Ultrasensitive Electroanalytical Detection of Pb2+ and H2O2 Using Bi and Fe—Based Nanoparticles Embedded into Porous Carbon Xerogel—The Influence of Nanocomposite Pyrolysis Temperatures
Abstract
:1. Introduction
2. Results and Discussion
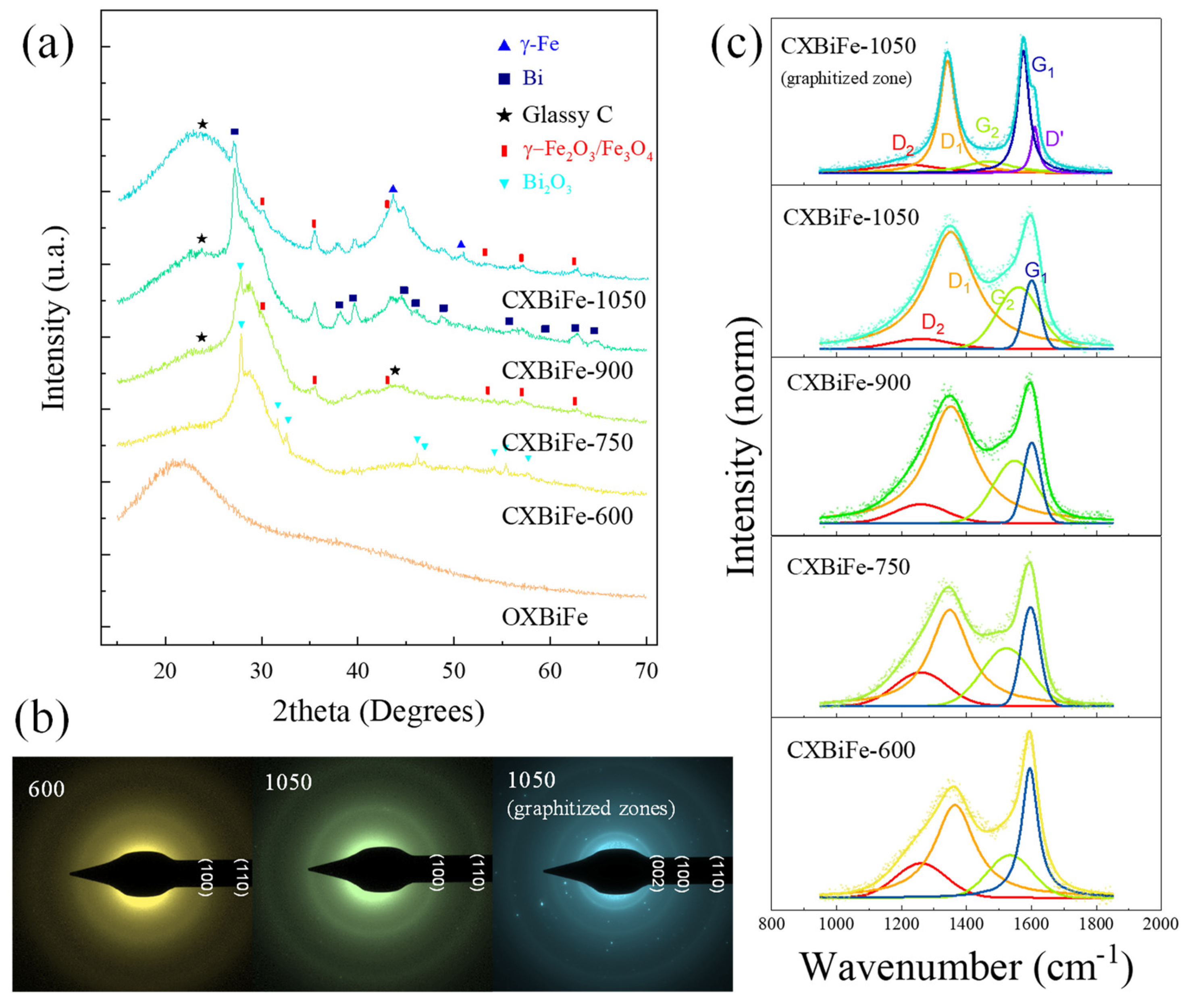
2.1. Morphological and Structural Characterization
2.1.1. Pyrolysis-Induced Changes in the Crystalline Structure
2.1.2. Effects Induced at Micro- and Nano-Scales
2.2. Electrochemical Behavior of GC/Chi-(CXBiFe-T) Modified Electrodes
2.2.1. EIS Measurements
2.2.2. Square Wave Voltammetry Measurements for H2O2 Detection
2.2.3. Square Wave Anodic Stripping Voltammetry Measurements for Pb2+ Detection
2.2.4. Correlations between the Morphological and Structural Characteristics and the Electroanalytical Behavior of BiFe-Carbon Xerogel Nanocomposites Modified Electrodes
Conductivity and Porosity
Nanoparticle Activation and Spatial Statistics of Diffusion Domains
2.2.5. Sensor—Long-Term Stability and Repeatability
2.2.6. Real Sample Analysis
3. Conclusions
4. Materials and Methods
4.1. Reagents
4.2. Synthesis and Temperature Program for CXBiFe-T Xerogel Ternary Composite
4.3. Characterization Methods
4.4. Preparation of the GC/Chi-(CXBiFe-T) Electrodes
4.5. Electrochemical Measurements
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Yang, Y.; Chiang, K.; Burke, N. Porous Carbon-Supported Catalysts for Energy and Environmental Applications: A Short Review. Catal. Today 2011, 178, 197–205. [Google Scholar] [CrossRef]
- Candelaria, S.L.; Shao, Y.; Zhou, W.; Li, X.; Xiao, J.; Zhang, J.G.; Wang, Y.; Liu, J.; Li, J.; Cao, G. Nanostructured Carbon for Energy Storage and Conversion. Nano Energy 2012, 1, 195–220. [Google Scholar] [CrossRef]
- Fort, C.I.; Pop, L.C. Heavy Metal and Metalloid Electrochemical Detection by Composite Nanostructures. In Advanced Nanostructures for Environmental Health; Elsevier: Amsterdam, The Netherlands, 2020; pp. 185–250. [Google Scholar]
- Maldonado-Hódar, F.J. Advances in the Development of Nanostructured Catalysts Based on Carbon Gels. Catal. Today 2013, 218–219, 43–50. [Google Scholar] [CrossRef]
- Cotet, L.C.; Gich, M.; Roig, A.; Popescu, I.C.; Cosoveanu, V.; Molins, E.; Danciu, V. Synthesis and Structural Characteristics of Carbon Aerogels with a High Content of Fe, Co, Ni, Cu, and Pd. J. Non-Cryst. Solids 2006, 352, 2772–2777. [Google Scholar] [CrossRef]
- Amaral-Labat, G.; Munhoz, M.G.C.; Fonseca, B.C.d.S.; Boss, A.F.N.; de Almeida-Mattos, P.; Braghiroli, F.L.; Bouafif, H.; Koubaa, A.; Lenz e Silva, G.F.B.; Baldan, M.R. Xerogel-like Materials from Sustainable Sources: Properties and Electrochemical Performances. Energies 2021, 14, 7977. [Google Scholar] [CrossRef]
- Maldonado-Hódar, F.J.; Moreno-Castilla, C.; Rivera-Utrilla, J.; Hanzawa, Y.; Yamada, Y. Catalytic Graphitization of Carbon Aerogels by Transition Metals. Langmuir 2000, 16, 4367–4373. [Google Scholar] [CrossRef]
- Liu, Z.; Lv, C.; Tan, X. One-Pot Synthesis of Fe, Co and Ni-Doped Carbon Xerogels and Their Magnetic Properties. J. Phys. Chem. Solids 2013, 74, 1275–1280. [Google Scholar] [CrossRef]
- Fort, C.I.; Cotet, L.C.; Danciu, V.; Turdean, G.L.; Popescu, I.C. Iron Doped Carbon Aerogel—New Electrode Material for Electrocatalytic Reduction of H2O2. Mater. Chem. Phys. 2013, 138, 893–898. [Google Scholar] [CrossRef]
- Prieto-simo, B.; Armatas, G.S.; Pomonis, P.J.; Nanos, C.G.; Prodromidis, M.I. Metal-Dispersed Xerogel-Based Composite Films for the Development of Interference Free Oxidase-Based Biosensors. Chem. Mater. 2004, 16, 1026–1034. [Google Scholar] [CrossRef]
- Tian, K.; Prestgard, M.; Tiwari, A. A Review of Recent Advances in Nonenzymatic Glucose Sensors. Mater. Sci. Eng. C 2014, 41, 100–118. [Google Scholar] [CrossRef]
- Adekunle, A.S.; Agboola, B.O.; Pillay, J.; Ozoemena, K.I. Electrocatalytic Detection of Dopamine at Single-Walled Carbon Nanotubes-Iron (III) Oxide Nanoparticles Platform. Sens. Actuators B Chem. 2010, 148, 93–102. [Google Scholar] [CrossRef]
- Chircov, C.; Grumezescu, A.M.; Holban, A.M. Magnetic Particles for Advanced Molecular Diagnosis. Materials 2019, 12, 2158. [Google Scholar] [CrossRef] [PubMed]
- Winkler, R.; Ciria, M.; Ahmad, M.; Plank, H.; Marcuello, C. A Review of the Current State of Magnetic Force Microscopy to Unravel the Magnetic Properties of Nanomaterials Applied in Biological Systems and Future Directions for Quantum Technologies. Nanomaterials 2023, 13, 2585. [Google Scholar] [CrossRef] [PubMed]
- Fort, C.I.; Cotet, L.C.; Vulpoi, A.; Turdean, G.L.; Danciu, V.; Baia, L.; Popescu, I.C. Bismuth Doped Carbon Xerogel Nanocomposite Incorporated in Chitosan Matrix for Ultrasensitive Voltammetric Detection of Pb(II) and Cd(II). Sens. Actuators B Chem. 2015, 220, 712–719. [Google Scholar] [CrossRef]
- Rusu, M.M.; Fort, C.I.; Cotet, L.C.; Vulpoi, A.; Todea, M.; Turdean, G.L.; Danciu, V.; Popescu, I.C.; Baia, L. Insights into the Morphological and Structural Particularities of Highly Sensitive Porous Bismuth-Carbon Nanocomposites Based Electrochemical Sensors. Sens. Actuators B Chem. 2018, 268, 398–410. [Google Scholar] [CrossRef]
- Švancara, I.; Prior, C.; Hočevar, S.B.; Wang, J. A Decade with Bismuth-Based Electrodes in Electroanalysis. Electroanalysis 2010, 22, 1405–1420. [Google Scholar] [CrossRef]
- Fort, C.I.; Rusu, M.M.; Cotet, L.C.; Vulpoi, A.; Florea, I.; Tuseau-Nenez, S.; Baia, M.; Baibarac, M.; Baia, L. Carbon Xerogel Nanostructures with Integrated Bi and Fe Components for Hydrogen Peroxide and Heavy Metal Detection. Molecules 2021, 26, 117. [Google Scholar] [CrossRef]
- Fort, C.I.; Rusu, M.M.; Pop, L.C.; Cotet, L.C.; Vulpoi, A.; Baia, M.; Baia, L. Preparation and Characterization of Carbon Xerogel Based Composites for Electrochemical Sensing and Photocatalytic Degradation. J. Nanosci. Nanotechnol. 2021, 21, 2323–2333. [Google Scholar] [CrossRef]
- Rusu, M.M.; Vulpoi, A.; Maurin, I.; Cotet, L.C.; Pop, L.C.; Fort, C.I.; Baia, M.; Baia, L.; Florea, I. Thermal Evolution of C–Fe–Bi Nanocomposite System: From Nanoparticle Formation to Heterogeneous Graphitization Stage. Microsc. Microanal. 2022, 28, 317–329. [Google Scholar] [CrossRef]
- Jurkiewicz, K.; Pawlyta, M.; Burian, A. Structure of Carbon Materials Explored by Local Transmission Electron Microscopy and Global Powder Diffraction Probes. C J. Carbon Res. 2018, 4, 68. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Robertson, J. Resonant Raman Spectroscopy of Disordered, Amorphous, and Diamondlike Carbon. Phys. Rev. B Condens. Matter Mater. Phys. 2001, 64, 075414. [Google Scholar] [CrossRef]
- Théry, A.; Béguin, F.; Kocon, L.; Lillo-Rodenas, M. Influence of the Carbonisation Temperature on the Structural and Electrochemical Properties of Carbon Aerogels. 2016; Volume 4. Available online: https://www.researchgate.net/publication/237627830_Influence_of_the_carbonisation_temperature_on_the_structural_and_electrochemical_properties_of_carbon_aerogels (accessed on 1 January 2023).
- Davies, T.J.; Moore, R.R.; Banks, C.E.; Compton, R.G. The Cyclic Voltammetric Response of Electrochemically Heterogeneous Surfaces. J. Electroanal. Chem. 2004, 574, 123–152. [Google Scholar] [CrossRef]
- Compton, R.G.; Banks, C.E. Understanding Voltammetry; Imperial College Press: London, UK, 2010; ISBN 978-1-84816-585-4. [Google Scholar]
- Wang, J. Study of Electrode Reactions and Interfacial Properties. In Analytical Electrochemistry; Wiley Online Books; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006; pp. 29–66. ISBN 9780471790303. [Google Scholar]
- Takai, K.; Oga, M.; Sato, H.; Enoki, T.; Ohki, Y.; Taomoto, A.; Suenaga, K.; Iijima, S. Structure and Electronic Properties of a Nongraphitic Disordered Carbon System and Its Heat-Treatment Effects. Phys. Rev. B Condens. Matter Mater. Phys. 2003, 67, 214202. [Google Scholar] [CrossRef]
- Chang, C.-T.; Lin, C.-Y. Electrochemical Reduction of Hydrogen Peroxide by Nanostructured Hematite Modified Electrodes. RSC Adv. 2016, 6, 67428–67434. [Google Scholar] [CrossRef]
- Zhang, R.; Chen, W. Fe3C-Functionalized 3D Nitrogen-Doped Carbon Structures for Electrochemical Detection of Hydrogen Peroxide. Sci. Bull. 2015, 60, 522–531. [Google Scholar] [CrossRef]
- Sheng, Z.M.; Gan, Z.Z.; Huang, H.; Niu, R.L.; Han, Z.W.; Jia, R.P. M-Nx (M = Fe, Co, Ni, Cu) Doped Graphitic Nanocages with High Specific Surface Area for Non-Enzymatic Electrochemical Detection of H2O2. Sens. Actuators B Chem. 2020, 305, 127550. [Google Scholar] [CrossRef]
- Gao, C.; Yu, X.Y.; Xu, R.X.; Liu, J.H.; Huang, X.J. AlOOH-Reduced Graphene Oxide Nanocomposites: One-Pot Hydrothermal Synthesis and Their Enhanced Electrochemical Activity for Heavy Metal Ions. ACS Appl. Mater. Interfaces 2012, 4, 4672–4682. [Google Scholar] [CrossRef] [PubMed]
- Gumpu, M.B.; Veerapandian, M.; Krishnan, U.M.; Rayappan, J.B.B. Simultaneous Electrochemical Detection of Cd(II), Pb(II), As(III) and Hg(II) Ions Using Ruthenium(II)-Textured Graphene Oxide Nanocomposite. Talanta 2017, 162, 574–582. [Google Scholar] [CrossRef] [PubMed]
- Pu, Y.; Wu, Y.; Yu, Z.; Lu, L.; Wang, X. Simultaneous Determination of Cd2+ and Pb2+ by an Electrochemical Sensor Based on Fe3O4/Bi2O3/C3N4 Nanocomposites. Talanta Open 2021, 3, 100024. [Google Scholar] [CrossRef]
- Zhao, G.; Liu, G. Synthesis of a Three-Dimensional (BiO)2CO3@single-Walled Carbon Nanotube Nanocomposite and Its Application for Ultrasensitive Detection of Trace Pb(II) and Cd(II) by Incorporating Nafion. Sens. Actuators B Chem. 2019, 288, 71–79. [Google Scholar] [CrossRef]
- Bandosz, T.J. Surface Chemistry of Carbon Materials. In Carbon Materials for Catalysis; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2009; pp. 45–92. [Google Scholar] [CrossRef]
- Najeh, I.; Ben Mansour, N.; Dahman, H.; Alyamani, A.; El Mir, L. DC and AC Characterizations of Electrical Conducting Nanoporous Carbon Structures Based on Resorcinol-Formaldehyde. J. Phys. Chem. Solids 2012, 73, 707–712. [Google Scholar] [CrossRef]
- Najeh, I.; Mansour, N.B.E.N.; Dahman, H.; Alyamani, A.; Mir, L.E.L. Electrical Properties of Partial Carbonized Nanoporous Resin Based on Resorcinol-Formaldehyde. Sens. Transducers 2014, 27, 285–289. [Google Scholar]
- Dai, X.; Wildgoose, G.G.; Salter, C.; Crossley, A.; Compton, R.G. Electroanalysis Using Macro-, Micro-, and Nanochemical Architectures on Electrode Surfaces. Bulk Surface Modification of Glassy Carbon Microspheres with Gold Nanoparticles and Their Electrical Wiring Using Carbon Nanotubes. Anal. Chem. 2006, 78, 6102–6108. [Google Scholar] [CrossRef] [PubMed]
- Guenther, G.; Theissmann, R.; Guillon, O. Size-Dependent Phase Transformations in Bismuth Oxide Nanoparticles. II. Melting and Stability Diagram. J. Phys. Chem. C 2014, 118, 27020–27027. [Google Scholar] [CrossRef]
- Lu, J.; Qiao, L.J.; Fu, P.Z.; Wu, Y.C. Phase Equilibrium of Bi2O3–Fe2O3 Pseudo-Binary System and Growth of BiFeO3 Single Crystal. J. Cryst. Growth 2011, 318, 936–941. [Google Scholar] [CrossRef]
- Henstridge, M.C.; Compton, R.G. Mass Transport to Micro- and Nanoelectrodes and Their Arrays: A Review. Chem. Rec. 2012, 12, 63–71. [Google Scholar] [CrossRef]
- Streeter, I.; Baron, R.; Compton, R.G. Voltammetry at Nanoparticle and Microparticle Modified Electrodes: Theory and Experiment. J. Phys. Chem. C 2007, 111, 17008–17014. [Google Scholar] [CrossRef]
- Aziz, S.B.; Brza, M.A.; Brevik, I.; Hafiz, M.H.; Asnawi, A.S.F.M.; Yusof, Y.M.; Abdulwahid, R.T.; Kadir, M.F.Z. Blending and Characteristics of Electrochemical Double-Layer Capacitor Device Assembled from Plasticized Proton Ion Conducting Chitosan:Dextran:NH4PF6 Polymer Electrolytes. Polymers 2020, 12, 2103. [Google Scholar] [CrossRef]
- Pele, K.G.; Amaveda, H.; Mora, M.; Marcuello, C.; Lostao, A.; Alamán-Díez, P.; Pérez-Huertas, S.; Ángeles Pérez, M.; García-Aznar, J.M.; García-Gareta, E. Hydrocolloids of Egg White and Gelatin as a Platform for Hydrogel-Based Tissue Engineering. Gels 2023, 9, 505. [Google Scholar] [CrossRef]
- Shimodaira, N.; Masui, A. Raman Spectroscopic Investigations of Activated Carbon Materials. J. Appl. Phys. 2002, 92, 902–909. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An Open-Source Platform for Biological-Image Analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Frank, E.; Hall, M.A.; Witten, I.H. The WEKA Workbench. In Data Mining; Elsevier: Amsterdam, The Netherlands, 2017; pp. 553–571. [Google Scholar] [CrossRef]
- Ward Jones, S.E.; Chevallier, F.G.; Paddon, C.A.; Compton, R.G. General Theory of Cathodic and Anodic Stripping Voltammetry at Solid Electrodes: Mathematical Modeling and Numerical Simulations. Anal. Chem. 2007, 79, 4110–4119. [Google Scholar] [CrossRef]







| Sample | dXRD (nm) | ID1/IG1 | SBET (m2/g) | DDLS (nm) | C/O/Bi/Fe (at%) | dnp (nm) | DV (nm) | |||
|---|---|---|---|---|---|---|---|---|---|---|
| Bi2O3 | Bi | Fe2O3/Fe3O4 | γ-Fe | |||||||
| OXBiFe | - | - | - | - | 0.86 | 10 | - | 74.0/25.5/0.2/0.2 | - | - |
| CXBiFe-600 | 33 | - | - | - | 0.72 | 229 | 1400 | 93.6/5.6/0.4/0.5 | 18 | 119 |
| CXBiFe-750 | 45 | - | 14 | - | 0.97 | 146 | 740 | 95.2/3.8/0.5/0.5 | 21 | 128 |
| CXBiFe-900 | - | 18 | 13 | 13 | 1.46 | 91 | 655 | 92.3/7.1/0.3/0.3 | 29 | 129 |
| CXBiFe-1050 | - | 22 | 18 | 13 | 1.70 | 16 | 570 | 94.4/4.8/0.1/0.7 | 33 | 154 |
| Electrode Type | GC | GC/Chi-CXBiFe-600 | GC/Chi-CXBiFe-750 | GC/Chi-CXBiFe-900 | GC/Chi-CXBiFe-1050 |
|---|---|---|---|---|---|
| Rel (Ω cm2) * | 5.98 ± 3 | 7.98 ± 0.9 | 22.6 ± 2.5 | 6.65 ± 1.5 | 12.2 ± 2.6 |
| CPE (µS sn/cm2) * n * | 25.2 ± 6 0.87 ± 1.0 | 84.1 ± 1 0.74 ± 0.22 | 58.2 ± 11 0.76 ± 2.1 | 50.2 ± 4 0.75 ± 0.79 | 40.9 ± 8 0.78 ± 1.4 |
| Rct (kΩ cm2) * | 6.2 ± 2 | 3.93 ± 1.6 | 1.5 ± 15.6 | 1.25 ± 5.4 | 0.91 ± 4.3 |
| W (mS s1/2/cm2) * | 4.89 ± 5 | 0.38 ± 1 | 0.30 ± 6.7 | 0.76 ± 3.6 | 1.05 ± 3.7 |
| C (µF/cm2) | 13.3 | 56.6 | 26.4 | 19.5 | 16.5 |
| χ2 | 0.003716 | 0.0002446 | 0.007471 | 0.002048 | 0.005647 |
| Electrode Type | Peak Potential (V*) | Linear Range (mM) | Sensitivity (µA/mM) | Detection Limit (µM) | Ref. |
|---|---|---|---|---|---|
| (Fe-CA)-CPE | −0.3 | 1–50 | 1.78 | 500.0 | [9] |
| α-Fe2O3NP|FePO4 | −0.3 | 1.66–4.95 | 225.00 | 3.4 | [28] |
| Fe3C/NG | −0.6 | 0.05–15 | 9.44 | 35.0 | [29] |
| Fe-NGCs | −0.3 | 0.001–5 | 13.30 | 0.5 | [30] |
| GC/Chi-BiFeCX | −0.3 | 0.005–0.05 | 1860.00 | 4.8 | [19] |
| GC/Chi-CXBiFe1.2 | −0.3 | 0.003–0.03 | 2350.00 | 0.2 | [18] |
| GC/Chi-(CXBiFe-900) | −0.50 | 0.05–1 | 2.10 | 5.7 | This work |
| GC/Chi-(CXBiFe-1050) | −0.46 | 0.05–1 | 4.55 | 2.5 |
| Electrode Type | Peak Potential (V*) | Linear Range | Sensitivity (µA/µM) | Detection Limit (pM) | Ref. | |
|---|---|---|---|---|---|---|
| µM | pM | |||||
| GC/AlOOH-rGO | −0.6 | 0.2–0.8 | 3.5 | 93.20 | [31] | |
| Au/[Ru(bpy)3]2+-GO | −0.38 | 0.1–1.5 | 24.1 | 35.00 | [32] | |
| Fe3O4/Bi2O3/C3N4/GC | −0.5 | 0.01–3 | 82.5 | 103 | [33] | |
| NiO/rGO/GCE | 0.03–0.6 | 92.8 | 104 | [34] | ||
| GC/Chi-(BiCX)Imp | −0.55 | 1–10 | 1.1 × 06 | 0.36 | [15] | |
| GC/Chi-(BiCX)Cos | −0.56 | 1–10 | 1.3 × 106 | 0.28 | [16] | |
| GC/Chi-(BiCA)Cos | −0.44 | 1–10 | 2.3 × 105 | 0.48 | [16] | |
| GC/Chi-(CXBiFe0.01) | −0.58 | 1–10 | 1.0 × 106 | 0.56 | [18] | |
| GC/Chi-(CXBiFe-600) | −0.53 | 10–90 | 2 × 104 | 6.19 | This work | |
| GC/Chi-(CXBiFe-750) | −0.48 | 10–110 | 4.9 × 104 | 5.67 | ||
| GC/Chi-(CXBiFe-900) | −0.44 | 1–12 | 2.5 × 105 | 0.30 | ||
| GC/Chi-(CXBiFe-1050) | −0.40 | 0.5–10 | 9.2 × 105 | 0.09 | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rusu, M.M.; Fort, C.I.; Vulpoi, A.; Barbu-Tudoran, L.; Baia, M.; Cotet, L.C.; Baia, L. Ultrasensitive Electroanalytical Detection of Pb2+ and H2O2 Using Bi and Fe—Based Nanoparticles Embedded into Porous Carbon Xerogel—The Influence of Nanocomposite Pyrolysis Temperatures. Gels 2023, 9, 868. https://doi.org/10.3390/gels9110868
Rusu MM, Fort CI, Vulpoi A, Barbu-Tudoran L, Baia M, Cotet LC, Baia L. Ultrasensitive Electroanalytical Detection of Pb2+ and H2O2 Using Bi and Fe—Based Nanoparticles Embedded into Porous Carbon Xerogel—The Influence of Nanocomposite Pyrolysis Temperatures. Gels. 2023; 9(11):868. https://doi.org/10.3390/gels9110868
Chicago/Turabian StyleRusu, Mihai M., Carmen I. Fort, Adriana Vulpoi, Lucian Barbu-Tudoran, Monica Baia, Liviu C. Cotet, and Lucian Baia. 2023. "Ultrasensitive Electroanalytical Detection of Pb2+ and H2O2 Using Bi and Fe—Based Nanoparticles Embedded into Porous Carbon Xerogel—The Influence of Nanocomposite Pyrolysis Temperatures" Gels 9, no. 11: 868. https://doi.org/10.3390/gels9110868










