A Systematic Review of Ebstein’s Anomaly with Left Ventricular Noncompaction
Abstract
1. Introduction
1.1. Ebstein’s Anomaly (EA)
1.2. Left Ventricular Noncompaction (LVNC)
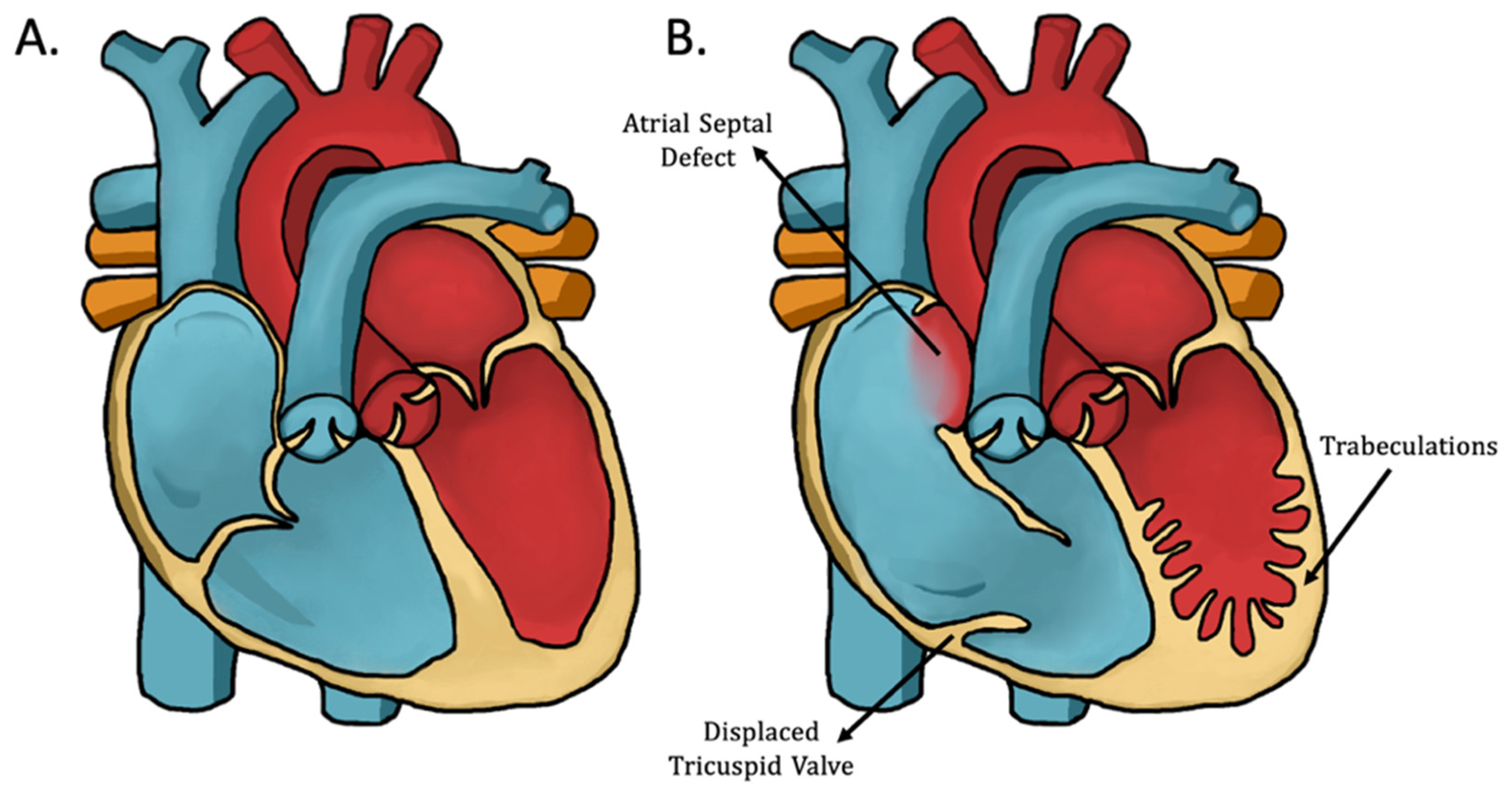
1.3. Ebstein’s Anomaly with Left Ventricular Noncompaction (EA/LVNC)
2. Methods
3. Results
3.1. We Identified 58 Peer-Reviewed Publications That Discuss EA/LVNC
3.2. EA/LVNC Patients Present with Clinical Signs of Both CHDs
3.3. EA/LVNC Patients Commonly Present with Arrhythmia
| Study | Published | Focus | Center | Subjects | Gender | EA/LVNC | Results |
|---|---|---|---|---|---|---|---|
| Attenhofer Jost et al. [78] | 2005 | Clinical study of EA | Mayo Clinic Rochester, MN, USA | 106 patients aged 0–52 years. | 39.7% M 60.3% F |
|
|
| El-Menyar et al. [18] | 2007 | Clinical study of LVNC in Qatar | Hamad General Hospital Doha, Qatar | 12 patients aged 0–37 years. | 33% M 67% F |
|
|
| Reemtsen et al. [7] | 2007 | Clinical study of EA | Children’s Hospital Los Angeles Los Angeles, CA, USA | 12 patients aged 0–17 years. | 58% M 42% F |
|
|
| Tsai et al. [17] | 2009 | Clinical study of LVNC | Riley Hospital for Children Indianapolis, IN, USA | 46 patients | 50% M 50% F |
|
|
| Stähli et al. [79] | 2013 | Clinical study of LVNC | University Children’s Hospital Zurich, Switzerland | 202 patients | 79% M 21% F |
|
|
| Pignatelli et al. [82] | 2014 | Clinical study of EA or EA/LVNC | Texas Children’s Hospital Houston, TX, USA | 61 infants |
|
| |
| Kumor et al. [80] | 2018 | Clinical study of EA | Institute of Cardiology Warsaw, Poland | 84 patients aged 16–71 years. | 41% M 59% F |
|
|
| Hirono et al. [83] | 2020 | Clinical and genetic study of LVNC | University of Toyama Toyama, Japan; | 53 Japanese probands aged 1–14 years. | 47% M 53% F |
|
|
| Marques et al. [81] | 2020 | CHD study of postnatal heart specimens | Heart Institute (InCor), University of Sao Paulo Medical School, Brazil | 259 postnatal hearts with 87.3% aged less than 18 years. | 49% M 51% F |
|
|
3.4. ECHO Is Preferred for EA Diagnosis While Cardiac MRI Is Preferred for LVNC
3.5. EA/LVNC Treatment Is Non-Specific and Heterogenous
3.6. Most Clinical Trials of EA or LVNC Seek to Assess CHD Genetic Etiology
3.7. Sarcomeric Variants Are Implicated in the Genetic Etiology of EA/LVNC
| Year | Publication | Sample Size | Occurrence | Findings |
|---|---|---|---|---|
| 2007 | Budde et al. [69] | 24 | Familial | MYH7 (p.R281T) |
| 2010 | Hoedemaekers et al. [104] | 58 | Sporadic | MYH7 (p.L301Q) |
| 2011 | Postma et al. [76] | 141 | Sporadic | MYH7; 7 mutations (5 novel) |
| 2014 | Hirono et al. [70] | 3 | Familial | MYH7 (p.M362R) |
| 2016 | Kelle et al. [50] | 1 | Sporadic | TPM1 (p.D159) |
| 2018 | Nijak et al. [71] | 5 | Familial | TPM1 (p.L131V) |
| 2019 | Carlston et al. [41] | 1 | Sporadic | NONO (p.N52Sfs) |
| 2020 | Hirono et al. [83] | 53 | Sporadic | 30 genetic variants in MYH7, TPM1, ACTC1, ANK2, COL4A1, DAAM1, DSG2, DSP, FGF16, FGFR2, HCN4, JUP, MYBPC3, MYH6, MYL2, PKP2, PRDM16, RYR2, and TAZ |
| 2020 | Samudrala et al. [72] | 17 | Familial | KLHL26 (p.R237C) |
| 2021 | Mehdi et al. [56] | 1 | Sporadic | Gain chromosome band 15q11.2 and 1q44 |
| 2022 | Coetzer et al. [42] | 1 | Sporadic | NONO (p.M389_T400del) |
| 2022 | Tu et al. [75] | 6 | Familial | MYH7 splicing variant |
4. Discussion
4.1. Shared Developmental Pathways of EA and LVNC
4.2. A Common Etiology for EA and LVNC?
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Attenhofer Jost, C.H.; Connolly, H.M.; Dearani, J.A.; Edwards, W.D.; Danielson, G.K. Ebstein’s anomaly. Circulation 2007, 115, 277–285. [Google Scholar] [CrossRef] [PubMed]
- Correa-Villasenor, A.; Ferencz, C.; Neill, C.A.; Wilson, P.D.; Boughman, J.A. Ebstein’s malformation of the tricuspid valve: Genetic and environmental factors. The Baltimore-Washington Infant Study Group. Teratology 1994, 50, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Lupo, P.J.; Langlois, P.H.; Mitchell, L.E. Epidemiology of Ebstein anomaly: Prevalence and patterns in Texas, 1999–2005. Am. J. Med. Genet. A 2011, 155, 1007–1014. [Google Scholar] [CrossRef]
- Fahed, A.C.; Gelb, B.D.; Seidman, J.G.; Seidman, C.E. Genetics of congenital heart disease: The glass half empty. Circ. Res. 2013, 112, 707–720. [Google Scholar] [CrossRef] [PubMed]
- Ho, S.Y.; Goltz, D.; McCarthy, K.; Cook, A.C.; Connell, M.G.; Smith, A.; Anderson, R.H. The atrioventricular junctions in Ebstein malformation. Heart 2000, 83, 444–449. [Google Scholar] [CrossRef]
- Chauvaud, S. Ebstein’s malformation. surgical treatment and results. Thorac. Cardiovasc. Surg. 2000, 48, 220–223. [Google Scholar] [CrossRef] [PubMed]
- Reemtsen, B.L.; Polimenakos, A.C.; Fagan, B.T.; Wells, W.J.; Starnes, V.A. Fate of the right ventricle after fenestrated right ventricular exclusion for severe neonatal Ebstein anomaly. J. Thorac. Cardiovasc. Surg. 2007, 134, 1406–1410. [Google Scholar] [CrossRef] [PubMed]
- Freedom, R.M.; Yoo, S.J.; Perrin, D.; Taylor, G.; Petersen, S.; Anderson, R.H. The morphological spectrum of ventricular noncompaction. Cardiol. Young 2005, 15, 345–364. [Google Scholar] [CrossRef] [PubMed]
- Voges, I.; Al-Mallah, M.H.; Scognamiglio, G.; Di Salvo, G. Right Heart-Pulmonary Circulation Unit in Congenital Heart Diseases. Heart Fail. Clin. 2018, 14, 283–295. [Google Scholar] [CrossRef]
- Celermajer, D.S.; Bull, C.; Till, J.A.; Cullen, S.; Vassillikos, V.P.; Sullivan, I.D.; Allan, L.; Nihoyannopoulos, P.; Somerville, J.; Deanfield, J.E. Ebstein’s anomaly: Presentation and outcome from fetus to adult. J. Am. Coll. Cardiol. 1994, 23, 170–176. [Google Scholar] [CrossRef]
- Cabrera, R.; Miranda-Fernandez, M.C.; Huertas-Quinones, V.M.; Carreno, M.; Pineda, I.; Restrepo, C.M.; Silva, C.T.; Quero, R.; Cano, J.D.; Manrique, D.C.; et al. Identification of clinically relevant phenotypes in patients with Ebstein anomaly. Clin. Cardiol. 2018, 41, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Carpentier, A.; Chauvaud, S.; Mace, L.; Relland, J.; Mihaileanu, S.; Marino, J.P.; Abry, B.; Guibourt, P. A new reconstructive operation for Ebstein’s anomaly of the tricuspid valve. J. Thorac. Cardiovasc. Surg. 1988, 96, 92–101. [Google Scholar] [CrossRef]
- Dearani, J.A.; Danielson, G.K. Congenital Heart Surgery Nomenclature and Database Project: Ebstein’s anomaly and tricuspid valve disease. Ann. Thorac. Surg. 2000, 69, S106–S117. [Google Scholar] [CrossRef]
- Zhang, W.; Chen, H.; Qu, X.; Chang, C.P.; Shou, W. Molecular mechanism of ventricular trabeculation/compaction and the pathogenesis of the left ventricular noncompaction cardiomyopathy (LVNC). Am. J. Med. Genet. C Semin. Med. Genet. 2013, 163, 144–156. [Google Scholar] [CrossRef] [PubMed]
- Towbin, J.A.; Lorts, A.; Jefferies, J.L. Left ventricular non-compaction cardiomyopathy. Lancet 2015, 386, 813–825. [Google Scholar] [CrossRef]
- Tarando, F.; Coisne, D.; Galli, E.; Rousseau, C.; Viera, F.; Bosseau, C.; Habib, G.; Lederlin, M.; Schnell, F.; Donal, E. Left ventricular non-compaction and idiopathic dilated cardiomyopathy: The significant diagnostic value of longitudinal strain. Int. J. Cardiovasc. Imaging 2017, 33, 83–95. [Google Scholar] [CrossRef]
- Tsai, S.F.; Ebenroth, E.S.; Hurwitz, R.A.; Cordes, T.M.; Schamberger, M.S.; Batra, A.S. Is left ventricular noncompaction in children truly an isolated lesion? Pediatr. Cardiol. 2009, 30, 597–602. [Google Scholar] [CrossRef]
- El-Menyar, A.A.; Gendi, S.M.; Numan, M.T. Noncompaction cardiomyopathy in the State of Qatar. Saudi Med. J. 2007, 28, 429–434. [Google Scholar]
- Chin, T.K.; Perloff, J.K.; Williams, R.G.; Jue, K.; Mohrmann, R. Isolated noncompaction of left ventricular myocardium. A study of eight cases. Circulation 1990, 82, 507–513. [Google Scholar] [CrossRef]
- Jenni, R.; Oechslin, E.; Schneider, J.; Attenhofer Jost, C.; Kaufmann, P.A. Echocardiographic and pathoanatomical characteristics of isolated left ventricular non-compaction: A step towards classification as a distinct cardiomyopathy. Heart 2001, 86, 666–671. [Google Scholar] [CrossRef]
- Petersen, S.E.; Selvanayagam, J.B.; Wiesmann, F.; Robson, M.D.; Francis, J.M.; Anderson, R.H.; Watkins, H.; Neubauer, S. Left ventricular non-compaction: Insights from cardiovascular magnetic resonance imaging. J. Am. Coll. Cardiol. 2005, 46, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Attenhofer Jost, C.H.; Connolly, H.M.; Warnes, C.A.; O’Leary, P.; Tajik, A.J.; Pellikka, P.A.; Seward, J.B. Noncompacted myocardium in Ebstein’s anomaly: Initial description in three patients. J. Am. Soc. Echocardiogr. 2004, 17, 677–680. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- Ashraf, H.; Pradhan, L.; Chang, E.I.; Terada, R.; Ryan, N.J.; Briggs, L.E.; Chowdhury, R.; Zarate, M.A.; Sugi, Y.; Nam, H.J.; et al. A mouse model of human congenital heart disease: High incidence of diverse cardiac anomalies and ventricular noncompaction produced by heterozygous Nkx2-5 homeodomain missense mutation. Circ. Cardiovasc. Genet. 2014, 7, 423–433. [Google Scholar] [CrossRef] [PubMed]
- Gittenberger-de Groot, A.C.; Winter, E.M.; Poelmann, R.E. Epicardium-derived cells (EPDCs) in development, cardiac disease and repair of ischemia. J. Cell Mol. Med. 2010, 14, 1056–1060. [Google Scholar] [CrossRef]
- Abu-Issa, R.; Waldo, K.; Kirby, M.L. Heart fields: One, two or more? Dev. Biol. 2004, 272, 281–285. [Google Scholar] [CrossRef] [PubMed]
- Basson, C.T.; Bachinsky, D.R.; Lin, R.C.; Levi, T.; Elkins, J.A.; Soults, J.; Grayzel, D.; Kroumpouzou, E.; Traill, T.A.; Leblanc-Straceski, J.; et al. Mutations in human TBX5 [corrected] cause limb and cardiac malformation in Holt-Oram syndrome. Nat. Genet. 1997, 15, 30–35. [Google Scholar] [CrossRef]
- Al-Wakeel-Marquard, N.; Seidel, F.; Kuhnisch, J.; Kuehne, T.; Berger, F.; Messroghli, D.R.; Klaassen, S. Midwall Fibrosis and Cardiac Mechanics: Rigid Body Rotation Is a Novel Marker of Disease Severity in Pediatric Primary Dilated Cardiomyopathy. Front. Cardiovasc. Med. 2021, 8, 810005. [Google Scholar] [CrossRef]
- Al-Wakeel-Marquard, N.; Degener, F.; Herbst, C.; Kuhnisch, J.; Dartsch, J.; Schmitt, B.; Kuehne, T.; Messroghli, D.; Berger, F.; Klaassen, S. RIKADA Study Reveals Risk Factors in Pediatric Primary Cardiomyopathy. J. Am. Heart Assoc. 2019, 8, e012531. [Google Scholar] [CrossRef]
- Renilla, A.; Santamarta, E.; Corros, C.; Martin, M.; Barreiro, M.; de la Hera, J. Isolated left ventricular non-compaction associated with Ebstein’s anomaly. Multimodality non-invasive imaging for the assessment of congenital heart disease. Arch. Cardiol. Mex. 2013, 83, 100–103. [Google Scholar] [CrossRef] [PubMed]
- Dima-Cozma, C.; Cojocaru, D.C.; Chiriac, S.; Negru, R.; Mitu, F. Ebstein’s anomaly. Rev. Med. Chir. Soc. Med. Nat. Iasi 2013, 117, 674–679. [Google Scholar]
- O’Leary, P.W.; Qureshi, M.Y.; Cetta, F.; Nelson, T.J.; Holst, K.A.; Dearani, J.A.; Wanek Program Clinical Pipeline, G. Cone Reconstruction for Ebstein Anomaly: Ventricular Remodeling and Preliminary Impact of Stem Cell Therapy. Mayo Clin. Proc. 2021, 96, 3053–3061. [Google Scholar] [CrossRef] [PubMed]
- Tulino, V.; Cacace, C.; Tulino, D.; Imbalzano, E.; Dattilo, G. Clinical variants in Ebstein’s anomaly. Int. J. Cardiol. 2013, 168, 4969–4970. [Google Scholar] [CrossRef] [PubMed]
- Andresen, H.; Kaag, N.; Potratz, J. Non-compaction of ventricular myocardium and contrast-enhanced echocardiography. Z Kardiol. 2005, 94, 483–485. [Google Scholar] [CrossRef] [PubMed]
- Arslan, S.; Gurlertop, H.Y.; Gundogdu, F.; Senocak, H. Left ventricular noncompaction and mid-caviter narrowing associated with Ebstein’s anomaly: Three-dimensional transthoracic echocardiographic image. Int. J. Cardiol. 2007, 115, e52–e55. [Google Scholar] [CrossRef] [PubMed]
- Aydin, C.; Engin, M. Ebstein’s anomaly associated with biventricular noncompaction. J. Echocardiogr. 2021. [Google Scholar] [CrossRef] [PubMed]
- Bagur, R.H.; Lederlin, M.; Montaudon, M.; Latrabe, V.; Corneloup, O.; Iriart, X.; Laurent, F. Images in cardiovascular medicine. Ebstein anomaly associated with left ventricular noncompaction. Circulation 2008, 118, e662–e664. [Google Scholar] [CrossRef]
- Baysan, O.; Yokusoglu, M.; Bugan, B.; Demirkol, S. E-page original images. A case of Ebstein anomaly and biventricular noncompaction. Anadolu Kardiyol. Derg. 2010, 10, E26. [Google Scholar] [CrossRef]
- Betrian Blasco, P.; Gallardo Agromayor, E. Ebstein’s anomaly and left ventricular noncompaction association. Int. J. Cardiol. 2007, 119, 264–265. [Google Scholar] [CrossRef]
- Carlston, C.M.; Bleyl, S.B.; Andrews, A.; Meyers, L.; Brown, S.; Bayrak-Toydemir, P.; Bale, J.F.; Botto, L.D. Expanding the genetic and clinical spectrum of the NONO-associated X-linked intellectual disability syndrome. Am. J. Med. Genet. A 2019, 179, 792–796. [Google Scholar] [CrossRef]
- Coetzer, K.C.; Moosa, S. Novel hemizygous loss-of-function variant in NONO identified in a South African boy. Am. J. Med. Genet. A 2022, 188, 373–376. [Google Scholar] [CrossRef]
- Das, B.B.; Chrisant, M.; Lavandosky, G.; Zakrzewski, M.; Winchester, R.; Turner, I.; Bibevski, S.; Scholl, F. An Adolescent with Left Ventricular Noncompaction and Ebstein Anomaly Presenting with Advanced Heart Failure: Discharge from Hospital with a Biventricular Assist Device. J. Pediatr. 2018, 202, 304–310. [Google Scholar] [CrossRef]
- Dogan, A.; Aksoy, H. Coexistence of congenital heart anomalies with noncompaction of the ventricular myocardium. Two case reports. Herz 2012, 37, 699–701. [Google Scholar] [CrossRef]
- Evans, E.; McDonald, M.; Adamson, D.L.; Khan, J.N. Cardiomyopathy in the peripartum period due to left ventricular non-compaction and association with Ebstein’s anomaly: A case report. Eur Heart J. Case Rep. 2021, 5, ytab088. [Google Scholar] [CrossRef]
- Fazio, G.; Visconti, C.; D’Angelo, L.; Grassedonio, E.; Lo Re, G.; D’Amico, T.; Sutera, L.; Novo, G.; Ferrara, F.; Midiri, M.; et al. Diagnosis and definition of biventricular non-compaction associated to Ebstein’s anomaly. Int. J. Cardiol. 2011, 150, e20–e24. [Google Scholar] [CrossRef]
- Geerdink, L.M.; Ter Heide, H.; Kapusta, L. Ebstein’s anomaly and aortic arch lesions: When right meets left. Int. J. Cardiol. 2016, 223, 568–570. [Google Scholar] [CrossRef]
- Ilercil, A.; Barack, J.; Malone, M.A.; Barold, S.S.; Herweg, B. Association of noncompaction of left ventricular myocardium with Ebstein’s anomaly. Echocardiography 2006, 23, 432–433. [Google Scholar] [CrossRef]
- Isilak, Z.; Tokatli, A.; Yalcin, M.; Uzun, M. Case images: A very rare case: Ebstein anomaly, biventricular noncompaction and pre-excitation in the same patient. Turk Kardiyol. Dern. Ars. 2012, 40, 557. [Google Scholar] [CrossRef]
- Kelle, A.M.; Bentley, S.J.; Rohena, L.O.; Cabalka, A.K.; Olson, T.M. Ebstein anomaly, left ventricular non-compaction, and early onset heart failure associated with a de novo alpha-tropomyosin gene mutation. Am. J. Med. Genet. A 2016, 170, 2186–2190. [Google Scholar] [CrossRef]
- Kharge, J.; Prasad, M.R.; Ramegowda, R.T. An unusual case of congenitally corrected transposition of the great arteries associated with noncompaction-like remodeling of the morphological right ventricle. Echocardiography 2011, 28, E212–E214. [Google Scholar] [CrossRef]
- Kiernan, T.J.; Fahy, G. Multiple accessory pathways, dual AV nodal physiology, non-compacted myocardium and patent foramen ovale in a patient with Ebstein’s anomaly: Report of a case. Int. J. Cardiol. 2007, 114, 412–413. [Google Scholar] [CrossRef]
- Kumar, N.; Troianos, C.A.; Baisden, J.S. Left Ventricular Assist Device Insertion in a Patient With Biventricular Noncompaction Cardiomyopathy, Ebstein Anomaly, and a Left Atrial Mass: A Case Report. A & A Case Rep. 2016, 7, 251–255. [Google Scholar] [CrossRef]
- Malagoli, A.; Rossi, L.; Mastrojanni, C.; Villani, G.Q. A perfect storm: Wolf Parkinson White syndrome, Ebstein’s anomaly, biventricular non-compaction, and bicuspid aortic valve. Eur. Heart J. Cardiovasc. Imaging 2014, 15, 827. [Google Scholar] [CrossRef][Green Version]
- McGee, M.; Warner, L.; Collins, N. Ebstein’s Anomaly, Left Ventricular Noncompaction, and Sudden Cardiac Death. Case Rep. Cardiol. 2015, 2015, 854236. [Google Scholar] [CrossRef]
- Mehdi, M.; Bhatia, S.; Patel, M.; Aly, A. Ebstein’s Anomaly, Left Ventricular Noncompaction and Gerbode-Like Defect Triad (Fetal Diagnosis and Neonatal Course). Case Rep. Pediatr. 2021, 2021, 9969588. [Google Scholar] [CrossRef]
- Nimeri, N.A.; Abou Nahia, F.F.; Ibrahim, A.S.; Khella, A.Y. The first reported case of non-compacted cardiomyopathy in a preterm infant with Ebstein’s anomaly. BMJ Case Rep. 2012, 2012, bcr0220125861. [Google Scholar] [CrossRef]
- Patra, S.; Singla, V.; Kharge, J.; Ravindranath, K.S.; Manjunath, C.N. A patient of Ebstein’s anomaly associated with biventricular noncompaction presented with Wolf Parkinson White syndrome—A rare presentation. J. Cardiovasc. Dis. Res. 2012, 3, 323–325. [Google Scholar] [CrossRef][Green Version]
- Saltik, L.; Eroglu, E.; Bayrak, F.; Degertekin, M. Left ventricular noncompaction associated with Ebstein’s anomaly. Turk Kardiyol. Dern. Ars. 2009, 37, 269–272. [Google Scholar]
- Scott, D.A.; Hernandez-Garcia, A.; Azamian, M.S.; Jordan, V.K.; Kim, B.J.; Starkovich, M.; Zhang, J.; Wong, L.J.; Darilek, S.A.; Breman, A.M.; et al. Congenital heart defects and left ventricular non-compaction in males with loss-of-function variants in NONO. J. Med. Genet. 2017, 54, 47–53. [Google Scholar] [CrossRef]
- Song, Z.Z. A combination of right ventricular hypertrabeculation/noncompaction and Ebstein’s anomaly. Int. J. Cardiol. 2010, 143, e30–e33. [Google Scholar] [CrossRef] [PubMed]
- Stollberger, C.; Kopsa, W.; Finsterer, J. Non-compaction of the right atrium and left ventricle in Ebstein’s malformation. J. Heart Valve Dis. 2006, 15, 719–720. [Google Scholar] [PubMed]
- Thakur, V.; Jaeggi, E.T.; Nield, L.E. A unique foetal case of left ventricular non-compaction associated with arrhythmia, structural cardiac anomalies, and agenesis of the ductus venosus. Cardiol. Young 2016, 26, 368–370. [Google Scholar] [CrossRef] [PubMed]
- Thani, K.B.; Khadivi, B.; Kahn, A.M.; Cotter, B.; Blanchard, D. Ebstein’s anomaly with left ventricular noncompaction and bicuspid aortic valve. J. Am. Coll. Cardiol. 2010, 56, 899. [Google Scholar] [CrossRef] [PubMed]
- Tourmousoglou, C.; Bogossian, H.; Ninios, V.; Ninios, E. A rare case of Ebstein’s anomaly with left ventricular noncompaction. Asian Cardiovasc. Thorac. Ann. 2019, 27, 208–209. [Google Scholar] [CrossRef] [PubMed]
- Wijesekera, V.; Yadav, R. Combined left ventricular non-compaction and Ebstein’s anomaly. Heart Asia 2012, 4, 37. [Google Scholar] [CrossRef] [PubMed]
- Zarrouk Mahjoub, S.; Mehri, S.; Ourda, F.; Boussaada, R.; Mechmeche, R.; Arab, S.B.; Finsterer, J. Transition m.3308T>C in the ND1 gene is associated with left ventricular hypertrabeculation/noncompaction. Cardiology 2011, 118, 153–158. [Google Scholar] [CrossRef]
- Bettinelli, A.L.; Mulder, T.J.; Funke, B.H.; Lafferty, K.A.; Longo, S.A.; Niyazov, D.M. Familial ebstein anomaly, left ventricular hypertrabeculation, and ventricular septal defect associated with a MYH7 mutation. Am. J. Med. Genet. A 2013, 161, 3187–3190. [Google Scholar] [CrossRef] [PubMed]
- Budde, B.S.; Binner, P.; Waldmuller, S.; Hohne, W.; Blankenfeldt, W.; Hassfeld, S.; Bromsen, J.; Dermintzoglou, A.; Wieczorek, M.; May, E.; et al. Noncompaction of the ventricular myocardium is associated with a de novo mutation in the beta-myosin heavy chain gene. PLoS ONE 2007, 2, e1362. [Google Scholar] [CrossRef]
- Hirono, K.; Hata, Y.; Ibuki, K.; Yoshimura, N. Familial Ebstein’s anomaly, left ventricular noncompaction, and ventricular septal defect associated with an MYH7 mutation. J. Thorac. Cardiovasc. Surg. 2014, 148, e223–e226. [Google Scholar] [CrossRef]
- Nijak, A.; Alaerts, M.; Kuiperi, C.; Corveleyn, A.; Suys, B.; Paelinck, B.; Saenen, J.; Van Craenenbroeck, E.; Van Laer, L.; Loeys, B.; et al. Left ventricular non-compaction with Ebstein anomaly attributed to a TPM1 mutation. Eur. J. Med. Genet. 2018, 61, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Samudrala, S.S.K.; North, L.M.; Stamm, K.D.; Earing, M.G.; Frommelt, M.A.; Willes, R.; Tripathi, S.; Dsouza, N.R.; Zimmermann, M.T.; Mahnke, D.K.; et al. Novel KLHL26 variant associated with a familial case of Ebstein’s anomaly and left ventricular noncompaction. Mol. Genet. Genom. Med. 2020, 8, e1152. [Google Scholar] [CrossRef]
- Sinkovec, M.; Kozelj, M.; Podnar, T. Familial biventricular myocardial noncompaction associated with Ebstein’s malformation. Int. J. Cardiol. 2005, 102, 297–302. [Google Scholar] [CrossRef] [PubMed]
- Neu, A.; Eiselt, M.; Paul, M.; Sauter, K.; Stallmeyer, B.; Isbrandt, D.; Schulze-Bahr, E. A homozygous SCN5A mutation in a severe, recessive type of cardiac conduction disease. Hum. Mutat. 2010, 31, E1609–E1621. [Google Scholar] [CrossRef] [PubMed]
- Tu, P.; Sun, H.; Zhang, X.; Ran, Q.; He, Y.; Ran, S. Diverse cardiac phenotypes among different carriers of the same MYH7 splicing variant allele (c.732+1G>A) from a family. BMC Med. Genom. 2022, 15, 36. [Google Scholar] [CrossRef] [PubMed]
- Postma, A.V.; van Engelen, K.; van de Meerakker, J.; Rahman, T.; Probst, S.; Baars, M.J.; Bauer, U.; Pickardt, T.; Sperling, S.R.; Berger, F.; et al. Mutations in the sarcomere gene MYH7 in Ebstein anomaly. Circ. Cardiovasc. Genet. 2011, 4, 43–50. [Google Scholar] [CrossRef] [PubMed]
- van Engelen, K.; Postma, A.V.; van de Meerakker, J.B.; Roos-Hesselink, J.W.; Helderman-van den Enden, A.T.; Vliegen, H.W.; Rahman, T.; Baars, M.J.; Sels, J.W.; Bauer, U.; et al. Ebstein’s anomaly may be caused by mutations in the sarcomere protein gene MYH7. Neth Heart J. 2013, 21, 113–117. [Google Scholar] [CrossRef][Green Version]
- Attenhofer Jost, C.H.; Connolly, H.M.; O’Leary, P.W.; Warnes, C.A.; Tajik, A.J.; Seward, J.B. Left heart lesions in patients with Ebstein anomaly. Mayo Clin. Proc. 2005, 80, 361–368. [Google Scholar] [CrossRef]
- Stahli, B.E.; Gebhard, C.; Biaggi, P.; Klaassen, S.; Valsangiacomo Buechel, E.; Attenhofer Jost, C.H.; Jenni, R.; Tanner, F.C.; Greutmann, M. Left ventricular non-compaction: Prevalence in congenital heart disease. Int. J. Cardiol. 2013, 167, 2477–2481. [Google Scholar] [CrossRef]
- Kumor, M.; Lipczynska, M.; Biernacka, E.K.; Klisiewicz, A.; Wojcik, A.; Konka, M.; Kozuch, K.; Szymanski, P.; Hoffman, P. Cardiac arrest and ventricular arrhythmia in adults with Ebstein anomaly and left ventricular non-compaction. J. Cardiol. 2018, 71, 484–487. [Google Scholar] [CrossRef]
- Marques, L.C.; Liguori, G.R.; Amarante Souza, A.C.; Aiello, V.D. Left Ventricular Noncompaction Is More Prevalent in Ventricular Septal Defect Than Other Congenital Heart Defects: A Morphological Study. J. Cardiovasc Dev. Dis 2020, 7, 39. [Google Scholar] [CrossRef] [PubMed]
- Pignatelli, R.H.; Texter, K.M.; Denfield, S.W.; Grenier, M.A.; Altman, C.A.; Ayres, N.A.; Chandra-Bose Reddy, S. LV Noncompaction in Ebstein’s anomaly in infants and outcomes. JACC Cardiovasc. Imaging 2014, 7, 207–209. [Google Scholar] [CrossRef]
- Hirono, K.; Hata, Y.; Miyao, N.; Okabe, M.; Takarada, S.; Nakaoka, H.; Ibuki, K.; Ozawa, S.; Yoshimura, N.; Nishida, N.; et al. Left Ventricular Noncompaction and Congenital Heart Disease Increases the Risk of Congestive Heart Failure. J. Clin. Med. 2020, 9, 785. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.M. Ebstein’s Anomaly: Genetics, Clinical Manifestations, and Management. Pediatr. Neonatol. 2017, 58, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Barra, S.N.; Providencia, R.; Paiva, L.; Nascimento, J.; Marques, A.L. A review on advanced atrioventricular block in young or middle-aged adults. Pacing Clin. Electrophysiol. 2012, 35, 1395–1405. [Google Scholar] [CrossRef]
- Gleva, M.J.; Wang, Y.; Curtis, J.P.; Berul, C.I.; Huddleston, C.B.; Poole, J.E. Complications Associated With Implantable Cardioverter Defibrillators in Adults With Congenital Heart Disease or Left Ventricular Noncompaction Cardiomyopathy (From the NCDR((R)) Implantable Cardioverter-Defibrillator Registry). Am. J. Cardiol. 2017, 120, 1891–1898. [Google Scholar] [CrossRef]
- Vermeer, A.M.; van Engelen, K.; Postma, A.V.; Baars, M.J.; Christiaans, I.; De Haij, S.; Klaassen, S.; Mulder, B.J.; Keavney, B. Ebstein anomaly associated with left ventricular noncompaction: An autosomal dominant condition that can be caused by mutations in MYH7. Am. J. Med. Genet. C Semin. Med. Genet. 2013, 163, 178–184. [Google Scholar] [CrossRef]
- Kayvanpour, E.; Sedaghat-Hamedani, F.; Gi, W.T.; Tugrul, O.F.; Amr, A.; Haas, J.; Zhu, F.; Ehlermann, P.; Uhlmann, L.; Katus, H.A.; et al. Clinical and genetic insights into non-compaction: A meta-analysis and systematic review on 7598 individuals. Clin. Res. Cardiol. 2019, 108, 1297–1308. [Google Scholar] [CrossRef]
- Monibi, A.A.; Neches, W.H.; Lenox, C.C.; Park, S.C.; Mathews, R.A.; Zuberbuhler, J.R. Left ventricular anomalies associated with Ebstein’s malformation of the tricuspid valve. Circulation 1978, 57, 303–306. [Google Scholar] [CrossRef] [PubMed]
- Daliento, L.; Angelini, A.; Ho, S.Y.; Frescura, C.; Turrini, P.; Baratella, M.C.; Thiene, G.; Anderson, R.H. Angiographic and morphologic features of the left ventricle in Ebstein’s malformation. Am. J. Cardiol. 1997, 80, 1051–1059. [Google Scholar] [CrossRef]
- Chin, K.T.; Xu, H.T.; Ching, Y.P.; Jin, D.Y. Differential subcellular localization and activity of kelch repeat proteins KLHDC1 and KLHDC2. Mol. Cell Biochem. 2007, 296, 109–119. [Google Scholar] [CrossRef]
- Qureshi, M.Y.; O’Leary, P.W.; Connolly, H.M. Cardiac imaging in Ebstein anomaly. Trends Cardiovasc. Med. 2018, 28, 403–409. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Lim, H.G.; Kim, W.H.; Lee, J.R.; Kim, Y.J. Long-Term Results after Surgical Treatment of Ebstein’s Anomaly: A 30-year Experience. Korean Circ. J. 2016, 46, 706–713. [Google Scholar] [CrossRef] [PubMed]
- Sainathan, S.; da Fonseca da Silva, L.; da Silva, J.P. Ebstein’s anomaly: Contemporary management strategies. J. Thorac. Dis. 2020, 12, 1161–1173. [Google Scholar] [CrossRef] [PubMed]
- Gioli-Pereira, L.; Pereira, A.C.; Mesquita, S.M.; Xavier-Neto, J.; Lopes, A.A.; Krieger, J.E. NKX2.5 mutations in patients with non-syndromic congenital heart disease. Int. J. Cardiol. 2010, 138, 261–265. [Google Scholar] [CrossRef]
- Digilio, M.C.; Bernardini, L.; Lepri, F.; Giuffrida, M.G.; Guida, V.; Baban, A.; Versacci, P.; Capolino, R.; Torres, B.; De Luca, A.; et al. Ebstein anomaly: Genetic heterogeneity and association with microdeletions 1p36 and 8p23.1. Am. J. Med. Genet. A 2011, 155, 2196–2202. [Google Scholar] [CrossRef]
- Bione, S.; D’Adamo, P.; Maestrini, E.; Gedeon, A.K.; Bolhuis, P.A.; Toniolo, D. A novel X-linked gene, G4.5. is responsible for Barth syndrome. Nat. Genet. 1996, 12, 385–389. [Google Scholar] [CrossRef]
- Kodo, K.; Ong, S.G.; Jahanbani, F.; Termglinchan, V.; Hirono, K.; InanlooRahatloo, K.; Ebert, A.D.; Shukla, P.; Abilez, O.J.; Churko, J.M.; et al. iPSC-derived cardiomyocytes reveal abnormal TGF-beta signalling in left ventricular non-compaction cardiomyopathy. Nat. Cell Biol. 2016, 18, 1031–1042. [Google Scholar] [CrossRef] [PubMed]
- van Waning, J.I.; Caliskan, K.; Hoedemaekers, Y.M.; van Spaendonck-Zwarts, K.Y.; Baas, A.F.; Boekholdt, S.M.; van Melle, J.P.; Teske, A.J.; Asselbergs, F.W.; Backx, A.; et al. Genetics, Clinical Features, and Long-Term Outcome of Noncompaction Cardiomyopathy. J. Am. Coll. Cardiol. 2018, 71, 711–722. [Google Scholar] [CrossRef]
- Kenton, A.B.; Sanchez, X.; Coveler, K.J.; Makar, K.A.; Jimenez, S.; Ichida, F.; Murphy, R.T.; Elliott, P.M.; McKenna, W.; Bowles, N.E.; et al. Isolated left ventricular noncompaction is rarely caused by mutations in G4.5, alpha-dystrobrevin and FK Binding Protein-12. Mol. Genet. Metab. 2004, 82, 162–166. [Google Scholar] [CrossRef]
- Luxan, G.; Casanova, J.C.; Martinez-Poveda, B.; Prados, B.; D’Amato, G.; MacGrogan, D.; Gonzalez-Rajal, A.; Dobarro, D.; Torroja, C.; Martinez, F.; et al. Mutations in the NOTCH pathway regulator MIB1 cause left ventricular noncompaction cardiomyopathy. Nat. Med. 2013, 19, 193–201. [Google Scholar] [CrossRef]
- Mercer, C.L.; Andreoletti, G.; Carroll, A.; Salmon, A.P.; Temple, I.K.; Ennis, S. Familial Ebstein Anomaly: Whole Exome Sequencing Identifies Novel Phenotype Associated With FLNA. Circ. Cardiovasc. Genet. 2017, 10, e001683. [Google Scholar] [CrossRef]
- Sicko, R.J.; Browne, M.L.; Rigler, S.L.; Druschel, C.M.; Liu, G.; Fan, R.; Romitti, P.A.; Caggana, M.; Kay, D.M.; Brody, L.C.; et al. Genetic Variants in Isolated Ebstein Anomaly Implicated in Myocardial Development Pathways. PLoS ONE 2016, 11, e0165174. [Google Scholar] [CrossRef]
- Hoedemaekers, Y.M.; Caliskan, K.; Majoor-Krakauer, D.; van de Laar, I.; Michels, M.; Witsenburg, M.; ten Cate, F.J.; Simoons, M.L.; Dooijes, D. Cardiac beta-myosin heavy chain defects in two families with non-compaction cardiomyopathy: Linking non-compaction to hypertrophic, restrictive, and dilated cardiomyopathies. Eur Heart J. 2007, 28, 2732–2737. [Google Scholar] [CrossRef]
- Stampfli, S.F.; Donati, T.G.; Hellermann, J.; Anwer, S.; Erhart, L.; Gruner, C.; Kaufmann, B.A.; Gencer, B.; Haager, P.K.; Muller, H.; et al. Right ventricle and outcome in left ventricular non-compaction cardiomyopathy. J. Cardiol. 2020, 75, 20–26. [Google Scholar] [CrossRef]
- Moorman, A.; Webb, S.; Brown, N.A.; Lamers, W.; Anderson, R.H. Development of the heart: (1) Formation of the cardiac chambers and arterial trunks. Heart 2003, 89, 806–814. [Google Scholar] [CrossRef]
- Sylva, M.; van den Hoff, M.J.; Moorman, A.F. Development of the human heart. Am. J. Med. Genet. A 2014, 164, 1347–1371. [Google Scholar] [CrossRef]
- Hinton, R.B.; Yutzey, K.E. Heart valve structure and function in development and disease. Annu. Rev. Physiol. 2011, 73, 29–46. [Google Scholar] [CrossRef]
- Combs, M.D.; Yutzey, K.E. Heart valve development: Regulatory networks in development and disease. Circ. Res. 2009, 105, 408–421. [Google Scholar] [CrossRef]
- Krishnan, A.; Samtani, R.; Dhanantwari, P.; Lee, E.; Yamada, S.; Shiota, K.; Donofrio, M.T.; Leatherbury, L.; Lo, C.W. A detailed comparison of mouse and human cardiac development. Pediatr. Res. 2014, 76, 500–507. [Google Scholar] [CrossRef]
- Anderson, R.H.; Webb, S.; Brown, N.A.; Lamers, W.; Moorman, A. Development of the heart: (2) Septation of the atriums and ventricles. Heart 2003, 89, 949–958. [Google Scholar] [CrossRef]
- Perez-Pomares, J.M.; de la Pompa, J.L. Signaling during epicardium and coronary vessel development. Circ. Res. 2011, 109, 1429–1442. [Google Scholar] [CrossRef]
- Gong, H.; Lyu, X.; Wang, Q.; Hu, M.; Zhang, X. Endothelial to mesenchymal transition in the cardiovascular system. Life Sci. 2017, 184, 95–102. [Google Scholar] [CrossRef]
- Lincoln, J.; Yutzey, K.E. Molecular and developmental mechanisms of congenital heart valve disease. Birth Defects Res. A Clin. Mol. Teratol. 2011, 91, 526–534. [Google Scholar] [CrossRef]
- de Vlaming, A.; Sauls, K.; Hajdu, Z.; Visconti, R.P.; Mehesz, A.N.; Levine, R.A.; Slaugenhaupt, S.A.; Hagege, A.; Chester, A.H.; Markwald, R.R.; et al. Atrioventricular valve development: New perspectives on an old theme. Differentiation 2012, 84, 103–116. [Google Scholar] [CrossRef]
- de Lange, F.J.; Moorman, A.F.; Anderson, R.H.; Manner, J.; Soufan, A.T.; de Gier-de Vries, C.; Schneider, M.D.; Webb, S.; van den Hoff, M.J.; Christoffels, V.M. Lineage and morphogenetic analysis of the cardiac valves. Circ. Res. 2004, 95, 645–654. [Google Scholar] [CrossRef]
- Lincoln, J.; Alfieri, C.M.; Yutzey, K.E. Development of heart valve leaflets and supporting apparatus in chicken and mouse embryos. Dev. Dyn. 2004, 230, 239–250. [Google Scholar] [CrossRef]
- Oechslin, E.; Jenni, R. Left ventricular non-compaction revisited: A distinct phenotype with genetic heterogeneity? Eur. Heart J. 2011, 32, 1446–1456. [Google Scholar] [CrossRef]
- Sedmera, D.; Pexieder, T.; Vuillemin, M.; Thompson, R.P.; Anderson, R.H. Developmental patterning of the myocardium. Anat. Rec. 2000, 258, 319–337. [Google Scholar] [CrossRef]
- Jimenez-Amilburu, V.; Rasouli, S.J.; Staudt, D.W.; Nakajima, H.; Chiba, A.; Mochizuki, N.; Stainier, D.Y.R. In Vivo Visualization of Cardiomyocyte Apicobasal Polarity Reveals Epithelial to Mesenchymal-like Transition during Cardiac Trabeculation. Cell Rep. 2016, 17, 2687–2699. [Google Scholar] [CrossRef]

| Age of Sporadic Case Presentation | Common Clinical Signs |
|---|---|
| Prenatal/Infancy (<1 year) [40,42,47,56,57,60,63] | Cyanosis |
| Childhood (1–12 years) [41,50] | Heart Failure Systolic Murmur |
| Adolescence (13–20 years) [22,39,43,44,49,54,59,67] OR Early Adulthood (21–40 years) [22,37,44,45,46,51,52,53,58,61] OR Middle Adulthood (41–60 years) [35,36,38,48,55,62,64,66] | Progressive or Exertional Dyspnea Systolic Murmur Palpitations or Tachycardia Chest Pain or Discomfort Arrhythmia |
| Late Adulthood (61 years+) [65] | Progressive or Exertional Dyspnea Asymptomatic/Incidental Finding |
| Clinical Signs | Imaging Modalities | Interventions/Therapies |
|---|---|---|
| Arrhythmia [35,37,43,45,51,55,61,63,65,73,74] Asymptomatic/Incidental Finding [36,65,66,75] Chest Pain or Discomfort [46,58,61,62] Cyanosis [47,57,74] Exertional Dyspnea [22,35,38,41,42,43,44,45,49,51,53,55,64,65,68,69,74] Heart Failure [50,73,74] Palpitations or Tachycardia [35,41,44,45,58,62] Systolic Murmur [39,40,45,48,49,54,59,66,67] | 2-D ECHO [7,22,35,36,37,38,39,40,41,42,43,44,45,46,47,48,49,50,51,52,53,54,55,56,57,58,59,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75] 3-D ECHO [36,39,46,49,53,61,65,75] Color Flow Doppler ECHO [36,37,44,46,47,48,51,54,58,59,61,63,68,69,70,73,75] Cardiac or Coronary CT Angiography [35,46,56,62] Cardiac MRI [37,38,40,41,45,50,51,53,55,56,62,64,65,66,69,73] Catheterization [53,73] Chest Radiograph [7,38,43,50,51,57,58,61] ECG [35,36,37,41,43,44,47,48,49,51,52,54,58,61,63,65,73,74,75] Electrophysiological Study [35,52,73,74] | Mechanical Ventilation [42,50,57] Radiofrequency Ablation [35,58,62] Arterioplasty/Annuloplasty [22] Other Cardiac Surgical Repair [7,41,47,53,56,70] Cardiac Transplantation [22,44,50,69] Atrial Septal Occluder (PFO Closure) [38] LVAD/BiVAD [43,53,74] Pacemaker/ICD [35,55,73,74] IABP [53] Inotropic Support (Epinephrine/Milirinone) [53] Anticoagulation (Aspirin/Warfarin/Heparin) [35,39,43,44,45,49,52,59,65,73] Heart Failure and Anti-arrhythmic Medications (Beta-Blockers/ACE inhibitors/Diuretics/Digoxin) [43,44,45,46,47,51,52,57,65,73,74] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Thareja, S.K.; Frommelt, M.A.; Lincoln, J.; Lough, J.W.; Mitchell, M.E.; Tomita-Mitchell, A. A Systematic Review of Ebstein’s Anomaly with Left Ventricular Noncompaction. J. Cardiovasc. Dev. Dis. 2022, 9, 115. https://doi.org/10.3390/jcdd9040115
Thareja SK, Frommelt MA, Lincoln J, Lough JW, Mitchell ME, Tomita-Mitchell A. A Systematic Review of Ebstein’s Anomaly with Left Ventricular Noncompaction. Journal of Cardiovascular Development and Disease. 2022; 9(4):115. https://doi.org/10.3390/jcdd9040115
Chicago/Turabian StyleThareja, Suma K., Michele A. Frommelt, Joy Lincoln, John W. Lough, Michael E. Mitchell, and Aoy Tomita-Mitchell. 2022. "A Systematic Review of Ebstein’s Anomaly with Left Ventricular Noncompaction" Journal of Cardiovascular Development and Disease 9, no. 4: 115. https://doi.org/10.3390/jcdd9040115
APA StyleThareja, S. K., Frommelt, M. A., Lincoln, J., Lough, J. W., Mitchell, M. E., & Tomita-Mitchell, A. (2022). A Systematic Review of Ebstein’s Anomaly with Left Ventricular Noncompaction. Journal of Cardiovascular Development and Disease, 9(4), 115. https://doi.org/10.3390/jcdd9040115







