Cardiovascular Risks Associated with Gender and Aging
Abstract
1. Introduction
2. Pathophysiology of CVD in Aged Adults
3. Prevalence of Cardiovascular Diseases in Aging and Elderly Adults
4. The Prevalence of Aging Adults Admitted to Critical Care
5. Management and Treatment of CVD in Older Adults
6. Risk Factors Associated with Cardiovascular Diseases among the Elderly
7. Diabetes in the Elderly Population
8. Obesity in the Elderly Population
9. Frailty in the Elderly Population
10. Sex Differences That Arise from Hormones in Aging Adults
11. The Impact of Estrogen on CVD in Aging Adults
12. The Impact of Testosterone on CVD in Aging Adults
13. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Curtis, A.B.; Karki, R.; Hattoum, A.; Sharma, U.C. Arrhythmias in Patients ≥ 80 Years of Age: Pathophysiology, Management, and Outcomes. J. Am. Coll. Cardiol. 2018, 71, 2041–2057. [Google Scholar] [CrossRef] [PubMed]
- North, B.J.; Sinclair, D.A. The intersection between aging and cardiovascular disease. Circ. Res. 2012, 110, 1097–1108. [Google Scholar] [CrossRef]
- Yazdanyar, A.; Newman, A.B. The burden of cardiovascular disease in the elderly: Morbidity, mortality, and costs. Clin. Geriatr. Med. 2009, 25, 563–577. [Google Scholar] [CrossRef]
- Garcia, M.; Mulvagh, S.L.; Merz, C.N.; Buring, J.E.; Manson, J.E. Cardiovascular Disease in Women: Clinical Perspectives. Circ. Res. 2016, 118, 1273–1293. [Google Scholar] [CrossRef] [PubMed]
- Keto, J.; Ventola, H.; Jokelainen, J.; Linden, K.; Keinanen-Kiukaanniemi, S.; Timonen, M.; Ylisaukko-Oja, T.; Auvinen, J. Cardiovascular disease risk factors in relation to smoking behaviour and history: A population-based cohort study. Open Heart 2016, 3, e000358. [Google Scholar] [CrossRef] [PubMed]
- Benjamin, E.J.; Muntner, P.; Alonso, A.; Bittencourt, M.S.; Callaway, C.W.; Carson, A.P.; Chamberlain, A.M.; Chang, A.R.; Cheng, S.; Das, S.R.; et al. Heart Disease and Stroke Statistics-2019 Update: A Report From the American Heart Association. Circulation 2019. [Google Scholar] [CrossRef]
- Nettleship, J.E.; Jones, R.D.; Channer, K.S.; Jones, T.H. Testosterone and coronary artery disease. Front Horm. Res. 2009, 37, 91–107. [Google Scholar] [CrossRef] [PubMed]
- Iorga, A.; Cunningham, C.M.; Moazeni, S.; Ruffenach, G.; Umar, S.; Eghbali, M. The protective role of estrogen and estrogen receptors in cardiovascular disease and the controversial use of estrogen therapy. Biol. Sex. Differ. 2017, 8, 33. [Google Scholar] [CrossRef]
- Steenman, M.; Lande, G. Cardiac aging and heart disease in humans. Biophys. Rev. 2017, 9, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Meschiari, C.A.; Ero, O.K.; Pan, H.; Finkel, T.; Lindsey, M.L. The impact of aging on cardiac extracellular matrix. Geroscience 2017, 39, 7–18. [Google Scholar] [CrossRef]
- Martos, R.; Baugh, J.; Ledwidge, M.; O’Loughlin, C.; Conlon, C.; Patle, A.; Donnelly, S.C.; McDonald, K. Diastolic heart failure: evidence of increased myocardial collagen turnover linked to diastolic dysfunction. Circulation 2007, 115, 888–895. [Google Scholar] [CrossRef]
- Burstein, B.; Nattel, S. Atrial fibrosis: Mechanisms and clinical relevance in atrial fibrillation. J. Am. Coll. Cardiol. 2008, 51, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Martin-Fernandez, B.; Gredilla, R. Mitochondria and oxidative stress in heart aging. Age (Dordr) 2016, 38, 225–238. [Google Scholar] [CrossRef] [PubMed]
- Nakou, E.S.; Parthenakis, F.L.; Kallergis, E.M.; Marketou, M.E.; Nakos, K.S.; Vardas, P.E. Healthy aging and myocardium: A complicated process with various effects in cardiac structure and physiology. Int. J. Cardiol. 2016, 209, 167–175. [Google Scholar] [CrossRef]
- Xie, W.; Santulli, G.; Reiken, S.R.; Yuan, Q.; Osborne, B.W.; Chen, B.X.; Marks, A.R. Mitochondrial oxidative stress promotes atrial fibrillation. Sci. Rep. 2015, 5, 11427. [Google Scholar] [CrossRef]
- Babusikova, E.; Lehotsky, J.; Dobrota, D.; Racay, P.; Kaplan, P. Age-associated changes in Ca(2+)-ATPase and oxidative damage in sarcoplasmic reticulum of rat heart. Physiol. Res. 2012, 61, 453–460. [Google Scholar]
- Carew, T.E. Role of biologically modified low-density lipoprotein in atherosclerosis. Am. J. Cardiol. 1989, 64, 18G–22G. [Google Scholar] [CrossRef]
- Kain, V.; Ingle, K.A.; Kachman, M.; Baum, H.; Shanmugam, G.; Rajasekaran, N.S.; Young, M.E.; Halade, G.V. Excess omega-6 fatty acids influx in aging drives metabolic dysregulation, electrocardiographic alterations, and low-grade chronic inflammation. Am. J. Physiol Heart Circ. Physiol 2018, 314, H160–H169. [Google Scholar] [CrossRef]
- Lu, Y.; Hajifathalian, K.; Ezzati, M.; Woodward, M.; Rimm, E.B.; Danaei, G.; Selmer, R.; Strand, B.H.; Fang, X.; et al. Metabolic mediators of the effects of body-mass index, overweight, and obesity on coronary heart disease and stroke: A pooled analysis of 97 prospective cohorts with 1·8 million participants. Lancet 2014, 383, 970–983. [Google Scholar] [PubMed]
- Lloyd-Sherlock, P.; Beard, J.; Minicuci, N.; Ebrahim, S.; Chatterji, S. Hypertension among older adults in low- and middle-income countries: prevalence, awareness and control. Int. J. Epidemiol. 2014, 43, 116–128. [Google Scholar] [CrossRef]
- Upadhya, B.; Taffet, G.E.; Cheng, C.P.; Kitzman, D.W. Heart failure with preserved ejection fraction in the elderly: Scope of the problem. J. Mol. Cell. Cardiol. 2015, 83, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.; Griffiths, M.; Lee, K.K.; McAllister, D.A.; Hunter, A.L.; Ferry, A.V.; Cruikshank, A.; Reid, A.; Stoddart, M.; Strachan, F.; et al. High sensitivity cardiac troponin and the under-diagnosis of myocardial infarction in women: Prospective cohort study. BMJ 2015, 350, g7873. [Google Scholar] [CrossRef] [PubMed]
- Elsaesser, A.; Hamm, C.W. Acute coronary syndrome—The risk of being female. Circulation 2004, 109, 565–567. [Google Scholar] [CrossRef] [PubMed]
- Docherty, A.B.; Alam, S.; Shah, A.S.; Moss, A.; Newby, D.E.; Mills, N.L.; Stanworth, S.J.; Lone, N.I.; Walsh, T.S.; Investigators, T. Unrecognised myocardial infarction and its relationship to outcome in critically ill patients with cardiovascular disease. Intensive Care Med. 2018, 44, 2059–2069. [Google Scholar] [CrossRef]
- Canto, J.G.; Goldberg, R.J.; Hand, M.M.; Bonow, R.O.; Sopko, G.; Pepine, C.J.; Long, T. Symptom presentation of women with acute coronary syndromes—Myth vs. reality. Arch. Intern. Med. 2007, 167, 2405–2413. [Google Scholar] [CrossRef]
- Afilalo, J.; Karunananthan, S.; Eisenberg, M.J.; Alexander, K.P.; Bergman, H. Role of Frailty in Patients With Cardiovascular Disease. Am. J. Cardiol. 2009, 103, 1616–1621. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, N.T.; Schilling, R.J. Sudden Cardiac Death and Arrhythmias. Arrhythm. Electrophysiol. Rev. 2018, 7, 111–117. [Google Scholar] [CrossRef]
- Ama Moor, V.J.; Nansseu, J.R.N.; Nouaga, M.E.D.; Noubiap, J.J.N.; Nguetsa, G.D.; Tchanana, G.; Ketcha, A.; Fokom-Domgue, J. Assessment of the 10-year risk of cardiovascular events among a group of Sub-Saharan African post-menopausal women. Cardiol. J. 2016, 23, 123–131. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wunsch, H.; Angus, D.C.; Harrison, D.A.; Linde-Zwirble, W.T.; Rowan, K.M. Comparison of medical admissions to intensive care units in the United States and United Kingdom. Am. J. Respir. Crit. Care Med. 2011, 183, 1666–1673. [Google Scholar] [CrossRef]
- Nielsson, M.S.; Christiansen, C.F.; Johansen, M.B.; Rasmussen, B.S.; Tonnesen, E.; Norgaard, M. Mortality in elderly ICU patients: A cohort study. Acta Anaesthesiol. Scand. 2014, 58, 19–26. [Google Scholar] [CrossRef]
- Fuchs, L.; Chronaki, C.E.; Park, S.; Novack, V.; Baumfeld, Y.; Scott, D.; McLennan, S.; Talmor, D.; Celi, L. ICU admission characteristics and mortality rates among elderly and very elderly patients. Intensive Care Med. 2012, 38, 1654–1661. [Google Scholar] [CrossRef]
- Peigne, V.; Somme, D.; Guérot, E.; Lenain, E.; Chatellier, G.; Fagon, J.-Y.; Saint-Jean, O. Treatment intensity, age and outcome in medical ICU patients: Results of a French administrative database. Ann. Intensive Care 2016, 6, 7. [Google Scholar] [CrossRef]
- Kasaoka, S. Evolved role of the cardiovascular intensive care unit (CICU). J. Intensive Care 2017, 5, 72. [Google Scholar] [CrossRef]
- Feng, Y.; Amoateng-Adjepong, Y.; Kaufman, D.; Gheorghe, C.; Manthous, C.A. Age, duration of mechanical ventilation, and outcomes of patients who are critically ill. Chest 2009, 136, 759–764. [Google Scholar] [CrossRef]
- Behrendt, C.E. Acute respiratory failure in the United States: Incidence and 31-day survival. Chest 2000, 118, 1100–1105. [Google Scholar] [CrossRef]
- El Solh, A.A.; Bhat, A.; Gunen, H.; Berbary, E. Extubation failure in the elderly. Respir. Med. 2004, 98, 661–668. [Google Scholar] [CrossRef]
- Su, K.-C.; Tsai, C.-C.; Chou, K.-T.; Lu, C.-C.; Liu, Y.-Y.; Chen, C.-S.; Wu, Y.-C.; Lee, Y.-C.; Perng, D.-W. Spontaneous breathing trial needs to be prolonged in critically ill and older patients requiring mechanical ventilation. J. Crit. Care 2012, 27, 324.e321–324.e327. [Google Scholar] [CrossRef]
- Suraseranivong, R.; Krairit, O.; Theerawit, P.; Sutherasan, Y. Association between age-related factors and extubation failure in elderly patients. PLoS ONE 2018, 13, e0207628. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Iyer, D.; Rodgers, L.E.; Vanthenapalli, S.; Panguluri, S.K. Impact of hyperoxia on cardiac pathophysiology. J. Cell. Physiol. 2019. [Google Scholar] [CrossRef]
- Han, C.H.; Guan, Z.B.; Zhang, P.X.; Fang, H.L.; Li, L.; Zhang, H.M.; Zhou, F.J.; Mao, Y.F.; Liu, W.W. Oxidative stress induced necroptosis activation is involved in the pathogenesis of hyperoxic acute lung injury. Biochem. Biophys. Res. Commun 2018, 495, 2178–2183. [Google Scholar] [CrossRef]
- Sato, T.; Paquet-Fifield, S.; Harris, N.C.; Roufail, S.; Turner, D.J.; Yuan, Y.; Zhang, Y.F.; Fox, S.B.; Hibbs, M.L.; Wilkinson-Berka, J.L.; et al. VEGF-D promotes pulmonary oedema in hyperoxic acute lung injury. J. Pathol. 2016, 239, 152–161. [Google Scholar] [CrossRef]
- Pannu, S.R. Too Much Oxygen: Hyperoxia and Oxygen Management in Mechanically Ventilated Patients. Semin. Respir. Crit. Care Med. 2016, 37, 16–22. [Google Scholar] [CrossRef]
- Smit, B.; Smulders, Y.M.; van der Wouden, J.C.; Oudemans-van Straaten, H.M.; Spoelstra-de Man, A.M.E. Hemodynamic effects of acute hyperoxia: Systematic review and meta-analysis. Crit. Care 2018, 22, 45. [Google Scholar] [CrossRef]
- Sinski, M.; Lewandowski, J.; Przybylski, J.; Zalewski, P.; Symonides, B.; Abramczyk, P.; Gaciong, Z. Deactivation of carotid body chemoreceptors by hyperoxia decreases blood pressure in hypertensive patients. Hypertens. Res. 2014, 37, 858–862. [Google Scholar] [CrossRef]
- Baleeiro, C.E.; Wilcoxen, S.E.; Morris, S.B.; Standiford, T.J.; Paine, R. Sublethal hyperoxia impairs pulmonary innate immunity. J. Immunol 2003, 171, 955–963. [Google Scholar] [CrossRef]
- Barazzone, C.; Horowitz, S.; Donati, Y.R.; Rodriguez, I.; Piguet, P.F. Oxygen toxicity in mouse lung: Pathways to cell death. Am. J. Respir. Cell Mol. Biol 1998, 19, 573–581. [Google Scholar] [CrossRef]
- Davis, D.P.; Meade, W.; Sise, M.J.; Kennedy, F.; Simon, F.; Tominaga, G.; Steele, J.; Coimbra, R. Both Hypoxemia and Extreme Hyperoxemia May Be Detrimental in Patients with Severe Traumatic Brain Injury. J. Neurotrauma 2009, 26, 2217–2223. [Google Scholar] [CrossRef]
- de Jonge, E.; Peelen, L.; Keijzers, P.; Joore, H.; de Lange, D.; van der Voort, P.; Bosman, R.; de Waal, R.; Wesselink, R.; de Keizer, N. Association between administered oxygen, arterial partial oxygen pressure and mortality in mechanically ventilated intensive care unit patients. Crit. Care 2008, 12, R156. [Google Scholar] [CrossRef]
- Farquhar, H.; Weatherall, M.; Wijesinghe, M.; Perrin, K.; Ranchord, A.; Simmonds, M.; Beasley, R. Systematic review of studies of the effect of hyperoxia on coronary blood flow. Am. Heart J. 2009, 158, 371–377. [Google Scholar] [CrossRef]
- Janz, D.R.; Hollenbeck, R.D.; Pollock, J.S.; McPherson, J.A.; Rice, T.W. Hyperoxia is associated with increased mortality in patients treated with mild therapeutic hypothermia after sudden cardiac arrest. Crit. Care Med. 2012, 40, 3135–3139. [Google Scholar] [CrossRef]
- Kilgannon, J.H.; Jones, A.E.; Shapiro, N.I.; Angelos, M.G.; Milcarek, B.; Hunter, K.; Parrillo, J.E.; Trzeciak, S. Association Between Arterial Hyperoxia Following Resuscitation From Cardiac Arrest and In-Hospital Mortality. Jama-J. Am. Med. Assoc. 2010, 303, 2165–2171. [Google Scholar] [CrossRef]
- Li, L.F.; Liao, S.K.; Ko, Y.S.; Lee, C.H.; Quinn, D.A. Hyperoxia increases ventilator-induced lung injury via mitogen-activated protein kinases: A prospective, controlled animal experiment. Crit. Care 2007, 11, R25. [Google Scholar] [CrossRef]
- Nagato, A.; Silva, F.L.; Silva, A.R.; Bezerra, F.S.; Oliveira, M.L.; Bello-Klein, A.; Porto, L.C.; Valenca, S.S. Hyperoxia-Induced Lung Injury Is Dose Dependent in Wistar Rats. Exp. Lung Res. 2009, 35, 713–728. [Google Scholar] [CrossRef]
- Page, D.; Ablordeppey, E.; Wessman, B.T.; Mohr, N.M.; Trzeciak, S.; Kollef, M.H.; Roberts, B.W.; Fuller, B.M. Emergency department hyperoxia is associated with increased mortality in mechanically ventilated patients: A cohort study. Crit. Care 2018, 22, 9. [Google Scholar] [CrossRef]
- Damiani, E.; Donati, A.; Girardis, M. Oxygen in the critically ill: Friend or foe? Curr. Opin. Anaesthesiol. 2018, 31, 129–135. [Google Scholar] [CrossRef]
- Damiani, E.; Adrario, E.; Girardis, M.; Romano, R.; Pelaia, P.; Singer, M.; Donati, A. Arterial hyperoxia and mortality in critically ill patients: A systematic review and meta-analysis. Crit. Care 2014, 18, 711. [Google Scholar] [CrossRef]
- Rincon, F.; Kang, J.; Maltenfort, M.; Vibbert, M.; Urtecho, J.; Athar, M.K.; Jallo, J.; Pineda, C.C.; Tzeng, D.; McBride, W.; et al. Association between hyperoxia and mortality after stroke: A multicenter cohort study. Crit. Care Med. 2014, 42, 387–396. [Google Scholar] [CrossRef]
- Stub, D.; Smith, K.; Bernard, S.; Nehme, Z.; Stephenson, M.; Bray, J.E.; Cameron, P.; Barger, B.; Ellims, A.H.; Taylor, A.J.; et al. Air Versus Oxygen in ST-Segment-Elevation Myocardial Infarction. Circulation 2015, 131, 2143–2150. [Google Scholar] [CrossRef]
- Peng, Y.W.; Mohammed, A.; Deatrick, K.B.; Major, T.; Cheng, D.; Charpie, I.; Charpie, J.R. Differential Effects of Normoxic and Hyperoxic Reperfusion on Global Myocardial Ischemia-Reperfusion Injury. Semin. Thorac. Cardiovasc. Surg. 2018. [Google Scholar] [CrossRef]
- de Visser, Y.P.; Walther, F.J.; Laghmani el, H.; Boersma, H.; van der Laarse, A.; Wagenaar, G.T. Sildenafil attenuates pulmonary inflammation and fibrin deposition, mortality and right ventricular hypertrophy in neonatal hyperoxic lung injury. Respir. Res. 2009, 10, 30. [Google Scholar] [CrossRef]
- Panguluri, S.K.; Tur, J.; Fukumoto, J.; Deng, W.; Sneed, K.B.; Kolliputi, N.; Bennett, E.S.; Tipparaju, S.M. Hyperoxia-induced hypertrophy and ion channel remodeling in left ventricle. Am. J. Physiol. Heart Circ. Physiol. 2013, 304, H1651–H1661. [Google Scholar] [CrossRef]
- Chapalamadugu, K.C.; Panguluri, S.K.; Bennett, E.S.; Kolliputi, N.; Tipparaju, S.M. High level of oxygen treatment causes cardiotoxicity with arrhythmias and redox modulation. Toxicol. Appl. Pharm. 2015, 282, 100–107. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Samal, E.; Mohapatra, S.; Panguluri, S.K. Hyperoxia-induced cardiotoxicity and ventricular remodeling in type-II diabetes mice. Heart Vessel. 2018, 33, 561–572. [Google Scholar] [CrossRef]
- Rodgers, J.L.; Rodgers, L.E.; Tian, Z.; Allen-Gipson, D.; Panguluri, S.K. Sex differences in murine cardiac pathophysiology with hyperoxia exposure. J. Cell. Physiol. 2019, 234, 1491–1501. [Google Scholar] [CrossRef]
- Moore, L.; Frost, J.; Britten, N. Context and complexity: The meaning of self-management for older adults with heart disease. Sociol. Health Illn. 2015, 37, 1254–1269. [Google Scholar] [CrossRef]
- Huynh-Hohnbaum, A.L.; Marshall, L.; Villa, V.M.; Lee, G. Self-Management of Heart Disease in Older Adults. Home Health Care Serv. Q 2015, 34, 159–172. [Google Scholar] [CrossRef]
- Bansal, M.; Chandra, K.S. The 2013 American College of Cardiology (ACC)/American Heart Association (AHA) guidelines for cholesterol management and for cardiovascular risk stratification: A reappraisal. Indian Heart J. 2014, 66, 1–4. [Google Scholar] [CrossRef]
- Anderson, T.J.; Grégoire, J.; Pearson, G.J.; Barry, A.R.; Couture, P.; Dawes, M.; Francis, G.A.; Genest, J.; Grover, S.; Gupta, M.; et al. 2016 Canadian Cardiovascular Society Guidelines for the Management of Dyslipidemia for the Prevention of Cardiovascular Disease in the Adult. Can. J. Cardiol. 2016, 32, 1263–1282. [Google Scholar] [CrossRef]
- Wang, Y.; Chun, O.K.; Song, W.O. Plasma and Dietary Antioxidant Status as Cardiovascular Disease Risk Factors: A Review of Human Studies. Nutrients 2013, 5, 2969–3004. [Google Scholar] [CrossRef]
- Rammos, C.; Hendgen-Cotta, U.B.; Sobierajski, J.; Bernard, A.; Kelm, M.; Rassaf, T. Dietary Nitrate Reverses Vascular Dysfunction in Older Adults With Moderately Increased Cardiovascular Risk. J. Am. Coll. Cardiol. 2014, 63, 1584–1585. [Google Scholar] [CrossRef]
- Booth, F.W.; Roberts, C.K.; Laye, M.J. Lack of Exercise Is a Major Cause of Chronic Diseases. Compr. Physiol. 2012, 2, 1143–1211. [Google Scholar] [CrossRef]
- Kimata, C.; Willcox, B.; Rodriguez, B. Effects of Walking on Coronary Heart Disease in Elderly Men with Diabetes. Geriatrics 2018, 3, 21. [Google Scholar] [CrossRef]
- Garatachea, N.; Pareja-Galeano, H.; Sanchis-Gomar, F.; Santos-Lozano, A.; Fiuza-Luces, C.; Moran, M.; Emanuele, E.; Joyner, M.J.; Lucia, A. Exercise Attenuates the Major Hallmarks of Aging. Rejuv Res. 2015, 18, 57–89. [Google Scholar] [CrossRef]
- Moreau, K.L.; Stauffer, B.L.; Kohrt, W.M.; Seals, D.R. Essential Role of Estrogen for Improvements in Vascular Endothelial Function With Endurance Exercise in Postmenopausal Women. J. Clin. Endocr. Metab. 2013, 98, 4507–4515. [Google Scholar] [CrossRef]
- Pierce, G.L.; Eskurza, I.; Walker, A.E.; Fay, T.N.; Seals, D.R. Sex-specific effects of habitual aerobic exercise on brachial artery flow-mediated dilation in middle-aged and older adults. Clin. Sci. 2011, 120, 13–23. [Google Scholar] [CrossRef]
- Hamilton, S.J.; Mills, B.; Birch, E.M.; Thompson, S.C. Smartphones in the secondary prevention of cardiovascular disease: A systematic review. Bmc Cardiovasc. Disord. 2018, 18, 25. [Google Scholar] [CrossRef]
- Piette, J.D.; Datwani, H.; Gaudioso, S.; Foster, S.M.; Westphal, J.; Perry, W.; Rodriguez-Saldana, J.; Mendoza-Avelares, M.O.; Marinec, N. Hypertension Management Using Mobile Technology and Home Blood Pressure Monitoring: Results of a Randomized Trial in Two Low/Middle-Income Countries. Telemed. E-Health 2012, 18, 613–620. [Google Scholar] [CrossRef]
- Still, C.H.; Jones, L.M.; Moss, K.O.; Variath, M.; Wright, K.D. African American Older Adults’ Perceived Use of Technology for Hypertension Self-Management. Res. Gerontol. Nurs. 2018, 11, 249–256. [Google Scholar] [CrossRef]
- Dietrich, E.; Davis, K. A Statin a Day to Keep the Doctor Away? Comparing Aspirin and Statins for Primary Prevention of Cardiovascular Disease. Ann. Pharm. 2014, 48, 1238–1241. [Google Scholar] [CrossRef]
- Duvernoy, C.S.; Blumenthal, R. The Numbers Are In Statins for the Primary Prevention of Cardiovascular Disease in Women. Circulation 2010, 121, 1063–1065. [Google Scholar] [CrossRef][Green Version]
- Taylor, F.; Huffman, M.D.; Macedo, A.F.; Moore, T.H.M.; Burke, M.; Smith, G.D.; Ward, K.; Ebrahim, S. Statins for the primary prevention of cardiovascular disease. Cochrane Db. Syst. Rev. 2011. [Google Scholar]
- Savarese, G.; Gotto, A.M.; Paolillo, S.; D’Amore, C.; Losco, T.; Musella, F.; Scala, O.; Marciano, C.; Ruggiero, D.; Marsico, F.; et al. Benefits of statins in elderly subjects without established cardiovascular disease: A meta-analysis. J. Am. Coll. Cardiol. 2013, 62, 2090–2099. [Google Scholar]
- Zhou, Z.; Albarqouni, L.; Breslin, M.; Curtis, A.J.; Nelson, M. Statin-associated muscle symptoms (SAMS) in primary prevention for cardiovascular disease in older adults: A protocol for a systematic review and meta-analysis of randomised controlled trials. Bmj Open 2017, 7, e017587. [Google Scholar] [CrossRef]
- Karp, I.; Chen, S.F.; Pilote, L. Sex differences in the effectiveness of statins after myocardial infarction. CMAJ 2007, 176, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Peters, S.A.E.; Colantonio, L.D.; Zhao, H.; Bittner, V.; Dai, Y.; Farkouh, M.E.; Monda, K.L.; Safford, M.M.; Muntner, P.; Woodward, M. Sex Differences in High-Intensity Statin Use Following Myocardial Infarction in the United States. J. Am. Coll. Cardiol. 2018, 71, 1729–1737. [Google Scholar] [CrossRef]
- Lloyd-Jones, D.M.; Leip, E.P.; Larson, M.G.; D’Agostino, R.B.; Beiser, A.; Wilson, P.W.; Wolf, P.A.; Levy, D. Prediction of lifetime risk for cardiovascular disease by risk factor burden at 50 years of age. Circulation 2006, 113, 791–798. [Google Scholar] [CrossRef]
- Tairova, M.S.; Graciolli, L.O.; Tairova, O.S.; De Marchi, T. Analysis of Cardiovascular Disease Risk Factors in Women. Open Access Maced. J. Med. Sci. 2018, 6. [Google Scholar] [CrossRef]
- Halter, J.B.; Musi, N.; McFarland Horne, F.; Crandall, J.P.; Goldberg, A.; Harkless, L.; Hazzard, W.R.; Huang, E.S.; Kirkman, M.S.; Plutzky, J.; et al. Diabetes and Cardiovascular Disease in Older Adults: Current Status and Future Directions. Diabetes 2014, 63, 2578–2589. [Google Scholar] [CrossRef]
- Corriere, M.; Rooparinesingh, N.; Kalyani, R.R. Epidemiology of diabetes and diabetes complications in the elderly: An emerging public health burden. Curr. Diab. Rep. 2013, 13, 805–813. [Google Scholar] [CrossRef]
- Toedebusch, R.; Belenchia, A.; Pulakat, L. Diabetic Cardiomyopathy: Impact of Biological Sex on Disease Development and Molecular Signatures. Front. Physiol. 2018, 9, 453. [Google Scholar] [CrossRef]
- Lee, P.G.; Halter, J.B. The Pathophysiology of Hyperglycemia in Older Adults: Clinical Considerations. Diabetes Care 2017, 40, 444–452. [Google Scholar] [CrossRef]
- Narayan, K.M.; Boyle, J.P.; Geiss, L.S.; Saaddine, J.B.; Thompson, T.J. Impact of recent increase in incidence on future diabetes burden: U.S., 2005-2050. Diabetes Care 2006, 29, 2114–2116. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-L.; Chiu, Y.-C.; Chang, H.-Y.; Hsu, K.-H.; Bai, Y.-B.; Wang, H.-H. Association of geriatric conditions and cardiovascular diseases with disability in older adults with diabetes: Findings from a nationally representative survey. Geriatr. Gerontol. Int. 2012, 13, 563–570. [Google Scholar] [CrossRef]
- Kautzky-Willer, A.; Harreiter, J.; Pacini, G. Sex and Gender Differences in Risk, Pathophysiology and Complications of Type 2 Diabetes Mellitus. Endocr. Rev. 2016, 37, 278–316. [Google Scholar] [CrossRef]
- Gale, E.A.; Gillespie, K.M. Diabetes and gender. Diabetologia 2001, 44, 3–15. [Google Scholar] [CrossRef]
- Natarajan, S.; Liao, Y.; Cao, G.; Lipsitz, S.R.; McGee, D.L. Sex differences in risk for coronary heart disease mortality associated with diabetes and established coronary heart disease. Arch. Intern. Med. 2003, 163, 1735–1740. [Google Scholar] [CrossRef]
- Krishnamurthy, J.; Ramsey, M.R.; Ligon, K.L.; Torrice, C.; Koh, A.; Bonner-Weir, S.; Sharpless, N.E. p16INK4a induces an age-dependent decline in islet regenerative potential. Nature 2006, 443, 453–457. [Google Scholar] [CrossRef]
- Kohlgruber, A.; Lynch, L. Adipose tissue inflammation in the pathogenesis of type 2 diabetes. Curr. Diab. Rep. 2015, 15, 92. [Google Scholar] [CrossRef]
- Liu, C.; Feng, X.; Li, Q.; Wang, Y.; Li, Q.; Hua, M. Adiponectin, TNF-alpha and inflammatory cytokines and risk of type 2 diabetes: A systematic review and meta-analysis. Cytokine 2016, 86, 100–109. [Google Scholar] [CrossRef]
- Tian, R.; Tian, M.; Wang, L.; Qian, H.; Zhang, S.; Pang, H.; Liu, Z.; Fang, L.; Shen, Z. C-reactive protein for predicting cardiovascular and all-cause mortality in type 2 diabetic patients: A meta-analysis. Cytokine 2019, 117, 59–64. [Google Scholar] [CrossRef]
- Assar, M.E.; Angulo, J.; Rodriguez-Manas, L. Diabetes and ageing-induced vascular inflammation. J. Physiol 2016, 594, 2125–2146. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.K.; Bartz, T.M.; Mukamal, K.J.; Djousse, L.; Kizer, J.R.; Tracy, R.P.; Zieman, S.J.; Rimm, E.B.; Siscovick, D.S.; Shlipak, M.; et al. Fetuin-A, Type 2 Diabetes, and Risk of Cardiovascular Disease in Older Adults: The Cardiovascular Health Study. Diabetes Care 2012, 36, 1222–1228. [Google Scholar] [CrossRef] [PubMed]
- Kalla, A.; Figueredo, V.M. Alcohol and cardiovascular disease in the geriatric population. Clin. Cardiol. 2017, 40, 444–449. [Google Scholar] [CrossRef]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative stress in obesity: A critical component in human diseases. Int. J. Mol. Sci 2014, 16, 378–400. [Google Scholar] [CrossRef] [PubMed]
- Mulero, J.; Zafrilla, P.; Martinez-Cacha, A. Oxidative stress, frailty and cognitive decline. J. Nutr. Health Aging 2011, 15, 756–760. [Google Scholar] [CrossRef]
- Namioka, N.; Hanyu, H.; Hirose, D.; Hatanaka, H.; Sato, T.; Shimizu, S. Oxidative stress and inflammation are associated with physical frailty in patients with Alzheimer’s disease. Geriatr. Gerontol. Int. 2017, 17, 913–918. [Google Scholar] [CrossRef]
- Roh, H.T.; Cho, S.Y.; So, W.Y. Obesity promotes oxidative stress and exacerbates blood-brain barrier disruption after high-intensity exercise. J. Sport Health Sci 2017, 6, 225–230. [Google Scholar] [CrossRef]
- Juonala, M.; Magnussen, C.G.; Berenson, G.S.; Venn, A.; Burns, T.L.; Sabin, M.A.; Srinivasan, S.R.; Daniels, S.R.; Davis, P.H.; Chen, W.; et al. Childhood adiposity, adult adiposity, and cardiovascular risk factors. N. Engl. J. Med. 2011, 365, 1876–1885. [Google Scholar] [CrossRef]
- Dudina, A.; Cooney, M.T.; Bacquer, D.D.; Backer, G.D.; Ducimetiere, P.; Jousilahti, P.; Keil, U.; Menotti, A.; Njolstad, I.; Oganov, R.; et al. Relationships between body mass index, cardiovascular mortality, and risk factors: a report from the SCORE investigators. Eur. J. Cardiovasc. Prev. Rehabil. 2011, 18, 731–742. [Google Scholar] [CrossRef] [PubMed]
- Khosravi, A.; Akhavan Tabib, A.; Golshadi, I.; Dana Siadat, Z.; Bahonar, A.; Zarfeshani, S.; Alikhasi, H.; Rezaee, S.; Noori, F.; Hashemi Jazi, M.; et al. The Relationship between Weight and CVD Risk Factors in a Sample Population from Central Iran (Based on IHHP). ARYA Atheroscler. 2012, 8, 82–89. [Google Scholar]
- Fan, H.; Li, X.; Zheng, L.; Chen, X.; Lan, Q.; Wu, H.; Ding, X.; Qian, D.; Shen, Y.; Yu, Z.; et al. Abdominal obesity is strongly associated with Cardiovascular Disease and its Risk Factors in Elderly and very Elderly Community-dwelling Chinese. Sci. Rep. 2016, 6, 21521. [Google Scholar] [CrossRef] [PubMed]
- Schneider, H.J.; Friedrich, N.; Klotsche, J.; Pieper, L.; Nauck, M.; John, U.; Dorr, M.; Felix, S.; Lehnert, H.; Pittrow, D.; et al. The predictive value of different measures of obesity for incident cardiovascular events and mortality. J. Clin. Endocrinol. Metab. 2010, 95, 1777–1785. [Google Scholar] [CrossRef]
- Guasch-Ferre, M.; Bullo, M.; Martinez-Gonzalez, M.A.; Corella, D.; Estruch, R.; Covas, M.-I.; Aros, F.; Warnberg, J.; Fiol, M.; Lapetra, J.; et al. Waist-to-height ratio and cardiovascular risk factors in elderly individuals at high cardiovascular risk. PLoS ONE 2012, 7, e43275. [Google Scholar] [CrossRef]
- Batsis, J.A.; Zagaria, A.B. Addressing Obesity in Aging Patients. Med. Clin North. Am. 2018, 102, 65–85. [Google Scholar] [CrossRef] [PubMed]
- Maas, A.H.E.M.; Appelman, Y.E.A. Gender differences in coronary heart disease. Neth. Heart J. 2010, 18, 598–602. [Google Scholar] [CrossRef] [PubMed]
- Decaria, J.E.; Sharp, C.; Petrella, R.J. Scoping review report: Obesity in older adults. Int. J. Obes. (Lond) 2012, 36, 1141–1150. [Google Scholar] [CrossRef]
- Afilalo, J.; Alexander, K.P.; Mack, M.J.; Maurer, M.S.; Green, P.; Allen, L.A.; Popma, J.J.; Ferrucci, L.; Forman, D.E. Frailty Assessment in the Cardiovascular Care of Older Adults. J. Am. Coll. Cardiol. 2014, 63, 747–762. [Google Scholar] [CrossRef]
- Sergi, G.; Veronese, N.; Fontana, L.; De Rui, M.; Bolzetta, F.; Zambon, S.; Corti, M.C.; Baggio, G.; Toffanello, E.D.; Crepaldi, G.; et al. Pre-frailty and risk of cardiovascular disease in elderly men and women: The Pro.V.A. study. J. Am. Coll. Cardiol 2015, 65, 976–983. [Google Scholar] [CrossRef] [PubMed]
- Kulmala, J.; Nykanen, I.; Hartikainen, S. Frailty as a predictor of all-cause mortality in older men and women. Geriatr. Gerontol. Int. 2014, 14, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Soysal, P.; Isik, A.T.; Carvalho, A.F.; Fernandes, B.S.; Solmi, M.; Schofield, P.; Veronese, N.; Stubbs, B. Oxidative stress and frailty: A systematic review and synthesis of the best evidence. Maturitas 2017, 99, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef] [PubMed]
- Villa, A.; Rizzi, N.; Vegeto, E.; Ciana, P.; Maggi, A. Estrogen accelerates the resolution of inflammation in macrophagic cells. Sci. Rep. 2015, 5, 15224. [Google Scholar] [CrossRef] [PubMed]
- Xue, B.; Singh, M.; Guo, F.; Hay, M.; Johnson, A.K. Protective actions of estrogen on angiotensin II-induced hypertension: Role of central nitric oxide. Am. J. Physiol. Heart Circ. Physiol. 2009, 297, H1638–H1646. [Google Scholar] [CrossRef] [PubMed]
- Jazbutyte, V.; Stumpner, J.; Redel, A.; Lorenzen, J.M.; Roewer, N.; Thum, T.; Kehl, F. Aromatase inhibition attenuates desflurane-induced preconditioning against acute myocardial infarction in male mouse heart in vivo. PLoS ONE 2012, 7, e42032. [Google Scholar] [CrossRef]
- Bell, J.R.; Bernasochi, G.B.; Varma, U.; Boon, W.C.; Ellem, S.J.; Risbridger, G.P.; Delbridge, L.M. Aromatase transgenic upregulation modulates basal cardiac performance and the response to ischemic stress in male mice. Am. J. Physiol. Heart Circ. Physiol. 2014, 306, H1265–H1274. [Google Scholar] [CrossRef] [PubMed]
- Cooke, P.S.; Nanjappa, M.K.; Ko, C.; Prins, G.S.; Hess, R.A. Estrogens in Male Physiology. Physiol. Rev. 2017, 97, 995–1043. [Google Scholar] [CrossRef] [PubMed]
- Baker, L.; Meldrum, K.K.; Wang, M.; Sankula, R.; Vanam, R.; Raiesdana, A.; Tsai, B.; Hile, K.; Brown, J.W.; Meldrum, D.R. The role of estrogen in cardiovascular disease. J. Surg. Res. 2003, 115, 325–344. [Google Scholar] [CrossRef]
- Costello, B.T.; Sprung, K.; Coulter, S.A. The Rise and Fall of Estrogen Therapy: Is Testosterone for “Manopause” Next? Tex. Heart Inst. J. 2017, 44, 338–340. [Google Scholar] [CrossRef]
- Parker, W.H.; Jacoby, V.; Shoupe, D.; Rocca, W. Effect of bilateral oophorectomy on women’s long-term health. Womens Health (Lond) 2009, 5, 565–576. [Google Scholar] [CrossRef] [PubMed]
- Klinge, C.M. Estrogenic control of mitochondrial function and biogenesis. J. Cell. Biochem. 2008, 105, 1342–1351. [Google Scholar] [CrossRef]
- Klinge, C.M. Estrogens regulate life and death in mitochondria. J. Bioenerg. Biomembr. 2017, 49, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Grodstein, F.; Manson, J.E.; Colditz, G.A.; Willett, W.C.; Speizer, F.E.; Stampfer, M.J. A prospective, observational study of postmenopausal hormone therapy and primary prevention of cardiovascular disease. Ann. Intern. Med. 2000, 133, 933–941. [Google Scholar] [CrossRef]
- Hulley, S.; Grady, D.; Bush, T.; Furberg, C.; Herrington, D.; Riggs, B.; Vittinghoff, E. Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women. Heart and Estrogen/progestin Replacement Study (HERS) Research Group. JAMA 1998, 280, 605–613. [Google Scholar] [CrossRef]
- Harman, S.M.; Black, D.M.; Naftolin, F.; Brinton, E.A.; Budoff, M.J.; Cedars, M.I.; Hopkins, P.N.; Lobo, R.A.; Manson, J.E.; Merriam, G.R.; et al. Arterial imaging outcomes and cardiovascular risk factors in recently menopausal women: A randomized trial. Ann. Intern. Med. 2014, 161, 249–260. [Google Scholar] [CrossRef]
- Schierbeck, L.L.; Rejnmark, L.; Tofteng, C.L.; Stilgren, L.; Eiken, P.; Mosekilde, L.; Kober, L.; Jensen, J.E. Effect of hormone replacement therapy on cardiovascular events in recently postmenopausal women: randomised trial. BMJ 2012, 345, e6409. [Google Scholar] [CrossRef]
- Ruige, J.B.; Mahmoud, A.M.; De Bacquer, D.; Kaufman, J.-M. Endogenous testosterone and cardiovascular disease in healthy men: A meta-analysis. Heart 2011, 97, 870. [Google Scholar] [CrossRef] [PubMed]
- Yeap, B.B.; Wu, F.C.W. Clinical practice update on testosterone therapy for male hypogonadism: Contrasting perspectives to optimize care. Clin Endocrinol (Oxf) 2019, 90, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Kloner, R.A.; Carson, C.; Dobs, A.; Kopecky, S.; Mohler, E.R. Testosterone and Cardiovascular Disease. J. Am. Coll. Cardiol. 2016, 67, 545. [Google Scholar] [CrossRef]
- Liao, P.-W.; Wu, C.-C.; Chen, K.-C.; Jaw, F.-S.; Yu, H.-J.; Liu, S.-P.; Ho, C.-H. Testosterone Threshold for Increased Cardiovascular Risk in Middle-Aged and Elderly Men: A Locally Weighted Regression Analysis. J. Sex. Med. 2016, 13, 1872–1880. [Google Scholar] [CrossRef]
- Daka, B.; Langer, R.D.; Larsson, C.A.; Rosen, T.; Jansson, P.A.; Rastam, L.; Lindblad, U. Low concentrations of serum testosterone predict acute myocardial infarction in men with type 2 diabetes mellitus. Bmc Endocr. Disord. 2015, 15, 35. [Google Scholar] [CrossRef][Green Version]
- Yeap, B.B.; Hyde, Z.; Almeida, O.P.; Norman, P.E.; Chubb, S.A.; Jamrozik, K.; Flicker, L.; Hankey, G.J. Lower testosterone levels predict incident stroke and transient ischemic attack in older men. J. Clin. Endocrinol. Metab. 2009, 94, 2353–2359. [Google Scholar] [CrossRef]
- Lin, J.W.; Lee, J.K.; Wu, C.K.; Caffrey, J.L.; Chang, M.H.; Hwang, J.J.; Dowling, N.; Lin, Y.S. Metabolic Syndrome, Testosterone, and Cardiovascular Mortality in Men. J. Sex. Med. 2011, 8, 2350–2360. [Google Scholar] [CrossRef] [PubMed]
- Kaczmarek, A.; Reczuch, K.; Majda, J.; Banasiak, W.; Ponikowski, P. The association of lower testosterone level with coronary artery disease in postmenopausal women. Int. J. Cardiol. 2003, 87, 53–57. [Google Scholar] [CrossRef]
- Vigen, R.; O’Donnell, C.I.; Baron, A.E.; Grunwald, G.K.; Maddox, T.M.; Bradley, S.M.; Barqawi, A.; Woning, G.; Wierman, M.E.; Plomondon, M.E.; et al. Association of testosterone therapy with mortality, myocardial infarction, and stroke in men with low testosterone levels. JAMA 2013, 310, 1829–1836. [Google Scholar] [CrossRef]
- Anderson, J.L.; May, H.T.; Lappe, D.L.; Bair, T.; Le, V.; Carlquist, J.F.; Muhlestein, J.B. Impact of Testosterone Replacement Therapy on Myocardial Infarction, Stroke, and Death in Men With Low Testosterone Concentrations in an Integrated Health Care System. Am. J. Cardiol. 2016, 117, 794–799. [Google Scholar] [CrossRef]
- Finkle, W.D.; Greenland, S.; Ridgeway, G.K.; Adams, J.L.; Frasco, M.A.; Cook, M.B.; Fraumeni, J.F., Jr.; Hoover, R.N. Increased risk of non-fatal myocardial infarction following testosterone therapy prescription in men. PLoS ONE 2014, 9, e85805. [Google Scholar] [CrossRef]
- Xu, L.; Freeman, G.; Cowling, B.J.; Schooling, C.M. Testosterone therapy and cardiovascular events among men: A systematic review and meta-analysis of placebo-controlled randomized trials. Bmc Med. 2013, 11, 108. [Google Scholar] [CrossRef] [PubMed]
- Bhasin, S.; Brito, J.P.; Cunningham, G.R.; Hayes, F.J.; Hodis, H.N.; Matsumoto, A.M.; Snyder, P.J.; Swerdloff, R.S.; Wu, F.C.; Yialamas, M.A. Testosterone Therapy in Men With Hypogonadism: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2018, 103, 1715–1744. [Google Scholar] [CrossRef]
- Snyder, P.J.; Bhasin, S.; Cunningham, G.R.; Matsumoto, A.M.; Stephens-Shields, A.J.; Cauley, J.A.; Gill, T.M.; Barrett-Connor, E.; Swerdloff, R.S.; Wang, C.; et al. Effects of Testosterone Treatment in Older Men. N. Engl. J. Med. 2016, 374, 611–624. [Google Scholar] [CrossRef]
- Snyder, P.J.; Bhasin, S.; Cunningham, G.R.; Matsumoto, A.M.; Stephens-Shields, A.J.; Cauley, J.A.; Gill, T.M.; Barrett-Connor, E.; Swerdloff, R.S.; Wang, C.; et al. Lessons From the Testosterone Trials. Endocr. Rev. 2018, 39, 369–386. [Google Scholar] [CrossRef] [PubMed]
 indicates upregulation. Tumor necrosis factor-α (TNFα), C-reactive protein (CRP), and interleukin-6 (IL-6).
indicates upregulation. Tumor necrosis factor-α (TNFα), C-reactive protein (CRP), and interleukin-6 (IL-6).
 indicates upregulation. Tumor necrosis factor-α (TNFα), C-reactive protein (CRP), and interleukin-6 (IL-6).
indicates upregulation. Tumor necrosis factor-α (TNFα), C-reactive protein (CRP), and interleukin-6 (IL-6).
 indicates upregulation and
indicates upregulation and  indicates downregulation. Coronary artery disease (CAD) and myocardial infarction (MI).
indicates downregulation. Coronary artery disease (CAD) and myocardial infarction (MI).
 indicates upregulation and
indicates upregulation and  indicates downregulation. Coronary artery disease (CAD) and myocardial infarction (MI).
indicates downregulation. Coronary artery disease (CAD) and myocardial infarction (MI).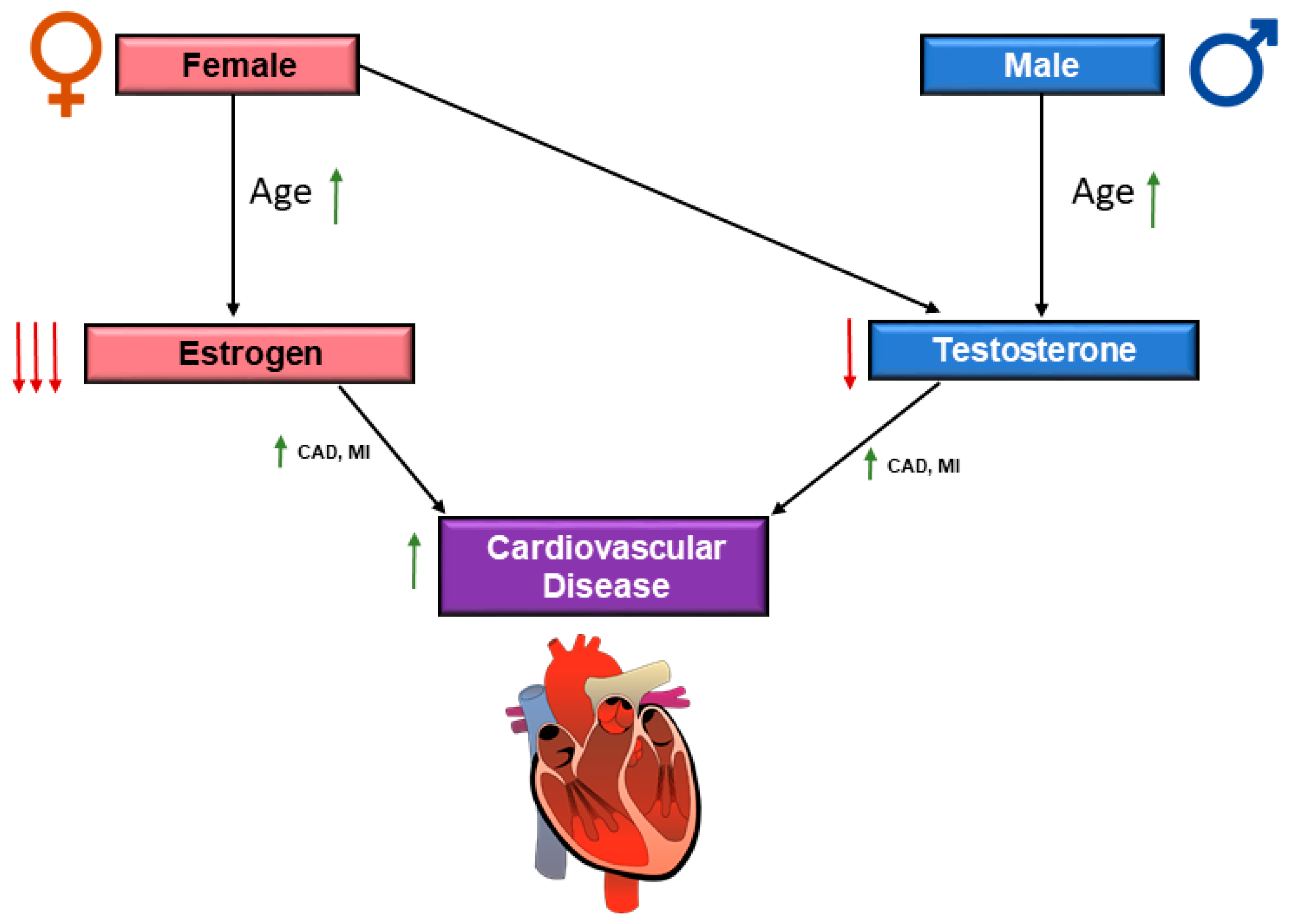
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodgers, J.L.; Jones, J.; Bolleddu, S.I.; Vanthenapalli, S.; Rodgers, L.E.; Shah, K.; Karia, K.; Panguluri, S.K. Cardiovascular Risks Associated with Gender and Aging. J. Cardiovasc. Dev. Dis. 2019, 6, 19. https://doi.org/10.3390/jcdd6020019
Rodgers JL, Jones J, Bolleddu SI, Vanthenapalli S, Rodgers LE, Shah K, Karia K, Panguluri SK. Cardiovascular Risks Associated with Gender and Aging. Journal of Cardiovascular Development and Disease. 2019; 6(2):19. https://doi.org/10.3390/jcdd6020019
Chicago/Turabian StyleRodgers, Jennifer L., Jarrod Jones, Samuel I. Bolleddu, Sahit Vanthenapalli, Lydia E. Rodgers, Kinjal Shah, Krishna Karia, and Siva K. Panguluri. 2019. "Cardiovascular Risks Associated with Gender and Aging" Journal of Cardiovascular Development and Disease 6, no. 2: 19. https://doi.org/10.3390/jcdd6020019
APA StyleRodgers, J. L., Jones, J., Bolleddu, S. I., Vanthenapalli, S., Rodgers, L. E., Shah, K., Karia, K., & Panguluri, S. K. (2019). Cardiovascular Risks Associated with Gender and Aging. Journal of Cardiovascular Development and Disease, 6(2), 19. https://doi.org/10.3390/jcdd6020019



