Tacticity-Dependent Interchain Interactions of Poly(N-Isopropylacrylamide) in Water: Toward the Molecular Dynamics Simulation of a Thermoresponsive Microgel
Abstract
:1. Introduction
2. Results and Discussion
2.1. Below the LCST
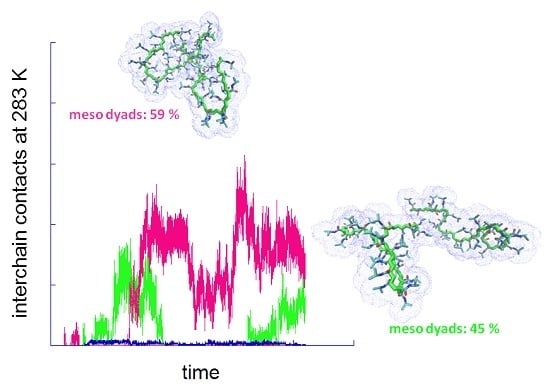
2.2. Above the LCST
3. Conclusions
4. Computing Details
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Fernandez-Nieves, A.; Wyss, H.M.; Mattsson, J.; Weitz, D.A. Microgel Suspensions, Fundamentals and Applications; Wiley-VCH Verlag GmbH & Co.: KGaA, Weinheim, Germany, 2011. [Google Scholar]
- Kwok, M.; Li, Z.; Ngai, T. Controlling the Synthesis and Characterization of Micrometer-Sized PNIPAM Microgels with Tailored Morphologies. Langmuir 2013, 29, 9581–9591. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Serpe, M.J. The Influence of Deposition Solution pH and Ionic Strength on the Quality of Poly(N-isopropylacrylamide) Microgel-Based Thin Films and Etalons. ACS Appl. Mater. Interfaces 2013, 5, 11977–11983. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Gao, Y.; Cao, C.; Chen, K.; Wen, Y.; Fang, D.; Li, L.; Guo, X. Binary Solvent Colloids of Thermosensitive Poly(N-isopropylacrylamide) Microgel for Smart Windows. Ind. Eng. Chem. Res. 2014, 53, 18462–18472. [Google Scholar] [CrossRef]
- Humphreys, B.A.; Willott, J.D.; Murdoch, T.J.; Webber, G.B.; Wanless, E.J. Specific ion modulated thermoresponse of poly(N-isopropylacrylamide) brushes. Phys. Chem. Chem. Phys. 2016, 18, 6037–6046. [Google Scholar] [CrossRef] [PubMed]
- Ito, M.; Ishizone, T. Living Anionic Polymerization of N-methoxymethyl-N-isopropylacrylamide: Synthesis of Well-defined poly(N-isopropylacrylamide) having various stereoregularity. J. Polym. Sci. A Polym. Chem. 2006, 44, 4832–4845. [Google Scholar] [CrossRef]
- Hirano, T.; Okumura, Y.; Kitajima, H.; Seno, M.; Sato, T. Dual Roles of Alkyl Alcohols as Syndiotactic-Specificity Inducers and Accelerators in the Radical Polymerization of N-Isopropylacrylamide and Some Properties of Syndiotactic Poly(N-isopropylacrylamide). J. Polym. Sci. A Polym. Chem. 2006, 44, 4450–4460. [Google Scholar] [CrossRef]
- Suito, Y.; Isobe, Y.; Habaue, S.; Okamoto, Y. Isotactic-specific Radical Polymerization of Methacrylamides in the Presence of Lewis Acids. J. Polym. Sci. A Polym. Chem. 2002, 40, 2496–2500. [Google Scholar] [CrossRef]
- Ray, B.; Okamoto, Y.; Kamigaito, M.; Sawamoto, M.; Seno, K.; Kanaoka, S.; Aoshima, S. Effect of Tacticity of Poly(N-isopropylacrylamide) on the Phase Separation Temperature of Its Aqueous Solutions. Polym. J. 2005, 37, 234–237. [Google Scholar] [CrossRef]
- Biswas, C.S.; Vishwakarma, N.K.; Patel, V.K.; Mishra, A.K.; Saha, S.; Ray, B. Synthesis and Study of the Properties of Stereocontrolled Poly(N-isopropylacrylamide) Gel and Its Linear Homopolymer Prepared in the Presence of a Y(OTf)3 Lewis Acid: Effect of the Composition of Methanol—Water Mixtures as Synthesis Media. Langmuir 2012, 28, 7014–7022. [Google Scholar] [CrossRef] [PubMed]
- Biswas, C.S.; Patel, V.K.; Vishwakarma, N.K.; Mishra, A.K.; Saha, S.; Ray, B. Synthesis and Characterization of Stereocontrolled Poly(N-isopropylacrylamide) Hydrogel Prepared in the Presence of Y(OTf)3 Lewis Acid. Langmuir 2010, 26, 6775–6782. [Google Scholar] [CrossRef] [PubMed]
- Tada, T.; Katsumoto, Y.; Goossens, K.; Uji-i, H.; Hofkens, J.; Shoji, T.; Kitamura, N.; Tsuboi, Y. Accelerating the Phase Separation in Aqueous Poly(N-isopropylacrylamide) Solutions by Slight Modification of the Polymer Stereoregularity. J. Phys. Chem. C 2013, 117, 10818–10824. [Google Scholar] [CrossRef]
- Nakano, S.; Ogiso, T.; Kita, R.; Shinyashiki, N.; Yagihara, S.; Yoneyama, M.; Katsumoto, Y. Thermoreversible Gelation of Isotactic-rich Poly(N-isopropylacrylamide) in Water. J. Chem. Phys. 2011, 135, 114903. [Google Scholar] [CrossRef] [PubMed]
- Hietala, S.; Nuopponen, M.; Kalliomäki, K.; Tenhu, H. Thermoassociating Poly(N-isopropylacrylamide) A–B–A Stereoblock Copolymers. Macromolecules 2008, 41, 2627–2631. [Google Scholar] [CrossRef]
- Nuopponen, M.; Kalliomäki, K.; Aseyev, V.; Tenhu, H. Spontaneous and Thermally Induced Self-Organization of A–B–A Stereoblock Polymers of N-Isopropylacrylamide in Aqueous Solutions. Macromolecules 2008, 41, 4881–4886. [Google Scholar] [CrossRef]
- De Oliveira, T.E.; Mukherji, D.; Kremer, K.; Netz, P.A. Effects of Stereochemistry and Copolymerization on the LCST of PNIPAm. J. Chem. Phys. 2017, 146, 034904. [Google Scholar] [CrossRef] [PubMed]
- Chiessi, E.; Paradossi, G. Influence of Tacticity on Hydrophobicity of Poly(N-isopropylacrylamide): A Single Chain Molecular Dynamics Simulation Study. J. Phys. Chem. B 2016, 120, 3765–3776. [Google Scholar] [CrossRef] [PubMed]
- Tucker, A.K.; Stevens, M.J. Study of the Polymer Length Dependence of the Single Chain Transition Temperature in Syndiotactic Poly(N-isopropylacrylamide) Oligomers in Water. Macromolecules 2012, 45, 6697–6703. [Google Scholar] [CrossRef]
- Deshmukh, S.A.; Sankaranarayanan, S.K.R.S.; Suthar, K.; Mancini, D.C. Role of Solvation Dynamics and Local Ordering of Water in Inducing Conformational Transitions in Poly(N-isopropylacrylamide) Oligomers through the LCST. J. Phys. Chem. B 2012, 116, 2651–2663. [Google Scholar] [CrossRef] [PubMed]
- Habaue, S.; Isobe, Y.; Okamoto, Y. Stereocontrolled radical polymerization of acrylamides and methacrylamides using Lewis acids. Tetrahedron 2002, 58, 8205–8209. [Google Scholar] [CrossRef]
- Ray, B.; Isobe, Y.; Matsumoto, K.; Habaue, S.; Okamoto, Y.; Kamigaito, M.; Sawamoto, M. RAFT Polymerization of N-Isopropylacrylamide in the Absence and Presence of Y(OTf)3: Simultaneous Control of Molecular Weight and Tacticity. Macromolecules 2004, 37, 1702–1710. [Google Scholar] [CrossRef]
- Nishi, K.; Hiroi, T.; Hashimoto, K.; Fujii, K.; Han, Y.-S.; Kim, T.-H.; Katsumoto, Y.; Shibayama, M. SANS and DLS Study of Tacticity Effects on Hydrophobicity and Phase Separation of Poly(N-isopropylacrylamide). Macromolecules 2013, 46, 6225–6232. [Google Scholar] [CrossRef]
- Richmond, T.J. Solvent Accessible Surface Area and Excluded Volume in Proteins. J. Mol. Biol. 1984, 178, 63–89. [Google Scholar] [CrossRef]
- Reynolds, J.A.; Gilbert, D.B.; Tanford, C. Empirical correlation between hydrophobic free energy and aqueous cavity surface area. Proc. Natl. Acad. Sci. USA 1974, 71, 2925–2927. [Google Scholar] [CrossRef] [PubMed]
- Katsumoto, Y.; Kubosaki, N.; Miyata, T. Molecular Approach to Understand the Tacticity Effects on the Hydrophilicity of Poly(N-isopropylacrylamide): Solubility of Dimer Model Compounds in Water. J. Phys. Chem. B 2010, 114, 13312–13318. [Google Scholar] [CrossRef] [PubMed]
- Katsumoto, Y.; Kubosaki, N. Tacticity Effects on the Phase Diagram for Poly(N-isopropylacrylamide) in Water. Macromolecules 2008, 41, 5955–5956. [Google Scholar] [CrossRef]
- De Gennes, P.G. A Second Type of Phase Separation in Polymer Solutions. C. R. Acad. Sci. 1991, 313, 1117–1122. [Google Scholar]
- Halperin, A.; Kröger, M.; Winnik, F.M. Poly(N-isopropylacrylamide) Phase Diagrams: Fifty Years of Research. Angew. Chem. Int. Ed. 2015, 54, 15342–15367. [Google Scholar] [CrossRef] [PubMed]
- Acciaro, R.; Gilányi, T.; Varga, I. Preparation of Monodisperse Poly(N-isopropylacrylamide) Microgel Particles with Homogenous Cross-Link Density Distribution. Langmuir 2011, 27, 7917–7925. [Google Scholar] [CrossRef] [PubMed]
- Flory, P.J.; Mark, J.E.; Abe, A. Random-Coil Configurations of Vinyl Polymer Chains. The Influence of Stereoregularity on the Average Dimensions. J. Am. Chem. Soc. 1966, 88, 639–650. [Google Scholar]
- Autieri, E.; Chiessi, E.; Lonardi, A.; Paradossi, G.; Sega, M. Conformation and Dynamics of Poly(N-isopropyl acrylamide) Trimers in Water: A Molecular Dynamics and Metadynamics Simulation Study. J. Phys. Chem. B 2011, 115, 5827–5839. [Google Scholar] [CrossRef] [PubMed]
- Jorgensen, W.L.; Maxwell, D.S.; Tirado-Rives, J. Development and Testing of the OPLS All-Atom Force Field on Conformational Energetics and Properties of Organic Liquids. J. Am. Chem. Soc. 1996, 118, 11225–11236. [Google Scholar] [CrossRef]
- Siu, S.W.I.; Pluhackova, K.; Böckmann, R.A. Optimization of the OPLS-AA Force Field for Long Hydrocarbons. J. Chem. Theory Comput. 2012, 8, 1459–1470. [Google Scholar] [CrossRef] [PubMed]
- Abbott, L.J.; Tucker, A.K.; Stevens, M.J. Single Chain Structure of a Poly(N-isopropylacrylamide) Surfactant in Water. J. Phys. Chem. B 2015, 119, 3837–3845. [Google Scholar] [CrossRef] [PubMed]
- Hockney, R.W. The Potential Calculation and Some Applications. Methods Comput. Phys. 1970, 9, 136–211. [Google Scholar]
- Hess, B.; Bekker, H.; Berendsen, H.J.C.; Fraaije, J.G.E.M. LINCS: A Linear Constraint Solver for Molecular Simulations. J. Comput. Chem. 1997, 18, 1463–1472. [Google Scholar] [CrossRef]
- Bussi, G.; Donadio, D.; Parrinello, M. Canonical Sampling through Velocity Rescaling. J. Chem. Phys. 2007, 126, 014101. [Google Scholar] [CrossRef] [PubMed]
- Parrinello, M.; Rahman, A. Polymorphic Transitions in Single Crystals: A New Molecular Dynamics Method. J. Appl. Phys. 1981, 52, 7182–7190. [Google Scholar] [CrossRef]
- Nosé, S.; Klein, M.L. Constant pressure molecular dynamics for molecular systems. Mol. Phys. 1983, 50, 1055–1076. [Google Scholar] [CrossRef]
- Essmann, U.; Perera, L.; Berkowitz, M.L.; Darden, T.; Lee, H.; Pedersen, L.G. A Smooth Particle Mesh Ewald Method. J. Chem. Phys. 1995, 103, 8577–8593. [Google Scholar] [CrossRef]
- Páll, S.; Abraham, M.J.; Kutzner, C.; Hess, B.; Lindahl, E. Tackling Exascale Software Challenges in Molecular Dynamics Simulations with GROMACS. In Solving Software Challenges for Exascale; Markidis, S., Laure, E., Eds.; Springer: London, UK, 2015; Volume 8759, pp. 3–27. [Google Scholar]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High Performance Molecular Simulations through Multi-level Parallelism from Laptops to Supercomputers. SoftwareX 2015, 1–2, 19–25. [Google Scholar] [CrossRef]
- Humphrey, W.; Dalke, A.; Schulten, K. VMD: Visual Molecular Dynamics. J. Mol. Graph. 1996, 14, 33–38. [Google Scholar] [CrossRef]
- Eisenhaber, F.; Lijnzaad, P.; Argos, P.; Sander, C.; Scharf, M. The Double Cube Lattice Method: Efficient Approaches to Numerical Integration of Surface Area and Volume and to Dot Surface Contouring of Molecular Assemblies. J. Comp. Chem. 1995, 16, 273–284. [Google Scholar] [CrossRef]
- Bondi, A. Van der Waals Volumes and Radii. J. Phys. Chem. 1964, 68, 441–451. [Google Scholar] [CrossRef]









| Stereoisomer | <r> (nm) | <NR> 1,2 | <NJ> 1,2 | <LJmean> 1,2 |
|---|---|---|---|---|
| Atactic | 3.1 ± 0.5 (A) 3.0 ± 0.7 (B) | 3 ± 2 (A) 2 ± 1 (B) | 3 ± 2 (A) 1.4 ± 0.6 (B) | 1.2 ± 0.4 (A) 1.6 ± 0.6 (B) |
| Isotactic-rich | 3 ± 1 (A) 1.7 ± 0.7 (B) | 8 ± 3 (A) 10 ± 3 (B) | 4 ± 1 (A) 5 ± 1 (B) | 1.9 ± 0.6 (A) 2.0 ± 0.6 (B) |
| Stereoisomer | <NR> 1,2 | <NJ> 1,2 | <LJmean> 1,2 |
|---|---|---|---|
| Atactic | 13 ± 4 (A) 12 ± 3 (B) | 5 ± 1 (A) 6 ± 2 (B) | 2.5 ± 0.8 (A) 1.9 ± 0.5 (B) |
| Isotactic-rich | 12 ± 2 (A) 11 ± 2 (B) | 6 ± 1 (A) 7 ± 1 (B) | 2.1 ± 0.6 (A) 1.5 ± 0.2 (B) |
| Meso Dyads Content (%) | Dyads Sequence 1 |
|---|---|
| 45 | rrrmmrmrrrrmmrrmmmmrmmrmrmrrr |
| 59 | rmmrrmrrmmrrmmmmrmmrrmmmrrmmm |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paradossi, G.; Chiessi, E. Tacticity-Dependent Interchain Interactions of Poly(N-Isopropylacrylamide) in Water: Toward the Molecular Dynamics Simulation of a Thermoresponsive Microgel. Gels 2017, 3, 13. https://doi.org/10.3390/gels3020013
Paradossi G, Chiessi E. Tacticity-Dependent Interchain Interactions of Poly(N-Isopropylacrylamide) in Water: Toward the Molecular Dynamics Simulation of a Thermoresponsive Microgel. Gels. 2017; 3(2):13. https://doi.org/10.3390/gels3020013
Chicago/Turabian StyleParadossi, Gaio, and Ester Chiessi. 2017. "Tacticity-Dependent Interchain Interactions of Poly(N-Isopropylacrylamide) in Water: Toward the Molecular Dynamics Simulation of a Thermoresponsive Microgel" Gels 3, no. 2: 13. https://doi.org/10.3390/gels3020013