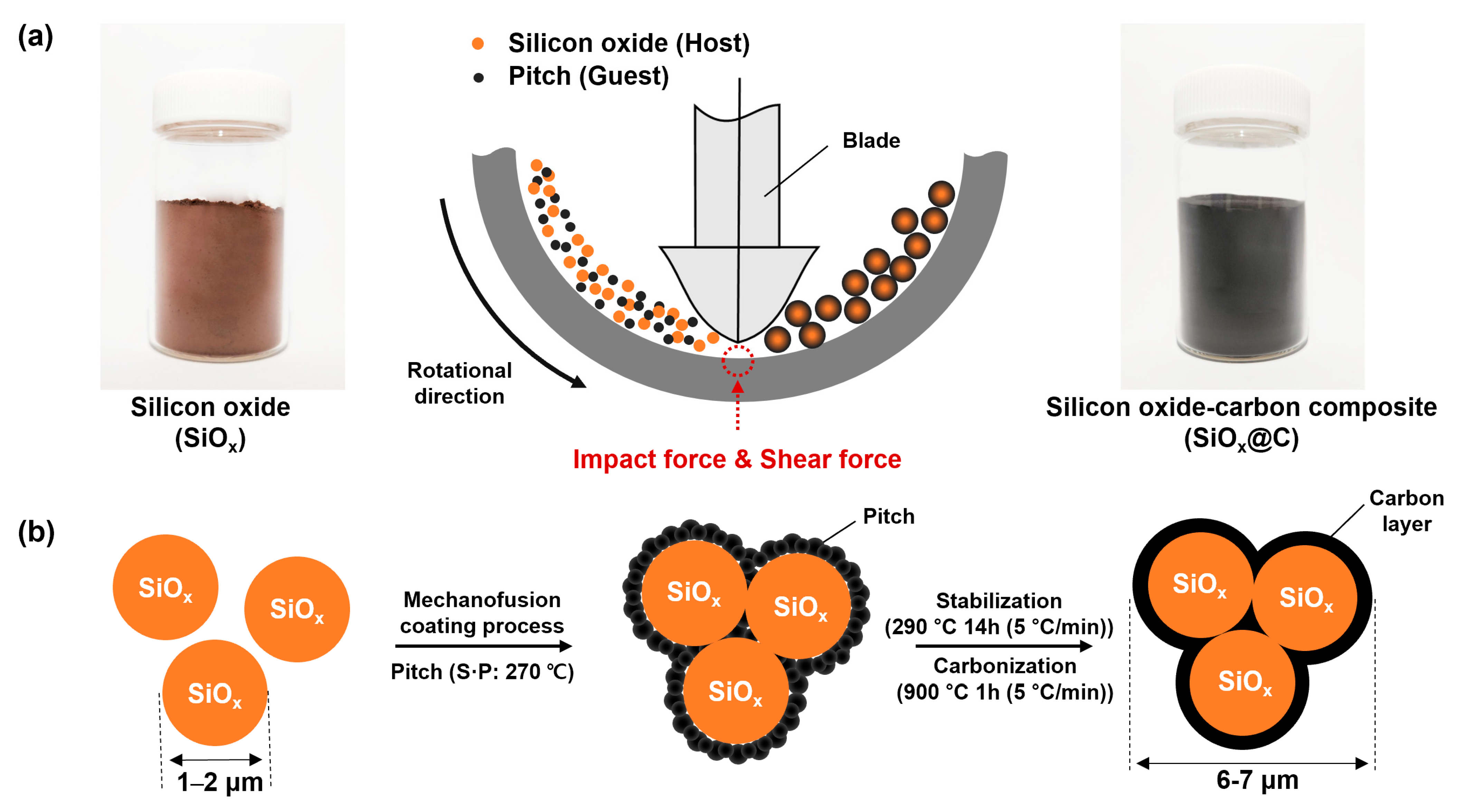
3.1. Mechanofusion-Derived SiOx@C Composites
The overall procedure for fabricating the SiO
x@C composite using mechanofusion process is represented in
Figure 1a. Mechanofusion is a method that efficiently combines different materials without the need for additional binders by applying three types of physical forces (compression, shear, and rotational forces) to internal particles [
28]. The mechanofusion process is relatively simple, inexpensive, and requires no solvents, thereby making it potentially attractive for environmentally responsible commercial manufacture. Under our experiment condition, SiO
x served as the host material, while a pitch was used as the guest material to facilitate composite formation. Irregularly shaped SiO
x particles, averaging 1.8 μm in size, were used in the experiment. Furthermore, as a result of analyzing the oxidation level of pristine SiO
x through X-ray photoelectron spectroscopy (XPS) measurement, it was found that the
x value of the SiO
x used in this work was 1.04 (see the
Supplementary Materials Figure S1). The carbon precursor—the pitch—was prepared through the thermal polymerization of residual petroleum oil, which was previously reported in our work [
29]. Due to the compression and shear forces generated between the blade and the rotating plate, independent SiO
x particles aggregate and combine with surrounding particles and pitch to form SiO
x-pitch composites (SiO
x@P). (
Figure 1b). During this process, the high heat generated from the high-speed rotating plate, exceeding the softening point (S.P) of the pitch, increases the cohesion between the SiO
x particles and pitch by neither coating the SiO
x surface nor promoting particle–particle adhesion. Following complexation, the SiO
x@P undergoes a transformation into a carbon layer through a stabilization and carbonization process. Generally, the thermoplastic feature of the pitch, the carbonization process, is performed after forming a crosslinking bond while undergoing a stabilization (insolubilization, oxidation) process at a temperature near the softening point to maintain the shape of the complex during the carbonization process [
30]. The pitch used in this experiment had an S.P. of 270 °C, and was stabilized at 290 °C for a sufficient period of time in an oxygen (O
2) atmosphere to form a more stable carbon structure. The carbonization yield of the coating pitch, as confirmed through the TGA curve, was 72% (see the
Supplementary Materials Figure S2). Consequently, it is expected that pores will form inside the carbon layer after carbonization, and these pores are anticipated to facilitate the creation of a connection path for the electrolyte’s active material and provide a buffering effect against volume expansion of SiO
x. A successful carbonization process was also visually confirmed by a color change in the sample. The initial dark brown SiO
x was transformed into black due to the presence of the carbon layer on the surface after composite formation and carbonization (
Figure 1a inset).
3.2. Characterization of SiOx@C Composites
Changes in the particle size and shape resulting from the mechanofusion process were confirmed through SEM analysis.
Figure 2a,b shows the particle shape before and after complexation, with the particle size analysis results shown in the inset. The pristine SiO
x particles showed a smooth surface with sharp edges, while the SiO
x@C composites exhibited a rougher surface and blunt edges. This is because the surface becomes rougher as the coating pitch present on the composite surface goes through the stabilization/carbonization process. The particle analysis revealed that the D
50 value of the pristine SiO
x was approximately 1.8 µm, whereas that of the SiO
x@C composite was around 7.6 µm. These alterations in the surface morphology and particle size can be attributed to the composite formation process. During the mechanofusion process, a single SiO
x particle aggregates with 2–3 surrounding particles and the pitch, leading to an increase in the average particle size. This result is consistent with the complexation process mechanism described above.
Generally, it is known that the size of the anode active material should be 15 μm or less to provide a sufficient reaction surface area and maintain ionic/electronic conductivity during charging/discharging [
31]. In the case of the SiO
x@C composite prepared in this experiment, the average particle size is under 8 μm; thus, it is considered that it may be applicable to the manufacturing process of an anode material without an additional particle sizing step.
EDS mapping analysis was also performed to confirm the formation of the carbon layer according to the stabilization and carbonization process.
Figure 2c illustrates the SEM images of the SiO
x@C composites along with the EDS mapping results. The elemental distribution analysis revealed the presence of silicon (76.9%), carbon (15.3%), and oxygen (7.6%) elements. It clearly shows that carbon elements are widely distributed throughout the whole SiO
x@C composite surface, indicating the uniform formation of the carbon layer.
The carbon layer on the surface of the SiOx@C composites functions as a buffer layer, effectively minimizing structural defects such as cracks and pulverization caused by the volume expansion and contraction of SiOx during charging/discharging. Additionally, it reduces contact resistance between active materials, facilitating rapid electron movement and providing an efficient pathway for electrons. The thickness of the carbon layer directly influences the electrochemical properties of the SiOx@C composites as it is closely related to the battery performance. Therefore, this study focuses on investigating the relation between the carbon layer thickness and the electrochemical properties by varying the amount of pitch during the fabrication of the SiOx@C composites via the mechanofusion method.
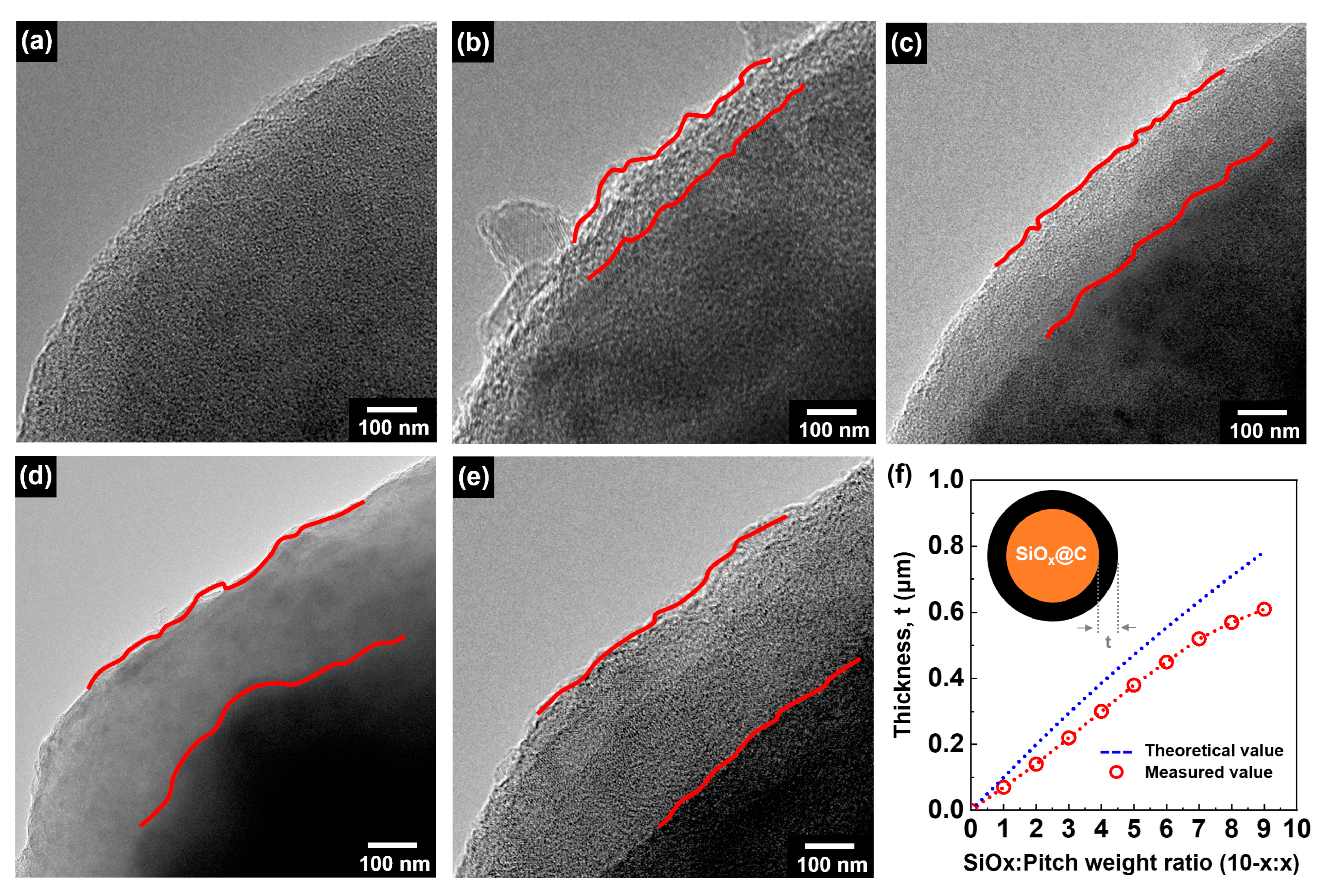
Changes in the carbon layer thickness of the SiO
x@C composites were analyzed using TEM in response to variations in the pitch content for complexation.
Figure 3a depicts the pristine SiO
x surface without the addition of the pitch.
Figure 3b–e shows images of the SiO
x@C composite surfaces with 10, 20, 30, and 40 wt% of pitch added compared to SiO
x, respectively. The pristine SiO
x shows a clean and smooth surface, whereas an increase in the coating pitch content leads to the roughening of the SiO
x@C composite surface due to the influence of the formed carbon layer. Additionally, with the increase in pitch content, the amount of carbon precursor capable of binding to the SiO
x surface during the mechanofusion process also increased. This led to the gradual thickening of the carbon layer on the SiO
x surface, forming an amorphous carbon layer through stabilization and carbonization. Notably, the sample with 40 wt% added pitch (SiO
x@C_6:4) exhibited a carbon layer thickness of around 400 nm.
Figure 3f shows the correlation between the mixing ratio and coating layer thickness. The theoretical value was calculated based on the particle size analysis results, assuming a SiO
x radius of 3.8 μm, and the actual measured value was obtained from the TEM analysis results. Both the theoretical and actual values indicated that as the amount of added pitch increased, the carbon layer thickness also tended to increase. However, it was observed that the actual thickness of the carbon layer was slightly lower than the theoretical value. This discrepancy can be attributed to factors such as the irregularity of the SiO
x particle shape and incomplete composite formation. In the case of the pristine SiO
x, which possesses an irregular morphology rather than spherical shape, it is anticipated that the carbon layer thickness might be uneven. Additionally, the difference between the theoretical and measured values is likely due to the influence of residual pitch that did not participate in the mechanofusion process, resulting in incomplete composite formation.
The degree of structural defects in the carbon layer on the SiO
x surface was determined through Raman analysis, and the results are presented in
Figure 4a. Both the pristine SiO
x and SiO
x@C composites show a peak at around 520 cm
−1, corresponding to the Si bulk single crystal, while the SiO
x@C composite displays carbon-related peaks at 1350 cm
−1 and 1600 cm
−1, corresponding to the D-band and G-band, respectively. The D-band indicates the degree of surface defect or disorder in the carbon layer, while the G-band represents unique characteristics related to the graphitization of the carbon layer [
32].
In general, the relative intensity of the D-band to the G-band (
ID/
IG) serves as an indicator of the degree of defects in the carbon bond structure. A decrease in the
ID/
IG value means reduced defectiveness or increased crystallinity of the carbon bonding structure, while an increase in the
ID/
IG value indicates increased defectiveness or decreased crystallinity [
33]. For the SiO
x@C composites, the
ID/
IG value was approximately 0.98, which was similar to the characteristic value of typical amorphous carbon. These results confirmed that the pitch composited with SiO
x particles was changed into an amorphous carbon layer, characterized by a mixed structure of sp
2 and sp
3 bonds, through the stabilization and carbonization process. In addition, the sp
2/sp
3 bonding ratio of the carbon layer in the SiO
x@C composite was also investigated (see the
Supplementary Materials Figure S3). From the C1s XPS peak, it is obvious that the carbon layer in the SiO
x@C composite exists in the sp
2 configuration more than the sp
3 configuration, and it is revealed that the sp
2/sp
3 ratio is about 2.26. Consequently, it was verified that the pitch with a low content of volatile hydrocarbons can serve as an effective precursor for forming a carbon layer due to its high coking value.
The carbon layer introduced into SiO
x is anticipated to improve the electrical conductivity of SiO
x. Changes in the electrical properties of the SiO
x@C composites prepared with varying amounts of pitch were analyzed using a powder resistance measurement (
Figure 4b). The SiO
x is a representative insulating material known for its high dielectric breakdown strength and low leakage current. As a result, the SiO
x exhibited insulating properties, with electrical conductivity converging to 0 S/cm. In contrast, the SiO
x@C composites showed an enhancement in electrical conductivity with the increasing content of added coating pitch. It was also confirmed that as the pitch content increases, the powder conductivity also increases faster at the same pressure. These phenomena are ascribed to the increased thickness of the carbon layer, which possesses excellent electrical conductivity. As the thickness of the carbon layer increases during the densification process under pressure, the reduction in the resistance per unit volume occurs due to the establishment of extended conductive channels. Under our experimental condition, the composite with 50 wt% added pitch (SiO
x@C_5:5) demonstrated superior electrical conductivity of 4.2 S/cm under a pressure of 200 MPa. This value is more than 14 times higher than that of the SiO
x@C_9:1 sample measured under the same conditions.
An electrochemical impedance spectroscopy (EIS) analysis was also performed for investigating the impedance of the cell containing SiO
x@C composites (see the
Supplementary Materials Figure S4). All SiO
x@C composite cells have a lower charge transfer resistance (
Rct) than that of the SiO
x cells, and the
Rct value of the SiO
x@C composites decreased with the increasing content of added coating pitch; this means that it has a smaller contact resistance and higher electrical conductivity as the thickness of the carbon layer increases. The
Rct value of the SiO
x@C_6:4 composite electrode is approximately 201 Ω, which is significantly lower than the
Rct of 372 Ω of the pristine SiO
x electrode.
The specific surface area of the anode material plays a crucial role in the initial charging and discharging processes of LIBs. The increased specific surface area of the active material provides advantages in terms of the capacity and rate characteristics. However, an excessive surface area can lead to the formation of irreversible compounds, such as SEI layers and Li
2Si
2O
5, Li
2SiO
3, Li
4SiO
4, resulting in reduced initial efficiency [
34].
To examine the changes in the specific surface area and pore structure of the SiO
x@C composite resulting from the carbon layer formation, nitrogen gas adsorption-desorption isotherms were conducted. As shown in
Figure 5a, both the SiO
x and SiO
x@C composites show similar adsorption isotherms, with minimal nitrogen adsorption at low relative pressures (
P/
P0 < 0.3), suggesting the development of mesopores and macropores rather than micropores. For the specific surface area (
SBET), pristine SiO
x was determined to have a value of 9.1 m
2/g. However, the
SBET of the SiO
x@C composite (SiO
x@C_8:2) was about 6.0 m
2/g, which represents a relatively reduced value compared to the pristine SiO
x. This reduction is attributed to the increase in the average particle size resulting from particle agglomeration during the mechanofusion process.
Generally, the specific surface area of electrode materials is considered suitable below 10 m
2/g, as larger values can negatively impact the battery life cycle [
35]. The
SBET values of the SiO
x@C composites prepared in this experiment ranged between 5 and 7 m
2/g, with no additional increase in the specific surface area observed through the mechanofusion composite process (
Table 1). Under our experimental condition, the pore size was calculated from the desorption isotherm using the BJH (Barrett-Joyner-Halenda) method. As a result of the BJH pore size distribution analysis (
Figure 5b), it can be seen that there are many pores with a diameter of 10 nm or more in the pristine SiO
x sample. On the other hand, in the case of the SiO
x@C composite, most pores were found to be concentratedly distributed in the range of 0.9 to 10 nm, with an average size of 8.68 nm. Interestingly, following the mechanofusion process, there was a slight decrease in the
SBET value, while the pore volume increased by over 450%. This phenomenon can be explained by the development of micro- and meso-pores as a result of carbon layer formation. During the carbonization process, the stabilized pitch introduces reactions that involve the removal of oxygen and hydrogen functional groups, promoting the development of micro- and meso-pores on the surface. As a result, it was found that in the case of the SiO
x@C composite, the microporosity and mesoporosity increased by 2.8% and 8.4%, respectively, while the macroporosity decreased to about 11.2% compared to the pristine SiO
x. Furthermore, the average pore size reduced by approximately 40% after the mechanofusion process (from 14.7 nm to 8.7 nm), indicating the development of smaller pores around the range of 2–10 nm. This observation coincides with the aforementioned increase in the mesopore volume and supports the explanation for the increase in the pore volume.
3.3. Battery Performance Evaluation of SiOx@C Composite Anode
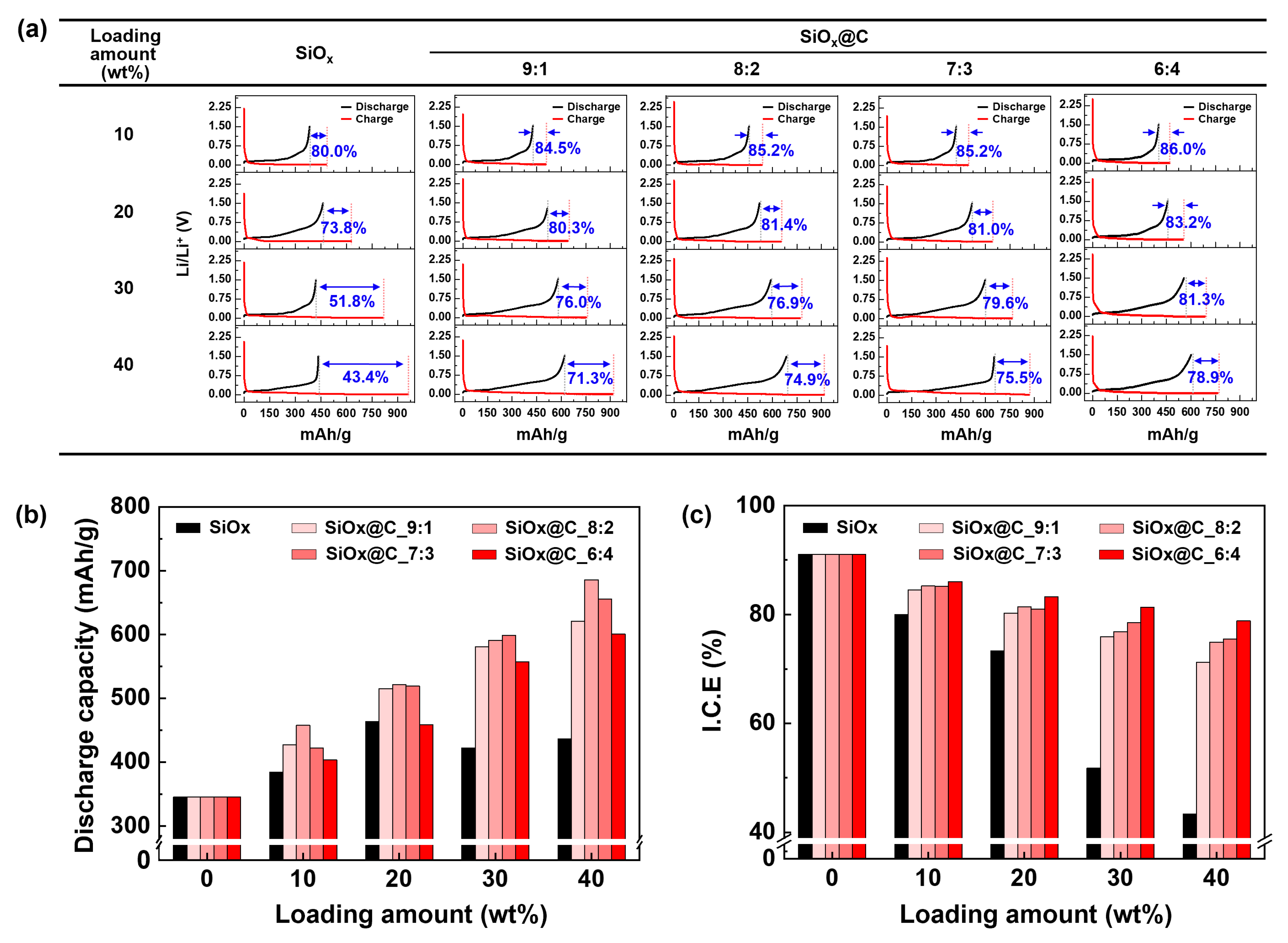
The electrochemical properties of the anode materials were evaluated under various fabricating conditions and active material composition ratios, with all the charge/discharge curves presented in
Figure 6a. The corresponding results for the discharge capacity and initial efficiency are summarized in
Figure 6b,c, respectively.
In the case of the anode electrode using only artificial graphite (AG) as the active material, a discharge capacity of 345.0 mAh/g and an initial efficiency of 91.0% were observed. This was comparable to the theoretical values of the typical AG-based anode materials (
Figure 6b). In the case of the electrode prepared by adding pristine SiO
x to the AG, the discharge capacity was found to increase to 463.3 mAh/g, up to a loading amount of 20 wt%, after which it began to decrease. This decrease was attributed to the volume expansion of SiO
x during lithium intercalation, leading to pulverization and loss of electrical contact due to the resulting structural transformation [
36].
For the SiO
x@C composites, the carbon layer on the SiO
x surface effectively alleviated the volume expansion of SiO
x caused by lithiation/delithiation and maintained the electrical conductivity, resulting in an increase in the discharge capacity with the increasing loading amount. Although a consistent causal relationship between the mixing ratio and discharge capacity was not observed at the same loading amount, as the content of pitch increased, the discharge capacity tended to decrease. Namely, the carbon layer thickness of the SiO
x@C composite and the discharge capacity showed an inverse relationship. This is attributed to SiO
x being the primary determinant of the discharge capacity in the SiO
x@C composites. Unlike graphite, which stores lithium ions through an intercalation reaction within the graphitic layers, an amorphous carbon is known to have a small contribution to securing the discharge capacity. To confirm the capacity contribution of the carbon layer in the SiO
x@C composite, the control experiments were carried out using carbonized pitch as a comparison material (see the
Supplementary Materials Figure S5). The carbonized pitch is expected to have a capacity of approximately 25 mAh/g, and the carbon layer of the SiO
x@C composite is believed to not contribute significantly to the capacity. The amorphous carbon layer introduced on the SiO
x surface only stored lithium ions in localized vacancies created by the unstable stacking structure of the carbon layer [
37]. Otherwise, when the pitch content was low (e.g., SiO
x@C_9:1), the capacity retention was relatively reduced due to the decreased role of the carbon layer described earlier. The sample with the highest discharge capacity was the SiO
x@C_8:2 composite with the loading amount of 40 wt%. This sample showed a discharge capacity of 685 mAh/g, which was approximately 1.57 times higher than that of the pristine SiO
x at the same loading amount. The detailed charge storage mechanism according to the lithiation/delithiation process of the SiO
x@C composite is shown in
Supplementary Materials Figure S6.
Figure 6c represents the ICE of the samples prepared with different mixing ratios and active material compositions. Both the SiO
x and SiO
x@C composites showed a tendency for the ICE value to decrease as the loading amount increased. In the case of the pristine SiO
x, the ICE value significantly decreased, reaching the lowest value of 43.4% when the loading amount was 40%. However, the SiO
x@C composites showed relatively high ICE values. At the 40 wt% loading amount, the SiO
x@C_6:4 composites demonstrated an ICE of 78.9%. This value was approximately 1.82 times higher than that of pristine SiO
x. This improvement was attributed to the effective suppression of irreversible reactions and SEI formation by the carbon layer introduced on the SiO
x surface. At the same loading amount, it was observed that the ICE tends to increase with the higher pitch content for fabricating SiO
x@C composites. In other words, an increased thickness of the carbon layer contributed to the enhanced stability of the active material. A thicker carbon layer introduced into the SiO
x@C composites is more advantageous in terms of the ICE value, but less advantageous in terms of securing the discharge capacity. Therefore, in this study, based on the various cell test results, the SiO
x@C_8:2 composite sample was selected as the optimal mixing ratio for the anode material, and cycle stability tests were conducted.
Cycling measurements were conducted on the anodes consisting of the pristine SiO
x and SiO
x@C_8:2 composites at a current density of 1.0 C for the initial two cycles, and 0.5 C for the subsequent cycles. A compilation of investigations carried out using various SiO
x@carbon composites is also presented in
Supplementary Materials Figure S7. As depicted in
Figure 7a, the SiO
x@C_8:2 composite shows a higher reversible capacity and more stable cycling performance compared to the pristine SiO
x. The anode containing the SiO
x@C_8:2 composite with the 20 wt% loading amount maintains about 60% of its initial charge capacity after 300 cycles, whereas the pristine SiO
x anode exhibits a significant decrease in capacity with a capacity retention rate of only 12.6% at 100 cycles.
Furthermore, the Coulombic efficiency of the SiO
x anode reaches 99% after 10 cycles, while the Coulombic efficiency of the SiO
x@C_8:2 anode achieves 99% after only 4 cycles (
Figure 7b). The delayed attainment of 99% Coulombic efficiency in the SiO
x anode suggests the continued formation of SEI and excessive side reactions on untreated surfaces, potentially leading to a substantial capacity reduction by consuming limited Li
+ ions.
Additionally, to compare the rate characteristics of the anode, the C-rate was measured at a current density of 0.1 C during the first cycle, followed by measurements at 0.2 C, 0.5 C, 1.0 C, 2.0 C, and 5.0 C for the subsequent five cycles (
Figure 7c). In particular, the SiO
x@C_8:2 composite demonstrated a stable specific capacity of 77.2% at a current density of 5.0 C compared to 0.2 C. Furthermore, as the current density was reduced from 5.0 C back to 0.2 C, the specific capacity recovered to 440 mAh/g. In contrast, the SiO
x anode showed relatively inferior rate characteristics, exhibiting a specific capacity of only 47.8% at a current density of 5.0 C compared to the established 0.2 C rate. Upon returning the current density from 5.0 C to 0.2 C, the SiO
x anode showed a specific capacity of only 189 mAh/g. Thus, it is believed that the carbon layer on the SiO
x@C composite induces the uniform formation of the SEI layer, thereby having a positive effect on battery cycle stability.