1. Introduction
Dendrobium officinale, a perennial herb of the genus
Dendrobium belonging to the family Orchidaceae, possesses abundant polysaccharides, alkaloids, phenols, and amino acids, which confer it with a high medicinal value [
1]. With improved living standards and healthcare awareness of people, the medicinal value of
D.
officinale is increasingly being recognized [
2]. However, with the market supply exceeding the demand, wild resources are gradually being depleted because of over-harvesting [
3], which has led breeders to develop synthetic varieties. Artificially produced
D.
officinale is poor quality and contains lower amounts of medicinal compounds than wild
D.
officinale. Therefore, attempts should be made to improve the content of medicinal ingredients of artificially produced
D.
officinale [
4].
Polyploids are organisms with three or more complete sets of chromosomes in their somatic cells [
5]. The increased number of chromosomes, additional genomic interactions, and genetic alterations affect epigenetic changes in various phenotypes and regulate gene expression [
6], resulting in polyploid plants that differ from diploid plants in their traits [
7]. Polyploid plants are typically characterized by enlarged organs [
8], greater resistance to stress [
9], increased secondary plant metabolites [
10], and improved utilization of beneficial components [
11]. Therefore, the yield of metabolites in plants can be increased by polyploidization and increasing the level of target compounds [
12,
13]. Various polyploidy induction pathways have been reported [
14], and chemical methods are among the most widely used mutagenesis techniques [
15]. Chemical mutagenesis is advantageous because it does not majorly change the plant genome, resulting in stable and heritable mutations. Colchicine and herbicides are the commonly used chemical mutagens [
16]. The main mechanism of action of chemical mutagens involves the inhibition of spindle formation during cell division to prevent the replicated chromosomes from being pulled toward the poles and inhibit the division of cells into two daughter cells. This leads to the doubling of chromosomes to form polyploid cells, eventually resulting in polyploid plants. Thus, polyploidy can be achieved by interfering with meiosis to allow for doubling of chromosomes within the cells by using chemical mutagens such as colchicine.
Diverse induction materials are used in polyploid breeding, and exophytes are available from various sources. Plant parts, such as seeds, stem segments, and clumped shoots, can be used as induction materials for polyploidization. Through different mutagenesis methods, the mutagenization rate can be increased by using different combinations of the mutagen concentration and induction time. While studying polyploidization induction of
D.
officinale, Zhang [
17] performed the induction of three types of explants, namely seed embryos, protocorms, and stem segments, by using the immersion method and medium addition method, and found that the effects of different induction methods using different explant materials varied greatly. Zhang et al. [
18] used different concentrations of colchicine and different treatment times to soak
D.
officinale protocorms and cluster buds, and reported that soaking the explants in 0.09% colchicine for 24 h was the most optimal condition, resulting in an induction rate of 48%. Wu [
19] used the medium addition method for the induction of protocorms and clumped shoots and reported that protocorms were more sensitive to colchicine than clumped shoots. The artificial induction of polyploids in different orchids is also particularly important [
20]. Induced polyploids in the genus Cardoso et al. [
21] can be used to obtain plants of compact size with increased floral longevity, higher number of flowers in bloom, and flowers with higher stiffness and more resistance to transport. Vilcherrez et al. [
22] studied different types of
Cattleya hybrida explants subjected to different exposure times and colchicine concentrations, and reported that the maximum number of polyploid plants were obtained from primary bulbs treated with 500–750 μM of colchicine for 18 h. In another study, most tetraploids were produced by treating the seeds of
Taraxacum kok-saghyz [
23] with 0.1% colchicine for 48 h after germination. The polyploid induction rate of the adventitious shoot segments of “Fengtou” ginger could reach 18% after treatment with colchicine at a concentration of 150 mg/L for 7 days [
24]. The highest tetraploid induction rate of 11.53% was achieved when the germinating seeds of
Trachyspermum ammi L. [
25] were treated with 0.05% colchicine for 24 h. As per previous studies of polyploidization, the criteria for determining plant variation differ based on plant morphological differences, plant stomatal differences, and chromosomal numbers [
17,
18,
19]. However, different determination criteria have yielded different rates of polyploidy, which necessitates extensive studies in the field. In this study, the variation rate and induction rate were determined on the basis of the stomatal differences of the treated plants and the chromosome doubling number, respectively.
In this study, different combinations of colchicine concentration and induction time were used to induce polyploidy in D. officinale, and materials from the proto-bulb period were used as explants. We determined the optimum treatment combinations for obtaining the highest mutation rate and the highest polyploid induction rate of D. officinale, and obtained the germplasm of tetraploid D. officinale plants. Furthermore, we obtained the karyotype data of diploid and tetraploid D. officinale plants by using the conventional squash karyotype analysis to verify their doubling characteristics. This study provides technical support and germplasm resources for creating polyploid germplasm of D. officinale and further plant breeding research.
2. Materials and Methods
2.1. Plant Material
The materials in this study were obtained from the capsules of artificially pollinated D. officinale, which were planted at the College of Horticulture, Hebei Agricultural University.
2.2. Preparation of D. officinale Protocorms
The capsules were rinsed under running tap water for 30 min and then disinfected in 0.1% HgCl2 for 15 min in an ultra-clean bench; they were rinsed again in sterile water and kept sterile. In the ultra-clean bench, the pods were cut open, and the seeds were sown into 1/2 MS + 0.1 mg·L−1 NAA + 30 g·L−1 sucrose + 7 g·L−1 agar (PH 5.8) medium. The seeds were spread evenly on the surface of the medium with an applicator stick and were allowed to germinate and expand in orchid culture bottles for almost 40 days to obtain D. officinale protocorms. The environmental conditions for tissue culture were: temperature (25 ± 2) °C, light intensity at 46 μmol·s−1·m−2, a photoperiod of 12 h.
2.3. Colchicine Configuration
Of note, 200 mg of colchicine (XiaSi Corporation, Beijing, China) was placed in a beaker and dissolved by adding a few drops of ethanol. After complete dissolution, a mother liquor was prepared at a concentration of 10%; it was filtered and sterilized using a disposable syringe and a sterile filter membrane with a pore size of 0.22 μm in an ultra-clean bench. Then, the mother liquor was aspirated into a sterile bottle and diluted with sterile water to prepare 0.1%, 0.2%, 0.3%, and 0.4% colchicine solutions (this concentration was determined by referring to the treatment concentration of previous studies [
14,
15,
16]). The whole process was performed under aseptic conditions. The prepared solutions were stored in the dark.
2.4. Induction of Protocorms of D. officinale
The protocorms in good growth condition, uniform size, tender green color, and without differentiation were soaked in colchicine at the concentrations of 0.1%, 0.2%, 0.3%, and 0.4% for 24 h, 36 h, and 48 h, respectively. The soaked protocorms were protected from light and placed on a shaker to ensure that the material was in full contact with the colchicine solution. The plants were then repeatedly rinsed three to four times with sterile water on an ultra-clean bench, and the water was blotted out with sterile filter paper. The plants were then transferred to MS + 0.2 mg·L−1 NAA + 1.5 mg·L−1 6-BA + 20 g·L−1 sucrose + 20% potato juice + 7 g·L−1 agar (PH 5.8) medium. The untreated plants were used as the control; a total of 12 treatment combinations were used, the number of protocorms in each treatment was 300, which were divided into three replicates. The growth status was observed weekly.
2.5. Morphological Observations
Eleven months after the colchicine treatment of protocorms, in the ultra-clean bench, 30 healthy and equal-sized plants were randomly selected each from the treatment groups and the control group. The morphological features, including plant height, internode length, stem diameter, and leaf shape index, were determined using vernier calipers, and the data were recorded.
2.6. Stomatal Observation
Stomatal measurements were performed on all of the selected plants, and the specific procedure for stomatal observations was as follows:
The leaves of sterile phytopathogenic seedlings of the treatment and control groups were collected, and a slide with a drop of distilled water in the middle was taken. About 3 mm
2 of the lower epidermis of the leaves of the treated and control plants was torn with pointed forceps and placed immediately in distilled water on the slide to prevent the stomata from losing water or closing. The coverslip was held on one side with forceps and gently lowered on the distilled water side to prevent the formation of air bubbles. Excess distilled water was aspirated, and the coverslip was pressed on the slide. The slides were observed using an OLYMPUS BX51 microscope (Olympus corporation, Tokyo, JAPAN). The size of guard cells was observed and recorded at 40 ×, and the number of stomata in the fixed field of view was counted. A study by Fang et al. [
26] on the induction of polyploidy in
Salvia miltiorrhiza Bge. reported that the plants were considered mutant if the stomatal length diameter of the treated group was ≥1.25 times that of the control group.
2.7. Chromosomal Observation
The specific procedures for chromosome observation are as follows:
Samples were obtained at around 10:00 a.m. (the time of peak division in the root tip meristem zone of D. officinale was determined by performing multiple sampling tests), and young root tips of 1 cm were cut from the treated and control plants. The root tips were immersed in 0.002 mol·L−1 of 8-hydroxyquinoline solution and treated at 15–20 °C for about 6 h to obtain more cells in the dividing phase. The pretreated root tips were removed, rinsed thrice with distilled water, immersed in Carnoy’s fixative (anhydrous ethanol–glacial acetic acid at a ratio of 3:1, ready to use), and treated for 24 h at 4 °C. The roots should be dissociated immediately or stored temporarily in 70% alcohol for later use. This kills the cells quickly and leaves the cell structure intact. The fixed root tips were removed, rinsed in 90% alcohol, and placed in 1 mol·L−1 of HCl for 4 min at 60 °C. This removes the pectin layer between the cells and weakens the cell wall. The dissociated root tips were washed thrice with distilled water, and the white phloem portion of the root tip was cut with a knife, pounded with knife forceps, and stained with modified phenol magenta for about 20 min at room temperature. One side of the coverslip was dipped in the staining solution, excess staining solution was removed with filter paper, and the coverslip was pressed with rubber to disperse the cells in the hyphal zone. The slide was observed using an OLYMPUS BX51 microscope, the number of chromosomes was counted, and the photographic images were obtained.
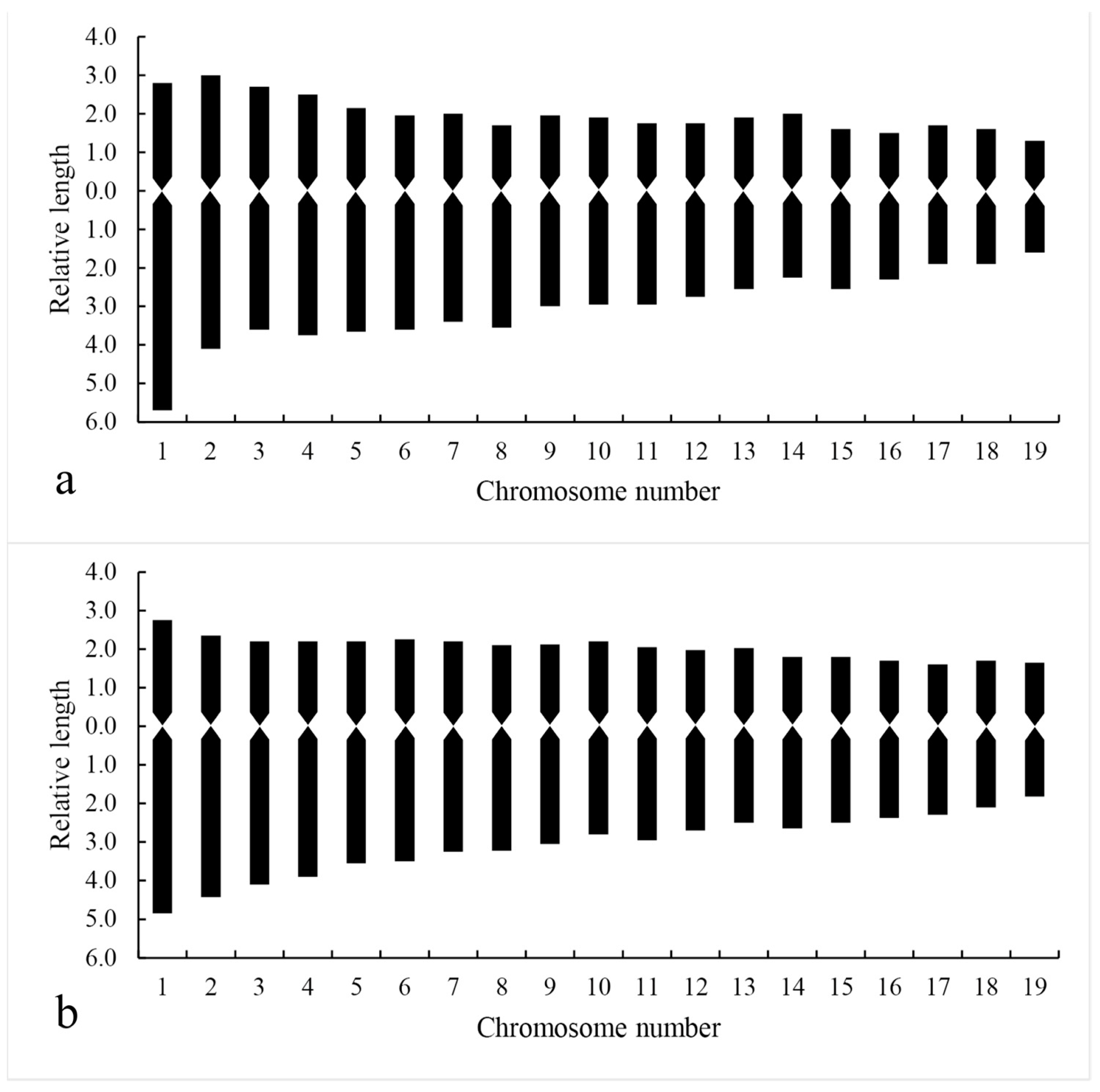
2.8. Chromosome Karyotype Analysis
Karyotypes of diploid and tetraploid
D.
officinale were observed and analyzed according to the features of the microscope system camera. Cells at mid-mitosis with well-dispersed chromosomes and minor overlap in the pressed slides were selected. Chromosomes were paired and measured using Adobe Photoshop software, and the chromosome data were analyzed by Excel software. Then, five cells from each material were taken to obtain the karyotype information, and the average value was taken as the karyotype analysis parameter. The criteria proposed by Li [
27] were used for analysis, the classification system proposed by Stebbins was used for the karyotype classification [
28], and the karyotype asymmetry coefficient was calculated by referring to the method given by Arano [
29].