- Shi, H.; He, Y.; Pan, Y.; Di, H.; Zeng, G.; Zhang, L.; Zhang, C. A modified mussel-inspired method to fabricate TiO2 decorated superhydrophilic PVDF membrane for oil/water separation. J. Membr. Sci. 2016, 506, 60–70. [Google Scholar] [CrossRef]
- Li, F.; Kong, W.; Zhao, X.; Pan, Y. Multifunctional TiO2-based superoleophobic/superhydrophilic coating for oil–water separation and oil purification. ACS Appl. Mater. Interfaces 2020, 12, 18074–18083. [Google Scholar] [CrossRef]
- Gao, C.; Sun, Z.; Li, K.; Chen, Y.; Cao, Y.; Zhang, S.; Feng, L. Integrated oil separation and water purification by a double-layer TiO2-based mesh. Energy Environ. Sci. 2013, 6, 1147–1151. [Google Scholar] [CrossRef]
- Banerjee, S.; Dionysiou, D.D.; Pillai, S.C. Self-cleaning applications of TiO2 by photo-induced hydrophilicity and photocatalysis. Appl. Catalysis B Environ. 2015, 176, 396–428. [Google Scholar] [CrossRef]
- Li, T.; Dong, C.; Liu, Y.; Wu, J.; Zhang, X.; Gong, X.; Zhao, W.; Wang, D.; Zhu, D. An anodized titanium/sol-gel composite coating with self-healable superhydrophobic and oleophobic property. Front. Mater. 2021, 8, 618674. [Google Scholar] [CrossRef]
- Wise, J.; Wise, J.P., Sr. A review of the toxicity of chemical dispersants. Rev. Envir. Health 2011, 26, 281–300. [Google Scholar] [CrossRef]
- Faksness, L.G.; Leirvik, F.; Taban, I.C.; Engen, F.; Jensen, H.V.; Holbu, J.W.; Dolva, H.; Bratveit, M. Offshore filed experiments with in-situ burning of oil: Emissions and burn efficiency. Environ. Res. 2022, 205, 112419. [Google Scholar] [CrossRef]
- Schneider, J.; Matsuoka, M.; Takeuchi, M.; Zhang, J.; Horiuchi, Y.; Anpo, M.; Bahnemann, D.W. Understanding TiO2 Photocatalysis: Mechanisms and Materials. Chem. Rev. 2014, 114, 9925–9933. [Google Scholar] [CrossRef] [PubMed]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysis for environmental applications. Appl. Catal. B Environ. 2012, 125, 332–334. [Google Scholar] [CrossRef]
- Vrakatseli, V.; Farsari, E.; Mataras, D. Wetting properties of transparent anatase/rutile mixed phase glancing angle magnetron sputtered nano-TiO2 films. Micromachines 2020, 11, 616. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, M.S.; Sadeghi, M.T. Improving oleophobicity and hydrophilicity of superhydrophobic surface by TiO2-based coatings. Mater. Res. Express 2018, 5, 085010. [Google Scholar] [CrossRef]
- Ao, Y.; Xu, J.; Fu, D.; Shen, X.; Yuan, C. Low temperature preparation of anatase TiO2-coated activated carbon. Colloids Surf. A Physicochem. Eng. Asp. 2007, 312, 125–127. [Google Scholar] [CrossRef]
- Sun, S.; Song, P.; Cui, J.; Liang, S. Amorphous TiO2 nanostructures: Synthesis, fundamental properties and photocatalytic applications. Catal. Sci. Technol. 2019, 16, 6–12. [Google Scholar] [CrossRef]
- Kameya, Y.; Yabe, H. Optical and Superhydrophilic Characteristics of TiO2 Coating with Subwavelength Surface Structure Consisting of Spherical Nanoparticle Aggregates. Coatings 2019, 9, 547. [Google Scholar] [CrossRef]
- Muller, P. Glossary of terms used in physical organic chemistry (IUPAC Recommendations 1994). Pure Appl. Chem. 1994, 66, 1077–1184. [Google Scholar] [CrossRef]
- Altawell, N. Financing for rural electrification. In Proceedings of the IEEE Conference on Computational Intelligence for Financial Engineering & Economics, New York, NY, USA, 29–30 March 2012. [Google Scholar]
- McKeen, L.W. Film Properties of Plastics and Elastomers, 4th ed.; William, A., Ed.; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1–23. [Google Scholar]
- Imoisili, P.E.; Jen, T.C.; Safaei, B. Microwave-assisted sol–gel synthesis of TiO2-mixed metal oxide nanocatalyst for degradation of organic pollutant. Nanotechnol. Rev. 2021, 10, 126–136. [Google Scholar] [CrossRef]
- Padrisi, E.; Rosa, R.; Baldi, G.; Dami, V.; Cioni, A.; Lorenzi, G.; Leonelli, C. Microwave-Assisted Vacuum Synthesis of TiO2 Nanocrystalline Powders in One-Pot, One-Step Prodedure. Nanomaterials 2021, 12, 149. [Google Scholar]
- Tao, Y.; Wu, C.Y.; Mazyck, D.W. Microwave-Assisted Preparation of TiO2/Activated Carbon Composite Photocatalyst for Removal of Methanol in Humid Air Stream. Ind. Eng. Chem. Res. 2006, 14, 5110–5116. [Google Scholar] [CrossRef]
- Guo, J.; Zhu, S.; Chen, Z.; Li, Y.; Yu, Z.; Liu, Q.; Li, J.; Feng, C.; Zhang, D. Sonochemical synthesis of TiO2 nanoparticles on graphene for use as photocatalyst. Ultrason. Sonochemistry 2011, 18, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Sadr, F.A.; Montazer, M. In situ sonosynthesis of nano TiO2 on cotton fabric. Ultrason. Sonochemistry 2014, 21, 681–691. [Google Scholar] [CrossRef]
- Lee, H.J. Improving superhydrophobic textiles materials. In Functional Textiles for Improved Performance, Protection and Health, 1st ed.; Pan, N., Sun, G., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 339–359. [Google Scholar]
- Moldoveanu, S.C.; David, V. RP-HPLC analytical columns. In Selection of the HPLC Method in Chemical Analysis; Elsevier: Boston, MA, USA, 2017; pp. 279–328. [Google Scholar]
- Sarkar, M.; Hasanuzzaman, M.; Gulshan, F.; Rashid, A. Surface, mechanical and shape memory properties of biodegradable polymers and their applications. Encycl. Mater. Plast. Polym. 2022, 2, 1092–1099. [Google Scholar]
- Contact Angle and Surface Tension—A Fascinating Liaison. Available online: https://www.cscscientific.com/csc-scientific-blog/how-does-contact-angle-relate-to-surface-tension#:~:text=A%20low%20contact%20angle%20(left,the%20quality%20of%20a%20liquid (accessed on 16 August 2022).
- Ye, L.; Zhang, Y.; Song, C.; Li, Y.; Jiang, B. A simple sol-gel method to prepare superhydrophilic silica coatings. Mater. Lett. 2017, 188, 316–318. [Google Scholar] [CrossRef]
- Wiranwetchayan, O.; Promnopas, S.; Thongtem, T.; Chaipanich, A.; Thongtem, S. Effect of alcohol solvents on TiO2 films prepared by sol–gel method. Surf. Coat. Technol. 2017, 326, 310–315. [Google Scholar] [CrossRef]
- Sadler, E.; Crick, C.R. Suction or gravity-fed oil-water separation using PDMS-coated glass filters. Sustain. Mater. Technol. 2021, 29, e00321. [Google Scholar] [CrossRef]
- Kroeger, R.M.; DeKay, H.G. Measurement of gravity filtration. J. Am. Pharm. Assoc. 1951, 40, 213–215. [Google Scholar] [CrossRef]
- Xue, C.H. Improving superhydrophobic coatings for textiles through chemical modifications. In Functional Textiles for Improved Performance, Protection and Health, 1st ed.; Pan, N., Sun, G., Eds.; Woodhead Publishing Limited: Cambridge, UK, 2011; Volume 2, pp. 320–338. [Google Scholar]
- Yan, X.; Ohno, T.; Nishijima, K.; Abe, R.; Ohtani, B. Is methylene blue an appropriate substrate for a photocatalytic activity test? A study with visible-light responsive titania. Chem. Phys. Lett. 2006, 429, 606–610. [Google Scholar] [CrossRef]
- Ungan, H.; Tekin, T. Effect of the sonication and coating time on the photocatalytic degradation of TiO2, TiO2-Ag, and TiO2-ZnO thin film photocatalysts. Chem. Eng. Commun. 2020, 207, 896–903. [Google Scholar] [CrossRef]
- Nishiyama, N.; Yokoyama, T. Permeability of porous media: Role of the critical pore size. J. Geophys. Res. Solid Earth 2017, 122, 6955–6971. [Google Scholar] [CrossRef]
- Drelich, J. The effect of drop (bubble) size on contact angle at solid surfaces. J. Adhes. 1997, 63, 31–51. [Google Scholar] [CrossRef]
- Ren, H.; Xu, S.; Wu, S.T. Effects of gravity on the shape of liquid droplets. Opt. Commun. 2010, 283, 3255–3258. [Google Scholar] [CrossRef]
- Nowak, B.; Bonora, M.; Zuzga, M.; Werner, L.; Jackiewicz-Zagorska, A.; Gac, J.M. MTMS-based aerogel structure deposition on propylene fibrous filter—Surface layer effect and distribution control for improvement of oil aerosol separation properties. J. Environ. Chem. Eng. 2022, 10, 108410. [Google Scholar] [CrossRef]

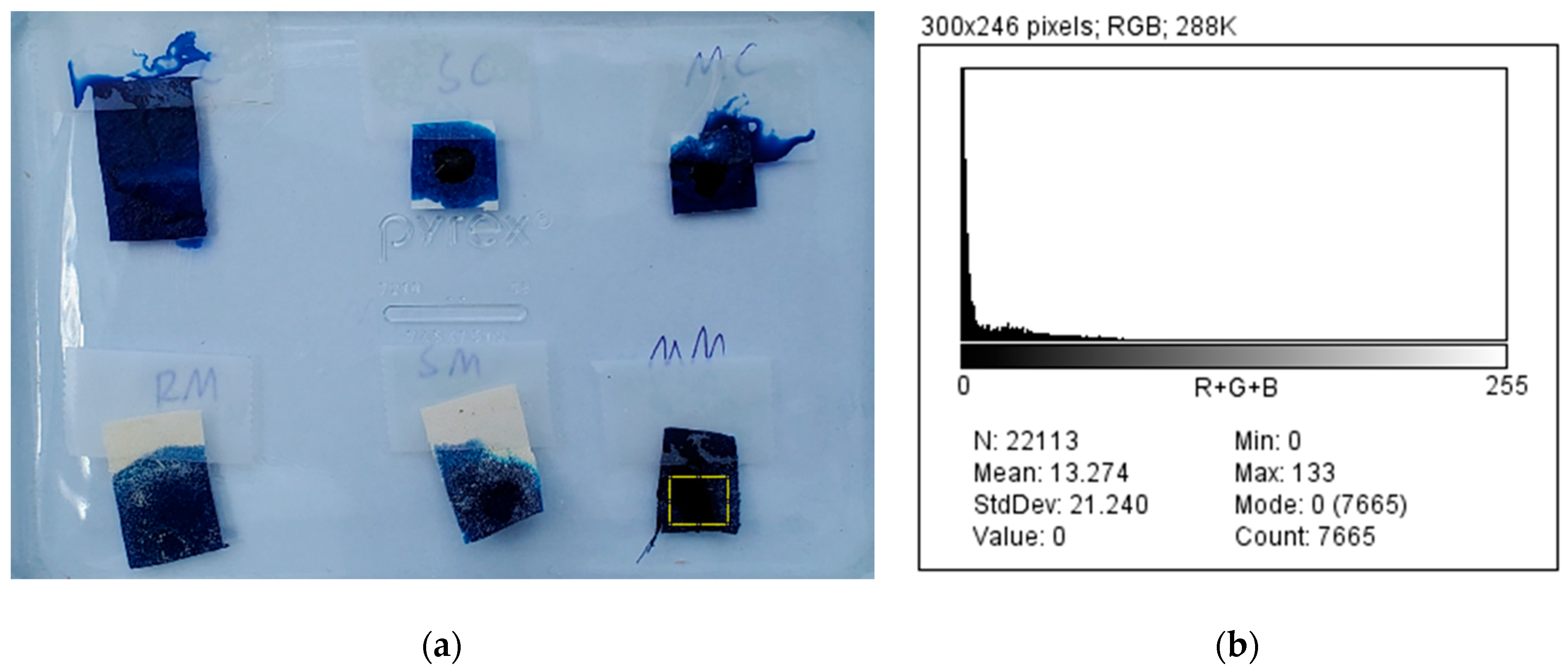

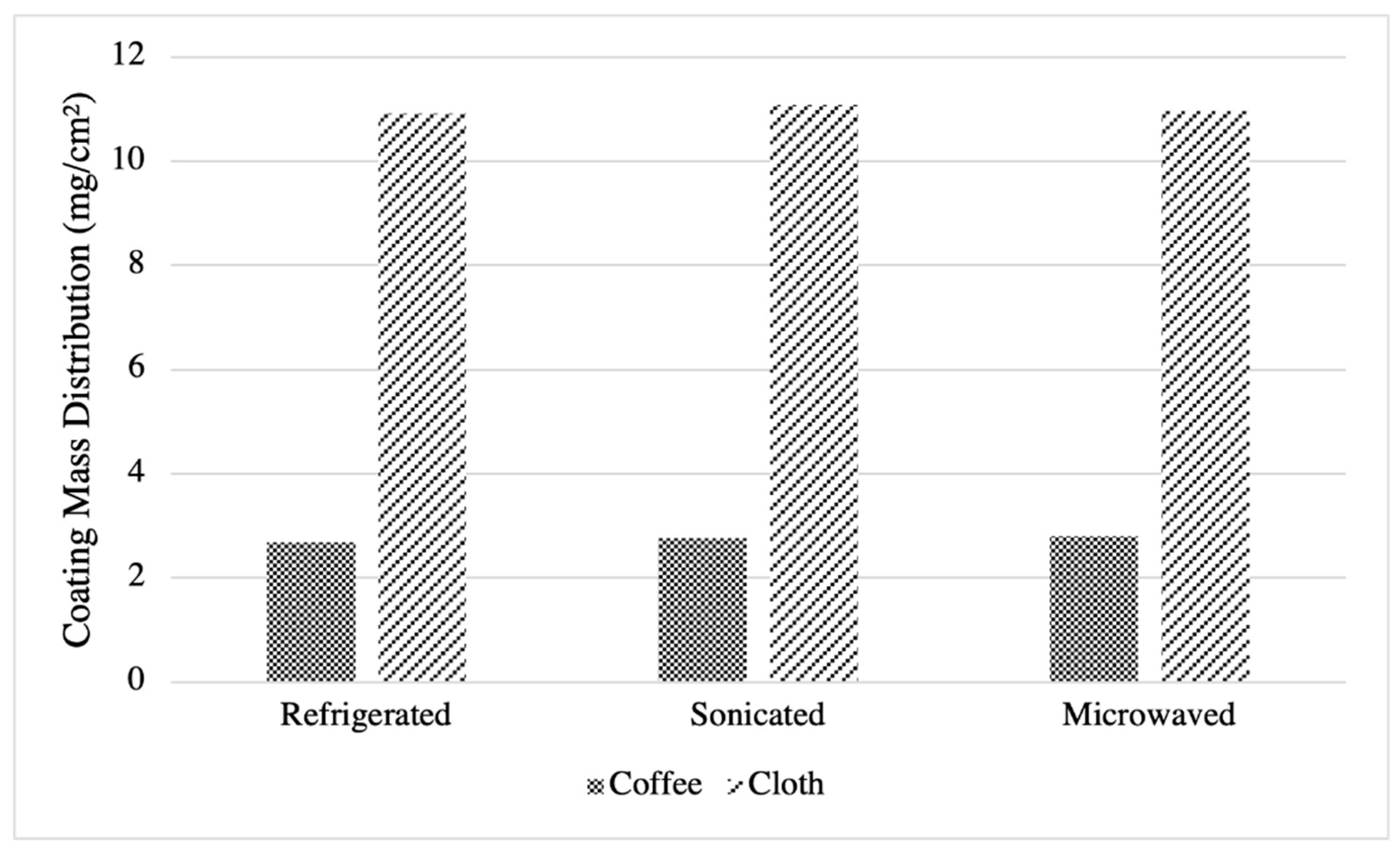

| Coffee Filter | Cloth | |
|---|---|---|
| Mass of soaked hexane (g) | 0.20 | 0.80 |
| Porosity ratio (φcoffee/φcloth) | 0.25 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ross, H.; Nguyen, H.; Nguyen, B.; Foster, A.; Salud, J.; Patino, M.; Gan, Y.X.; Li, M. Filter Modified with Hydrophilic and Oleophobic Coating for Efficient and Affordable Oil/Water Separation. Separations 2022, 9, 269. https://doi.org/10.3390/separations9100269
Ross H, Nguyen H, Nguyen B, Foster A, Salud J, Patino M, Gan YX, Li M. Filter Modified with Hydrophilic and Oleophobic Coating for Efficient and Affordable Oil/Water Separation. Separations. 2022; 9(10):269. https://doi.org/10.3390/separations9100269
Chicago/Turabian StyleRoss, Hunter, Huyen Nguyen, Brian Nguyen, Ashton Foster, James Salud, Mike Patino, Yong X. Gan, and Mingheng Li. 2022. "Filter Modified with Hydrophilic and Oleophobic Coating for Efficient and Affordable Oil/Water Separation" Separations 9, no. 10: 269. https://doi.org/10.3390/separations9100269
APA StyleRoss, H., Nguyen, H., Nguyen, B., Foster, A., Salud, J., Patino, M., Gan, Y. X., & Li, M. (2022). Filter Modified with Hydrophilic and Oleophobic Coating for Efficient and Affordable Oil/Water Separation. Separations, 9(10), 269. https://doi.org/10.3390/separations9100269