3.1. Electrolysis of SWCNTs in CuSO4 Solution
Several copper composite formulations bearing a range of calculated SWCNT percentages were prepared using the electroplating method by varying the SWCNT concentration in the electroplating bath, while maintaining a [CuSO
4] of 0.1 M. Based upon our previous work it is likely that a fraction of the copper ions from the bath were complexed to the SWCNT [
9,
19], forming nuclei for growth of electroplated copper grains among the bundles [
23]. Prior studies have reported on the benefits of seeding copper moieties among SWCNT aggregates via chemical deposition methods, especially via organic media that exfoliate nanomaterial aggregates to promote improved adhesion of electroplated copper coatings on CNT surfaces [
3]. Vortexing in the bath, caused by rapid stirring, and the applied potential difference between the electrodes likely caused migration of copper ions along with SWCNT aggregates (and their metallic catalyst impurities) toward the cathode. This was also observed in our prior study on electromigration [
19].
The mixed deposit of copper and SWCNT grew as a layer on the surface of the copper cathode until it visually reached a thickness of 2–3 mm, at which point it sloughed off and fell to the bottom of the bath. This process was repeated for the durations stated in
Table 1 for each sample. The contents of the plating bath, and any remaining deposit adhering loosely to the copper cathode were collected, washed in DI water and acetone, and air-dried (
Figure S4). This deposit could be easily removed from the electrode surface, washed with DI water and acetone, and dried for further characterization. Literature reports that higher voltages can speed up electrochemical reactions [
24]. Hence, the choice of 14.7 V, which was the highest setting in the DC voltage apparatus used for the study. The enhanced rate of reaction coupled with agitation used to disperse the hydrophobic nanotubes in the plating bath also likely resulted in non-adherent coatings on the cathode, but this was not of major concern as the intention was to first collect any cathodic deposits for subsequent analysis and processing. The cathodic sludge can be collected and processed further into compacts through compression and/or extrusion. Ongoing experiments in our group are attempting to optimize the electroplating process wherein copper is deposited as a smooth layer onto the CNT fiber/cable surfaces directly, rather than onto individual CNT bundles dispersed in the plating bath. Several approaches to preparing copper–nanotube composites are possible and the approach outlined in this paper is one of them, with the uniqueness centered on the avoidance of surfactants despite the fact that the nanotubes are hydrophobic.
Figure 1 shows representative SEM images (at two magnifications) of Cu–SCWNT composites containing varying proportions of SWCNTs produced by direct current electroplating using the chosen constant voltage (14 V) power supply. Based on calculations, composites containing 0, 2.3, 3.8, and 6.2% SWCNTs were obtained. In the absence of SWCNTs (
Figure 1a) the Cu deposited as a dendritic (“fern-like”) structure comprising of individual crystallites of 0.25–0.5 μm in size (
Figure 1b). A similar morphology has been observed previously for Cu electrolysis under conditions of high concentration and high overpotential [
25].
With the addition of 2.3 wt% SWCNTs to the electrolysis solution, the dendritic nature appeared to visually decline while the structure became more porous (
Figure 1c). At this concentration the SWCNT bundles appear to be coating the Cu crystallites in a highly heterogeneous manner (
Figure 1d). The approach postulated previously, where CNT aggregates are first exfoliated in an organic medium followed by reacting with copper species, can potentially be used to overcome the problem of heterogeneous nature of Cu–CNT composites [
3]. Further increasing the SWCNT concentration results in the complete loss of the dendritic structure (
Figure 1e,g), and the resulting material is heterogeneous but uniform mix of Cu particles and SWCNTs (
Figure 1f). Finally, at the highest concentrations studied herein, the individual Cu crystallites have SWCNTs encapsulating them (
Figure 1h). A similar morphology was observed in the electrodeposition of Cu and surfactant dispersed MWCNTs [
26,
27]. Irrespectively, it is clear that the SWCNTs have altered the nucleation of copper particles during the electroplating process. Unfortunately, as in previous work, it is clear that without subsequent densification the resulting composites will remain heterogeneous [
27].
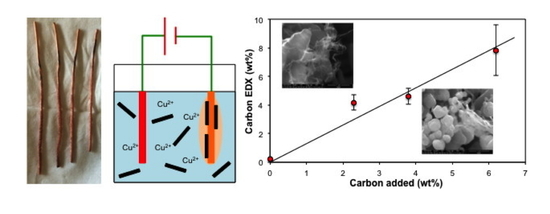
The EDX elemental analysis of the Cu–SWCNT composites is shown in
Table 2.
Table 2 is intended to show a trend in terms of carbon content for the sake of discussion only, and not to present a statistically validated observation. As would be expected the copper composition is inversely proportional to carbon content (
Figure S5), and the EDX analysis of carbon (wt%) is close to quantitative with respect to the wt% of SWCNTs added (
Figure 2) suggesting that all the SWCNTs are incorporated into the composite. This is possible considering that all dispersed solids (including any CNTs unbound to the copper during the plating process) in the plating bath were collected (after physical mixing) onto a filter membrane and dried. The presence of oxygen can be due to native oxide layers on the copper formed by self-passivation, along with oxygen-containing defect groups on nanotube surfaces [
28]. It is also likely that copper oxides may have formed in the plating bath prior to deposition in the cathode, and prior researchers have reported on the electrodeposition of copper oxides [
29,
30]. It is interesting to note, however, that the oxygen content decreases dramatically with addition of SWCNT (
Table 2), suggesting that the presence of the hydrophobic SWCNTs decreases the oxidation of Cu (see below). However, increased SWCNT content beyond 2.3 wt% did not result in further decrease in oxygen content. The observed trends could likely be due to passivation [
31,
32], increased corrosion potential [
33], or reduction [
20] of the Cu; however, we note that under non-electolysis conditions CuO is the preferred composition when copper is deposited on CNTs [
34].
The presence of Fe is associated with the catalyst residue present in the SWCNTs [
22,
35], and hence the iron content is generally proportional to carbon (
Figure S6). Surprisingly, even the SWCNT-free control copper deposit was found to contain iron. EDX characterization of copper wire electrode material confirmed the presence of up to 4 wt% of iron. It is likely that some of this iron could have leached into the deposit during the electroplating process.
The surface composition of the composites may be verified by XPS analysis (
Table 3); Cu 2p spectra corresponding to electrodeposited copper and Cu–SWCNT composite deposited with 4 wt% SWCNTs are displayed in
Figure 3a,b, respectively. Whilst it is difficult to separate the contributions from Cu
2O and metallic Cu due to the close proximity of their components, each of which appear at a binding energy of ca. 932.7 eV, the presence of a component at ca. 933.2 eV in
Figure 3a is consistent with the CuO chemical environment [
36,
37,
38]. Conversely, no CuO component was observed in the case of the Cu–SWCNT composite, indicating that co-deposition with SWCNTs yielded a higher degree of copper reduction than electrodeposition of copper alone. Our hypothesizes is that the possible inhibition of oxidation of copper in the presence of the SWCNTs due to their acting as potential reducing agents or redox catalysts [
39,
40]. The presence of hydroxides in the samples was likely due to the partial reaction of copper oxide with water from either the bath, or from atmospheric humidity. In both cases, a large component measured at a binding energy of ca. 934.7 eV may be attributed to the presence of Cu(OH)
2 in the samples [
36,
38]. It should be noted that due to small chemical shifts between the Cu/Cu
2O and CuO environments, it is difficult to accurately determine the position and relative area of the CuO component in
Figure 3a; for this reason, the estimated atomic percentage of CuO given in
Table 3 should be treated as approximate.
X-ray diffraction (XRD) of all the electroplated samples (
Table 4) reveals crystalline Cu as the major component in all cases. As is suggested by EDX and XPS there is a minor component of oxide in each sample; however, the percentage of oxide was not proportional to percentage of SWCNT added (consistent with EDX measurements, see
Table 2), it is possibly due changes in the morphology (
Figure 1) and hence surface area of the samples. Alternatively, Liu et al. have shown that during electrodeposition, the thicker the copper plated layer, the thicker was the oxide film [
41]. An alternative explanation would be that the presence of SWCNTs alters the plating current density, that is known to affect the concentrations of impurities [
42]. We note that unlike XPS (
Figure 3) no Cu(OH)
2 is observed in the XRD (
Figure 4) [
43]; however, given the surface nature of XPS, this would suggest that the Cu(OH)
2 is formed by surface hydrolysis rather than as a bulk material.
The most interesting observation is that, as suggested by XPS, the identity of the oxide is dependent on the presence of the SWCNTs. The SWCNT-free control displayed the presence of CuO (ICDD #04-007-1375), but the oxide in all the samples containing SWCNTs is Cu
2O (ICDD #04-002-3214), see
Figure 4. This change suggests that the presence of added SWCNTs modifies the nature of the native oxide layers formed. Additives such as water and acids promote the rate of formation of Cu
2O by the oxidation of Cu metal, as well as the further oxidation to CuO. So presumably, under the present conditions the SWCNTs inhibit the subsequent conversion of Cu
2O to CuO; however, we have previously observed the opposite under higher energy conditions [
34]. An alternative explanation could involve the thickness of the Cu coating.
It was observed previously [
42] that when Cu coating is less than 0.165 μm the oxide is CuO (with a Cu
2O interface to Cu); however, when the coating is thicker than 0.165 μm the oxide is Cu
2O. Based upon this observation, it would suggest that the presence of the SWCNTs acts as a seed for Cu growth. Seeding usually occurs at native or induced defect sites on the nanotube surfaces [
44,
45]. The crystal grain size for the Cu and the oxides showed no correlation between crystallite size and SWCNT content (
Table 4).
The Raman spectra of the Cu–SWCNTs composites show the presence D and G bands and the radial breathing modes associated with SWCNTs (e.g.,
Figure 5a). The D band represents the presence of sp
3 carbon centers (as opposed to sp
2 carbon of a pristine SWCNT), thus, the I
G:I
D ratio is often used as a good indicator of quality in bulk samples [
46]. As seen in
Table 5, the I
G:I
D ratios appeared consistent in the samples containing added SWCNTs, but were lower than for the SWCNTs before electrolysis (I
G:I
D = 15.52 ± 1.52,
Figure 5b). This observation suggests that the structure of the SWCNTs underwent a consistent change during the electroplating process, possibly due to the presence of acetic acid in the plating bath. Prior research has suggested that nanotubes with low diameters and initial I
G:I
D > 1 are significantly susceptible to structural modification during acid treatment [
47].
3.2. Electrolysis in the Presence of Cu Micropowder
The Cu–SWCNT composites generated consisted of porous deposit that sloughed off the cathode into the bath, and had to be filtered, washed, and dried, yielding a powdery residue. In an attempt to induce harder, adherent coatings on the cathode surface, microscale copper particles were suspended in the bath in addition to SWCNTs. Addition of such metal particles has been found to promote the formation of hard coatings through a combination of mechanisms: impact adhesion on electrode surface, followed by nucleation of electroplated coatings on the surfaces of the impacted particles, cementing them onto the electrode surface [
48,
49]. Hence the addition of Cu powder (<425 μm) was investigated in an attempt to create denser, more adherent coatings.
Figure 6 illustrates an SEM image of Cu micropowder (Sigma Aldrich, St Louis, MO, USA) and a physical mixture with SWCNTs. The Cu micropowder comprises aggregates (50–300 μm) of individual crystallites (263 (14) Å), while in the physically mixed sample, the SWCNTs are highly bundled and existed as physically separate domains surrounded by copper particles (
Figure 6a,b).
Electrolysis of SWCNTs/CuSO
4 was performed in the presence of microscale copper powder dispersed in the stirring plating bath. A hard adherent coating thickness of 0.20 ± 0.05 mm was achieved over 2.75” coated lengths of the 16-gauge copper wire cathode (
Figure 7). This is likely due to grain stabilization and dispersion hardening of copper grains by dispersed copper microparticles [
48,
49]. The adherent coatings held on to the electrode surface despite manual abrasion between fingers, or mild abrasion using tissue paper. Multiple cathodes were coated simultaneously (
Figure S7), and on average, 0.11 ± 0.03 g of coating was deposited per wire cathode, translating to a total hard deposit of about 0.444 g over 27.5 cm of cathode surface (for the 4 copper wire cathodes combined). In addition, a porous deposit was also obtained, which appeared to contain the majority of the Cu micro powder.
SEM images of the hard coatings (
Figure 8a) revealed large features formed from fused particles (10–50 μm), on which were smaller (1–2 μm) particles (
Figure 8b). It is unclear from the SEM images if the SWCNTs are incorporated; however, the fine deposit obtained during the plating process was comprised more of fibrous bundles of SWCNTs (
Figure 8c). The associated EDX data (
Table 6) confirms the presence of C (and the Fe associated with the catalyst residue in the SWCNTs) in both samples. The carbon content (12.3 ± 7.4 at%) for the hard coating is higher than in samples prepared by electrolysis in the absence of added Cu micropowder. However, the porous deposit has significantly greater oxygen content than previously observed (c.f.,
Table 2;
Table 6).