Application of Polymer Hydrogels in the Prevention of Postoperative Adhesion: A Review
Abstract
:1. Introduction
2. Overview of Hydrogel
2.1. Definition of Hydrogel
2.2. Classification of Hydrogels
- (1)
- Natural hydrogels and synthetic hydrogels
- (2)
- Physical and chemical crosslinking hydrogels
- (3)
- Homopolymer hydrogels, copolymerized hydrogels, and polymer hydrogels
- (4)
- Solid hydrogels, semi-solid hydrogels, and liquid hydrogels
| Basis | Classify | Advantages | Disadvantages | References |
|---|---|---|---|---|
| Polymer sources | Natural hydrogel | Biocompatible, low irritation | Low strength, toughness | [41] |
| Synthetic hydrogel | High gel strength, long life performance | Low biocompatibility, irritation to tissues | [43] | |
| Crosslinking degree | Physical crosslinking hydrogels | Easy to produce, high biosafety, reversible | Susceptible to outside influences, lack of stability | [47] |
| Chemical crosslinking hydrogels | Stable, long-lasting mechanical strength | Irreversible, low biocompatibility, difficult to degrade | [57] | |
| Polymer composition | Homopolymer hydrogels | Pure product, low impurities | Limited performance improvements | [75] |
| Copolymerized hydrogels | Excellent properties | High cost of raw materials | [78] | |
| Polymer hydrogels | High flexibility, diverse properties | Difficult to synthesize | [16,80] | |
| Physical state | Solid hydrogel | Robust and flexible, with a strong crosslinked network structure | Weak tissue adhesion | [29,53,84,90] |
| Semi-solid hydrogel | Strong adhesion, good wetting, adsorption properties | Synthetic complexity | [29,86] | |
| Liquid hydrogel | Biocompatible, functional, excellent loading capacity | Hard to surface modify | [89] |
3. Advantages of Hydrogels for the Prevention of Postoperative Adhesion
3.1. Biocompatibility and Biodegradability
3.1.1. Hyaluronic Acid
3.1.2. Gelatin
3.1.3. Carboxymethyl Cellulose
3.1.4. Chitosan
3.1.5. Alginate
3.2. Delivery Performance
3.2.1. Anti-inflammatory Drugs
3.2.2. Tissue Plasminogen Activator
3.2.3. Anticoagulant Drugs
3.2.4. Anti-microbial Drugs
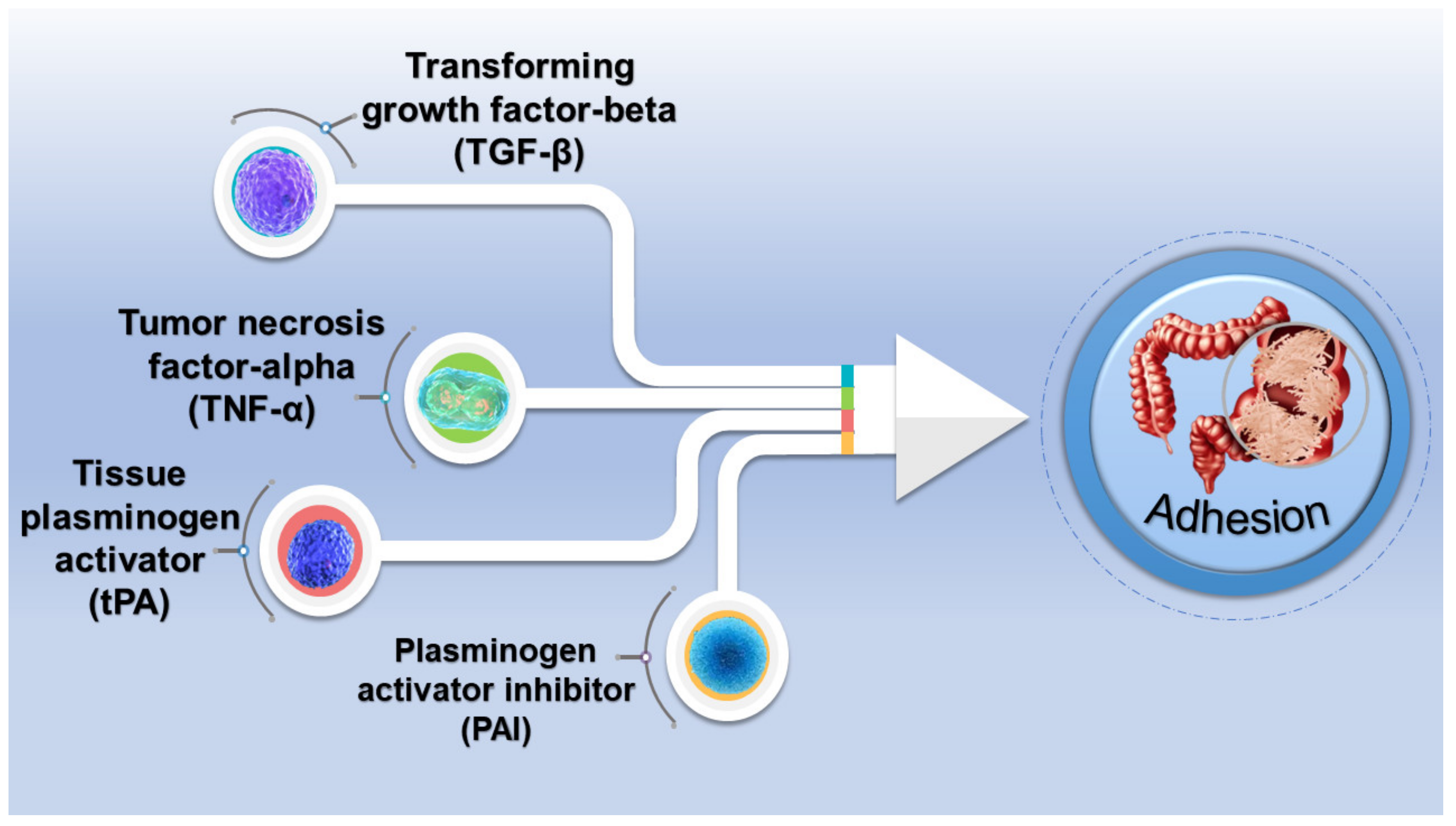
3.3. Adhesion Performance
4. New Progress in the Application of Hydrogel in Anti-Postoperative Adhesions
4.1. Physical Crosslinking Polymerization
4.1.1. Ionic–Electrostatic Crosslinked Hydrogel
4.1.2. Hydrogen Bonds Crosslinked Hydrogel
4.2. Chemical Crosslinking Polymerization
4.2.1. Chemical Crosslinking Hydrogel
4.2.2. Photoinitiated Hydrogel
4.2.3. ‘Click’ Chemically Reactive Hydrogel
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lauder, C.I.W.; Garcea, G.; Strickland, A.; Maddern, G.J. Abdominal adhesion prevention: Still a sticky subject? Dig. Surg. 2010, 27, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Diamond, M.P.; Freeman, M.L. Clinical implications of postsurgical adhesions. Hum. Reprod. Update 2001, 7, 567–576. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hassanabad, A.F.; Zarzycki, A.N.; Jeon, K.; Dundas, J.A.; Vasanthan, V.; Deniset, J.F.; Fedak, P.W.M. Prevention of post-operative adhesions: A comprehensive review of present and emerging strategies. Biomolecules 2021, 11, 1027. [Google Scholar] [CrossRef] [PubMed]
- Coccolini, F.; Ansaloni, L.; Manfredi, R.; Campanati, L.; Poiasina, E.; Bertoli, P.; Capponi, M.G.; Sartelli, M.; Di Saverio, S.; Cucchi, M.; et al. Peritoneal adhesion index (PAI): Proposal of a score for the “ignored iceberg” of medicine and surgery. World J. Emerg. Surg. 2013, 8, 6. [Google Scholar] [CrossRef] [Green Version]
- Binda, M.M.; Molinas, C.R.; Koninckx, P.R. Reactive oxygen species and adhesion formation—Clinical implications in adhesion prevention. Hum. Reprod. 2003, 18, 2503–2507. [Google Scholar] [CrossRef] [Green Version]
- Sun, Z.; Sessler, D.I.; Dalton, J.E.; Devereaux, P.J.; Shahinyan, A.; Naylor, A.J.; Hutcherson, M.T.; Finnegan, P.S.; Tandon, V.; Darvish-Kazem, S.; et al. Postoperative hypoxemia is common and persistent: A prospective blinded observational study. Anesth. Analg. 2015, 121, 709–715. [Google Scholar] [CrossRef] [Green Version]
- ten Broek, R.P.G.; Strik, C.; Issa, Y.; Bleichrodt, R.P.; van Goor, H. Adhesiolysis-related morbidity in abdominal surgery. Ann. Surg. 2013, 258, 98–106. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Zhao, J.; Chen, Z.; Wu, H.; Wang, S. A molybdenum-based nanoplatform with multienzyme mimicking capacities for oxidative stress-induced acute liver injury treatment. Inorg. Chem. Front. 2023. [Google Scholar] [CrossRef]
- Yang, L.B.; Chu, J.S.; Fix, J.A. Colon-specific drug delivery: New approaches and in vitro/in vivo evaluation. Int. J. Pharm. 2002, 235, 1–15. [Google Scholar] [CrossRef]
- Al Brahim, A.; Bai, B.J.; Schuman, T. Comprehensive review of polymer and polymer gel treatments for natural gas-related conformance control. Gels 2022, 8, 353. [Google Scholar] [CrossRef]
- Agnihotri, S.A.; Mallikarjuna, N.N.; Aminabhavi, T.M. Recent advances on chitosan-based micro- and nanoparticles in drug delivery. J. Control. Release 2004, 100, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Ward, B.C.; Panitch, A. Abdominal adhesions: Current and novel therapies. J. Surg. Res. 2011, 165, 91–111. [Google Scholar] [CrossRef] [PubMed]
- Chiorescu, S.; Grad, N.O.; Mironiuc, I.A.; Andercou, O.A. Rosuvastatin administered intraperitoneally reduces the formation of postoperative adhesions in rats. Ann. Ital. Chir. 2018, 89, 95–100. [Google Scholar]
- Caliari, S.R.; Burdick, J.A. A practical guide to hydrogels for cell culture. Nat. Methods 2016, 13, 405–414. [Google Scholar] [CrossRef] [Green Version]
- Xie, M.; Zeng, Y.; Wu, H.; Wang, S.; Zhao, J. Multifunctional carboxymethyl chitosan/oxidized dextran/sodium alginate hydrogels as dressing for hemostasis and closure of infected wounds. Int. J. Biol. Macromol. 2022, 219, 1337–1350. [Google Scholar] [CrossRef]
- Wang, T.; Yi, W.; Zhang, Y.; Wu, H.; Fan, H.; Zhao, J.; Wang, S. Sodium alginate hydrogel containing platelet-rich plasma for wound healing. Colloids Surf. B 2023, 222, 113096. [Google Scholar] [CrossRef]
- Han, W.; Wang, S. Advances in hemostatic hydrogels that can adhere to wet surfaces. Gels 2023, 9, 2. [Google Scholar] [CrossRef]
- Larchenko, E.Y.; Khonina, T.G.; Shadrina, E.V.; Pestov, A.V.; Chupakhin, O.N.; Menshutina, N.V.; Lebedev, A.E.; Lovskaya, D.D.; Larionov, L.P.; Chigvintsev, S.A. Pharmacologically active hydrogels derived from silicon glycerolates and chitosan. Russ. Chem. Bull. 2014, 63, 1225–1231. [Google Scholar] [CrossRef]
- Fan, X.M.; Wang, S.B.; Fang, Y.; Li, P.Y.; Zhou, W.K.; Wang, Z.C.; Chen, M.F.; Liu, H.Q. Tough polyacrylamide-tannic acid-kaolin adhesive hydrogels for quick hemostatic application. Mat. Sci. Eng. C-Mater. 2020, 109, 110649. [Google Scholar] [CrossRef]
- Zhang, H.; Xu, Y.A.; Lei, Y.Y.; Wen, X.Y.; Liang, J.S. Tourmaline nanoparticles modifying hemostatic property of chitosan/ polyvinyl alcohol hydrogels. Mater. Lett. 2022, 324, 132718. [Google Scholar] [CrossRef]
- Fundueanu, G.; Constantin, M.; Bucatariu, S.; Mocanu, G. Cation exchange hydrogels as platform for loading and controlled delivery of drug. J. Nanosci. Nanotechnol. 2017, 17, 4643–4648. [Google Scholar] [CrossRef]
- Hu, J.L.; Liu, S.W.; He, Y.L.; Deng, Y.Y. Drug-loaded physical cross-linking sodium alginate/chitosan hydrogels as wound dressings. Lat. Am. J. Pharm. 2017, 36, 1952–1956. [Google Scholar]
- Zhang, Z.F.; Wang, S.; Waterhouse, G.I.N.; Zhang, Q.H.; Li, L.F. Poly(N-isopropylacrylamide)/mesoporous silica thermosensitive composite hydrogels for drug loading and release. J. Appl. Polym. Sci. 2020, 137, 48391. [Google Scholar] [CrossRef]
- Yang, Y.L.; Liu, X.L.; Li, Y.; Wang, Y.; Bao, C.Y.; Chen, Y.F.; Lin, Q.N.; Zhu, L.Y. A postoperative anti-adhesion barrier based on photoinduced imine-crosslinking hydrogel with tissue-adhesive ability. Acta Biomater. 2017, 62, 199–209. [Google Scholar] [CrossRef]
- Shou, Y.F.; Zhang, J.H.; Yan, S.F.; Xia, P.F.; Xu, P.L.; Li, G.F.; Zhang, K.X.; Yin, J.B. Thermoresponsive chitosan/DOPA-based hydrogel as an injectable therapy approach for tissue-adhesion and hemostasis. ACS Biomater. Sci. Eng. 2020, 6, 3619–3629. [Google Scholar] [CrossRef]
- Park, J.K.; Nah, J.W.; Choi, C. Thermosensitive chitosan-based hydrogel with growth factor as adhesion barrier. Polymer (Korea) 2015, 39, 480–486. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Zhang, Y.; Wu, H.; Tang, J.; Zhou, J.; Zhao, J.; Wang, S. A conductive gelatin methacrylamide hydrogel for synergistic therapy of osteosarcoma and potential bone regeneration. Int. J. Biol. Macromol. 2023, 228, 111–122. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, Y.; Zhao, J.; Wang, S. Multifunctional hydrogels based on chitosan, hyaluronic acid and other biological macromolecules for the treatment of inflammatory bowel disease: A review. Int. J. Biol. Macromol. 2023, 227, 505–523. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Zhang, Y.; Wu, H.; Wang, S.; Li, X. An oxidative polymerized carboxymethyl cellulose hydrogel for the combined anti-tumor recurrence. J. Mater. Sci. 2023, 58, 369–382. [Google Scholar] [CrossRef]
- Li, Z.; Zhao, Y.; Ouyang, X.; Yang, Y.; Chen, Y.; Luo, Q.; Zhang, Y.; Zhu, D.; Yu, X.; Li, L. Biomimetic hybrid hydrogel for hemostasis, adhesion prevention and promoting regeneration after partial liver resection. Bioact. Mater. 2022, 11, 41–51. [Google Scholar] [CrossRef]
- Chou, P.-Y.; Chen, S.-H.; Chen, C.-H.; Chen, S.-H.; Fong, Y.T.; Chen, J.-P. Thermo-responsive in-situ forming hydrogels as barriers to prevent post-operative peritendinous adhesion. Acta Biomater. 2017, 63, 85–95. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhu, C.; Zhang, Z.; Zhao, J.; Yuan, Y.; Wang, S. Oxidation triggered formation of polydopamine-modified carboxymethyl cellulose hydrogel for anti-recurrence of tumor. Colloids Surf. B 2021, 207, 112025. [Google Scholar] [CrossRef]
- Zhao, X.; Yang, J.; Liu, Y.; Gao, J.; Wang, K.; Liu, W. An injectable and antifouling self-fused supramolecular hydrogel for preventing postoperative and recurrent adhesions. Chem. Eng. J. 2021, 404, 85–95. [Google Scholar] [CrossRef]
- Bang, S.; Lee, E.; Ko, Y.-G.; Kim, W.I.; Kwon, O.H. Injectable pullulan hydrogel for the prevention of postoperative tissue adhesion. Int. J. Biol. Macromol. 2016, 87, 155–162. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Jin, X.; Cong, Y.; Liu, Y.Y.; Fu, J. Degradable natural polymer hydrogels for articular cartilage tissue engineering. J. Chem. Technol. Biotechnol. 2013, 88, 327–339. [Google Scholar] [CrossRef]
- Taylor, D.L.; Panhuis, M.I.H. Self-healing hydrogels. Adv. Mater. 2016, 28, 9060–9093. [Google Scholar] [CrossRef]
- Jeong, J.J.; Lee, D.W.; Song, S.Y.; Park, Y.; Kim, J.H.; Kim, J.I.; Kim, H.G.; Nam, K.T.; Lee, W.J.; Nam, K.H.; et al. Development of novel biocompatible thermosensitive anti-adhesive agents using human-derived acellular dermal matrix. PLoS ONE 2019, 14, 0212583. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.X.; Peng, C.R.; Chen, Z.P.; Sun, S.D.; Shi, Z.Q.; Jin, L.Q.; Zhao, W.F.; Zhao, C.S. Engineering antimicrobial and biocompatible electrospun PLGA fibrous membranes by irradiation grafting polyvinylpyrrolidone and periodate. Colloids Surf. B Biointerfaces 2019, 181, 918–926. [Google Scholar] [CrossRef]
- Hoesli, C.A.; Luu, M.; Piret, J.M. A novel alginate hollow fiber bioreactor process for cellular therapy applications. Biotechnol. Prog. 2009, 25, 1740–1751. [Google Scholar] [CrossRef]
- Voorhaar, L.; Hoogenboom, R. Supramolecular polymer networks: Hydrogels and bulk materials. Chem. Soc. Rev. 2016, 45, 4013–4031. [Google Scholar] [CrossRef]
- Sergi, R.; Bellucci, D.; Cannillo, V. A review of bioactive glass/natural polymer composites: State of the art. Materials 2020, 13, 5560. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Cui, C.; Fan, C.; Xu, Z.; Liu, Y.; Liu, W. Tea eggs-inspired high-strength natural polymer hydrogels. Bioact. Mater. 2021, 6, 2820–2828. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.; Corrigan, N.; Wong, E.H.H.; Boyer, C. Bioactive synthetic polymers. Adv. Mater. 2022, 34, 2105063. [Google Scholar] [CrossRef] [PubMed]
- Asliyuce, S.; Idil, N.; Mattiasson, B. Upgrading of bio-separation and bioanalysis using synthetic polymers: Molecularly imprinted polymers (MIPs), cryogels, stimuli-responsive polymers. Eng. Life Sci. 2022, 22, 204–216. [Google Scholar] [CrossRef] [PubMed]
- Mansouri, S.; Lavigne, P.; Corsi, K.; Benderdour, M.; Beaumont, E.; Fernandes, J.C. Chitosan-DNA nanoparticles as non-viral vectors in gene therapy: Strategies to improve transfection efficacy. Eur. J. Pharm. Sci. 2004, 57, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Zhao, J.; Chen, Y.; Zheng, Y.; Li, J.; Zhao, J.; Zhang, J.; Liu, Y.; Liu, X.; Wang, S. MoS2-ALG-Fe/GOx hydrogel with Fenton catalytic activity for combined cancer photothermal, starvation, and chemodynamic therapy. Colloids Surf. B 2020, 195, 111243. [Google Scholar] [CrossRef] [PubMed]
- Li, S.S.; Xia, Y.Z.; Qiu, Y.; Chen, X.N.; Shi, S.X. Preparation and property of starch nanoparticles reinforced aldehyde-hydrazide covalently crosslinked PNIPAM hydrogels. J. Appl. Polym. Sci. 2018, 135, 45761. [Google Scholar] [CrossRef]
- Berger, J.; Reist, M.; Mayer, J.M.; Felt, O.; Gurny, R. Structure and interactions in chitosan hydrogels formed by complexation or aggregation for biomedical applications. Eur. J. Pharm. Sci. 2004, 57, 35–52. [Google Scholar] [CrossRef]
- Zhang, D.K.; Duan, J.J.; Wang, D.G.; Ge, S.R. Effect of preparation methods on mechanical properties of PVA/HA composite hydrogel. J.Bionic.Eng 2010, 7, 235–243. [Google Scholar] [CrossRef]
- Tai, Z.X.; Yan, X.B.; Xue, Q.J. Three-dimensional graphene/polyaniline composite hydrogel as supercapacitor electrode. J. Electrochem. Soc. 2012, 159, A1702–A1709. [Google Scholar] [CrossRef]
- Lai, E.P.; Yue, X.X.; Ning, W.E.; Huang, J.W.; Ling, X.L.; Lin, H.T. Three-dimensional graphene-based composite hydrogel materials for flexible supercapacitor electrodes. Front. Chem. 2019, 7, 660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malhotra, A.; Bera, T.; Zhai, L. Bioinspired metal ion coordinated polyelectrolyte fibrous nanoreactors. Adv. Mater. Interfaces 2016, 3, 1600692. [Google Scholar] [CrossRef]
- Ul Abdeen, Z.; Saeed, R. Ionic interactions in cross-linked poly(vinyl alcohol) hydrogel blended with starch. Rev. Roum. Chim. 2019, 64, 233–240. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhao, C.Z.; Xiang, N.P.; Li, W. Chain entanglements and hydrogen bonds in carbopol microgel reinforced hydrogel. Macromol. Chem. Phys. 2016, 217, 2139–2144. [Google Scholar] [CrossRef]
- Zhang, G.Z.; Wang, C.Y.; Ngai, T. Injectable hydrogel cross-linked by quadruple hydrogen bonding for drug encapsulation and delivery. J. Control. Release 2017, 259, E36–E37. [Google Scholar] [CrossRef]
- Wang, M.Y.; Wang, L.; Xu, Y.D.; Fu, L.H.; Yang, H. One-pot fabrication of triple shape memory hydrogel based on coordination bond, the dynamic borate ester bonds, and hydrogen bond. Soft Mater. 2019, 17, 342–349. [Google Scholar] [CrossRef]
- Echalier, C.; Valot, L.; Martinez, J.; Mehdi, A.; Subra, G. Chemical cross-linking methods for cell encapsulation in hydrogels. Mater. Today Commun. 2019, 20, 100536. [Google Scholar] [CrossRef] [Green Version]
- Akhtar, M.F.; Hanif, M.; Ranjha, N.M. Methods of synthesis of hydrogels—A review. Saudi Pharm. J. 2016, 24, 554–559. [Google Scholar] [CrossRef] [Green Version]
- Rebers, L.; Reichsollner, R.; Regett, S.; Tovar, G.E.M.; Borchers, K.; Baudis, S.; Southan, A. Differentiation of physical and chemical cross-linking in gelatin methacryloyl hydrogels. Sci. Rep. 2021, 11, 3256. [Google Scholar] [CrossRef]
- Marshall, L.J.; Matsarskaia, O.; Schweins, R.; Adams, D.J. Enhancement of the mechanical properties of lysine-containing peptide-based supramolecular hydrogels by chemical cross-linking. Soft Mater. 2021, 17, 8459–8464. [Google Scholar] [CrossRef]
- Wang, X.G.; Zhao, F.; Qin, X.P.; Feng, S.Y. Triple networks hydrogel with high strength made by chemical and physical cross-linking. Colloids Surf. A Physicochem. Eng. Asp. 2020, 589, 124428. [Google Scholar] [CrossRef]
- Sun, W.R.; Hu, Y.C.; Cheng, Y.Z.; Yang, S.; Kang, Z.Q. Effect of cross-linking methods on stress relaxation of PVA/PAM-co-PAA-based hydrogels. Int. J. Polym. Anal. Charact. 2021, 26, 330–341. [Google Scholar] [CrossRef]
- Jung, D.; Lee, K.M.; Chang, J.Y.; Yun, M.; Choi, H.J.; Kim, Y.A.; Yoon, H.; Kim, H. Selective de-cross-linking of transformable, double-network hydrogels: Preparation, structural conversion, and controlled release. ACS Appl. Mater. Interfaces 2018, 10, 42985–42991. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, Y.M.; Yang, Z.Y.; Chen, X.L.; Zhao, Y.S. Preparation of MPASP-PAA/Fe3+ composite conductive hydrogel with physical and chemical double crosslinking structure and its application in flexible strain sensors. Macromol. Chem. Phys. 2022, 223, 2100467. [Google Scholar] [CrossRef]
- Xu, J.Y.; Liu, X.; Ren, X.Y.; Gao, G.H. The role of chemical and physical crosslinking in different deformation stages of hybrid hydrogels. Eur. Polym. J. 2018, 100, 86–95. [Google Scholar] [CrossRef]
- Zhao, Z.; Li, Z.; Xia, Q.; Xi, H.; Lin, Y. Fast synthesis of temperature-sensitive PNIPAAm hydrogels by microwave irradiation. Eur. Polym. J. 2008, 44, 1217–1224. [Google Scholar] [CrossRef]
- Sun, T.Y.; Zhan, B.; Qin, D.; Wang, B.X.; Gao, Y.Y. Evaluation on the anti-oxidation effects of a thermosensitive chitosan-based hydrogel loaded antioxidin-RL. J. Control. Release 2017, 259, E129–E130. [Google Scholar] [CrossRef]
- Komeri, R.; Thankam, F.G.; Muthu, J. Free radical scavenging injectable hydrogels for regenerative therapy. Mater. Sci. Eng. C 2017, 71, 100–110. [Google Scholar] [CrossRef]
- Ding, H.Y.; Liang, X.X.; Zheng, S.Y.; Wang, Q.; Li, Z.J.; Sun, G.X. Actuators assembled from hydrogel blocks of various shapes via condensation reactions. Mater. Chem. Phys. 2020, 253, 123332. [Google Scholar] [CrossRef]
- Franconetti, A.; Dominguez-Rodriguez, P.; Lara-Garcia, D.; Prado-Gotor, R.; Cabrera-Escribano, F. Native and modified chitosan-based hydrogels as green heterogeneous organocatalysts for imine-mediated Knoevenagel condensation. Appl. Catal. A 2016, 517, 176–186. [Google Scholar] [CrossRef]
- Eguchi, Y.; Kato, T.; Tanaka, T.; Maruyama, T. A DNA-gold nanoparticle hybrid hydrogel network prepared by enzymatic reaction. Chem. Commun. 2017, 53, 5802–5805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, H.; Gu, H.C.; Chen, Z.Y.; Shang, L.R.; Zhao, Z.; Gu, Z.Z.; Zhao, Y.J. Enzymatic inverse opal hydrogel particles for biocatalyst. ACS Appl. Mater. Interfaces 2017, 9, 12914–12918. [Google Scholar] [CrossRef] [PubMed]
- Simon, D.; Obst, F.; Haefner, S.; Heroldt, T.; Peiter, M.; Simon, F.; Richter, A.; Voit, B.; Appelhans, D. Hydrogel/enzyme dots as adaptable tool for non-compartmentalized multi-enzymatic reactions in microfluidic devices. React. Chem. Eng. 2019, 4, 67–77. [Google Scholar] [CrossRef]
- Tian, T.; Wei, X.F.; Jia, S.S.; Zhang, R.H.; Li, J.X.; Zhu, Z.; Zhang, H.M.; Ma, Y.L.; Lin, Z.Y.; Yang, C.J. Integration of target responsive hydrogel with cascaded enzymatic reactions and microfluidic paper-based analytic devices (mu PADs) for point-of-care testing (POCT). Biosens. Bioelectron. 2016, 77, 537–542. [Google Scholar] [CrossRef]
- Nakan, U.; Tolkyn, B.; Adikanova, D.B.; Negim, E.S.; Yeligbayeva, G.Z.; Seilkhanov, T.M.; Kenzhebayeva, B.A.; Abdiyev, K.Z. Characterization and swelling properties of copolymer poly(N, N-dimethyl acrylamide-co-acrylic acid) and homopolymer poly(acrylic acid). Egypt. J. Chem. 2022, 65, 767–773. [Google Scholar] [CrossRef]
- Lin, X.X.; Zhang, H.T.; Qin, Y.Q.; Hu, X.H. Design and fabrication of photo-responsive hydrogel for the application of functional contact lens. Front. Mater 2021, 8, 680359. [Google Scholar] [CrossRef]
- Wakayama, H.; Henares, T.G.; Jigawa, K.; Funano, S.; Sueyoshi, K.; Endo, T.; Hisamoto, H. Design of a single-step immunoassay principle based on the combination of an enzyme-labeled antibody release coating and a hydrogel copolymerized with a fluorescent enzyme substrate in a microfluidic capillary device. Lab Chip 2013, 13, 4304–4307. [Google Scholar] [CrossRef]
- Gan, D.L.; Xing, W.S.; Jiang, L.L.; Fang, J.; Zhao, C.C.; Ren, F.Z.; Fang, L.M.; Wang, K.F.; Lu, X. Plant-inspired adhesive and tough hydrogel based on Ag-Lignin nanoparticles-triggered dynamic redox catechol chemistry. Nat. Commun. 2019, 10, 49. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chowdhury, P.; Ali, M.A. Preparation of poly(vinyl alcohol) hydrogel film with acrylic acid for selective absorption of heavy metal ions. J. Polym. Mater. 2005, 22, 277–281. [Google Scholar]
- Muratore, L.M.; Davis, T.P. Self-reinforcing hydrogels comprised of hydrophobic methyl methacrylate macromers copolymerized with N,N-dimethylacrylamide. J. Polym. Sci. Pol. Chem. 2000, 38, 810–817. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, H.; Huang, J.; Xia, N.; Li, T.; Xia, Q. Self-double-emulsifying drug delivery system incorporated in natural hydrogels: A new way for topical application of vitamin C. J. Microencapsul. 2018, 35, 90–101. [Google Scholar] [CrossRef] [PubMed]
- Qian, K.; Wang, J.; He, F.; Wu, Y.; Zhou, Z. An experimental investigation on transpiration cooling performances using solid hydrogel as coolant. Appl. Therm. Eng. 2019, 158, 113753. [Google Scholar] [CrossRef]
- Guetta, O.; Rittel, D. Hyperelastic modeling of solid methyl cellulose hydrogel under quasi-static compression. J. Mech. Behav. Biomed. Mater. 2021, 124, 104857. [Google Scholar] [CrossRef]
- Buntum, T.; Kongprayoon, A.; Mungyoi, W.; Charoenram, P.; Kiti, K.; Thanomsilp, C.; Supaphol, P.; Suwantong, O. Wound-aided semi-solid poly(vinyl alcohol) hydrogels incorporating essential oil-loaded chitosan nanoparticles. Int. J. Biol. Macromol. 2021, 189, 135–141. [Google Scholar] [CrossRef]
- Lee, M.S.; Kim, J.C. Photodependent release from poly(vinyl alcohol)/epoxypropoxy coumarin hydrogels. J. Appl. Polym. Sci. 2012, 124, 4339–4345. [Google Scholar] [CrossRef]
- Ngampunwetchakul, L.; Toonkaew, S.; Supaphol, P.; Suwantong, O. Semi-solid poly(vinyl alcohol) hydrogels containing ginger essential oil encapsulated in chitosan nanoparticles for use in wound management. J. Polym. Res. 2019, 26, 224. [Google Scholar] [CrossRef]
- Aouada, F.A.; de Moura, M.R.; Rubira, A.F.; Muniz, E.C.; Fernandes, P.R.G.; Mukai, H.; da Silveira, A.C.F.; Itri, R. Birefringent hydrogels based on PAAm and lyotropic liquid crystal: Optical, morphological and hydrophilic characterization. Eur. Polym. J. 2006, 42, 2781–2790. [Google Scholar] [CrossRef]
- Kularatne, R.S.; Kim, H.; Ammanamanchi, M.; Hayenga, H.N.; Ware, T.H. Shape-morphing chromonic liquid crystal hydrogels. Chem. Mater. 2016, 28, 8489–8492. [Google Scholar] [CrossRef]
- Dong, L.; Agarwal, A.K.; Beebe, D.J.; Jiang, H.R. Variable-focus liquid microlenses and microlens arrays actuated by thermoresponsive hydrogels. Adv. Mater. 2007, 19, 401–405. [Google Scholar] [CrossRef]
- Yu, Y.; Zheng, X.; Liu, X.; Zhao, J.; Wang, S. Injectable carboxymethyl chitosan-based hydrogel for simultaneous anti-tumor recurrence and anti-bacterial applications. Int. J. Biol. Macromol. 2023, 230, 123196. [Google Scholar] [CrossRef]
- Xie, P.; Zhang, L.; Shen, H.; Wu, H.; Zhao, J.; Wang, S.; Hu, L. Biodegradable MoSe2-polyvinylpyrrolidone nanoparticles with multi-enzyme activity for ameliorating acute pancreatitis. J. Nanobiotechnol. 2022, 20, 113. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.; Wang, S.; Zhang, X.; Ye, C.; Wang, S.; An, X. Development of PVA-based microsphere as a potential embolization agent. Mater. Sci. Eng. C 2022, 135, 112677. [Google Scholar] [CrossRef] [PubMed]
- Constantin, C.P.; Aflori, M.; Damian, R.F.; Rusu, R.D. Biocompatibility of polyimides: A mini-review. Materials 2019, 12, 3166. [Google Scholar] [CrossRef] [Green Version]
- Xiong, Y.; Zhang, X.; Ma, X.; Wang, W.; Yan, F.; Zhao, X.; Chu, X.; Xu, W.; Sun, C. A review of the properties and applications of bioadhesive hydrogels. Polym. Chem. 2021, 12, 3721–3739. [Google Scholar] [CrossRef]
- Kim, J.; Park, Y.; Tae, G.; Lee, K.B.; Hwang, C.M.; Hwang, S.J.; Kim, I.S.; Noh, I.; Sun, K. Characterization of low-molecular-weight hyaluronic acid-based hydrogel and differential stem cell responses in the hydrogel microenvironments. J. Biomed. Mater. Res. Part A 2009, 88A, 967–975. [Google Scholar] [CrossRef]
- Chocholata, P.; Kulda, V.; Dvorakova, J.; Supova, M.; Zaloudkova, M.; Babuska, V. In situ hydroxyapatite synthesis enhances biocompatibility of PVA/HA hydrogels. Int. J. Mol. Sci. 2021, 22, 9335. [Google Scholar] [CrossRef]
- Jeong, J.Y.; Chung, P.K.; Yoo, J.C. Effect of sodium hyaluronate/carboxymethyl cellulose (Guardix-sol) on retear rate and postoperative stiffness in arthroscopic rotator cuff repair patients: A prospective cohort study. J. Orthop. Surg. 2017, 25, 1–8. [Google Scholar] [CrossRef]
- Wang, Q.Q.; Liu, Y.; Zhang, C.J.; Zhang, C.; Zhu, P. Alginate/gelatin blended hydrogel fibers cross-linked by Ca2+ and oxidized starch: Preparation and properties. Mat. Sci. Eng. C-Mater. 2019, 99, 1469–1476. [Google Scholar] [CrossRef]
- Yin, O.S.; Ahmad, I.; Amin, M. Effect of cellulose nanocrystals content and ph on swelling behaviour of gelatin based hydrogel. Sains Malays. 2015, 44, 793–799. [Google Scholar]
- Piao, Y.Z.; Chen, B.Q. Synthesis and mechanical properties of double cross-linked gelatin-graphene oxide hydrogels. Int. J. Biol. Macromol. 2017, 101, 791–798. [Google Scholar] [CrossRef]
- Milakin, K.A.; Capakova, Z.; Acharya, U.; Vajdak, J.; Moravkova, Z.; Hodan, J.; Humpolicek, P.; Bober, P. Biocompatible and antibacterial gelatin-based polypyrrole cryogels. Polymer 2020, 197, 122491. [Google Scholar] [CrossRef]
- Bang, S.; Ko, Y.-G.; Kim, W.I.; Cho, D.; Park, W.H.; Kwon, O.H. Preventing postoperative tissue adhesion using injectable carboxymethyl cellulose-pullulan hydrogels. Int. J. Biol. Macromol. 2017, 105, 886–893. [Google Scholar] [CrossRef] [PubMed]
- Dahlan, N.A.; Pushpamalar, J.; Veeramachineni, A.K.; Muniyandy, S. Smart hydrogel of carboxymethyl cellulose grafted carboxymethyl polyvinyl alcohol and properties studied for future material applications. J. Surg. Res. 2018, 26, 2061–2071. [Google Scholar] [CrossRef]
- Kou, S.; Peters, L.M.; Mucalo, M.R. Chitosan: A review of sources and preparation methods. Int. J. Biol. Macromol. 2021, 169, 85–94. [Google Scholar] [CrossRef]
- Kas, H.S. Chitosan: Properties, preparations and application to microparticulate systems. J. Microencapsul. 1997, 14, 689–711. [Google Scholar] [CrossRef]
- Zhang, S.; Li, J.; Li, J.; Du, N.; Li, D.; Li, F.; Man, J. Application status and technical analysis of chitosan-based medical dressings: A review. RSC Adv. 2020, 10, 34308–34322. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Tavakoli, E.; Motamedi, A.; Salimi, H. Facile synthesis of extremely biocompatible double-network hydrogels based on chitosan and poly(vinyl alcohol) with enhanced mechanical properties. J. Appl. Polym. Sci. 2018, 135, 45752. [Google Scholar] [CrossRef]
- Lee, K.Y.; Mooney, D.J. Alginate: Properties and biomedical applications. Prog. Polym. Sci. 2012, 37, 106–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, S.; Park, H.; Chen, K.; Park, H.; Lee, D. Development of an implantable PCL/alginate bilayer scaffold to prevent secondary infections. Korean J. Chem. Eng. 2020, 37, 677–687. [Google Scholar] [CrossRef]
- Kim, D.; Choi, G.J.; Baek, S.; Abdullah, A.; Jang, S.; Hong, S.A.; Kim, B.G.; Lee, J.; Kang, H.; Lee, D. Characterization of anti-adhesion properties of alginate/polyethylene oxide film to reduce postsurgical peritoneal adhesions. Sci. Adv. Mater. 2017, 9, 1669–1677. [Google Scholar] [CrossRef]
- Song, X.; Guo, J.; Liu, Y.; Li, F.; Yang, Q.; Guan, F.; Di, C. Preparation and characterization of multi-network hydrogels based on sodium alginate/krill protein/polyacrylamide-Strength, shape memory, conductivity and biocompatibility. Int. J. Biol. Macromol. 2022, 207, 140–151. [Google Scholar] [CrossRef] [PubMed]
- Shi, B.; Ding, J.X.; Wei, J.C.; Fu, C.F.; Zhuang, X.L.; Chen, X.S. Drug-incorporated electrospun fibers efficiently prevent postoperative adhesion. Curr. Pharm. Des. 2015, 21, 1960–1966. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, S.; Mehrabani, D.; Tavakolian, A.R.; Tanideh, N. Role of an anti-inflammatory drug in prevention of intra-abdominal adhesions: An experimental study on guinea pigs. J. Appl. Anim. Res. 2004, 26, 123–125. [Google Scholar] [CrossRef]
- Buckenmaier, C.C.; Pusateri, A.E.; Harris, R.A.; Hetz, S.P. Comparison of antiadhesive treatments using an objective rat model. Am. Surg. 1999, 65, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Hellebrekers, B.W.J.; Trimbos-Kemper, T.C.M.; Trimbos, J.; Emeis, J.J.; Kooistra, T. Use of fibrinolytic agents in the prevention of postoperative adhesion formation. Fertil. Steril. 2000, 74, 203–212. [Google Scholar] [CrossRef]
- Ahmad, G.; Mackie, F.L.; Iles, D.A.; O’Flynn, H.; Dias, S.; Metwally, M.; Watson, A. Fluid and pharmacological agents for adhesion prevention after gynaecological surgery. Cochrane Database Syst. Rev. 2014, 7, 1465–1858. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dufay, M.; Jimenez, M.; Casetta, M.; Chai, F.; Blanchemain, N.; Stoclet, G.; Cazaux, F.; Bellayer, S.; Degoutin, S. PCL covered PP meshes plasma-grafted by sulfonated monomer for the prevention of postoperative abdominal adhesions. Mater. Today Commun. 2021, 26, 101968. [Google Scholar] [CrossRef]
- Sanchez-Gomez, S.; Martinez-de-Tejada, G. Antimicrobial peptides as anti-biofilm agents in medical implants. Curr. Top. Med. Chem. 2017, 17, 590–603. [Google Scholar] [CrossRef]
- Wang, T.; Zhang, X.; Wang, Z.C.; Zhu, X.Z.; Liu, J.; Min, X.; Cao, T.; Fan, X.D. Smart composite hydrogels with pH-responsiveness and electrical conductivity for flexible sensors and logic gates. Polymers 2019, 11, 1564. [Google Scholar] [CrossRef] [Green Version]
- Avais, M.; Chattopadhyay, S. Waterborne pH responsive hydrogels: Synthesis, characterization and selective pH responsive behavior around physiological pH. Polymer 2019, 180, 121701. [Google Scholar] [CrossRef]
- Haidari, H.; Kopecki, Z.; Sutton, A.T.; Garg, S.; Cowin, A.J.; Vasilev, K. pH-responsive “smart” hydrogel for controlled delivery of silver nanoparticles to infected wounds. Antibiotics 2021, 10, 49. [Google Scholar] [CrossRef] [PubMed]
- Sehgal, S.N. Sirolimus: Its discovery, biological properties, and mechanism of action. Transplant. Proc. 2003, 35, 7S–14S. [Google Scholar] [CrossRef] [PubMed]
- Maciver, A.H.; McCall, M.D.; Edgar, R.L.; Thiesen, A.L.; Bigam, D.L.; Churchill, T.A.; Shapiro, A.M.J. Sirolimus drug-eluting, hydrogel-impregnated polypropylene mesh reduces intra-abdominal adhesion formation in a mouse model. Surgery 2011, 150, 907–915. [Google Scholar] [CrossRef]
- Song, X.; Wang, Y.; Gao, L. Mechanism of antioxidant properties of quercetin and quercetin-DNA complex. J. Mol. Model. 2020, 26, 133. [Google Scholar] [CrossRef]
- Zeng, H.; Liu, X.; Zhang, Z.; Song, X.; Quan, J.; Zheng, J.; Shen, Z.; Ni, Y.; Liu, C.; Zhang, Y.; et al. Self-healing, injectable hydrogel based on dual dynamic covalent cross-linking against postoperative abdominal cavity adhesion. Acta Biomater. 2022, 16, 49. [Google Scholar]
- He, T.; Zou, C.; Song, L.J.; Wang, N.; Yang, S.L.X.; Zeng, Y.; Wu, Q.J.; Zhang, W.L.; Chen, Y.T.; Gong, C.Y. Improving antiadhesion effect of thermosensitive hydrogel with sustained release of tissue-type plasminogen activator in a rat repeated-injury model. ACS Appl. Mater. Interfaces 2016, 8, 33514–33520. [Google Scholar] [CrossRef]
- Arikan, S.; Adas, G.; Barut, G.; Toklu, A.S.; Kocakusak, A.; Uzun, H.; Kemik, O.; Daduk, Y.; Aydin, S.; Purisa, S. An evaluation of low molecular weight heparin and hyperbaric oxygen treatment in the prevention of intra-abdominal adhesions and wound healing. Am. J. Surg. 2005, 189, 155–160. [Google Scholar] [CrossRef]
- Almamar, A.; Schlachta, C.M.; Alkhamesi, N.A. The systemic effect and the absorption rate of aerosolized intra-peritoneal heparin with or without hyaluronic acid in the prevention of postoperative abdominal adhesions. Surg. Endosc. 2019, 33, 2517–2520. [Google Scholar] [CrossRef] [PubMed]
- Hemker, H.C. A century of heparin: Past, present and future. J. Thromb. Haemost. 2016, 14, 2329–2338. [Google Scholar] [CrossRef]
- Gockel, L.M.; Ponert, J.M.; Schwarz, S.; Schlesinger, M.; Bendas, G. The low molecular weight heparin tinzaparin attenuates platelet activation in terms of metastatic niche formation by coagulation-dependent and independent pathways. Molecules 2018, 23, 2753. [Google Scholar] [CrossRef] [Green Version]
- Reyhan, E.; Irkoroco, O.; Surmelioglu, A.; Ozkara, S.; Deger, K.C.; Aziret, M.; Erdem, H.; Cetinkunar, S.; Tilki, M.; Demirturk, P.; et al. Abolition of anti-adhesiogenic effect of heparin by protamine sulfate. Int. J. Surg. 2014, 12, 729–733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fayez, J.A.; Schneider, P.J. Prevention of pelvic adhesion formation by different modalities of treatment. Am. J. Obstet. Gynecol. 1987, 157, 1184–1188. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.-S.; Xu, X.-X.; Xiang, W.-W.; Zhang, H.-H.; Lin, H.-L.; Shen, L.-E.; Lin, Q.; Lin, F.; Zhou, Z.-Y. Using 17β-estradiol heparin-poloxamer thermosensitive hydrogel to enhance the endometrial regeneration and functional recovery of intrauterine adhesions in a rat model. FASEB J. 2020, 34, 446–457. [Google Scholar] [CrossRef] [Green Version]
- Zindel, J.; Mittner, J.; Bayer, J.; April-Monn, S.L.; Kohler, A.; Nusse, Y.; Dosch, M.; Buchi, I.; Sanchez-Taltavull, D.; Dawson, H.; et al. Intraperitoneal microbial contamination drives post-surgical peritoneal adhesions by mesothelial EGFR-signaling. Nat. Commun. 2021, 12, 7316. [Google Scholar] [CrossRef] [PubMed]
- Singh, H.; Miyamoto, S.; Darshi, M.; Torralba, M.G.; Kwon, K.; Sharma, K.; Pieper, R. Gut microbial changes in diabetic db/db mice and recovery of microbial diversity upon pirfenidone treatment. Microorganisms 2020, 8, 1347. [Google Scholar] [CrossRef] [PubMed]
- Ruwanpura, S.M.; Thomas, B.J.; Bardin, P.G. Pirfenidone: Molecular mechanisms and potential clinical applications in lung disease. Am. J. Respir. Cell Mol. Biol. 2020, 62, 413–422. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.; Cheng, J.; Chen, C.; Lu, Y.; Chen, X.; Zhang, F. Pirfenidone-loaded hyaluronic acid methacryloyl hydrogel for preventing epidural adhesions after laminectomy. Drug Delivery Transl. Res. 2022, 1, 1–12. [Google Scholar] [CrossRef]
- Chausheva, S.; Redwan, B.; Sharma, S.; Marella, N.; Schossleitner, K.; Mueller, A.C.; Petzelbauer, P.; Morris, T.; Lang, I.M. Synthetic Fibrin-Derived B beta(15–42) Peptide Delays Thrombus Resolution in a Mouse Model. Arterioscler. Thromb. Vasc. Biol. 2021, 41, 2168–2180. [Google Scholar] [CrossRef]
- Yakovlev, S.; Medved, L. Effect of fibrinogen, fibrin, and fibrin degradation products on transendothelial migration of leukocytes. Thromb. Res. 2018, 162, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Sato, N.; Aoyama, Y.; Yamanaka, J.; Toyotama, A.; Okuzono, T. Particle adsorption on hydrogel surfaces in aqueous media due to van der waals attraction. Sci. Rep. 2017, 7, 6099. [Google Scholar] [CrossRef] [Green Version]
- Aoyama, Y.; Sato, N.; Toyotama, A.; Okuzono, T.; Yamanaka, J. Particle adsorption on polymer gel surface driven by van der waals attraction. Bull. Chem. Soc. Jpn. 2022, 95, 314–324. [Google Scholar] [CrossRef]
- Fujita, M.; Policastro, G.M.; Burdick, A.; Lam, H.T.; Ungerleider, J.L.; Braden, R.L.; Huang, D.; Osborn, K.G.; Omens, J.H.; Madani, M.M.; et al. Preventing post-surgical cardiac adhesions with a catechol-functionalized oxime hydrogel. Nat. Commun. 2021, 12, 3764. [Google Scholar] [CrossRef] [PubMed]
- Elliott, W.H.; Bonani, W.; Maniglio, D.; Motta, A.; Tan, W.; Migliaresi, C. Silk hydrogels of tunable structure and viscoelastic properties using different chronological orders of genipin and physical cross-linking. ACS Appl. Mater. Interfaces 2015, 7, 12099–12108. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.J.; Yoo, J.J.; Atala, A. Recent applications of polymeric biomaterials and stem cells in tissue engineering and regenerative medicine. Polymer 2014, 38, 113–128. [Google Scholar]
- Li, Z.Y.; Zhou, Y.Z.; Li, T.Y.; Zhang, J.J.; Tian, H. Stimuli-responsive hydrogels: Fabrication and biomedical applications. View 2022, 3, 20200112. [Google Scholar] [CrossRef]
- Ma, Z.W.; Mao, Z.W.; Gao, C.Y. Surface modification and property analysis of biomedical polymers used for tissue engineering. Colloids Surf. B Biointerfaces 2007, 60, 137–157. [Google Scholar] [CrossRef]
- Cao, J.; Cai, Y.; Yu, L.; Zhou, J. Dual physically crosslinked hydrogels based on the synergistic effects of electrostatic and dipole-dipole interactions. J. Mater. Chem. B 2019, 7, 676–683. [Google Scholar] [CrossRef]
- Shi, C.; Yang, F.; Hu, L.; Wang, H.; Wang, Y.; Wang, Z.; Pan, S.; Chen, J. Construction of polysaccharide based physically crosslinked double-network antibacterial hydrogel. Mater. Lett. 2022, 316, 1. [Google Scholar] [CrossRef]
- Jing, H.; He, L.; Feng, J.; Fu, H.; Guan, S.; Guo, P. High strength hydrogels with multiple shape-memory ability based on hydrophobic and electrostatic interactions. Soft Mater. 2019, 15, 5264–5270. [Google Scholar] [CrossRef]
- Wu, Y.; Mu, Y.; Luo, Y.; Menon, C.; Zhou, Z.; Chu, P.K.; Feng, S.-P. Hofmeister effect and electrostatic interaction enhanced ionic conductive organohydrogels for electronic applications. Adv. Funct. Mater. 2022, 32, 2110859. [Google Scholar] [CrossRef]
- Zhou, Y.F.; Lei, L.; Zhang, Z.L.; Zhang, R.S.; Song, Q.Q.; Li, X.Y. Cation instructed steroidal prodrug supramolecular hydrogel. J. Colloid Interface Sci. 2018, 528, 10–17. [Google Scholar] [CrossRef] [PubMed]
- Monica Bravo-Anaya, L.; Gricelda Fernandez-Solis, K.; Rosselgong, J.; Elena Nano-Rodriguez, J.L.; Carvajal, F.; Rinaudo, M. Chitosan-DNA polyelectrolyte complex: Influence of chitosan characteristics and mechanism of complex formation. Int. J. Biol. Macromol. 2019, 126, 1037–1049. [Google Scholar] [CrossRef]
- Kulikouskaya, V.I.; Lazouskaya, M.E.; Agabekov, V.E. Features of the Formation of Interpolyelectrolyte Complexes Based on Chitosan and Pectin. Theor. Exp. Chem. 2019, 54, 375–385. [Google Scholar] [CrossRef]
- Voron’ko, N.G.; Derkach, S.R.; Kuchina, Y.A.; Sokolan, N.I. The chitosan-gelatin (bio)polyelectrolyte complexes formation in an acidic medium. Carbohydr. Polym. 2016, 138, 265–272. [Google Scholar] [CrossRef] [PubMed]
- Wu, D.; Zhu, L.; Li, Y.; Zhang, X.; Xu, S.; Yang, G.; Delair, T. Chitosan-based colloidal polyelectrolyte complexes for drug delivery: A review. Carbohydr. Polym. 2020, 238, 116126. [Google Scholar] [CrossRef] [PubMed]
- Potas, J.; Szymanska, E.; Winnicka, K. Challenges in developing of chitosan—Based polyelectrolyte complexes as a platform for mucosal and skin drug delivery. Eur. Polym. 2020, 140, 110020. [Google Scholar] [CrossRef]
- Ishihara, M.; Kishimoto, S.; Nakamura, S.; Sato, Y.; Hattori, H. Polyelectrolyte complexes of natural polymers and their biomedical applications. Polymers 2019, 11, 672. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruiz-Esparza, G.U.; Wang, X.C.; Zhang, X.C.; Jimenez-Vazquez, S.; Diaz-Gomez, L.; Lavoie, A.M.; Afewerki, S.; Fuentes-Baldemar, A.A.; Parra-Saldivar, R.; Jiang, N.; et al. Nanoengineered shear-thinning hydrogel barrier for preventing postoperative abdominal adhesions. Nano-Micro Lett. 2021, 13, 212. [Google Scholar] [CrossRef]
- Cui, C.Y.; Wu, T.L.; Chen, X.Y.; Liu, Y.; Li, Y.; Xu, Z.Y.; Fan, C.C.; Liu, W.G. A janus hydrogel wet adhesive for internal tissue repair and anti-postoperative adhesion. Adv. Funct. Mater. 2020, 30, 2005689. [Google Scholar] [CrossRef]
- Yan, T.; Zou, Y.; Zhang, X.; Li, D.; Guo, X.; Yang, D. Hydrogen bond interpenetrated agarose/pva network: A highly ionic conductive and flame-retardant gel polymer electrolyte. ACS Appl. Mater. Interfaces 2021, 13, 9856–9864. [Google Scholar] [CrossRef]
- Wang, R.; Chen, X.; Yang, Y.; Xu, Y.; Zhang, Q.; Zhang, Y.; Cheng, Y. Imidazolidinyl urea reinforced polyacrylamide hydrogels through the formation of multiple hydrogen bonds. React. Funct. Polym. 2022, 172, 105183. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, L.; Wang, L.; Yu, J. Hydrogen-bonded supramolecular hydrogels derived from 3,3’,4,4’-benzophenonetetracarboxylic acid and hydroxylpyridines. Chin. J. Chem. 2009, 27, 2279–2283. [Google Scholar] [CrossRef]
- Hongyao, D.; Xin Ning, Z.; Si Yu, Z.; Yihu, S.; Zi Liang, W.; Qiang, Z. Hydrogen bond reinforced poly(1-vinylimidazole-co-acrylic acid) hydrogels with high toughness, fast self-recovery, and dual pH-responsiveness. Polymer 2017, 131, 95–103. [Google Scholar]
- Choi, J.; Kim, S.; Yoo, J.; Choi, S.H.; Char, K. Self-healable antifreeze hydrogel based on dense quadruple hydrogen bonding. Macromolecules 2021, 54, 6389–6399. [Google Scholar] [CrossRef]
- Song, X.W.; Zhang, Z.Q.; Shen, Z.L.; Zheng, J.; Liu, X.; Ni, Y.Q.; Quan, J.; Li, X.R.; Hu, G.; Zhang, Y. Facile preparation of drug-releasing supramolecular hydrogel for preventing postoperative peritoneal adhesion. ACS Appl. Mater. Interfaces 2021, 13, 56881–56891. [Google Scholar] [CrossRef]
- Yang, Y.; Zhao, X.; Yu, J.; Chen, X.; Chen, X.; Cui, C.; Zhang, J.; Zhang, Q.; Zhang, Y.; Wang, S.; et al. H-bonding supramolecular hydrogels with promising mechanical strength and shape memory properties for postoperative antiadhesion application. ACS Appl. Mater. Interfaces 2020, 12, 34161–34169. [Google Scholar] [CrossRef]
- Kim, Y.; Song, J.; Park, S.C.; Ahn, M.; Park, M.J.; Song, S.H.; Yoo, S.-Y.; Hong, S.G.; Hong, B.H. Photoinitiated polymerization of hydrogels by graphene quantum dots. Nanomaterials 2021, 11, 2169. [Google Scholar] [CrossRef]
- Wei, G.; Wei, J.; Zhou, J.; Chen, Y.; Wu, D.; Wang, Q. MoS2 nanosheet initiated smart polymeric hydrogel for NIR-driven Ag(I) enrichment. Chem. Eng. J. 2020, 382, 123018. [Google Scholar] [CrossRef]
- Huang, Y.-C.; Liu, Z.-H.; Kuo, C.-Y.; Chen, J.-P. Photo-Crosslinked Hyaluronic Acid/Carboxymethyl Cellulose Composite Hydrogel as a Dural Substitute to Prevent Post-Surgical Adhesion. Int. J. Mol. Sci. 2022, 23, 6177. [Google Scholar] [CrossRef]
- Mi, Y.; Yang, F.F.; Bloomquist, C.; Xia, Y.L.; Sun, B.; Qi, Y.F.; Wagner, K.; Montgomery, S.A.; Zhang, T.; Wang, A.Z. Biologically Targeted Photo-Crosslinkable Nanopatch to Prevent Postsurgical Peritoneal Adhesion. Adv. Sci. 2019, 6, 1900809. [Google Scholar] [CrossRef] [Green Version]
- Jagtap, A.; More, A. A review on self-initiated and photoinitiator-free system for photopolymerization. Polym. Bull. 2022, 79, 8057–8091. [Google Scholar] [CrossRef]
- Wang, Z.J.; Li, C.Y.; Zhao, X.Y.; Wu, Z.L.; Zheng, Q. Thermo- and photo-responsive composite hydrogels with programmed deformations. J. Mater. Chem. B 2019, 7, 1674–1678. [Google Scholar] [CrossRef] [PubMed]
- Wu, W.; Ni, Q.; Xiang, Y.; Dai, Y.; Jiang, S.; Wan, L.P.; Liu, X.N.; Cui, W.G. Fabrication of a photo-crosslinked gelatin hydrogel for preventing abdominal adhesion. RSC Adv. 2016, 6, 92449–92453. [Google Scholar] [CrossRef]
- Mao, S.-Y.; Peng, H.-W.; Wei, S.-Y.; Chen, C.-S.; Chen, Y.-C. Dynamically and spatially controllable albumin-based hydrogels for the prevention of postoperative adhesion. ACS Biomater. Sci. Eng. 2021, 7, 3293–3305. [Google Scholar] [CrossRef]
- Zhang, T.; Zheng, Z.H.; Cheng, X.; Ding, X.B.; Peng, Y.X. “Click” chemistry in materials science. Prog.Chem 2008, 20, 1090–1101. [Google Scholar]
- Ding, X.Y.; Li, G.; Xiao, C.S.; Chen, X.S. Enhancing the stability of hydrogels by doubling the schiff base linkages. Macromol. Chem. Phys. 2019, 220, 1800484. [Google Scholar] [CrossRef]
- Gondia, N.K.; Priya, J.; Sharma, S.K. Synthesis and physico-chemical characterization of a Schiff base and its zinc complex. Res. Chem. Intermed. 2017, 43, 1165–1178. [Google Scholar] [CrossRef]
- Segura, J.L.; Mancheno, M.J.; Zamora, F. Covalent organic frameworks based on Schiff-base chemistry: Synthesis, properties and potential applications. Chem. Soc. Rev. 2016, 45, 5635–5671. [Google Scholar] [CrossRef]
- Abdalla, T.H.; Nasr, A.S.; Bassioni, G.; Harding, D.R.; Kandile, N.G. Fabrication of sustainable hydrogels-based chitosan Schiff base and their potential applications. Arab. J. Chem. 2022, 15, 103511. [Google Scholar] [CrossRef]







Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cai, J.; Guo, J.; Wang, S. Application of Polymer Hydrogels in the Prevention of Postoperative Adhesion: A Review. Gels 2023, 9, 98. https://doi.org/10.3390/gels9020098
Cai J, Guo J, Wang S. Application of Polymer Hydrogels in the Prevention of Postoperative Adhesion: A Review. Gels. 2023; 9(2):98. https://doi.org/10.3390/gels9020098
Chicago/Turabian StyleCai, Jie, Jiaming Guo, and Shige Wang. 2023. "Application of Polymer Hydrogels in the Prevention of Postoperative Adhesion: A Review" Gels 9, no. 2: 98. https://doi.org/10.3390/gels9020098









