Effects of Transglutaminase on Myofibrillar Protein Composite Gels with Addition of Non-Meat Protein Emulsion
Abstract
:1. Introduction
2. Results and Discussion
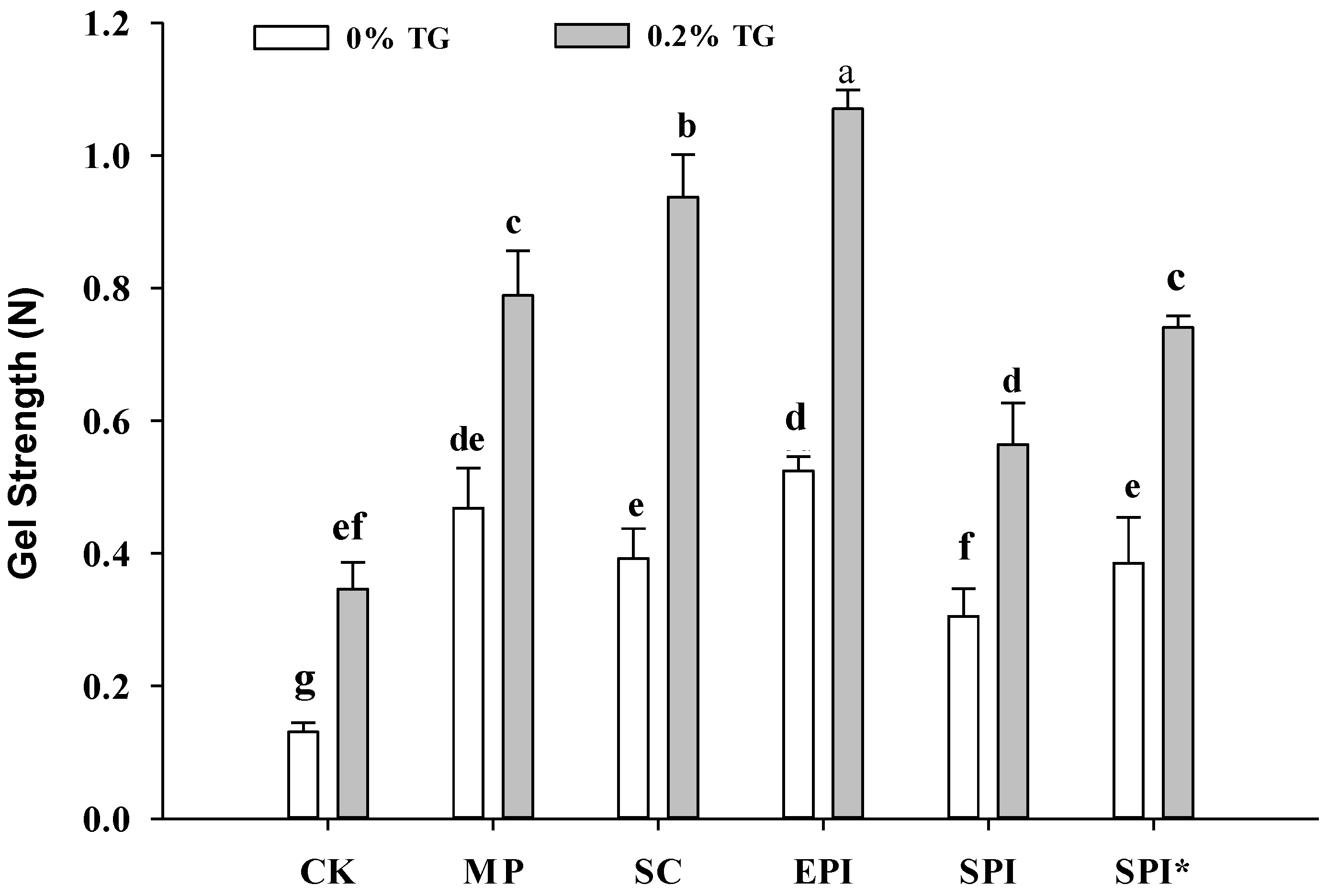
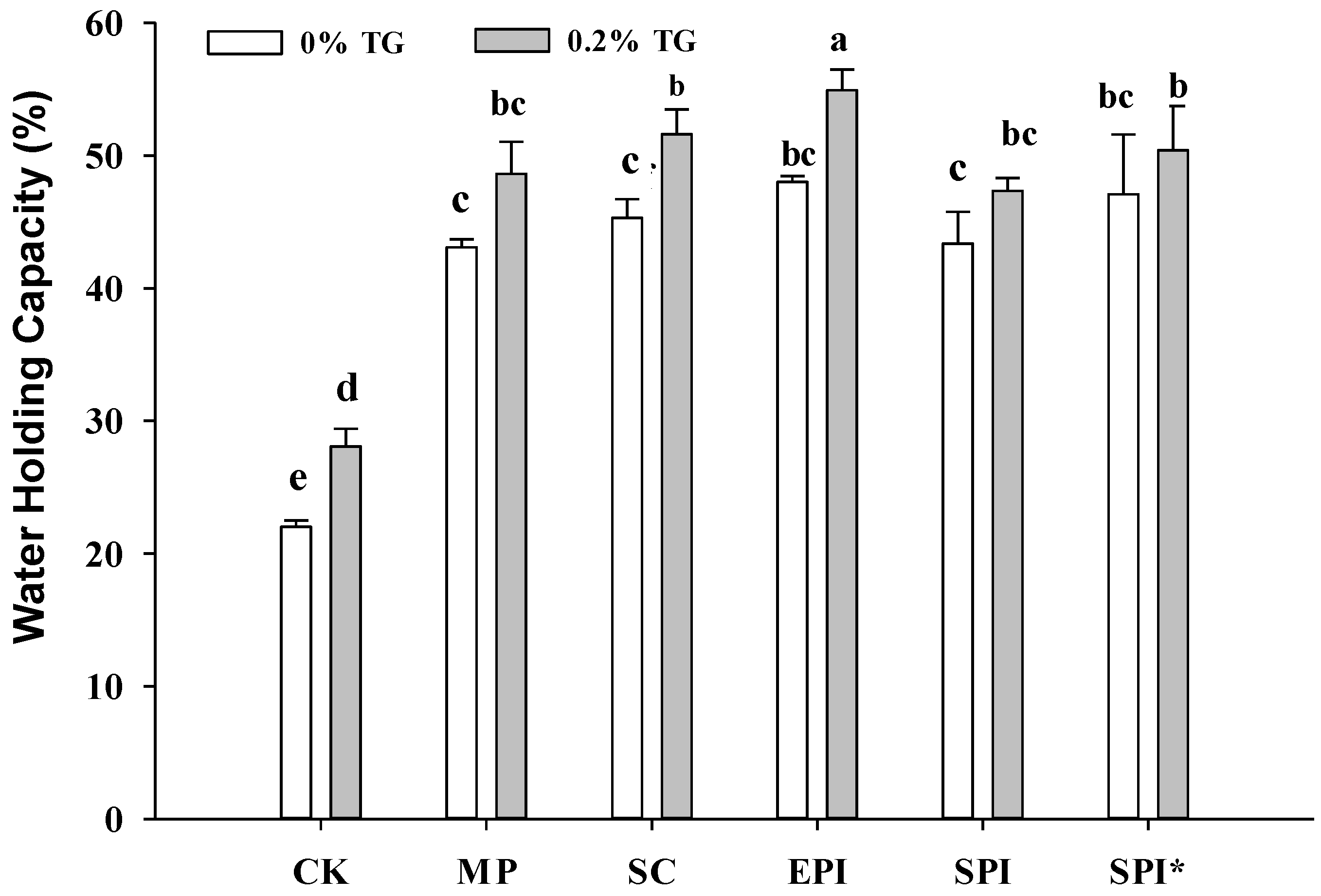
2.1. Effects of TGase on Gel Strength and Water Holding Capacity of the Composite Gels
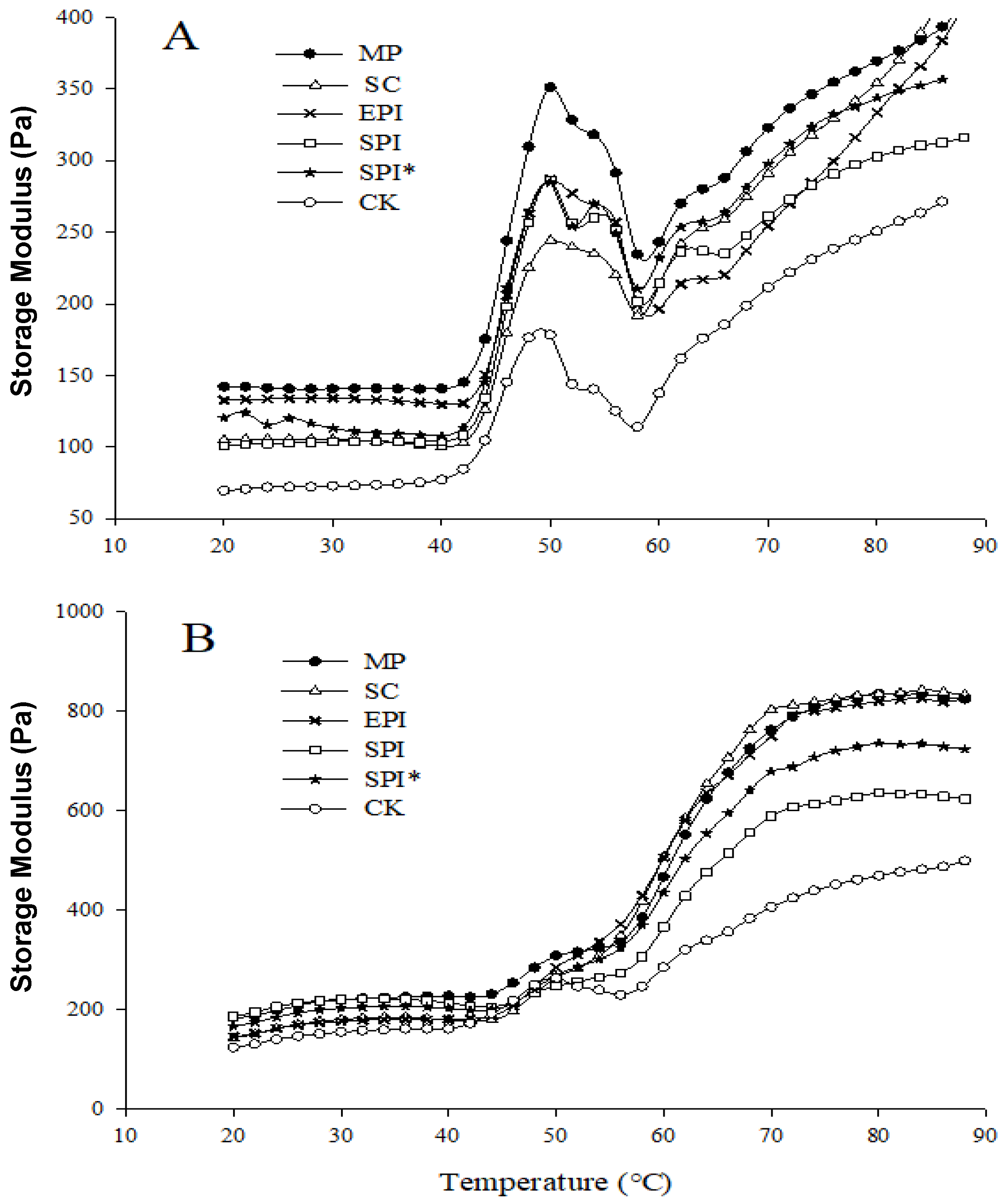
2.2. Rheological Properties of MP Composite Gelling Solutions
2.3. Analysis of Protein Not Involved in Shangqing Liquid
2.4. Effects of TGase on the Microstructure of Composite Gels (ESEM)
2.5. Low-Field NMR Analysis of Water Phase Distribution in Gel
3. Conclusions
4. Materials and Methods
4.1. Materials
4.2. Extraction of Myofibrillar Protein (MP)
4.3. Preparation of Emulsions
4.4. Preparation of MP-Emulsion Composite Sols and Gels
4.5. Determination of Gel Strength and Water Holding Capacity
4.6. Determination of Rheological Properties
4.7. SDS-PAGE Analysis of Protein Involved in the Supernatant of Liquid
4.8. Observation of Microstructure
4.9. Composite Gel Relaxation Time Test
4.10. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maltais, A.; Remondetto, G.E.; Subirade, M. Mechanisms involved in the formation and structure of soya protein cold-set gels: A molecular and supramolecular investigation. Food Hydrocoll. 2008, 22, 550–559. [Google Scholar] [CrossRef]
- Wu, M.; Xiong, Y.L.; Chen, J.; Tang, X.; Zhou, G. Rheological and Microstructural Properties of Porcine Myofibrillar Protein–Lipid Emulsion Composite Gels. J. Food Sci. 2009, 74, E207–E217. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Xiong, Y.L.; Chen, J. Rheology and microstructure of myofibrillar protein–plant lipid composite gels: Effect of emulsion droplet size and membrane type. J. Food Eng. 2011, 106, 318–324. [Google Scholar] [CrossRef]
- Jian, H.; Qiao, F.; Yang, P.; Guo, F.; Huang, X.; Adhikari, B.; Chen, J. Roles of soluble and insoluble aggregates induced by soy protein processing in the gelation of myofibrillar protein. Int. J. Food Sci. Technol. 2015, 51, 480–489. [Google Scholar] [CrossRef]
- Sindayikengera, S.; Xia, W.-S. Nutritional evaluation of caseins and whey proteins and their hydrolysates from Protamex. J. Zhejiang Univ. B 2006, 7, 90–98. [Google Scholar] [CrossRef] [PubMed]
- Mine, Y. Recent advances in egg protein functionality in the food system. World’s Poult. Sci. J. 2002, 58, 31–39. [Google Scholar] [CrossRef]
- Mine, Y. Recent advances in the understanding of egg white protein functionality. Trends Food Sci. Technol. 1995, 6, 225–232. [Google Scholar] [CrossRef]
- Croguennec, T.; Nau, F.; Brule, G. Influence of pH and Salts on Egg White Gelation. J. Food Sci. 2002, 67, 608–614. [Google Scholar] [CrossRef]
- Seki, N.; Uno, H.; Lee, N.-H.; Kimura, I.; Toyoda, K.; Fujita, T.; Arai, K.-I. Transglutaminase activity in Alaska pollack muscle and surimi, and its reaction with myosin B. Nippon. Suisan Gakkaishi 1990, 56, 125–132. [Google Scholar] [CrossRef]
- Li, Q.; Gui, P.; Huang, Z.; Feng, L.; Luo, Y. Effect of transglutaminase on quality and gel properties of pork and fish mince mixtures. J. Texture Stud. 2017, 49, 56–64. [Google Scholar] [CrossRef]
- DeJong, G.; Koppelman, S. Transglutaminase Catalyzed Reactions: Impact on Food Applications. J. Food Sci. 2002, 67, 2798–2806. [Google Scholar] [CrossRef]
- Nonaka, M.; Toiguchi, S.; Sakamoto, H.; Kawajiri, H.; Soeda, T.; Motoki, M. Changes caused by microbial transglutaminase on physical properties of thermally induced soy protein gels. Food Hydrocoll. 1994, 8, 1–8. [Google Scholar] [CrossRef]
- Ramírez-Suárez, J.; Xiong, Y. Effect of transglutaminase-induced cross-linking on gelation of myofibrillar/soy protein mixtures. Meat Sci. 2003, 65, 899–907. [Google Scholar] [CrossRef] [PubMed]
- Ionescu, A.; Aprodu, I.; Darabă, A.; Porneală, L. The effects of transglutaminase on the functional properties of the myofibrillar protein concentrate obtained from beef heart. Meat Sci. 2008, 79, 278–284. [Google Scholar] [CrossRef]
- Jiang, J.; Xiong, Y.L. Role of interfacial protein membrane in oxidative stability of vegetable oil substitution emulsions applicable to nutritionally modified sausage. Meat Sci. 2015, 109, 56–65. [Google Scholar] [CrossRef]
- Xiong, Y.; Blanchard, S.; Means, W. Properties of Broiler Myofibril Gels Containing Emulsified Lipids. Poult. Sci. 1992, 71, 1548–1555. [Google Scholar] [CrossRef]
- Chang-Lee, M.; Lampila, L.; Crawford, D. Yield and Composition of Surimi from Pacific Whiting (Merluccius productus) and the Effect of Various Protein Additives on Gel Strength. J. Food Sci. 1990, 55, 83–86. [Google Scholar] [CrossRef]
- Sakamoto, H.; Kumazawa, Y.; Motoki, M. Strength of Protein Gels Prepared with Microbial Transglutaminase as Related to Reaction Conditions. J. Food Sci. 1994, 59, 866–871. [Google Scholar] [CrossRef]
- Ker, Y.; Toledo, R. Influence of Shear Treatments on Consistency and Gelling Properties of Whey Protein Isolate Suspensions. J. Food Sci. 1992, 57, 82–85. [Google Scholar] [CrossRef]
- Chen, H.; Huang, J.; Zhou, W.; Chen, J. Comparison of gel quality between catfish surimi and common surimi. Food Mach. 2022, 38, 19–23+137. [Google Scholar] [CrossRef]
- Yuan, D.; Yin, Y.; Chang, J.; Kong, B.; Liu, Q. Research progress of hydrocolloids in improving the quality of low-fat emulsion meat products. Food Ind. Sci. Technol. 2022, 43, 426–433. [Google Scholar] [CrossRef]
- Luo, K.; Liu, S.; Miao, S.; Adhikari, B.; Wang, X.; Chen, J. Effects of transglutaminase pre-crosslinking on salt-induced gelation of soy protein isolate emulsion. J. Food Eng. 2019, 263, 280–287. [Google Scholar] [CrossRef]
- Wang, X.; Luo, K.; Liu, S.; Adhikari, B.; Chen, J. Improvement of gelation properties of soy protein isolate emulsion induced by calcium cooperated with magnesium. J. Food Eng. 2018, 244, 32–39. [Google Scholar] [CrossRef]
- Mao, L.; Roos, Y.H.; Miao, S. Study on the Rheological Properties and Volatile Release of Cold-Set Emulsion-Filled Protein Gels. J. Agric. Food Chem. 2014, 62, 11420–11428. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.; Xu, J.; Ji, F.; Tan, M.; Luo, S.; Zhong, X.; Zheng, Z. Properties of transglutaminase-induced myofibrillar/wheat gluten gels. J. Food Sci. 2021, 86, 2387–2397. [Google Scholar] [CrossRef]
- Qin, X.-S.; Luo, S.-Z.; Cai, J.; Zhong, X.-Y.; Jiang, S.-T.; Zhao, Y.-Y.; Zheng, Z. Transglutaminase-induced gelation properties of soy protein isolate and wheat gluten mixtures with high intensity ultrasonic pretreatment. Ultrason. Sonochem. 2016, 31, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.D.; Arntfield, S.D. Gelation properties of myofibrillar/pea protein mixtures induced by transglutaminase crosslinking. Food Hydrocoll. 2012, 27, 394–400. [Google Scholar] [CrossRef]
- Chin, K.B.; Go, M.Y.; Xiong, Y.L. Konjac flour improved textural and water retention properties of transglutaminase-mediated, heat-induced porcine myofibrillar protein gel: Effect of salt level and transglutaminase incubation. Meat Sci. 2009, 81, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Trespalacios, P.; Pla, R. Simultaneous application of transglutaminase and high pressure to improve functional properties of chicken meat gels. Food Chem. 2007, 100, 264–272. [Google Scholar] [CrossRef]
- Belton, P.S.; Jackson, R.R.; Packer, K.J. Pulsed NMR studies of water in striated muscle: I. Transverse nuclear spin relaxation times and freezing effects. Biochim. Biophys. Acta (BBA)-Gen. Subj. 1972, 286, 16–25. [Google Scholar] [CrossRef]
- Han, M.; Wang, P.; Xu, X.; Zhou, G. Low-field NMR study of heat-induced gelation of pork myofibrillar proteins and its relationship with microstructural characteristics. Food Res. Int. 2014, 62, 1175–1182. [Google Scholar] [CrossRef]
- Shaarani, S.M.; Nott, K.P.; Hall, L.D. Combination of NMR and MRI quantitation of moisture and structure changes for convection cooking of fresh chicken meat. Meat Sci. 2006, 72, 398–403. [Google Scholar] [CrossRef] [PubMed]
- Simpson, N.E.; Grant, S.C.; Blackband, S.J.; Constantinidis, I. NMR properties of alginate microbeads. Biomaterials 2003, 24, 4941–4948. [Google Scholar] [CrossRef]
- Hinrichs, R.; Götz, J.; Noll, M.; Wolfschoon, A.; Eibel, H.; Weisser, H. Characterisation of different treated whey protein concentrates by means of low-resolution nuclear magnetic resonance. Int. Dairy J. 2004, 14, 817–827. [Google Scholar] [CrossRef]
- Youssef, M.; Barbut, S. Physicochemical Effects of the Lipid Phase and Protein Level on Meat Emulsion Stability, Texture, and Microstructure. J. Food Sci. 2010, 75, S108–S114. [Google Scholar] [CrossRef]
- Xiong, Y.; Brekke, C. gelation properties of chicken myofibrils treated with calcium and magnesium chlorides. J. Muscle Foods 1991, 2, 21–36. [Google Scholar] [CrossRef]
- Wu, M.; Hu, J.; Gu, X.; Wang, Q.; Wei, R.; Wang, J.; Li, Z.; Liu, R.; Ge, Q.; Yu, H. Myofibrillar protein composite gels: Effect of esterified potato starch, lard and peanut oil on the gel properties. J. Sci. Food Agric. 2021, 102, 2731–2740. [Google Scholar] [CrossRef] [PubMed]


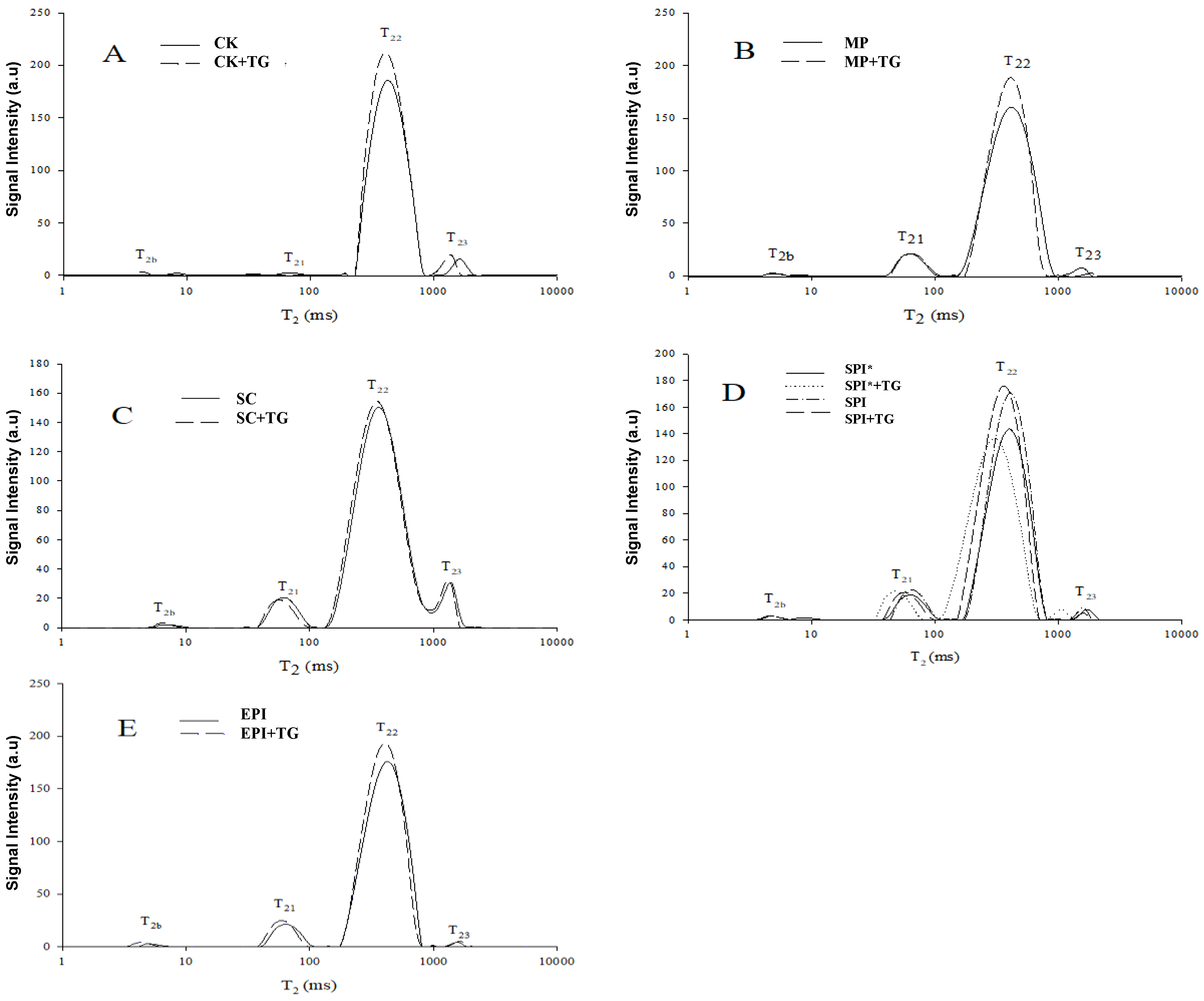
| Combinations | CK | CK + TG | MP | MP + TG | SC | SC + TG |
|---|---|---|---|---|---|---|
| T2b (ms) | 8.23 ± 7.34 a | 4.32 ± 0.65 e | 4.96 ± 1.08 de | 5.75 ± 6.54 d | 7.29 ± 1.51 b | 6.65 ± 1.52 c |
| T21 (ms) | 69.04 ± 8.02 a | 35.15 ± 6.09 f | 63.93 ± 0.00 c | 66.03 ± 0.00 b | 62.27 ± 4.95 c | 58.24 ± 0.00 d |
| T22 (ms) | 431.68 ± 0.00 a | 423.07 ± 0.00 b | 401.51 ± 0.00 c | 388.92 ± 0.00 d | 364.04 ± 0.00 e | 353.29 ± 0.00 f |
| T23 (ms) | 1611.50 ± 122.56 b | 1357.80 ± 0.00 f | 1482.60 ± 122.56 e | 1818.40 ± 262.55 a | 1313.60 ± 106.59 f | 1260.70 ± 0.00 g |
| Combinations | SPI* | SPI* + TG | SPI | SPI + TG | EPI | EPI + TG |
| T2b (ms) | 4.79 ± 1.22 de | 4.89 ± 1.08 de | 8.88 ± 1.84 a | 6.79 ± 4.82 c | 5.37 ± 1.51 d | 4.41 ± 0.99 e |
| T21 (ms) | 66.37 ± 0.00 b | 58.52 ± 0.00 d | 64.19 ± 0.00 bc | 49.47 ± 3.74 e | 64.89 ± 0.00 bc | 59.85 ± 0.00 cd |
| T22 (ms) | 393.01 ± 0.00 cd | 343.44 ± 0.00 a | 385.03 ± 0.00 d | 293.02 ± 0.00 g | 403.21 ± 19.97 c | 388.79 ± 0.00 d |
| T23 (ms) | 1552.10 ± 122.56 d | 1567.50 ± 122.56 d | 1690.10 ± 122.56 b | 1052.50 ± 92.71 h | 1517.70 ± 0.00 d | 1635.50 ± 140.91 c |
| Combinations | CK | CK + TG | MP | MP + TG | SC | SC + TG |
|---|---|---|---|---|---|---|
| A2b/A (%) | 0.38 ± 0.15 e | 0.44 ± 0.09 de | 0.46 ± 0.08 d | 0.69 ± 0.39 b | 0.49 ± 0.05 d | 0.57 ± 0.01 c |
| A21/A (%) | 0.66 ± 0.16 f | 0.31 ± 0.13 f | 6.71 ± 0.14 b | 6.78 ± 0.03 ab | 6.86 ± 0.26 d | 5.87 ± 0.07 e |
| A22/A (%) | 95.90 ± 0.30 a | 96.12 ± 0.27 a | 91.60 ± 0.34 c | 92.18 ± 0.26 c | 93.59 ± 0.40 b | 93.87 ± 0.34 b |
| A23/A (%) | 3.03 ± 0.30 a | 3.12 ± 0.25 a | 1.21 ± 0.13 c | 0.34 ± 0.15 d | - | - |
| Combinations | SPI* | SPI* + TG | SPI | SPI + TG | EPI | EPI + TG |
| A2b/A (%) | 0.66 ± 0.09 b | 0.59 ± 0.09 c | 0.44 ± 0.09 de | 0.09 ± 0.06 f | 0.60 ± 0.04 c | 0.83 ± 0.09 a |
| A21/A (%) | 7.65 ± 0.26 b | 6.80 ± 0.23 d | 7.86 ± 0.26 a | 7.68 ± 0.54 b | 7.24 ± 0.25 c | 7.52 ± 0.22 b |
| A22/A (%) | 90.38 ± 0.24 d | 91.96 ± 0.18 c | 90.21 ± 0.14 d | 90.73 ± 0.94 d | 91.56 ± 0.23 c | 90.88 ± 0.49 d |
| A23/A (%) | 1.31 ± 0.13 b | 0.65 ± 0.12 d | 1.49 ± 0.17 b | 1.50 ± 0.89 b | 0.60 ± 0.09 d | 0.77 ± 0.21 d |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, M.; Yin, Q.; Bian, J.; Xu, Y.; Gu, C.; Jiao, J.; Yang, J.; Zhang, Y. Effects of Transglutaminase on Myofibrillar Protein Composite Gels with Addition of Non-Meat Protein Emulsion. Gels 2023, 9, 910. https://doi.org/10.3390/gels9110910
Wu M, Yin Q, Bian J, Xu Y, Gu C, Jiao J, Yang J, Zhang Y. Effects of Transglutaminase on Myofibrillar Protein Composite Gels with Addition of Non-Meat Protein Emulsion. Gels. 2023; 9(11):910. https://doi.org/10.3390/gels9110910
Chicago/Turabian StyleWu, Mangang, Qing Yin, Junjie Bian, Yuyu Xu, Chen Gu, Junying Jiao, Jingjing Yang, and Yunlin Zhang. 2023. "Effects of Transglutaminase on Myofibrillar Protein Composite Gels with Addition of Non-Meat Protein Emulsion" Gels 9, no. 11: 910. https://doi.org/10.3390/gels9110910
APA StyleWu, M., Yin, Q., Bian, J., Xu, Y., Gu, C., Jiao, J., Yang, J., & Zhang, Y. (2023). Effects of Transglutaminase on Myofibrillar Protein Composite Gels with Addition of Non-Meat Protein Emulsion. Gels, 9(11), 910. https://doi.org/10.3390/gels9110910






