VIS-NIR Modeling of Hydrangenol and Phyllodulcin Contents in Tea-Hortensia (Hydrangea macrophylla subsp. serrata)
Abstract
1. Introduction
2. Materials and Methods
2.1. Spectrometer Set-Up
2.2. Plant Material and Cultivation
2.3. Experimental Design
2.4. Analysis of Hydrangenol and Phyllodulcin
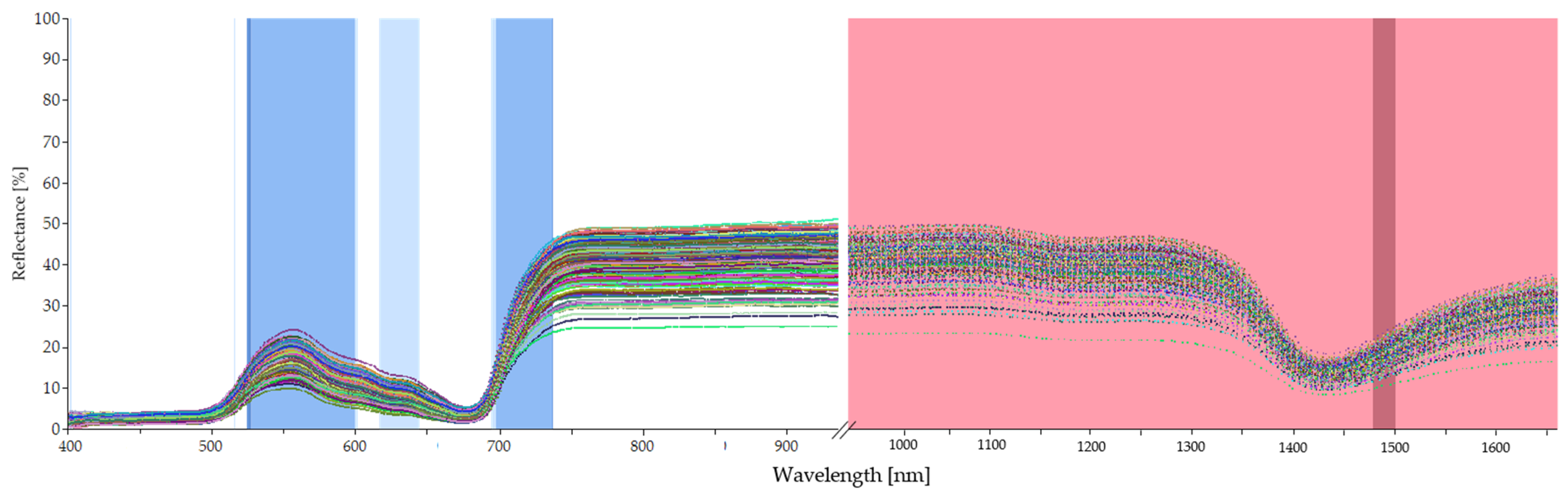
2.5. Outlier Detection and Spectra Pre-Processing
2.6. Model Development
2.7. Statistical Analysis
- n = number of samples (spectra), yi = measured reference value of the sample i,
- ŷ = predicted value of the sample i, ȳ = mean value of all samples.
3. Results
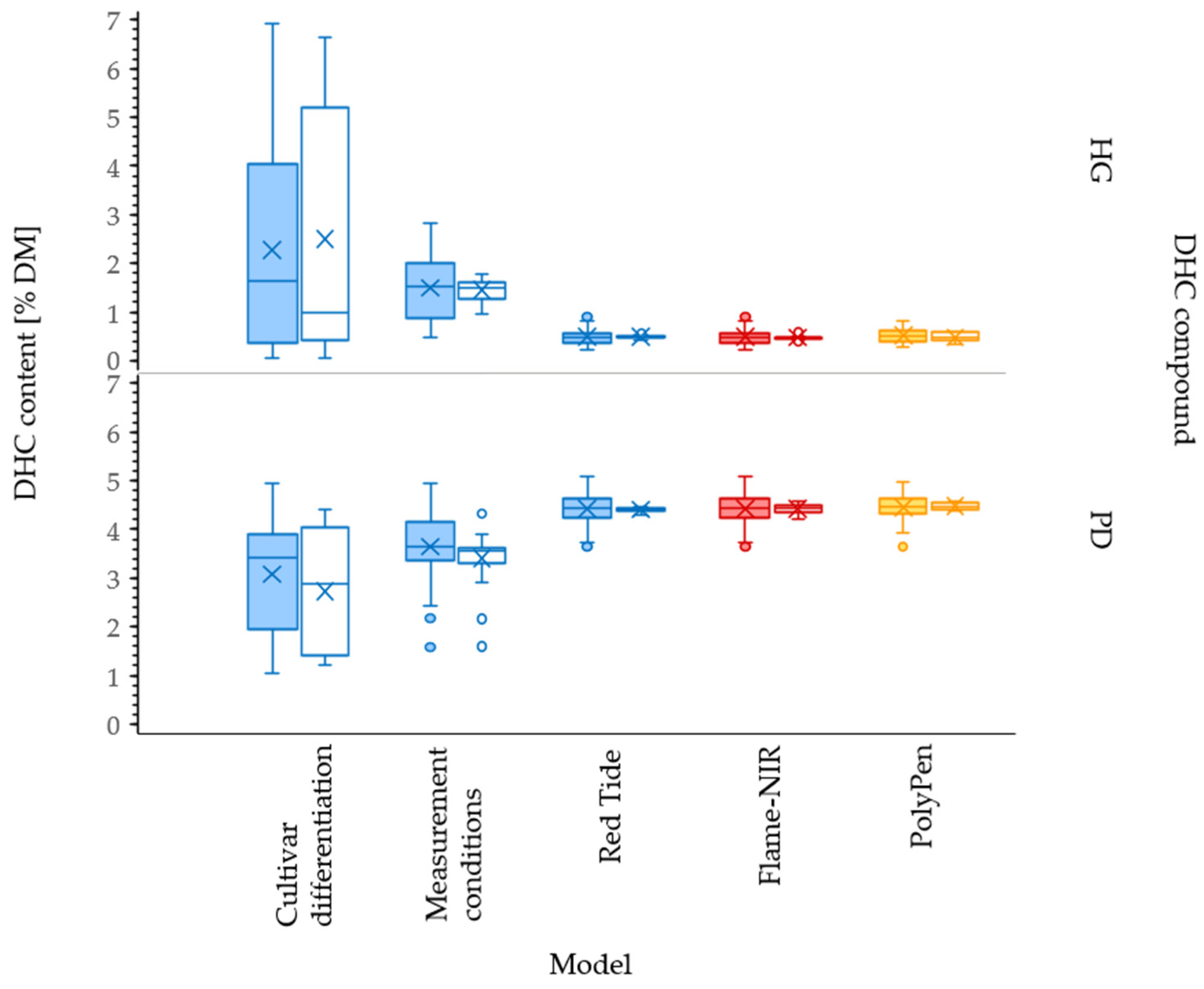
3.1. Hydrangenol and Phyllodulcin Contents
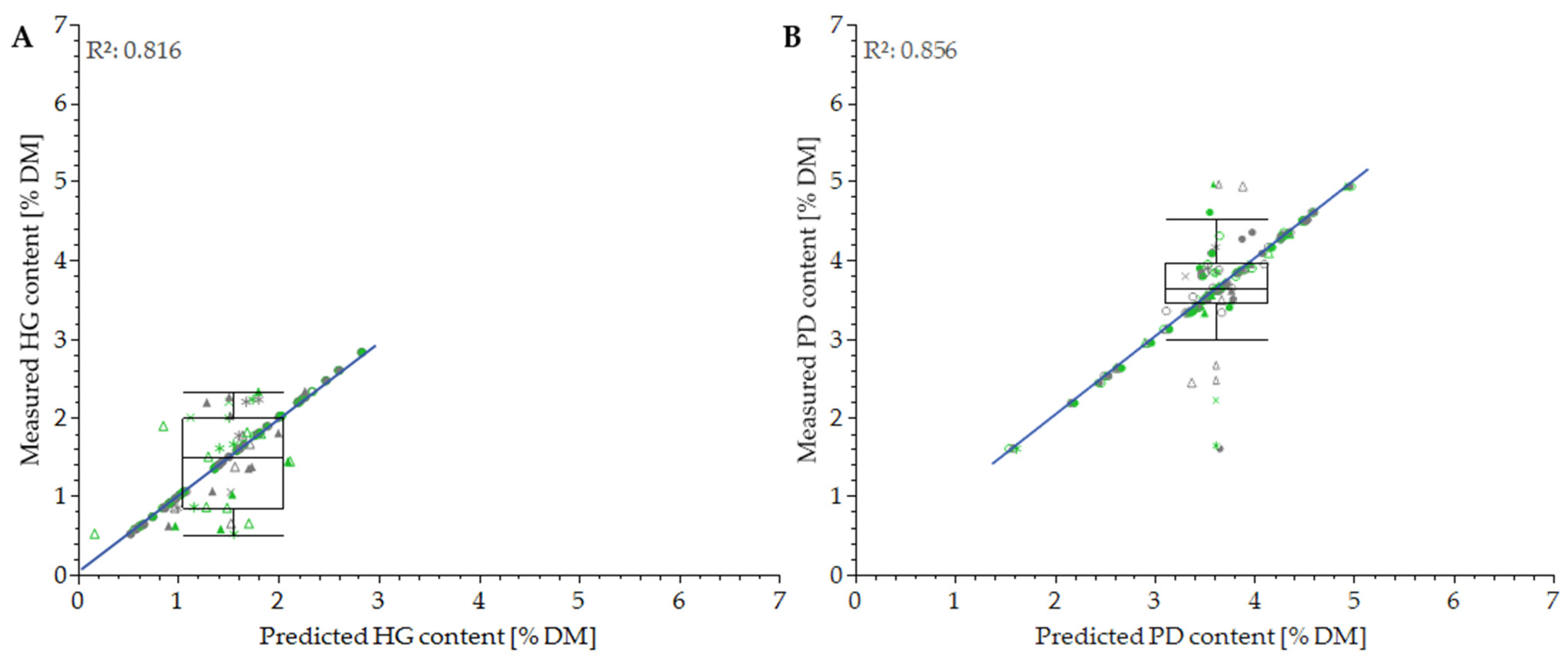
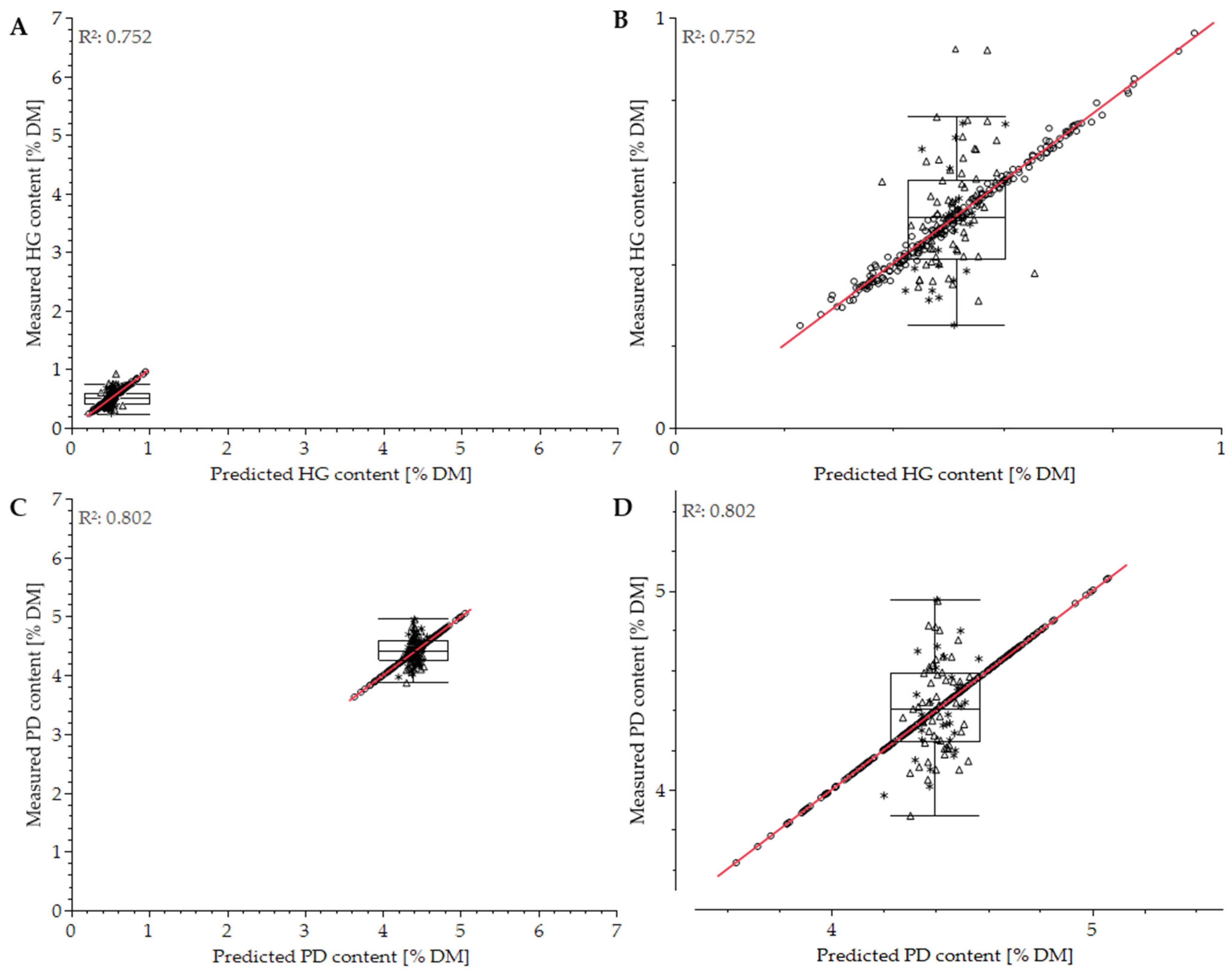
3.2. Differentiation of Cultivars
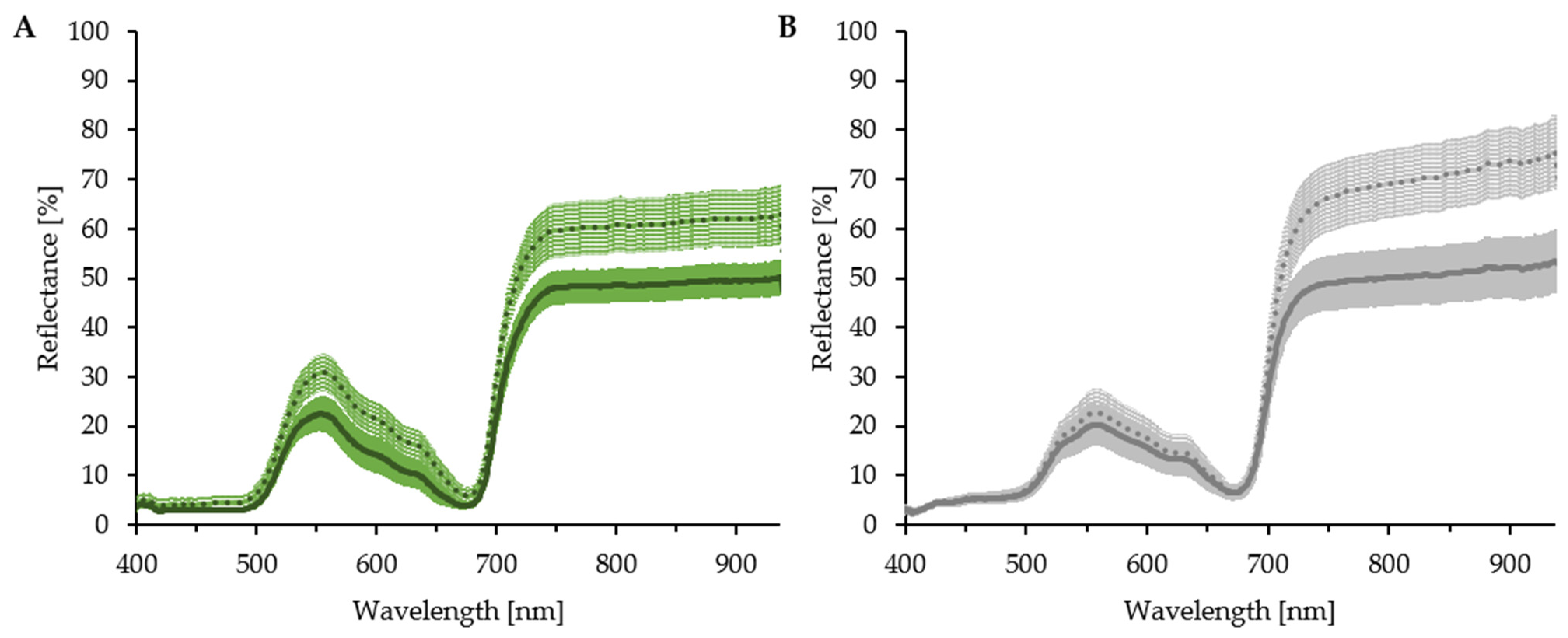
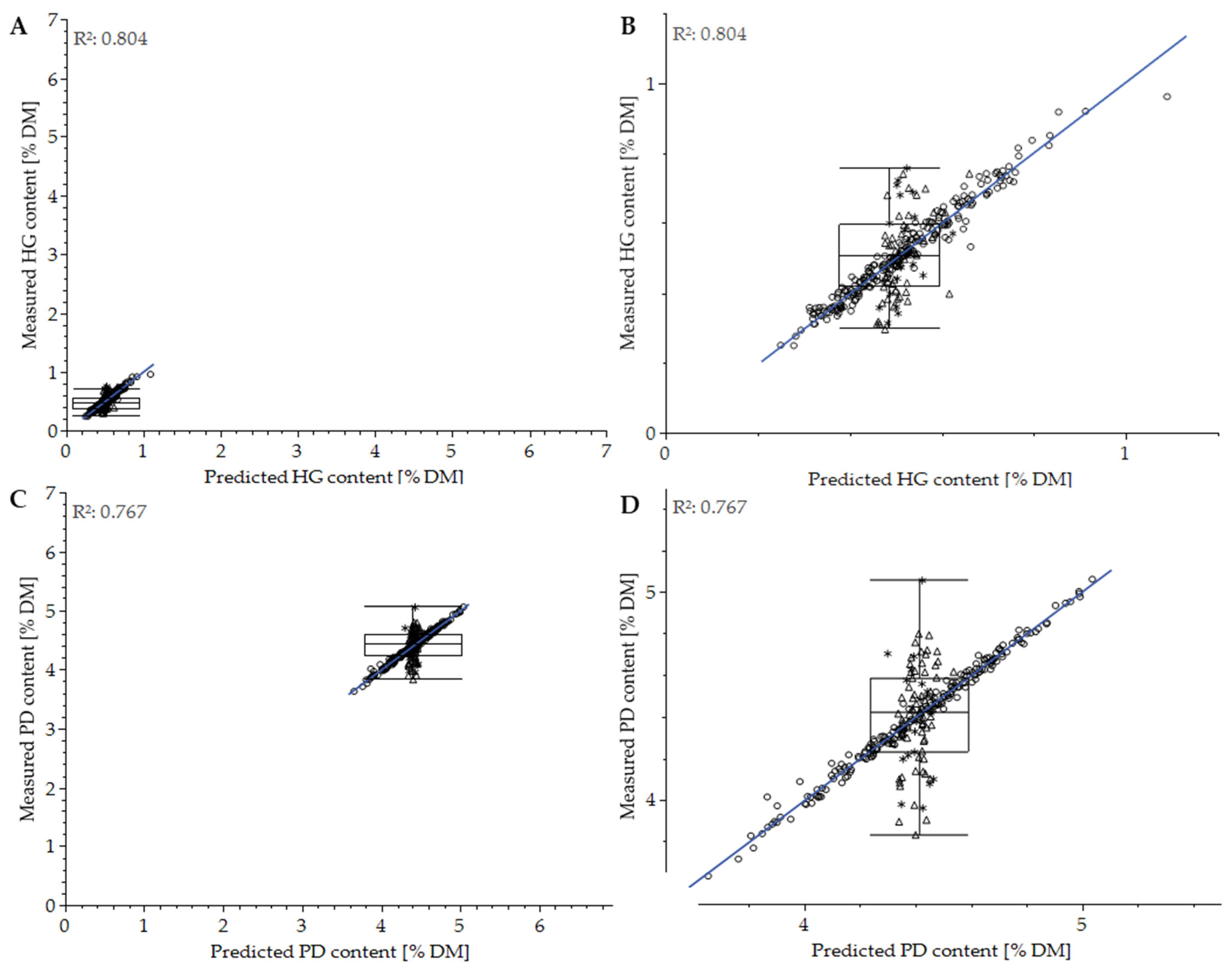
3.3. Impact of Measurement Conditions
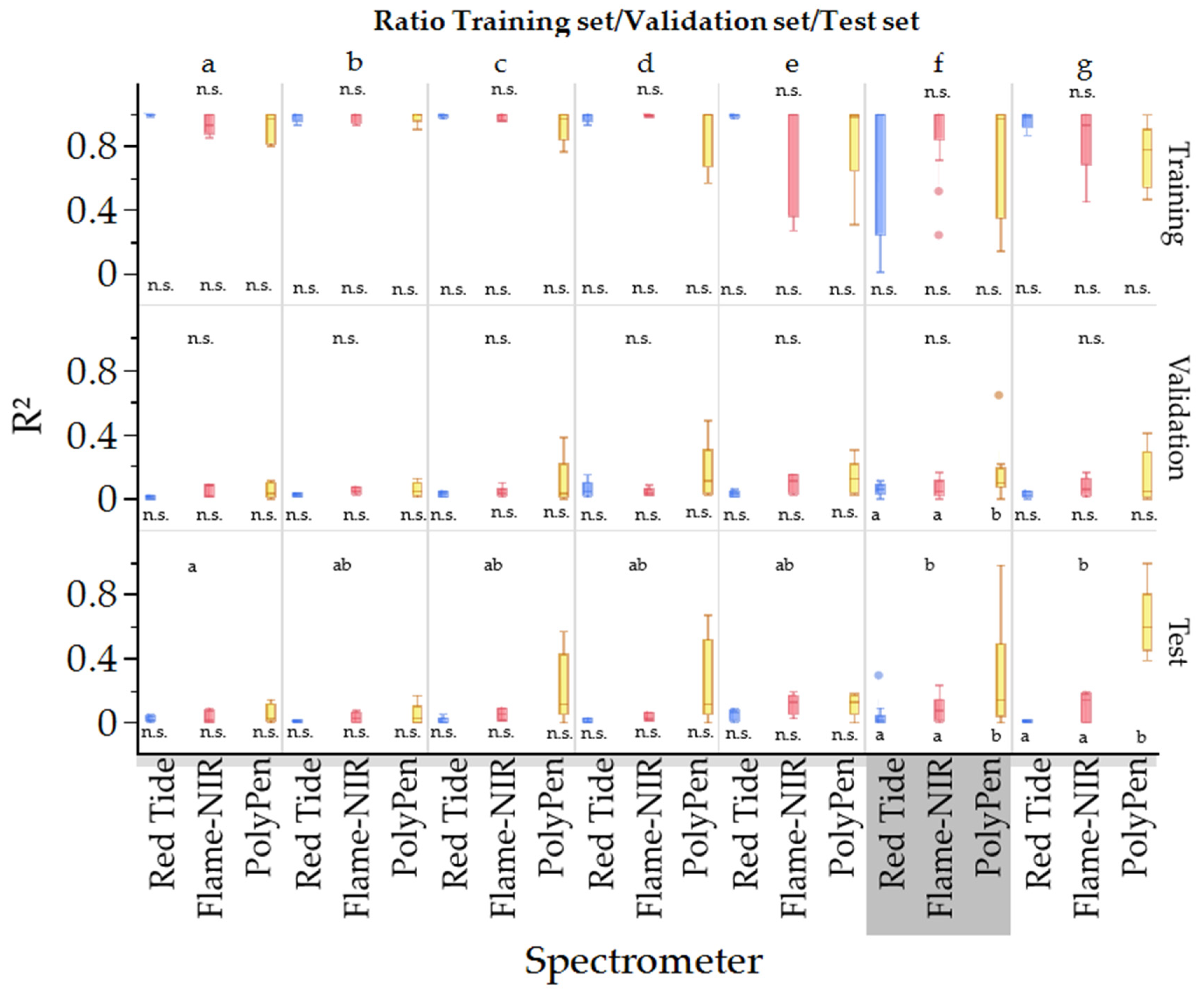
3.4. Effect of Spectrometer
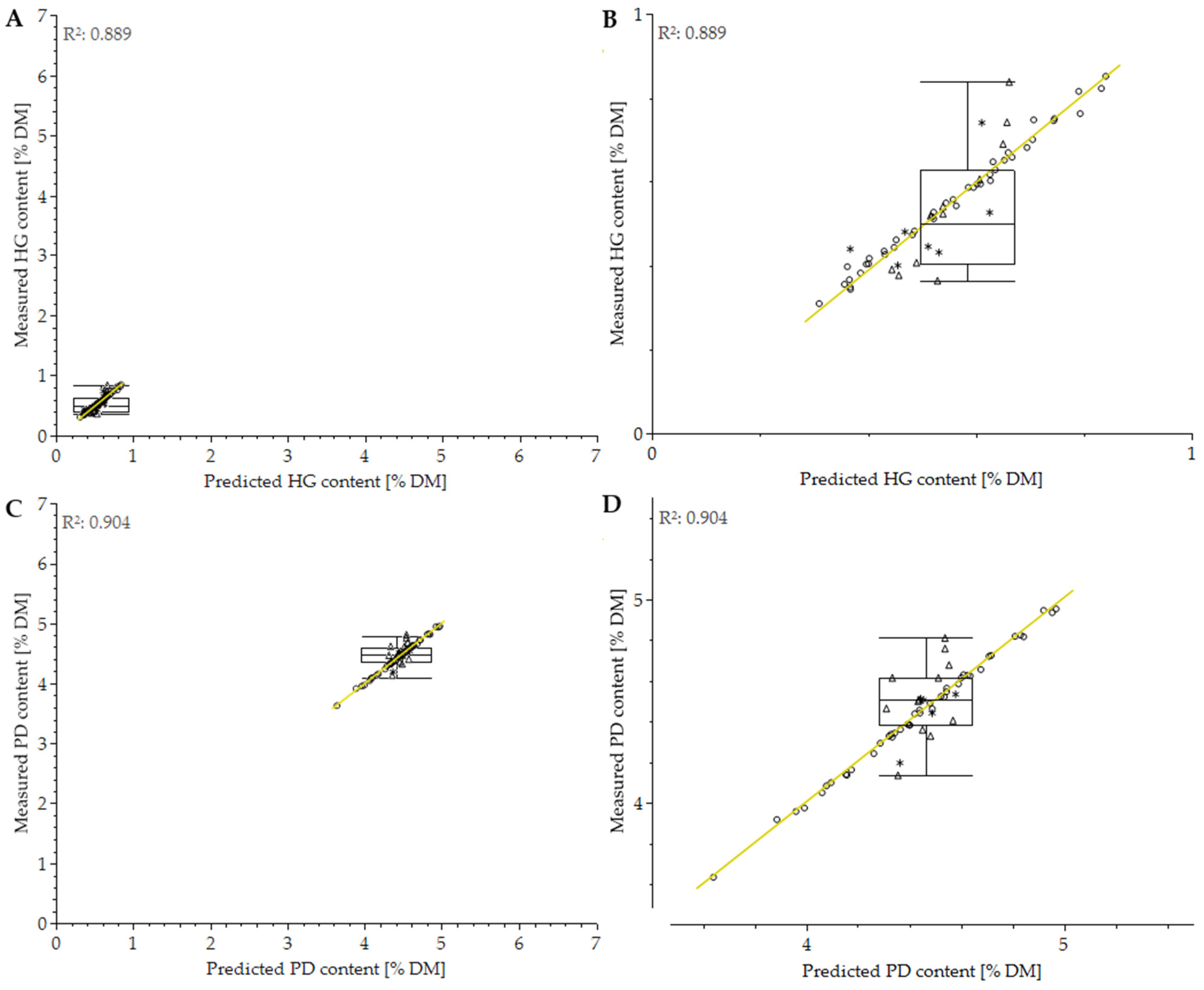
3.5. Use of Handheld PolyPen RP400
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kindl, H. Formation of a Stilbene in Isolated Chloroplasts. Hoppy Seylers Z. Physiol. Chem. 1971, 325, 767–768. [Google Scholar]
- Yagi, A.; Ogata, Y.; Yamauchi, T.; Nishoka, I. Metabolism of Phenylpropanoids in Hydrangea serrata var. thunbergii and the Biosynthesis of Phyllodulcin. Phytochemistry 1977, 16, 1098–1100. [Google Scholar] [CrossRef]
- Ujihara, M.; Shinozaki, M.; Kato, M. Accumulation of Phyllodulcin in Sweet-Leaf Plants of Hydrangea serrata and Its Neutrality in the Defence against a Specialist Leafmining Herbivore. Res. Popul. Ecol. 1995, 37, 249–257. [Google Scholar] [CrossRef]
- Moll, M.D.; Vieregge, A.S.; Wiesbaum, C.; Blings, M.; Vana, F.; Hillebrand, S.; Ley, J.; Kraska, T.; Pude, R. Dihydroisocoumarin Content and Phenotyping of Hydrangea macrophylla subsp. serrata Cultivars under Different Shading Regimes. Agronomy 2021, 11, 1743. [Google Scholar] [CrossRef]
- Suzuki, H.; Ikeda, T.; Matsumoto, T.; Noguchi, M. Polyphenol Components in Cultured Cells of Amacha (Hydrangea macrophylla seringe var. thunbergii Makino). Agric. Biol. Chem. 1978, 42, 1133–1137. [Google Scholar] [CrossRef]
- Shin, W.; Kim, S.J.; Shin, J.M.; Kim, S.H. Structure-taste correlations in sweet dihydrochalcone, sweet dihydroisocoumarin, and bitter flavone compounds. J. Med. Chem. 1995, 38, 4325–4331. [Google Scholar] [CrossRef]
- Asen, S.; Cathey, H.M.; Stuart, N.W. Enhancement of Gibberellin Growth-Promoting Activity by Hydrangenol Isolated From Leaves of Hydrangea macrophylla. Plant Physiol. 1960, 35, 816–819. [Google Scholar] [CrossRef]
- Nozawa, K.; Yamada, M.; Tsuda, Y.; Kawai, K.-I.; Nakajima, S. Antifungal Activity of Oosponol, Oospolactone, Phyllodulcin, Hydrangenol, and Some Other Related Compounds. Chem. Pharm. Bull. 1981, 29, 2689–2691. [Google Scholar] [CrossRef]
- Kakegawa, H.; Matsumoto, H.; Satoh, T. Inhibitory Effects of Hydrangenol Derivatives on the Activation of Hyaluronidase and ther Antiallergic Ectivities. Planta Med. 1988, 385–389. [Google Scholar] [CrossRef]
- Kim, H.-J.; Kang, C.-H.; Jayasooriya, R.G.P.T.; Dilshara, M.G.; Lee, S.; Choi, Y.H.; Seo, Y.T.; Kim, G.-Y. Hydrangenol inhibits lipopolysaccharide-induced nitric oxide production in BV2 microglial cells by suppressing the NF-κB pathway and activating the Nrf2-mediated HO-1 pathway. Int. Immunopharmacol. 2016, 35, 61–69. [Google Scholar] [CrossRef]
- Shin, S.-S.; Ko, M.-C.; Park, Y.-J.; Hwang, B.; Park, S.L.; Kim, W.-J.; Moon, S.-K. Hydrangenol inhibits the proliferation, migration, and invasion of EJ bladder cancer cells via p21WAF1-mediated G1-phase cell cycle arrest, p38 MAPK activation, and reduction in Sp-1-induced MMP-9 expression. EXCLI J. 2018, 17, 531–543. [Google Scholar] [CrossRef] [PubMed]
- Myung, D.-B.; Han, H.-S.; Shin, J.-S.; Park, J.Y.; Hwang, H.J.; Kim, H.J.; Ahn, H.S.; Lee, S.H.; Lee, K.-T. Hydrangenol Isolated from the Leaves of Hydrangea serrata Attenuates Wrinkle Formation and Repairs Skin Moisture in UVB-Irradiated Hairless Mice. Nutrients 2019, 11, 2354. [Google Scholar] [CrossRef]
- Sanjeewani, W.C.; Sutharsan, S.; Srikrishnah, S. Effects of Hydrangea (Hydrangea macrophylla L.) Leaf Extract Foliar Application on Growth and Yield of Mungbean (Vigna radiata L.). Int. J. Bot. Stud. 2019, 4, 180–184. [Google Scholar]
- Nakajima, S.; Sugiyama, S.; Suto, M. Synthesis of Antifungal Isocoumarins (I). Org. Prep. Proced. Int. 1979, 11, 77–86. [Google Scholar] [CrossRef]
- Yamahara, J.; Matsuda, H.; Shimoda, H.; Ishikawa, H.; Kawamori, S.; Wariishi, N.; Harada, E.; Murakami, N.; Yoshikawa, M. Development of bioactive functions in hydrangeae dulcis folium. II. Antiulcer, antiallergy, and cholagoic effects of the extract from hydrangeae dulcis folium. Yakugaku Zasshi 1994, 114, 401–413. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Braca, A.; Bader, A.; de Tommasi, N. Plant and Fungi 3,4-Dihydroisocoumarins: Structures, Biological Activity, and Taxonomic Relationships. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2012; Volume 37, pp. 191–215. ISBN 978-0-44459-514-0. [Google Scholar]
- Dilshara, M.G.; Jayasooriya, R.G.P.T.; Lee, S.; Jeong, J.B.; Seo, Y.T.; Choi, Y.H.; Jeong, J.-W.; Jang, Y.P.; Jeong, Y.-K.; Kim, G.-Y. Water extract of processed Hydrangea macrophylla (Thunb.) Ser. leaf attenuates the expression of pro-inflammatory mediators by suppressing Akt-mediated NF-κB activation. Environ. Toxicol. Pharmacol. 2013, 35, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Kawamura, M.; Kagata, M.; Masaki, E.; Nishi, H. Phyllodulcin, a Constituent of “Amacha”, Inhibits Phosphodiesterase in Bovine Adrenocortical Cells. Pharmacol. Toxicol. 2002, 90, 106–108. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.; Lim, S.M.; Kim, M.S.; Yoo, S.H.; Kim, Y. Phyllodulcin, a Natural Sweetener, Regulates Obesity-Related Metabolic Changes and Fat Browning-Related Genes of Subcutaneous White Adipose Tissue in High-Fat Diet-Induced Obese Mice. Nutrients 2017, 9, 1049. [Google Scholar] [CrossRef]
- Kim, E.; Shin, J.H.; Seok, P.R.; Kim, M.S.; Yoo, S.H.; Kim, Y. Phyllodulcin, a natural functional sweetener, improves diabetic metabolic changes by regulating hepatic lipogenesis, inflammation, oxidative stress, fibrosis, and gluconeogenesis in db/db mice. J. Funct. Foods 2018, 42, 1–11. [Google Scholar] [CrossRef]
- Kamei, K.; Matsuoka, H.; Furuhata, S.I.; Fujisaki, R.I.; Kawakami, T.; Mogi, S.; Yoshihara, H.; Aoki, N.; Ishii, A.; Shibuya, T. Anti-Malarial Activity of Leaf-Extract of Hydrangea macrophylla, a Common Japanese Plant. Acta Med. Okayama 2000, 54, 227–232. [Google Scholar] [CrossRef]
- Hu, C.-L.; Ge, L.; Tang, Y.; Li, J.; Wu, C.-H.; Hu, J.-H.; Yuan, J.-T.; Fan, Y.-Z. Phyllodulcin Protects PC12 Cells against the Injury Induced by Oxygen and Glucose Deprivation-Restoration. Acta Pol. Pharm. Drug Res. 2019, 76, 1043–1050. [Google Scholar] [CrossRef]
- Swartz, M.E. UPLC™: An Introduction and Review. J. Liq. Chromatogr. Relat. Technol. 2005, 28, 1253–1263. [Google Scholar] [CrossRef]
- Nováková, L.; Matysová, L.; Solich, P. Advantages of application of UPLC in pharmaceutical analysis. Talanta 2006, 68, 908–918. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Saini, G.; Nair, A.; Sharma, R. UPLC: A Preeminent Technique In Pharmaceutical Analysis. Acta Pol. Pharm. Drug Res. 2012, 69, 371–380. [Google Scholar]
- Cozzolino, D. Near infrared spectroscopy in natural products analysis. Planta Med. 2009, 75, 746–756. [Google Scholar] [CrossRef]
- Schulz, H. Analysis of Coffee, Tea, Cocoa, Tobacco, Spices, Medicinal and Aromatic Plants, and Related Products. In Near-Infrared Spectroscopy in Agriculture; Roberts, C.A., Ed.; Wiley: Hoboken, NJ, USA, 2004; pp. 345–376. [Google Scholar]
- Giovenzana, V.; Beghi, R.; Buratti, S.; Civelli, R.; Guidetti, R. Monitoring of fresh-cut Valerianella locusta Laterr. shelf life by electronic nose and VIS-NIR spectroscopy. Talanta 2014, 120, 368–375. [Google Scholar] [CrossRef]
- Ercioglu, E.; Velioglu, H.M.; Boyaci, I.H. Chemometric Evaluation of Discrimination of Aromatic Plants by Using NIRS, LIBS. Food Anal. Methods 2018, 11, 1656–1667. [Google Scholar] [CrossRef]
- Wulandari, L.; Permana, B.D.; Kristiningrum, N. Determination of Total Flavonoid Content in Medicinal Plant Leaves Powder Using Infrared Spectroscopy and Chemometrics. Indones. J. Chem. 2020, 20, 1044–1051. [Google Scholar] [CrossRef]
- Ercioglu, E.; Velioglu, H.M.; Boyaci, I.H. Determination of terpenoid contents of aromatic plants using NIRS. Talanta 2018, 178, 716–721. [Google Scholar] [CrossRef]
- Schulz, H.; Drews, H.-H.; Krüger, H. Rapid NIRS Determination of Quality Parameters in Leaves and Isolated Essential Oils of Mentha Species. J. Essent. Oil Res. 1999, 11, 185–190. [Google Scholar] [CrossRef]
- Noland, R.L.; Wells, M.S.; Coulter, J.A.; Tiede, T.; Baker, J.M.; Martinson, K.L.; Sheaffer, C.C. Estimating alfalfa yield and nutritive value using remote sensing and air temperature. Field Crops Res. 2018, 222, 189–196. [Google Scholar] [CrossRef]
- Pantazi, X.E.; Moshou, D.; Alexandridis, T.; Whetton, R.L.; Mouazen, A.M. Wheat yield prediction using machine learning and advanced sensing techniques. Comput. Electron. Agric. 2016, 121, 57–65. [Google Scholar] [CrossRef]
- Stroppiana, D.; Migliazzi, M.; Chiarabini, V.; Crema, A.; Musanti, M.; Franchino, C.; Villa, P. Rice Yield Estimation Using Multispectral Data From UAV: A Preliminary Experiment In Northern Italy. In Proceedings of the International Geoscience and Remote Sensing Symposium (IGARSS), Milan, Italy, 26–31 July 2015; pp. 4664–4667. [Google Scholar]
- Landgrebe, D. On Information Extraction Principles for Hyperspectral Data: A White Paper; School of Electrical and Computer Engineering, Purdue University: West Lafayette, IN, USA, 1997. [Google Scholar]
- Famili, A.; Shen, W.-M.; Weber, R.; Simoudis, E. Data Preprocessing and Intelligent Data Analysis. Intell. Data Anal. 1997, 1, 3–23. [Google Scholar] [CrossRef]
- Aguilera-Morillo, M.C.; Aguilera, A.M. P-spline Estimation of Functional Classification Methods for Improving the Quality in the Food Industry. Commun. Stat. B Simul. Comput. 2015, 44, 2513–2534. [Google Scholar] [CrossRef]
- Aguilera, A.M.; Aguilera-Morillo, M.C. Comparative study of different B-spline approaches for functional data. Math. Comput. Model. 2013, 58, 1568–1579. [Google Scholar] [CrossRef]
- Haaland, D.M.; Thomas, E.V. Partial least-squares methods for spectral analyses. 1. Relation to other quantitative calibration methods and the extraction of qualitative information. Anal. Chem. 1988, 60, 1193–1202. [Google Scholar] [CrossRef]
- Hotelling, H. The Generalization of Student’s Ratio. Ann. Math. Stat. 1931, 2, 360–378. [Google Scholar] [CrossRef]
- Barnes, R.J.; Dhanoa, M.S.; Lister, S.J. Standard Normal Variate Transformation and De-Trending of Near-Infrared Diffuse Reflectance Spectra. Appl. Spectrosc. 1989, 43, 772–777. [Google Scholar] [CrossRef]
- Barak, P. Smoothing and Differentiation by an Adaptive-Degree Polynomial Filter. Anal. Chem. 1995, 67, 2758–2762. [Google Scholar] [CrossRef]
- Savitzky, A.; Golay, M. Smoothing and Differentiation of Data by Simplified Least Squares Procedures. Anal. Chem. 1964, 36, 1627–1639. [Google Scholar] [CrossRef]
- Luo, J.; Ying, K.; He, P.; Bai, J. Properties of Savitzky–Golay digital differentiators. Digit. Signal Process. 2005, 15, 122–136. [Google Scholar] [CrossRef]
- O’Haver, T.C. Potential Clinical Applications of Derivative and Wavelength-Modulation Spectrometry. Clin. Chem. 1979, 25, 1548–1553. [Google Scholar] [CrossRef]
- Barbin, D.F.; ElMasry, G.; Sun, D.-W.; Allen, P. Predicting quality and sensory attributes of pork using near-infrared hyperspectral imaging. Anal. Chim. Acta 2012, 719, 30–42. [Google Scholar] [CrossRef] [PubMed]
- Aleixandre-Tudo, J.L.; Nieuwoudt, H.; Aleixandre, J.L.; Du Toit, W.J. Robust Ultraviolet-Visible (UV-Vis) Partial Least-Squares (PLS) Models for Tannin Quantification in Red Wine. J. Agric. Food Chem. 2015, 63, 1088–1098. [Google Scholar] [CrossRef]
- Huang, Y.; Dong, W.; Sanaeifar, A.; Wang, X.; Luo, W.; Zhan, B.; Liu, X.; Li, R.; Zhang, H.; Li, X. Development of simple identification models for four main catechins and caffeine in fresh green tea leaf based on visible and near-infrared spectroscopy. Comput. Electron. Agric. 2020, 173, 105388. [Google Scholar] [CrossRef]
- Sanaeifar, A.; Huang, X.; Chen, M.; Zhao, Z.; Ji, Y.; Li, X.; He, Y.; Zhu, Y.; Chen, X.; Yu, X. Nondestructive monitoring of polyphenols and caffeine during green tea processing using Vis-NIR spectroscopy. Food Sci. Nutr. 2020, 8, 5860–5874. [Google Scholar] [CrossRef] [PubMed]
- Wold, S.; Sjöström, M.; Eriksson, L. PLS-regression: A basic tool of chemometrics. Chemom. Intell. Lab. Syst. 2001, 58, 109–130. [Google Scholar] [CrossRef]
- Dunn, W.J.; Wold, S.; Edlund, U.; Hellberg, S.; Gasteiger, J. Multivariate structure-activity relationships between data from a battery of biological tests and an ensemble of structure descriptors: The PLS method. Quant. Struct. Act. Relat. 1984, 3, 131–137. [Google Scholar] [CrossRef]
- Geladi, P.; Kowalski, B.R. Partial least-squares regression: A tutorial. Anal. Chim. Acta 1986, 185, 1–17. [Google Scholar] [CrossRef]
- Hu, Y.; Zhang, H.; Liang, W.; Xu, P.; Lou, K.; Pu, J. Rapid and Simultaneous Measurement of Praeruptorin A, Praeruptorin B, Praeruptorin E, and Moisture Contents in Peucedani Radix Using Near-Infrared Spectroscopy and Chemometrics. J. AOAC Int. 2020, 103, 504–512. [Google Scholar] [CrossRef]
- Wold, S.; Johansson, E.; Cocchi, M. PLS: Partial least squares projections to latent structures. In 3D QSAR in Drug Design: Theory, Methods and Applications; Kubinyi, H., Ed.; ESCOM: Leiden, The Netherlands, 1993; pp. 523–550. ISBN 90-72199-14-6. [Google Scholar]
- Udelhoven, T.; Jarmer, T.; Hostert, P.; Hill, J. The aquisition of spectral reflectance measurements under field and laboratory conditions as support for hyperspectral applications in precision farming. In Proceedings of the 2nd EARSeL Workshop on Imaging Spectroscopy Co-Organized with the SIG “Imaging Spectroscopy”, Enschede, The Netherlands, 11–13 July 2000. [Google Scholar]
- Ge, H.; Lu, S.; Zhao, Y. Effects of Leaf Hair on Leaf Reflectance and Hyperspectral Vegetation Indices. Spectrosc. Spec. Anal. 2012, 32, 439–444. [Google Scholar]
- Lu, S. Effects of Leaf Surface Wax on Leaf Spectrum and Hyperspectral Vegetation Indices. In Proceedings of the International Geoscience and Remote Sensing Symposium IGARSS 2013, Melbourne, Australia, 21–26 July 2013. [Google Scholar]
- Lu, X.; Lu, S. Effects of adaxial and abaxial surface on the estimation of leaf chlorophyll content using hyperspectral vegetation indices. Int. J. Remote Sens. 2015, 36, 1447–1469. [Google Scholar] [CrossRef]
- Yamashita, H.; Sonobe, R.; Hirono, Y.; Morita, A.; Ikka, T. Dissection of hyperspectral reflectance to estimate nitrogen and chlorophyll contents in tea leaves based on machine learning algorithms. Sci. Rep. 2020, 10, 17360. [Google Scholar] [CrossRef] [PubMed]
- Gong, P.; Pu, R.; Heald, R.C. Analysis of in situ hyperspectral data for nutrient estimation of giant sequoia. Int. J. Remote Sens. 2002, 23, 1827–1850. [Google Scholar] [CrossRef]
- Saviano, A.M.; Madruga, R.O.G.; Lourenço, F.R. Measurement uncertainty of a UPLC stability indicating method for determination of linezolid in dosage forms. Measurement 2015, 59, 1–8. [Google Scholar] [CrossRef]
- Tschannerl, J.; Ren, J.; Jack, F.; Krause, J.; Zhao, H.; Huang, W.; Marshall, S. Potential of UV and SWIR hyperspectral imaging for determination of levels of phenolic flavour compounds in peated barley malt. Food Chem. 2019, 270, 105–112. [Google Scholar] [CrossRef]



| Spectrometer (Year of Experiment) | Wavelength Range [nm] for SNV Transformation | Wavelength Range [nm] for SG Filter | Preprocessing Method (DHC Compound) |
|---|---|---|---|
| Red Tide (2019) | 350–938 | 360–928 | SNV + SG smoothing: 5th polynomial order; distance to right/left filter edge = 10 (HG, PD) |
| Red Tide (2021) | 350–1000 | 420–918 | SNV + SG smoothing: 5th polynomial order, distance to right/left filter edge = 20 (HG, PD) |
| Flame-NIR (2021) | 940–1664 | - | SNV |
| Red Tide + Flame-NIR (2021) | 400–1664 | - | SNV |
| PolyPen (2021) | 325–792 | 353–765 | SNV + SG 2nd derivative: 7th polynomial order; distance to right/left filter edge = 15 (HG) |
| 382–740 | SNV + SG 1st derivative: 7th polynomial order; distance to right/left filter edge = 30 (PD) |
| Model | DHC Compound | Calibration | Validation | Prediction | Overall Model | ||||
|---|---|---|---|---|---|---|---|---|---|
| Rc2 | RMSEC | Rv2 | RMSEV | Rp2 | RMSEP | Rtotal2 | RMSEtotal | ||
| Cultivar differentiation | HG | 0.919 | 0.569 | 0.998 | 0.099 | 0.998 | 0.116 | 0.941 | 0.496 |
| PD | 0.910 | 0.340 | 0.893 | 0.344 | 0.910 | 0.340 | 0.921 | 0.305 | |
| Measurement conditions | HG | 1.000 | 0.007 | 0.239 | 0.523 | 0.253 | 0.549 | 0.816 | 0.273 |
| PD | 0.861 | 0.261 | 0.762 | 0.297 | 0.935 | 0.196 | 0.856 | 0.261 | |
| Red Tide 2021 | HG | 0.959 | 0.028 | 0.276 | 0.101 | 0.105 | 0.121 | 0.804 | 0.059 |
| PD | 0.989 | 0.029 | 0.114 | 0.221 | 0.006 | 0.274 | 0.767 | 0.128 | |
| Flame-NIR 2021 | HG | 0.989 | 0.014 | 0.087 | 0.124 | 0.236 | 0.127 | 0.752 | 0.066 |
| PD | 1.000 | 0.001 | 0.024 | 0.229 | 0.115 | 0.230 | 0.802 | 0.118 | |
| Red Tide + Flame-NIR | HG | 1.000 | 0.001 | 0.173 | 0.129 | 0.118 | 0.134 | 0.753 | 0.066 |
| PD | 0.998 | 0.012 | 0.076 | 0.219 | 0.230 | 0.225 | 0.820 | 0.112 | |
| PolyPen 2021 | HG | 0.991 | 0.015 | 0.863 | 0.062 | 0.422 | 0.096 | 0.889 | 0.049 |
| PD | 0.998 | 0.015 | 0.194 | 0.182 | 0.582 | 0.104 | 0.904 | 0.084 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Moll, M.D.; Kahlert, L.; Gross, E.; Schwarze, E.-C.; Blings, M.; Hillebrand, S.; Ley, J.; Kraska, T.; Pude, R. VIS-NIR Modeling of Hydrangenol and Phyllodulcin Contents in Tea-Hortensia (Hydrangea macrophylla subsp. serrata). Horticulturae 2022, 8, 264. https://doi.org/10.3390/horticulturae8030264
Moll MD, Kahlert L, Gross E, Schwarze E-C, Blings M, Hillebrand S, Ley J, Kraska T, Pude R. VIS-NIR Modeling of Hydrangenol and Phyllodulcin Contents in Tea-Hortensia (Hydrangea macrophylla subsp. serrata). Horticulturae. 2022; 8(3):264. https://doi.org/10.3390/horticulturae8030264
Chicago/Turabian StyleMoll, Marcel Dieter, Liane Kahlert, Egon Gross, Esther-Corinna Schwarze, Maria Blings, Silke Hillebrand, Jakob Ley, Thorsten Kraska, and Ralf Pude. 2022. "VIS-NIR Modeling of Hydrangenol and Phyllodulcin Contents in Tea-Hortensia (Hydrangea macrophylla subsp. serrata)" Horticulturae 8, no. 3: 264. https://doi.org/10.3390/horticulturae8030264
APA StyleMoll, M. D., Kahlert, L., Gross, E., Schwarze, E.-C., Blings, M., Hillebrand, S., Ley, J., Kraska, T., & Pude, R. (2022). VIS-NIR Modeling of Hydrangenol and Phyllodulcin Contents in Tea-Hortensia (Hydrangea macrophylla subsp. serrata). Horticulturae, 8(3), 264. https://doi.org/10.3390/horticulturae8030264







