Lithium Iron Phosphate Battery Regeneration and Recycling: Techniques and Efficiency
Abstract
:1. Introduction
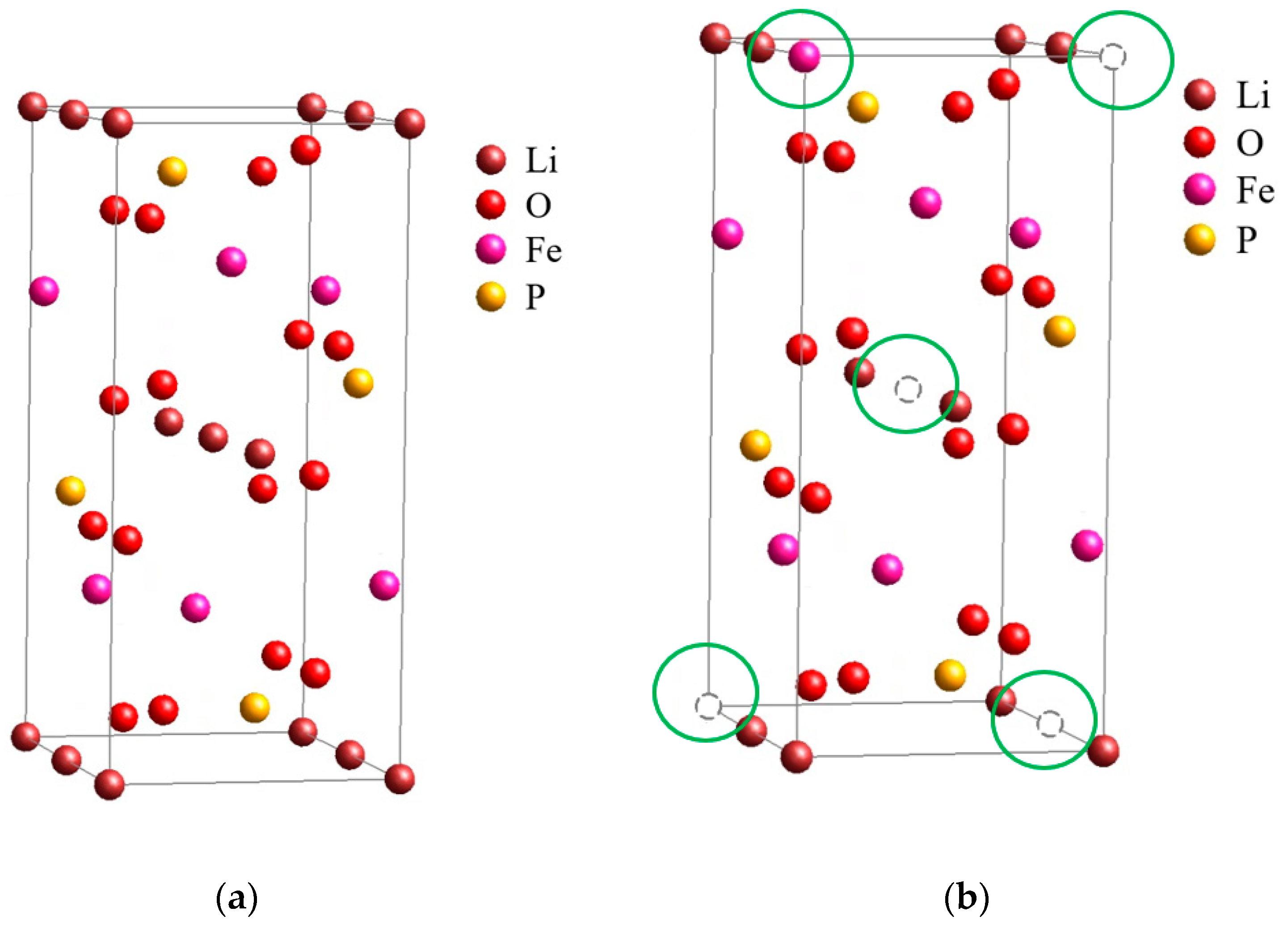
2. Lithium-Ion Battery, LFP Structure and Its Degradation
- Positive electrode: Typically made from lithium oxides, such as lithium cobalt oxide (LiCoO2), lithium iron phosphate (LiFePO4), lithium manganese oxide (LiMn2O4), lithium nickel cobalt aluminum oxide (NCA, LiNiCoAlO2), or lithium nickel manganese cobalt oxide (NCM or NMC, LiNiMnCoO2). During charging, the positive electrode releases lithium ions, which it then reabsorbs during discharge.
- Negative electrode: Usually made from graphite or other carbon-based materials, except in lithium-titanate (LTO) batteries. During discharge, lithium ions move from the negative electrode to the positive electrode.
- Electrolyte: A substance containing lithium ions that facilitates ion transfer between the negative electrode and positive electrode. It is typically an organic solvent, such as a lithium salt dissolved in carbonates.
- Separator: A thin porous material positioned between the negative electrode and positive electrode. It prevents electrode contact and short-circuiting while allowing lithium ions to pass through. Separator materials: polyethylene (PE) and polypropylene (PP) are the most common polymer materials for the manufacture of separators. Multilayer structures are often used, such as PP/PE/PP. Cellulose, polyamide, ceramic coatings, or gel polymers are found as alternatives, less frequently in mass production, but are in demand in specialized solutions (for example, to increase thermal stability or improve electrolyte properties).
- Current collectors in lithium-ion batteries (Li-ion) are important components that ensure the collection and transfer of charge from the material of the active electrode to the external circuit. Current collectors are usually made of metals that have high permeability, corrosion resistance, and chemical stability in the electrolyte environment. In lithium-iron-phosphate (LFP) batteries, as in most other lithium-ion systems, two basic metals are usually used as current collectors (metal foil serving as a “base” for the active material). Aluminum (Al) is used for the positive electrode (in this case, it is a layer of LiFePO4 active material), and copper (Cu) for the negative electrode (most often graphite-based).
Degradation Aspects and Mechanisms of LFP Battery Failure
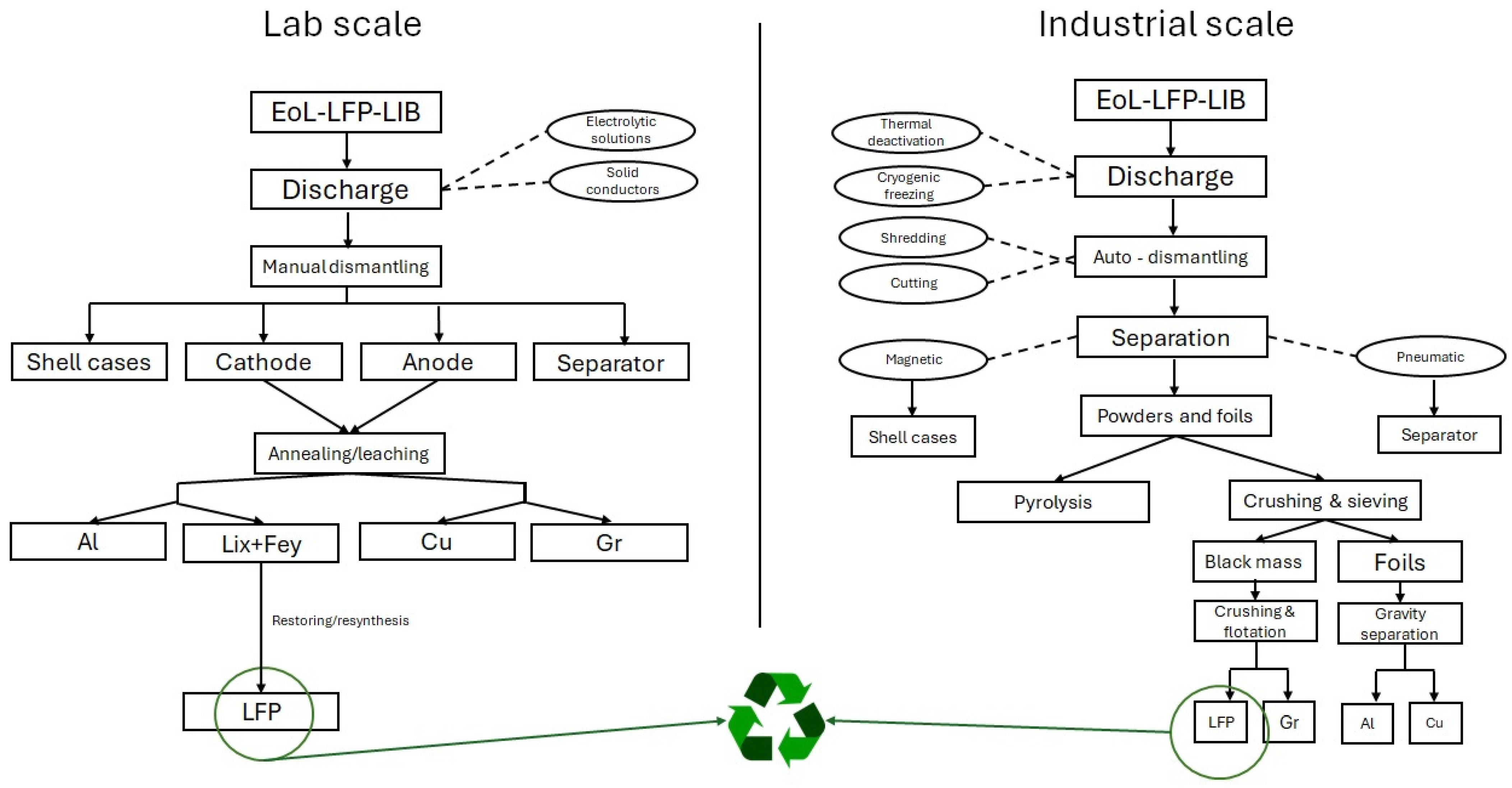
3. Pretreatment
4. Direct Regeneration
4.1. Solid-State Methods
4.2. Liquid-State Methods
4.3. Electrochemical Methods
5. Recycling
5.1. Material Recovery Methods
5.1.1. Hydrometallurgy
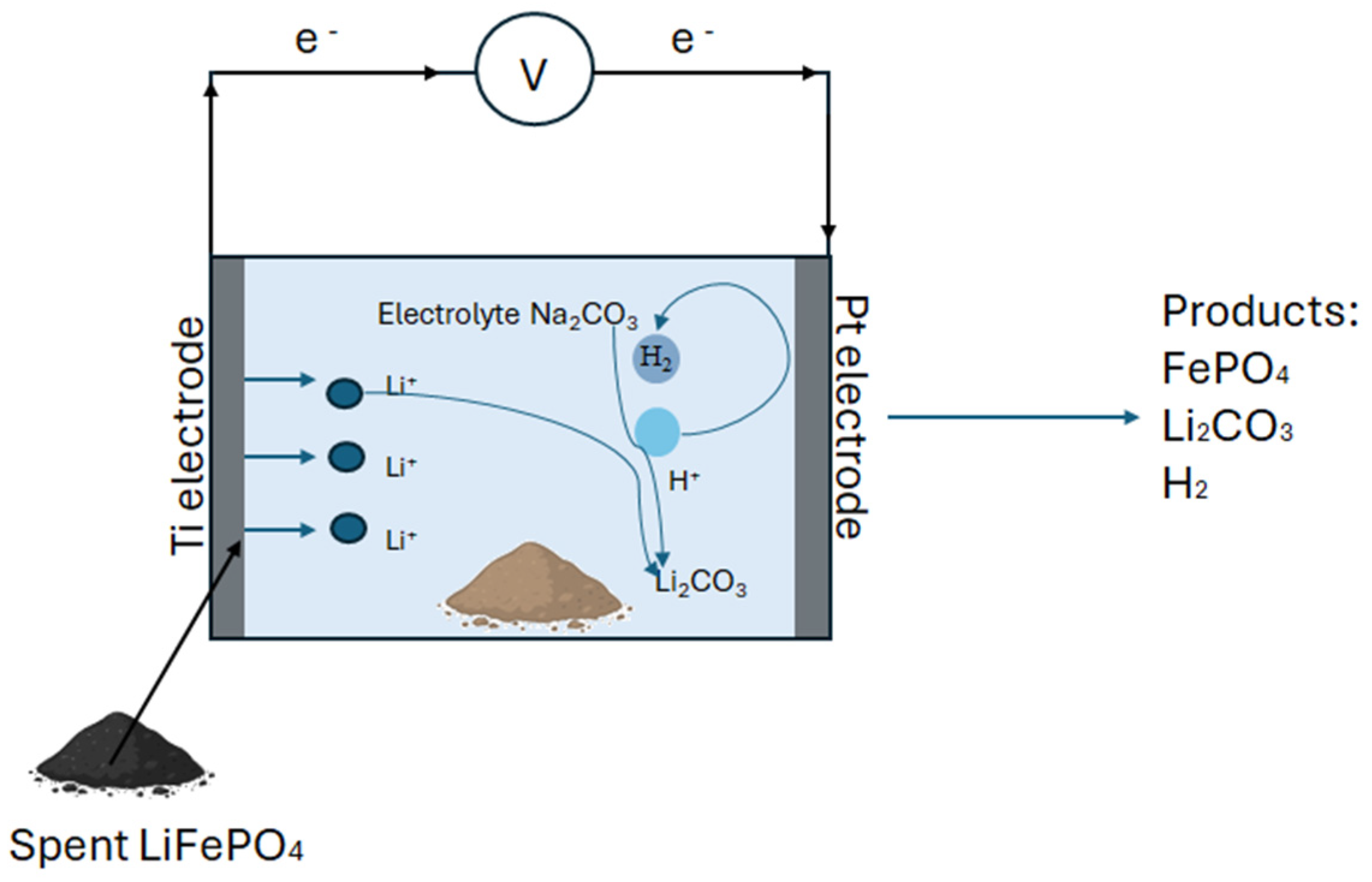
5.1.2. Electrochemical Approach
5.1.3. Pyrometallurgy
5.2. LFP Resynthesis Methods
5.2.1. Pyrometallurgy
5.2.2. Hydrometallurgy
5.2.3. Solvometallurgy
- High Performance and Stability:
- 2.
- Eco-Friendliness and Sustainability:
- 3.
- Innovative Techniques:
- Lower Initial Capacities:
- 2.
- Complexity and Refinement Needs:
- 3.
- Process Specificity and Optimization:
6. Conclusions and Perspectives
6.1. Perspectives
6.1.1. Optimization of Closed-Loop Recycling
6.1.2. Integration of Automation and AI
6.1.3. Development of Green Chemistry Solutions
6.1.4. Economic Feasibility and Policy Support
6.1.5. Advanced Material Design
Author Contributions
Funding
Conflicts of Interest
References
- Rogelj, J.; Den Elzen, M.; Höhne, N.; Fransen, T.; Fekete, H.; Winkler, H.; Schaeffer, R.; Sha, F.; Riahi, K.; Meinshausen, M. Paris Agreement Climate Proposals Need a Boost to Keep Warming Well below 2 °C. Nature 2016, 534, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Martins, F.; Felgueiras, C.; Smitkova, M.; Caetano, N. Analysis of Fossil Fuel Energy Consumption and Environmental Impacts in European Countries. Energies 2019, 12, 964. [Google Scholar] [CrossRef]
- Lieskoski, S.; Koskinen, O.; Tuuf, J.; Björklund-Sänkiaho, M. A Review of the Current Status of Energy Storage in Finland and Future Development Prospects. J. Energy Storage 2024, 93, 112327. [Google Scholar] [CrossRef]
- Zou, Y.; Lin, Z.; Li, D.; Liu, Z. Advancements in Artificial Neural Networks for Health Management of Energy Storage Lithium-Ion Batteries: A Comprehensive Review. J. Energy Storage 2023, 73, 109069. [Google Scholar] [CrossRef]
- Shareef, H.; Islam, M.M.; Mohamed, A. A Review of the Stage-of-the-Art Charging Technologies, Placement Methodologies, and Impacts of Electric Vehicles. Renew. Sustain. Energy Rev. 2016, 64, 403–420. [Google Scholar] [CrossRef]
- Choudhari, T.P.; Illahi, U.; Al-Hosni, M.; Caulfield, B.; O’Mahony, M. Decarbonising Shared Mobility: The Potential of Shared Electric Vehicles. Transp. Res. Part. D Transp. Environ. 2024, 133, 104313. [Google Scholar] [CrossRef]
- Available online: https://greenstartpoint.ru/tesla-energy-perehodit-na-novye-akb/?ysclid=m2x7xe4vxm563778359 (accessed on 15 November 2024).
- Wu, X.; Ma, J.; Wang, J.; Zhang, X.; Zhou, G.; Liang, Z. Progress, Key Issues, and Future Prospects for Li-Ion Battery Recycling. Glob. Chall. 2022, 6, 2200067. [Google Scholar] [CrossRef]
- Meshram, P.; Mishra, A.; Abhilash; Sahu, R. Environmental Impact of Spent Lithium Ion Batteries and Green Recycling Perspectives by Organic Acids—A Review. Chemosphere 2020, 242, 125291. [Google Scholar] [CrossRef]
- LFP Battery Is Returning, Driving Chinese LFP Battery Manufacturers Going Overseas. Available online: http://www.iccsino.com/news/show-htm-itemid-16446.html (accessed on 15 November 2024).
- Miao, Y.; Liu, L.; Zhang, Y.; Tan, Q.; Li, J. An Overview of Global Power Lithium-Ion Batteries and Associated Critical Metal Recycling. J. Hazard. Mater. 2022, 425, 127900. [Google Scholar] [CrossRef]
- Rkhis, M.; Nabil, N. The Art of Strain Engineering: Fine-Tuning the Electrochemical Performance of Olivine Ferrophosphate LiFePO4 as Positive electrode Materials for Li-Ion Batteries. J. Energy Storage 2024, 97, 112815. [Google Scholar] [CrossRef]
- Zhang, B.; Xu, Y.; Silvester, D.S.; Banks, C.E.; Deng, W.; Zou, G.; Hou, H.; Ji, X. Direct Regeneration of Positive electrode Materials in Spent Lithium-Ion Batteries toward Closed-Loop Recycling and Sustainability. J. Power Sources 2024, 589, 233728. [Google Scholar] [CrossRef]
- Fang, J.; Ding, Z.; Ling, Y.; Li, J.; Zhuge, X.; Luo, Z.; Ren, Y.; Luo, K. Green Recycling and Regeneration of LiNi0.5Co0.2Mn0.3O2 from Spent Lithium-Ion Batteries Assisted by Sodium Sulfate Electrolysis. Chem. Eng. J. 2022, 440, 135880. [Google Scholar] [CrossRef]
- Aannir, M.; Hakkou, R.; Levard, C.; Taha, Y.; Ghennioui, A.; Rose, J.; Saadoune, I. Towards a Closed Loop Recycling Process of End-of-Life Lithium-Ion Batteries: Recovery of Critical Metals and Electrochemical Performance Evaluation of a Regenerated LiCoO2. J. Power Sources 2023, 580, 233341. [Google Scholar] [CrossRef]
- Yoshino, A. The Birth of the Lithium-Ion Battery. Angew. Chem. Int. Ed. 2012, 51, 5798–5800. [Google Scholar] [CrossRef]
- Scrosati, B.; Garche, J. Lithium Batteries: Status, Prospects and Future. J. Power Sources 2010, 195, 2419–2430. [Google Scholar] [CrossRef]
- Jorgensen, A.B.; Mu, A.G.; Qin, T.F. Recycling of Lithium Iron Phosphate Batteries: Challenges and Opportunities. J. Sustain. Mater. Technol. 2020, 15, 1–10. [Google Scholar]
- Lee, M.Y.; Yoo, J.K.; Kang, H.S. Economic and Environmental Assessment of Recycling Strategies for Lithium-Ion Batteries. Resour. Conserv. Recycl. 2021, 174, 105775. [Google Scholar]
- Sun, F.; Wang, J.; Zhang, G. Recycling Processes for Lithium-Ion Batteries: A Review of Recent Advances and Future Prospects. Chem. Eng. J. 2021, 419, 130019. [Google Scholar]
- Xu, C.; Dai, Y.; Zhang, X. A Review of the Current Progress and Challenges in Recycling Lithium-Ion Batteries. Green Energy Environ. 2022, 7, 10–30. [Google Scholar]
- Ohzuku, T.; Brodd, R.J. An Overview of Positive-Electrode Materials for Advanced Lithium-Ion Batteries. J. Power Sources 2007, 174, 449–456. [Google Scholar] [CrossRef]
- Padhi, A.K.; Nanjundaswamy, K.S.; Goodenough, J.B. Phospho-olivines as Positive-Electrode Materials for Rechargeable Lithium Batteries. J. Electrochem. Soc. 1997, 144, 1188–1194. [Google Scholar] [CrossRef]
- Yamada, A.; Hosoya, M.; Chung, S.-C.; Kudo, Y.; Hinokuma, K.; Liu, K.-Y.; Nishi, Y. Olivine-Type Positive electrodes. J. Power Sources 2003, 119–121, 232–238. [Google Scholar] [CrossRef]
- Amine, K. Olivine LiCoPO4 as 4.8 V Electrode Material for Lithium Batteries. Electrochem. Solid-State Lett. 1999, 3, 178. [Google Scholar] [CrossRef]
- Chen, M.; Ma, X.; Chen, B.; Arsenault, R.; Karlson, P.; Simon, N.; Wang, Y. Recycling End-of-Life Electric Vehicle Lithium-Ion Batteries. Joule 2019, 3, 2622–2646. [Google Scholar] [CrossRef]
- Park, K.-Y.; Park, I.; Kim, H.; Lim, H.; Hong, J.; Kim, J.; Kang, K. Anti-Site Reordering in LiFePO4: Defect Annihilation on Charge Carrier Injection. Chem. Mater. 2014, 26, 5345–5351. [Google Scholar] [CrossRef]
- Zheng, H.; Tan, L.; Zhang, L.; Qu, Q.; Wan, Z.; Wang, Y.; Shen, M.; Zheng, H. Correlation between Lithium Deposition on Graphite Electrode and the Capacity Loss for LiFePO4/Graphite Cells. Electrochim. Acta 2015, 173, 323–330. [Google Scholar] [CrossRef]
- Zhang, H.; Yang, Y.; Ren, D.; Wang, L.; He, X. Graphite as Negative electrode Materials: Fundamental Mechanism, Recent Progress and Advances. Energy Storage Mater. 2021, 36, 147–170. [Google Scholar] [CrossRef]
- Zinth, V.; Von Lüders, C.; Hofmann, M.; Hattendorff, J.; Buchberger, I.; Erhard, S.; Rebelo-Kornmeier, J.; Jossen, A.; Gilles, R. Lithium Plating in Lithium-Ion Batteries at Sub-Ambient Temperatures Investigated by in Situ Neutron Diffraction. J. Power Sources 2014, 271, 152–159. [Google Scholar] [CrossRef]
- Zhang, L.; Zhang, Y.; Xu, Z.; Zhu, P. The Foreseeable Future of Spent Lithium-Ion Batteries: Advanced Upcycling for Toxic Electrolyte, Positive electrode, and Negative electrode from Environmental and Technological Perspectives. Environ. Sci. Technol. 2023, 57, 13270–13291. [Google Scholar] [CrossRef]
- Piątek, J.; Afyon, S.; Budnyak, T.M.; Budnyk, S.; Sipponen, M.H.; Slabon, A. Sustainable Li-Ion Batteries: Chemistry and Recycling. Adv. Energy Mater. 2021, 11, 2003456. [Google Scholar] [CrossRef]
- Zhang, J.; Zou, J.; He, D.; Hu, W.; Peng, D.; Li, Y.; Zhao, Z.; Wang, S.; Li, P.; Su, S.; et al. Molten Salt Infiltration–Oxidation Synergistic Controlled Lithium Extraction from Spent Lithium Iron Phosphate Batteries: An Efficient, Acid Free, and Closed-Loop Strategy. Green Chem. 2023, 25, 6057–6066. [Google Scholar] [CrossRef]
- Zeng, Z.; Xu, P.; Li, J.; Yi, C.; Zhao, W.; Sun, W.; Ji, X.; Yang, Y.; Ge, P. Large-Scale and Homogenized Strategies of Spent LiFePO4 Recycling: Reconstruction of Targeted Lattice. Adv. Funct. Mater. 2024, 34, 2308671. [Google Scholar] [CrossRef]
- Liu, K.; Wang, J.; Wang, M.; Zhang, Q.; Cao, Y.; Huang, L.; Valix, M.; Tsang, D.C.W. Low-Carbon Recycling of Spent Lithium Iron Phosphate Batteries via a Hydro-Oxygen Repair Route. Green Chem. 2023, 25, 6642–6651. [Google Scholar] [CrossRef]
- Kim, S.; Bang, J.; Yoo, J.; Shin, Y.; Bae, J.; Jeong, J.; Kim, K.; Dong, P.; Kwon, K. A Comprehensive Review on the Pretreatment Process in Lithium-Ion Battery Recycling. J. Clean. Prod. 2021, 294, 126329. [Google Scholar] [CrossRef]
- Lv, W.; Wang, Z.; Cao, H.; Sun, Y.; Zhang, Y.; Sun, Z. A Critical Review and Analysis on the Recycling of Spent Lithium-Ion Batteries. ACS Sustain. Chem. Eng. 2018, 6, 1504–1521. [Google Scholar] [CrossRef]
- Yu, D.; Huang, Z.; Makuza, B.; Guo, X.; Tian, Q. Pretreatment Options for the Recycling of Spent Lithium-Ion Batteries: A Comprehensive Review. Miner. Eng. 2021, 173, 107218. [Google Scholar] [CrossRef]
- Zhang, G.; Yuan, X.; He, Y.; Wang, H.; Zhang, T.; Xie, W. Recent Advances in Pretreating Technology for Recycling Valuable Metals from Spent Lithium-Ion Batteries. J. Hazard. Mater. 2021, 406, 124332. [Google Scholar] [CrossRef]
- Li, L.; Qu, W.; Zhang, X.; Lu, J.; Chen, R.; Wu, F.; Amine, K. Succinic Acid-Based Leaching System: A Sustainable Process for Recovery of Valuable Metals from Spent Li-Ion Batteries. J. Power Sources 2015, 282, 544–551. [Google Scholar] [CrossRef]
- Wu, J.; Zheng, M.; Liu, T.; Wang, Y.; Liu, Y.; Nai, J.; Zhang, L.; Zhang, S.; Tao, X. Direct Recovery: A Sustainable Recycling Technology for Spent Lithium-Ion Battery. Energy Storage Mater. 2023, 54, 120–134. [Google Scholar] [CrossRef]
- Zhuang, L.; Sun, C.; Zhou, T.; Li, H.; Dai, A. Recovery of Valuable Metals from LiNi0.5Co0.2Mn0.3O2 Positive electrode Materials of Spent Li-Ion Batteries Using Mild Mixed Acid as Leachant. Waste Manag. 2019, 85, 175–185. [Google Scholar] [CrossRef]
- Makuza, B.; Tian, Q.; Guo, X.; Chattopadhyay, K.; Yu, D. Pyrometallurgical Options for Recycling Spent Lithium-Ion Batteries: A Comprehensive Review. J. Power Sources 2021, 491, 229622. [Google Scholar] [CrossRef]
- Brückner, L.; Frank, J.; Elwert, T. Industrial Recycling of Lithium-Ion Batteries—A Critical Review of Metallurgical Process Routes. Metals 2020, 10, 1107. [Google Scholar] [CrossRef]
- Zhao, Y.; Yuan, X.; Jiang, L.; Wen, J.; Wang, H.; Guan, R.; Zhang, J.; Zeng, G. Regeneration and Reutilization of Positive electrode Materials from Spent Lithium-Ion Batteries. Chem. Eng. J. 2020, 383, 123089. [Google Scholar] [CrossRef]
- Yao, L.P.; Zeng, Q.; Qi, T.; Li, J. An Environmentally Friendly Discharge Technology to Pretreat Spent Lithium-Ion Batteries. J. Clean. Prod. 2020, 245, 118820. [Google Scholar] [CrossRef]
- Ku, H.; Jung, Y.; Jo, M.; Park, S.; Kim, S.; Yang, D.; Rhee, K.; An, E.-M.; Sohn, J.; Kwon, K. Recycling of Spent Lithium-Ion Battery Positive electrode Materials by Ammoniacal Leaching. J. Hazard. Mater. 2016, 313, 138–146. [Google Scholar] [CrossRef]
- Xiao, J.; Guo, J.; Zhan, L.; Xu, Z. A Cleaner Approach to the Discharge Process of Spent Lithium Ion Batteries in Different Solutions. J. Clean. Prod. 2020, 255, 120064. [Google Scholar] [CrossRef]
- Ojanen, S.; Lundström, M.; Santasalo-Aarnio, A.; Serna-Guerrero, R. Challenging the Concept of Electrochemical Discharge Using Salt Solutions for Lithium-Ion Batteries Recycling. Waste Manag. 2018, 76, 242–249. [Google Scholar] [CrossRef]
- Wang, F.; Zhang, T.; He, Y.; Zhao, Y.; Wang, S.; Zhang, G.; Zhang, Y.; Feng, Y. Recovery of Valuable Materials from Spent Lithium-Ion Batteries by Mechanical Separation and Thermal Treatment. J. Clean. Prod. 2018, 185, 646–652. [Google Scholar] [CrossRef]
- Fang, Z.; Duan, Q.; Peng, Q.; Wei, Z.; Cao, H.; Sun, J.; Wang, Q. Comparative Study of Chemical Discharge Strategy to Pretreat Spent Lithium-Ion Batteries for Safe, Efficient, and Environmentally Friendly Recycling. J. Clean. Prod. 2022, 359, 132116. [Google Scholar] [CrossRef]
- Chen, X.; Hua, W.; Yuan, L.; Ji, S.; Wang, S.; Yan, S. Evolution Fate of Battery Chemistry during Efficient Discharging Processing of Spent Lithium-Ion Batteries. Waste Manag. 2023, 170, 278–286. [Google Scholar] [CrossRef]
- Yao, Y.; Zhu, M.; Zhao, Z.; Tong, B.; Fan, Y.; Hua, Z. Hydrometallurgical Processes for Recycling Spent Lithium-Ion Batteries: A Critical Review. ACS Sustain. Chem. Eng. 2018, 6, 13611–13627. [Google Scholar] [CrossRef]
- Lombardo, G.; Ebin, B.; Foreman, M.R.S.J.; Steenari, B.-M.; Petranikova, M. Incineration of EV Lithium-Ion Batteries as a Pretreatment for Recycling—Determination of the Potential Formation of Hazardous by-Products and Effects on Metal Compounds. J. Hazard. Mater. 2020, 393, 122372. [Google Scholar] [CrossRef]
- Liu, C.; Lin, J.; Cao, H.; Zhang, Y.; Sun, Z. Recycling of Spent Lithium-Ion Batteries in View of Lithium Recovery: A Critical Review. J. Clean. Prod. 2019, 228, 801–813. [Google Scholar] [CrossRef]
- Zhang, G.; He, Y.; Feng, Y.; Wang, H.; Zhang, T.; Xie, W.; Zhu, X. Enhancement in Liberation of Electrode Materials Derived from Spent Lithium-Ion Battery by Pyrolysis. J. Clean. Prod. 2018, 199, 62–68. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, C.; Feng, P.; Yang, X.; Yang, X. A Novel Pulsated Pneumatic Separation with Variable-Diameter Structure and Its Application in the Recycling Spent Lithium-Ion Batteries. Waste Manag. 2021, 131, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Zhan, R.; Oldenburg, Z.; Pan, L. Recovery of Active Positive electrode Materials from Lithium-Ion Batteries Using Froth Flotation. Sustain. Mater. Technol. 2018, 17, e00062. [Google Scholar] [CrossRef]
- Liang, Q.; Yue, H.; Wang, S.; Yang, S.; Lam, K.; Hou, X. Recycling and Crystal Regeneration of Commercial Used LiFePO4 Positive electrode Materials. Electrochim. Acta 2020, 330, 135323. [Google Scholar] [CrossRef]
- Sun, J.; Jiang, Z.; Jia, P.; Li, S.; Wang, W.; Song, Z.; Mao, Y.; Zhao, X.; Zhou, B. A Sustainable Revival Process for Defective LiFePO4 Positive electrodes through the Synergy of Defect-Targeted Healing and in-Situ Construction of 3D-Interconnected Porous Carbon Networks. Waste Manag. 2023, 158, 125–135. [Google Scholar] [CrossRef]
- Zhao, T.; Mahandra, H.; Marthi, R.; Ji, X.; Zhao, W.; Chae, S.; Traversy, M.; Li, W.; Yu, F.; Li, L.; et al. An Overview on the Life Cycle of Lithium Iron Phosphate: Synthesis, Modification, Application, and Recycling. Chem. Eng. J. 2024, 485, 149923. [Google Scholar] [CrossRef]
- Song, L.; Qi, C.; Wang, S.; Zhu, X.; Zhang, T.; Jin, Y.; Zhang, M. Direct Regeneration of Waste LiFePO4 Positive electrode Materials with a Solid-Phase Method Promoted by Activated CNTs. Waste Manag. 2023, 157, 141–148. [Google Scholar] [CrossRef]
- Yao, T.; Zhang, H.; Qi, C.; Ma, J.; Zhou, Z.; Sun, Q.; Song, L.; Jin, Y.; Zhang, M. Effective Regeneration of Waste LiFePO4 Positive electrode Material by Cu Doping Modification. Appl. Surf. Sci. 2024, 659, 159920. [Google Scholar] [CrossRef]
- Liu, P.; Zhang, Y.; Dong, P.; Zhang, Y.; Meng, Q.; Zhou, S.; Yang, X.; Zhang, M.; Yang, X. Direct Regeneration of Spent LiFePO4 Positive electrode Materials with Pre-Oxidation and V-Doping. J. Alloys Compd. 2021, 860, 157909. [Google Scholar] [CrossRef]
- Zou, Y.; Cao, J.; Li, H.; Wu, W.; Liang, Y.; Zhang, J. Large-Scale Direct Regeneration of LiFePO4@C Based on Spray Drying. Ind. Chem. Mater. 2023, 1, 254–261. [Google Scholar] [CrossRef]
- Wang, Z.; Xu, H.; Liu, Z.; Jin, M.; Deng, L.; Li, S.; Huang, Y. A Recrystallization Approach to Repairing Spent LiFePO4 Black Mass. J. Mater. Chem. A 2023, 11, 9057–9065. [Google Scholar] [CrossRef]
- Jiang, Z.; Sun, J.; Jia, P.; Wang, W.; Song, Z.; Zhao, X.; Mao, Y. A Sustainable Strategy for Spent Li-Ion Battery Regeneration: Microwave-Hydrothermal Relithiation Complemented with Negative electrode-Revived Graphene to Construct a LiFePO4/MWrGO Positive electrode Material. Sustain. Energy Fuels 2022, 6, 2207–2222. [Google Scholar] [CrossRef]
- Chen, B.; Liu, M.; Cao, S.; Hu, H.; Chen, G.; Guo, X.; Wang, X. Direct Regeneration and Performance of Spent LiFePO4 via a Green Efficient Hydrothermal Technique. J. Alloys Compd. 2022, 924, 166487. [Google Scholar] [CrossRef]
- Xu, Y.; Zhang, B.; Ge, Z.; Wang, H.; Hong, N.; Xiao, X.; Song, B.; Zhang, Y.; Tian, Y.; Deng, W.; et al. Direct Recovery of Degraded LiFePO4 Positive electrode via Mild Chemical Relithiation Strategy. Chem. Eng. J. 2023, 477, 147201. [Google Scholar] [CrossRef]
- Tang, D.; Ji, G.; Wang, J.; Liang, Z.; Chen, W.; Ji, H.; Ma, J.; Liu, S.; Zhuang, Z.; Zhou, G. A Multifunctional Amino Acid Enables Direct Recycling of Spent LiFePO4 Positive electrode Material. Adv. Mater. 2024, 36, 2309722. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, Z.; Zhang, J.; Chen, Y.; Wang, C. Economical and Low-Carbon Regeneration of Spent LiFePO4 Materials by Hydrothermal Relithiation. J. Alloys Compd. 2023, 947, 169660. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, J.; Zhang, H.; Wang, Y.; Chen, Y.; Wang, C. Simultaneous Anodic De-Lithiation/Cathodic Lithium-Embedded Regeneration Method for Recycling of Spent LiFePO4 Battery. Energy Storage Mater. 2024, 65, 103081. [Google Scholar] [CrossRef]
- Jia, K.; Ma, J.; Wang, J.; Liang, Z.; Ji, G.; Piao, Z.; Gao, R.; Zhu, Y.; Zhuang, Z.; Zhou, G.; et al. Long-Life Regenerated LiFePO4 from Spent Positive electrode by Elevating the d-Band Center of Fe. Adv. Mater. 2023, 35, 2208034. [Google Scholar] [CrossRef]
- Yang, J.; Zhou, K.; Gong, R.; Meng, Q.; Zhang, Y.; Dong, P. Direct Regeneration of Spent LiFePO4 Materials via a Green and Economical One-Step Hydrothermal Process. J. Environ. Manag. 2023, 348, 119384. [Google Scholar] [CrossRef]
- Fan, M.; Chang, X.; Meng, X.-H.; Gu, C.-F.; Zhang, C.-H.; Meng, Q.; Wan, L.-J.; Guo, Y.-G. Structural Restoration of Degraded LiFePO4 Positive electrode with Enhanced Kinetics Using Residual Lithium in Spent Graphite Negative electrodes. CCS Chem. 2023, 5, 1189–1201. [Google Scholar] [CrossRef]
- Zhou, S.; Du, J.; Xiong, X.; Liu, L.; Wang, J.; Fu, L.; Ye, J.; Chen, Y.; Wu, Y. Direct Recovery of Scrapped LiFePO4 by a Green and Low-Cost Electrochemical Re-Lithiation Method. Green Chem. 2022, 24, 6278–6286. [Google Scholar] [CrossRef]
- Liu, W.; Li, K.; Wang, W.; Hu, Y.; Ren, Z.; Zhou, Z. Selective Leaching of Lithium Ions from LiFePO4 Powders Using Hydrochloric Acid and Sodium Hypochlorite System. Can. J. Chem. Eng. 2023, 101, 1831–1841. [Google Scholar] [CrossRef]
- Li, X.; Benstead, M.; Peeters, N.; Binnemans, K. Recycling of Metals from LiFePO4 Battery Positive electrode Material by Using Ionic Liquid Based-Aqueous Biphasic Systems. RSC Adv. 2024, 14, 9262–9272. [Google Scholar] [CrossRef]
- Mahandra, H.; Ghahreman, A. A Sustainable Process for Selective Recovery of Lithium as Lithium Phosphate from Spent LiFePO4 Batteries. Resour. Conserv. Recycl. 2021, 175, 105883. [Google Scholar] [CrossRef]
- Jiang, Z.; Zhu, P.; Yang, Y.; Jin, W.; Zou, G.; Hou, H.; Hu, J.; Sun, W.; Ji, X. Sustainable Recovery of Lithium from Spent LiFePO4 via Proton Circulation. Chem. Eng. J. 2023, 466, 143186. [Google Scholar] [CrossRef]
- Dai, Y.; Xu, Z.; Hua, D.; Gu, H.; Wang, N. Theoretical-Molar Fe3+ Recovering Lithium from Spent LiFePO4 Batteries: An Acid-Free, Efficient, and Selective Process. J. Hazard. Mater. 2020, 396, 122707. [Google Scholar] [CrossRef]
- Yu, F.; Xu, X.; Guo, Y. Recovery of Metal Ions in Lithium Iron Phosphate Powder and Lithium Nickel-Cobalt-Manganate Powder by Electrochemical Oxidation. Sep. Purif. Technol. 2024, 344, 127134. [Google Scholar] [CrossRef]
- Zhao, J.; Zhou, F.; Wang, H.; Gao, S.; Wang, D.; Yin, H. Recovery of Lithium Iron Phosphate Batteries through Electrochemical Oxidation in Na2CO3 Solutions. J. Power Sources 2023, 582, 233562. [Google Scholar] [CrossRef]
- Li, R.; Li, Y.; Dong, L.; Yang, Q.; Tian, S.; Ren, Z.; Zhou, Z. Study on Selective Recovery of Lithium Ions from Lithium Iron Phosphate Powder by Electrochemical Method. Sep. Purif. Technol. 2023, 310, 123133. [Google Scholar] [CrossRef]
- Zhang, B.; Qu, X.; Chen, X.; Liu, D.; Zhao, Z.; Xie, H.; Wang, D.; Yin, H. A Sodium Salt-Assisted Roasting Approach Followed by Leaching for Recovering Spent LiFePO4 Batteries. J. Hazard. Mater. 2022, 424, 127586. [Google Scholar] [CrossRef]
- Qu, X.; Ma, J.; Zhang, B.; Zhao, J.; Qiu, B.; Chen, X.; Zhou, F.; Li, X.; Gao, S.; Wang, D.; et al. Fast Ammonium Sulfate Salt Assisted Roasting for Selectively Recycling Degraded LiFePO4 Positive electrode. J. Clean. Prod. 2024, 435, 140428. [Google Scholar] [CrossRef]
- Bian, D.; Sun, Y.; Li, S.; Tian, Y.; Yang, Z.; Fan, X.; Zhang, W. A Novel Process to Recycle Spent LiFePO4 for Synthesizing LiFePO4/C Hierarchical Microflowers. Electrochim. Acta 2016, 190, 134–140. [Google Scholar] [CrossRef]
- Wen, G.; Yuan, S.; Dong, Z.; Gao, P.; Ding, H.; Lei, S.; Liu, Q. Mechanism and Process Study of Spent Lithium Iron Phosphate Batteries by Medium-Temperature Oxidation Roasting Strategy. Sep. Purif. Technol. 2025, 356, 129987. [Google Scholar] [CrossRef]
- Zhang, L.; Teng, T.; Xiao, L.; Shen, L.; Ran, J.; Zheng, J.; Zhu, Y.; Chen, H. Recovery of LiFePO4 from Used Lithium-Ion Batteries by Sodium-Bisulphate-Assisted Roasting. J. Clean. Prod. 2022, 379, 134748. [Google Scholar] [CrossRef]
- Yang, L.; Wang, D.; Zhang, J.; Chen, Y.; Wang, C. An Economical and Closed-Loop Hydrometallurgical Method to Prepare Battery-Grade Iron Phosphate from Delithiated LiFePO4 Positive electrode Scrap. J. Clean. Prod. 2024, 444, 141194. [Google Scholar] [CrossRef]
- Zhou, H.; Luo, Z.; Wang, S.; Ma, X.; Cao, Z. A Mild Closed-Loop Process for Lithium–Iron Separation and Positive electrode Materials Regeneration from Spent LiFePO4 Batteries. Sep. Purif. Technol. 2023, 315, 123742. [Google Scholar] [CrossRef]
- Li, P.; Luo, S.; Zhang, L.; Wang, Y.; Zhang, H.; Wang, J.; Yan, S.; Hou, P.; Wang, Q.; Zhang, Y.; et al. Study on Efficient and Synergistic Leaching of Valuable Metals from Spent Lithium Iron Phosphate Using the Phosphoric Acid-Oxalic Acid System. Sep. Purif. Technol. 2022, 303, 122247. [Google Scholar] [CrossRef]
- Yadav, P.; Jie, C.J.; Tan, S.; Srinivasan, M. Recycling of Positive electrode from Spent Lithium Iron Phosphate Batteries. J. Hazard. Mater. 2020, 399, 123068. [Google Scholar] [CrossRef]
- Kumar, J.; Shen, X.; Li, B.; Liu, H.; Zhao, J. Selective Recovery of Li and FePO4 from Spent LiFePO4 Positive electrode Scraps by Organic Acids and the Properties of the Regenerated LiFePO4. Waste Manag. 2020, 113, 32–40. [Google Scholar] [CrossRef] [PubMed]
- Guan, L.; Liu, M.; Yu, F.; Qiu, T.; Zhou, T.; Lin, X. A LiFePO4 Regeneration Method Based on PVAc Alcoholysis Reaction. Renew. Energy 2021, 175, 559–567. [Google Scholar] [CrossRef]

| Method | Key Characteristics | Advantages | Challenges |
|---|---|---|---|
| Solid-state | High-temperature sintering with lithium sources and reducing agents under reducing conditions | Simple, cost-effective, suitable for large-scale production, enhanced with doping options | High energy consumption, non-uniform lithium distribution, potential toxic emissions |
| Liquid-state | Hydrothermal treatment with lithium-containing solutions under reducing conditions | Superior morphology, enhanced performance, uniform lithium distribution | High energy consumption, non-uniform lithium distribution, potential toxic emissions |
| Electrochemical | Utilizes electrochemical cells with external energy input, uses inorganic salt solutions as electrolytes | Eco-friendly, high lithium utilization, recyclable electrolytes, potential for clean regeneration | Complex operation, less studied, requires specialized equipment, not yet suitable for large-scale application |
| Method | Key Reagents | Process Overview | Products | Outcomes | Environmental Impact | Ref. |
|---|---|---|---|---|---|---|
| Redox Leaching | HCl, NaOCl | Oxidation of Fe(II) to Fe(III); leaching at low temperatures | FePO4, Li2CO3 | High leaching efficiency; preservation of olivine structure | Mild conditions; minimal degradation | [77] |
| Ionic Liquid Extraction | [P44414]Cl, HCl/NaCl, NH3 | Two-phase system for selective metal separation; Fe precipitation | Li3PO4, Fe(OH)2, Fe(OH)3 | High selectivity for Li and Fe | No oxidizers; eco-friendly separation | [78] |
| Acid and Oxidizer Leaching | HCOOH, H2O2 | Two-stage Li precipitation; oxidation of Fe2+ | Li3PO4, FePO4 | Optimized leaching conditions; high purity | Low formic acid usage; eco-friendly | [73] |
| Proton Circulation | Monocarboxylic acids | Leaching Via proton exchange; FePO4 precipitation | Li3PO4, FePO4 | Stable, scalable method | Acid-based; stable recycling method | [80] |
| Acid-Free Leaching | Fe2(SO4)3, H2O2 | Fe3+ and Li leaching; NaOH precipitation | FePO4, Li2CO3 | High Fe oxidation; lithium recovery | No harmful by-products | [81] |
| Electro-oxidation (Fenton) | H2O2, OH- | Fe2+ oxidation; Li release Via hydroxyl radicals | FePO4, Li+ | 98% lithium leaching | Mild reagents; low waste | [76] |
| Electrolysis | Na2CO3 | Li extraction Via negative electrode oxidation; FePO4 retention | Li2CO3, H2, NaOH | 99% leaching efficiency | Acid-free; eco-friendly | [83] |
| Electrochemical and Leaching | H2SO4 | Negative electrode oxidation; FePO4 as by-product | FePO4, Li+ | Near-complete Li recovery | Minimal by-products | [84]. |
| Carbothermic Reduction | Na2CO3/NaOH, C | High-temperature roasting; Fe reduction | Li3PO4, Fe | 99.2% efficiency | Scalable; low carbon use | [79] |
| Pyrometallurgy | (NH4)2SO4 | Firing under oxygen or vacuum; FePO4, Li leaching | FePO4, Li(NH4)SO4 | Rapid, simple process | Few reagents; suitable for industry | [80] |
| Method | Key Processes | Outcomes | Advantages | Challenges | Ref. |
|---|---|---|---|---|---|
| Carbothermic Reduction | FePO4·2H2O microflowers synthesized; LiFePO4/C mixed with acetylene black and PVDF. | High structural integrity confirmed via XRD. Discharge capacity 150–160 mAh/g at 0.1 C; 93.7% capacity retention at 0.1 C. | Simple method; good electrochemical performance. | Carbon interference needs further study. | [87] |
| Oxidative Roasting | Roasting at 500 °C; minimal graphite oxidation. | >99% lithium leaching; optimal graphite removal at 800 °C. | High lithium extraction efficiency. | Balancing temperature for graphite removal and sintering. | [88] |
| Sulfation Roasting | NaHSO4·H2O used; sintering at 600 °C. | Li3PO4 with high purity; 162.25 mAh/g at 0.1 C. Discharge capacities R-LFP and F-LFP after 200 cycles 140.99 and 144.78 mAh/g; retention rates of 96.57% and 97.83% | High purity and performance; eco-friendly. | Optimization of sodium salt addition. | [89] |
| Hydrometallurgical Approach | FePO4·2H2O leached with H3PO4; no alkali used. | Discharge capacity of 157.6 mAh/g at 0.1 C; 100% capacity retention after 100 cycles at 1 C. | Eco-friendly, no wastewater discharge. | Requires further scalability. | [90] |
| Buffered System Leaching | NaH2PO4 and H2O2 used for leaching; Li3PO4 crystallized via evaporation. | Capacities of the order of 150–160 mAh/g; superior initial capacity; 99.2% retention after 200 cycles at 0.5 C. | Closed-loop recycling; high efficiency. | Buffer optimization needed. | [91] |
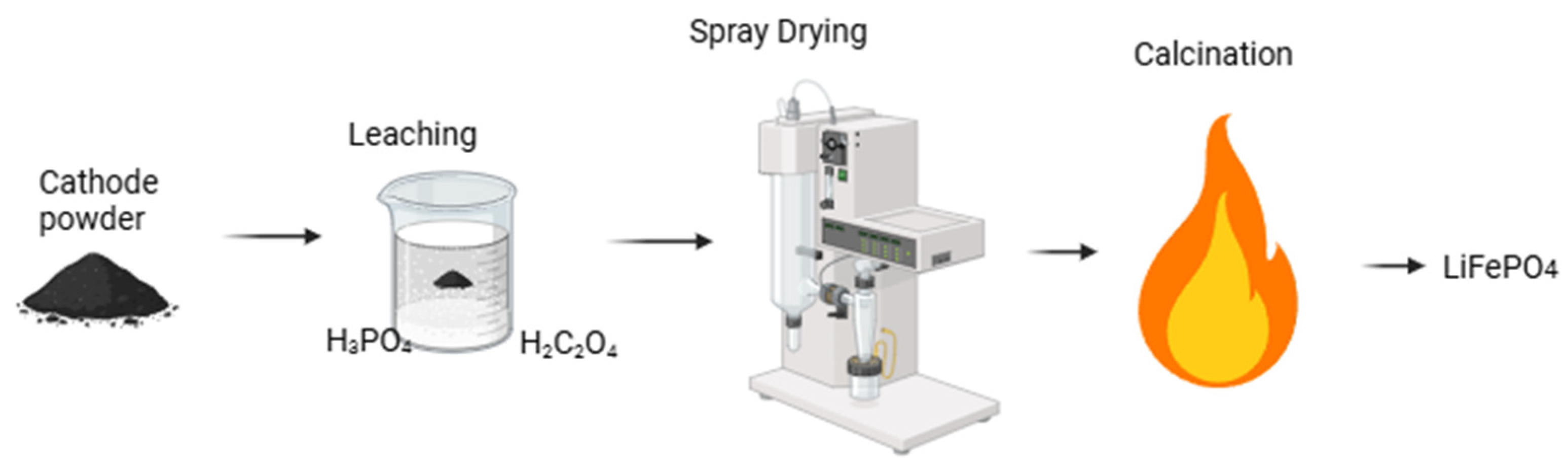
| Mixed Acid Leaching and Spray Drying | H3PO4 and H2C2O4 used; spray drying employed. | Well-crystallized LFP; improved ion diffusion. | Enhanced lithium-ion diffusion; good morphology. | No electrochemical tests performed. | [92] |
| Weak Organic Acids Leaching | MSA and TSA used at room temperature. | Initial capacity of 93 mAh/g at 0.2 C; stable at high rates. | Green chemistry approach. | Carbon content higher than commercial standards. | [93] |
| Fruit Juice Leaching | Leaching with lemon, orange, and apple juices. | Discharge capacity of 155.3 mAh/g at 0.1 C; 98.3% retention after 100 cycles. | Green chemistry using food by-products. | Scaling up for industrial use. | [94] |
| Ammonium Persulfate Leaching | Ammonium persulfate used; carbothermic reduction with glucose. | 161.9 mAh/g at 0.1 C; enhanced crystallinity. The storage capacity of the LFP-G12 reaches 97.9%. | High crystallinity and performance. | Requires fine-tuning of carbon coatings. | [86] |
| Alcoholysis and High-Temperature | PVAc as carbon source; sintering in methanol–ammonia solution. | Discharge capacity of 163.2 mAh/g at 0.1 C. Capacity retention is 97.08% at 0.5 C after 100 cycles. | Simple, impurity-free process. | Requires optimization of PVAc dosing. | [95] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kosenko, A.; Bolotova, A.; Pushnitsa, K.; Novikov, P.; Popovich, A.A. Lithium Iron Phosphate Battery Regeneration and Recycling: Techniques and Efficiency. Batteries 2025, 11, 136. https://doi.org/10.3390/batteries11040136
Kosenko A, Bolotova A, Pushnitsa K, Novikov P, Popovich AA. Lithium Iron Phosphate Battery Regeneration and Recycling: Techniques and Efficiency. Batteries. 2025; 11(4):136. https://doi.org/10.3390/batteries11040136
Chicago/Turabian StyleKosenko, Alexandra, Antonina Bolotova, Konstantin Pushnitsa, Pavel Novikov, and Anatoliy A. Popovich. 2025. "Lithium Iron Phosphate Battery Regeneration and Recycling: Techniques and Efficiency" Batteries 11, no. 4: 136. https://doi.org/10.3390/batteries11040136
APA StyleKosenko, A., Bolotova, A., Pushnitsa, K., Novikov, P., & Popovich, A. A. (2025). Lithium Iron Phosphate Battery Regeneration and Recycling: Techniques and Efficiency. Batteries, 11(4), 136. https://doi.org/10.3390/batteries11040136









