The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries
Abstract
:1. Introduction
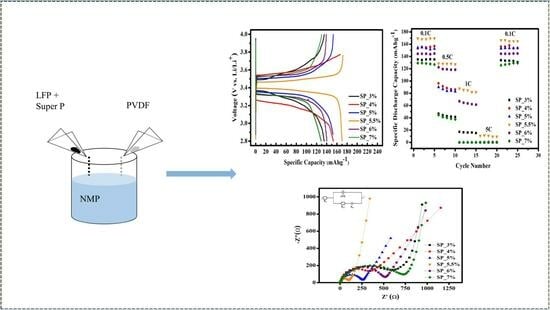
2. Results and Discussion
3. Materials and Methods
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Deng, D. Li-ion batteries: Basics, progress, and challenges. Energy Sci. Eng. 2015, 3, 385–418. [Google Scholar] [CrossRef]
- Yang, K.; Liu, Z.; Chai, J.; Zheng, Y.; Fu, X.; Shen, Y.H.; Chen, J.; Liu, Z.; Shi, S. High performance polyimide-based separator for 4.5 V high voltage LiCoO2 battery with superior safety. Mater. Chem. Phys. 2022, 282, 125975–125983. [Google Scholar] [CrossRef]
- Mohanty, D.; Huang, P.H.; Hung, I.M. Preparation and Characterization of a LiFePO4-Lithium Salt Composite Cathode for All-Solid-State Li-Metal Batteries. Batteries 2023, 9, 236. [Google Scholar] [CrossRef]
- Guo, J.; Zhang, J.; Chen, C.; Lan, Y. Rapid photodegradation of methyl orange by oxalic acis assisted with cathode material of lithium ion batteries LiFePO4. J. Taiwan Inst. Chem. Eng. 2016, 62, 187–191. [Google Scholar] [CrossRef]
- Geng, J.; Zou, Z.; Wang, T.; Zhang, S.; Ling, W.; Peng, X.; Liang, F. Synthesis and modification mechanism of vanadium oxide coated LiFePO4 cathode materials with excellent electrochemical performance. Nanotechnology 2023, 34, 445403–445412. [Google Scholar] [CrossRef]
- Yang, C.C.; Jang, J.H.; Jiang, J.R. Preparation of carbon and oxide co-modified LiFePO4 cathode material for high performance lithium-ion battery. Mater. Chem. Phys. 2015, 165, 196–206. [Google Scholar] [CrossRef]
- Kraytsberg, A.; Ein-Eli, Y. Higher, Stronger, Better… A Review of 5 Volt Cathode Materials for Advanced Lithium-Ion Batteries. Adv. Energy Mater. 2012, 2, 922–939. [Google Scholar] [CrossRef]
- Liu, H.; Liu, Y.; An, L.; Zhao, X.; Wang, L.; Liang, G. High Energy Density LiFePO4/C Cathode Material Synthesized by Wet Ball Milling Combined with Spray Drying Method. J. Electrochem. Soc. 2017, 164, A3666–A3672. [Google Scholar] [CrossRef]
- Nie, Z.; Liu, Y.; Yang, L.; Li, S.; Pan, F. Construction and Application of Materials Knowledge Graph Based on Author Disambiguation: Revisiting the Evolution of LiFePO4. Adv. Energy Mater. 2021, 11, 2003580–2003584. [Google Scholar] [CrossRef]
- Cramer, C.L.; Lonescu, E.; Zajac, M.G.; Nelson, A.T.; Katoh, Y.; Haslam, J.J.; Wondraczek, L.; Aguirre, T.G.; LeBlanc, S.; Wang, H.; et al. Additive manufacturing of ceramic materials for energy applications: Road map and opportunities. J. Eur. Ceram. Soc. 2022, 42, 3049–3088. [Google Scholar] [CrossRef]
- Zhang, F.; Qi, L. Recent Progress in Self-Supported Metal Oxide Nanoarray Electrodes for Advanced Lithium-Ion Batteries. Adv. Sci. 2016, 3, 1600049–1600077. [Google Scholar] [CrossRef]
- Tang, X.C.; Li, L.X.; Lai, Q.L.; Song, X.W.; Jiang, L.H. Investigation on diffusion behavior of Li+ in LiFePO4 by capacity intermittent titration technique (CITT). Electrochim. Acta 2009, 54, 2329–2334. [Google Scholar] [CrossRef]
- Gao, C.; Zhou, J.; Liu, G.; Wang, L. Lithium-ions diffusion kinetic in LiFePO4/carbon nanoparticles synthesized by microwave plasma chemical vapor deposition for lithium-ion batteries. App. Surf. Sci. 2018, 433, 35–44. [Google Scholar] [CrossRef]
- Li, J.; Yuan, C.F.; Guo, Z.H.; Zhang, Z.A.; Lai, Y.Q.; Liu, J. Limiting factors for low-temperature performance of electrolytes in LiFePO4/Li and graphite/Li half cells. Electrochim. Acta 2012, 59, 69–74. [Google Scholar] [CrossRef]
- Zhang, S.S.; Jow, T.R. Aluminum corrosion in electrolyte of Li-ion battery. J. Power Sources 2002, 109, 458–464. [Google Scholar] [CrossRef]
- Zhang, S.S.; Xu, K.; Jow, T.R. The low temperature performance of Li-ion batteries. J. Power Sources 2003, 115, 137–140. [Google Scholar] [CrossRef]
- Zhang, S.S.; Xu, K.; Jow, T.R. Electrochemical impedance study on the low temperature of Li-ion batteries. Electrochim. Acta 2004, 49, 1057–1061. [Google Scholar] [CrossRef]
- Chang, W.; Kim, S.J.; Park, I.T.; Cho, B.W.; Chung, K.Y.; Shin, H.C. Low temperature performance of LiFePO4 cathode material for Li-ion batteries. J. Alloys Compd. 2013, 563, 249–253. [Google Scholar] [CrossRef]
- Shu, H.; Chen, M.; Fu, Y.; Yang, X.; Yi, X.; Bai, Y.; Liang, Q.; Wei, Q.; Hu, B.; Tan, J.; et al. Improvement of electrochemical performance for spherical LiFePO4 via hybrid coated with electron conductive carbon and fast Li ion conductive La0.56Li0.33TiO3. J. Power Sources 2014, 252, 73–78. [Google Scholar] [CrossRef]
- Wang, K.X.; Li, X.H.; Chen, J.S. Surface and Interface Engineering of Electrode Materials for Lithium-Ion Batteries. Adv. Mater. 2015, 27, 527–545. [Google Scholar] [CrossRef]
- Sangeeta; Agnihotri, S.; Arya, A.; Sharma, A.L. Improved electrochemical performance of the Cr doped cathode materials for energy storage/conversion devices. AIP Conf. Proc. 2016, 1728, 020380. [Google Scholar] [CrossRef]
- Yao, J.; Wu, F.; Qiu, X.; Li, N.; Su, Y. Effect of CeO2-coating on the electrochemical performances of LiFePO4/C cathode material. Electrochim. Acta 2011, 56, 5587–5592. [Google Scholar] [CrossRef]
- Weiss, M.; Ruess, R.; Kasnatcheew, J.; Levartovsky, Y.; Lexy, N.R.; Minnmann, P.; Stolz, L.; Waldmann, T.; Mehrens, M.; Aurbach, D.; et al. Fast Charging of Lithium-Ion Batteries: A Review of Materials Aspects. Adv. Energy Mater. 2021, 11, 2101126–2101162. [Google Scholar] [CrossRef]
- Weng, J.; Peng, L. Improving the electrochemical performance of LiFePO4 cathode with novel water-soluble binders. Mater. Chem. Phys. 2022, 290, 126530–126534. [Google Scholar] [CrossRef]
- Örnek, A.; Bulut, E.; Özacar, M. The chemical, physical and electrochemical effects of carbon sources on the nano-scale LiFePO4 cathode surface. Ceram. Int. 2014, 40, 15727–15736. [Google Scholar] [CrossRef]
- Miao, C.; Bai, P.; Jiang, Q.; Sun, S.; Wang, X. A novel synthesis and characterization of LiFePO4 and LiFePO4/C as a cathode material for lithium-ion battery. J. Power Sources 2014, 246, 232–238. [Google Scholar] [CrossRef]
- Lin, Y.; Gao, M.X.; Zhu, D.; Liu, Y.F.; Pan, H.G. Effects of carbon coating and iron phosphides on the electrochemical properties of LiFePO4/C. J. Power Sources 2008, 184, 444–448. [Google Scholar] [CrossRef]
- Al-Salih, H.; Houache, M.S.E.; Baranova, E.A.; Abu-Lebdeh, Y. Composite Cathodes for Solid-State Lithium Batteries: “Catholytes” the Underrated Giants. Adv. Energy Sustain. Res. 2022, 3, 2200032–2200046. [Google Scholar] [CrossRef]
- Nguyen, V.A.; Kuss, C. Review—Conducting Polymer-Based Binders for Lithium-Ion Batteries and Beyond. J. Electrochem. Soc. 2020, 167, 065501–065516. [Google Scholar] [CrossRef]
- Wei, M.; Zhang, F.; Wang, W.; Alexandridis, P.; Zhou, C.; Wu, G. 3D direct writing fabrication of electrodes for electrochemical storage devices. J. Power Sources 2017, 354, 134–147. [Google Scholar] [CrossRef]
- Zhao, N.; Li, Y.; Zhao, X.; Zhi, X.; Liang, G. Effect of particle size and purity on the low temperature electrochemical performance of LiFePO4/C cathode material. J. Alloys Compd. 2016, 683, 123–132. [Google Scholar] [CrossRef]
- Cheng, F.; Wan, W.; Tan, Z.; Huang, Y.; Zhou, H.; Chen, J.; Zhang, X. High power performance of nano-LiFePO4/C cathode material synthesized via lauric acid-assisted solid-state reaction. Electrochim. Acta 2011, 56, 2999–3005. [Google Scholar] [CrossRef]
- Yang, C.C.; Hung, Y.W.; Lue, S.J. The Carbon Additive Effect on Electrochemical Performance of LiFe0.5Mn0.5PO4/C Composites by a Simple Solid-State Method for Lithium Ion Batteries. Batteries 2016, 2, 18. [Google Scholar] [CrossRef]
- Liu, H.; Miao, C.; Meng, Y.; He, Y.B.; Xu, Q.; Zhang, X.; Tang, Z. Optimized synthesis of nano-sized LiFePO4/C particles with excellent rate capability for lithium ion batteries. Electrochim. Acta 2014, 130, 322–328. [Google Scholar] [CrossRef]
- Eftekhari, A. LiFePO4/C nanocomposites for lithium-ion batteries. J. Power Sources 2017, 343, 395–411. [Google Scholar] [CrossRef]
- Zhang, B.; Xu, Y.; Wang, J.; Lin, J.; Wang, C.; Chen, Y. Lanthanum and cerium Co-doped LiFePO4: Morphology, electrochemical performance and kinetic study from −30–+50 °C. Electrochim. Acta 2019, 322, 134686–134695. [Google Scholar] [CrossRef]
- Yang, W.; Bi, Y.; Qin, Y.; Liu, Y.; Zhang, X.; Yang, B.; Wu, Q.; Wang, D.; Shi, S. LiMn0.8Fe0.2PO4/C cathode material synthesized via co-precipitation method with superior high-rate and low-temperature performances for lithium-ion batteries. J. Power Sources 2015, 275, 785–791. [Google Scholar] [CrossRef]
- Lin, Y.; Lin, Y.; Zhou, T.; Zhao, G.; Huang, Y.; Huang, Z. Enhanced electrochemical performances of LiFePO4/C by surface modification with Sn nanoparticles. J. Power Sources 2013, 226, 20–26. [Google Scholar] [CrossRef]
- Jiang, Q.; Zhang, H.; Ren, Z.; Ma, H.; Xue, M. Recent progresses of metal-organic framework-based materials in electrochemical energy storage. Mater. Today Sustain. 2022, 19, 100174–100205. [Google Scholar] [CrossRef]
- Li, X.; Sun, X.; Hu, X.; Fan, F.; Cai, S.; Zheng, C.; Stucky, G.D. Review on comprehending and enhancing the initial Coulombic efficiency of anode materials in lithium-ion/sodium-ion batteries. Nano Energy 2020, 77, 105143–105162. [Google Scholar] [CrossRef]
- Mauger, A.; Julien, C.; Paolella, A.; Armand, M.; Zaghib, K. Recent Progress on Organic Electrodes Materials for Rechargeable Batteries and Supercapacitors. Materials 2019, 12, 1770. [Google Scholar] [CrossRef] [PubMed]
- Song, M.K.; Park, S.; Alamgir, F.M.; Cho, J.; Liu, M. Nanostructured electrodes for lithium-ion and lithium-air batteries: The latest developments, challenges, and perspectives. Mater. Sci. Eng. R Rep. 2011, 72, 203–252. [Google Scholar] [CrossRef]
- Ellis, B.L.; Lee, K.T.; Nazar, L.F. Positive Electrode Materials for Li-Ion and Li-Batteries. Chem. Mater. 2010, 22, 691–714. [Google Scholar] [CrossRef]
- Yousaf, M.; Shi, H.T.H.; Wang, Y.; Chen, Y.; Ma, Z.; Cao, A.; Naguib, H.A.; Han, R.P.S. Novel Pliable Electrodes for Flexible Electrochemical Energy Storage Devices: Recent Progress and Challenges. Adv. Energy Mater. 2016, 6, 1600490–1600512. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, Z.; Wang, Z.; Jiang, Y.; Wen, G.; Gao, P.; Zhu, Y. Electrochemical impedance spectroscopy study of lithium-rich material 0.5Li2MnO3·0.5LiNi1/3Co1/3Mn1/3O2 in the first two charge-discharge cycles. Electrochim. Acta 2019, 310, 136–145. [Google Scholar] [CrossRef]
- Mohanty, D.; Chen, S.Y.; Hung, I.M. Effect of Lithium Salt Concentration on Materials Characteristics and Electrochemical Performance of Hybrid Inorganic/Polymer Solid Electrolyte for Solid-State Lithium-Ion Batteries. Batteries 2022, 8, 173. [Google Scholar] [CrossRef]
- Mohanty, D.; Lu, Z.L.; Hung, I.M. Effect of carbon coating on electrochemical properties of Li3V2(PO4)3 cathode synthesized by citric-acid gel method for lithium-ion batteries. J. Appl. Electrochem. 2023, 53, 1003–1013. [Google Scholar] [CrossRef]
- Rudy, A.; Mironenko, A.; Naumov, V.; Novozhilova, A.; Skundin, A.; Fedorov, I. Determination of Diffusion Coefficients of Lithium in Solid Electrolyte LiPON. Batteries 2021, 7, 21. [Google Scholar] [CrossRef]
- Ngamchuea, K.; Eloul, S.; Tschulik, K.; Compton, R.G. Planar diffusion to macro disc electrodes—What electrode size is required for the Cottrell and Randles-Sevcik equations to apply quantitatively? J. Solid State Electrochem. 2014, 18, 3251–3257. [Google Scholar] [CrossRef]








| ISO Level | ASTM Grade | Test Results |
|---|---|---|
| 0 | 5B | The edge of the incision is completely smooth, and the edge of the grid has not peeled off. |
| 1 | 4B | There are small pieces peeling off at the intersection of the cuts, and the actual damage in the scribed area does not exceed 5%. |
| 2 | 3B | The edges and/or intersections of the incisions have peeled off, with an area of more than 5%, but less than 15%. |
| 3 | 2B | Partially chipped or completely chipped along the edge of the notch, and the partially chipped the was stripped by more than 15%, but less than 35%. |
| 4 | 1B | The edge of the cut is flaking or/or partly squared or partially peeled off, and the area is larger than the 35% mark of the grid area, but not more than 65%. |
| 5 | 0B | Beyond the previous level. |
| Sample | ISO Level | ASTM Grade |
|---|---|---|
| SP_3% | 1 | 4B |
| SP_4% | 2 | 3B |
| SP_5% | 2 | 3B |
| SP_5.5% | 2 | 3B |
| SP_6% | 4 | 1B |
| SP_7% | 5 | 0B |
| Sample | Re (Ω) | Rct (Ω) | DEIS (cm2 s−1) |
|---|---|---|---|
| SP_3% | 2.33 | 338 | 1.08 × 10−14 |
| SP_4% | 9.97 | 284 | 1.45 × 10−16 |
| SP_5% | 2.71 | 199 | 6.99 × 10−15 |
| SP_5.5% | 1.87 | 67 | 5.76 × 10−14 |
| SP_6% | 2.26 | 372 | 7.13 × 10−15 |
| SP_7% | 2.13 | 412 | 2.89 × 10−14 |
| ∆V * | Scanning Rate (mVs−1) | ||||||
|---|---|---|---|---|---|---|---|
| Sample | 0.1 | 0.2 | 0.25 | 0.5 | 0.75 | 1.0 | |
| SP_3% | 0.77 | --- | --- | --- | --- | --- | |
| SP_4% | 0.30 | 0.39 | 0.42 | 0.60 | 1.00 | --- | |
| SP_5% | 0.26 | 0.35 | 0.39 | 0.54 | 0.64 | 0.73 | |
| SP_5.5% | 0.23 | 0.28 | 0.29 | 0.39 | 0.43 | 0.49 | |
| SP_6% | 0.44 | 0.75 | 0.92 | --- | --- | --- | |
| SP_7% | 0.91 | --- | --- | --- | --- | --- | |
| Sample | DCV (cm2 s−1) Oxidation | DCV (cm2 s−1) Reduction |
|---|---|---|
| SP_4% | 7.95 × 10−11 | 5.78 × 10−11 |
| SP_5% | 8.18 × 10−11 | 7.09 × 10−11 |
| SP_5.5% | 2.60 × 10−10 | 1.14 × 10−10 |
| SP_6% | 1.63 × 10−11 | 1.57 × 10−11 |
| Sample Name | LiFePO4 (wt%) | Super P (wt%) | PVDF (wt%) |
|---|---|---|---|
| SP_3% | 88 | 3 | 9 |
| SP_4% | 88 | 4 | 8 |
| SP_5% | 88 | 5 | 7 |
| SP_5.5% | 88 | 5.5 | 6.5 |
| SP_6% | 88 | 6 | 6 |
| SP_7% | 88 | 7 | 5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mohanty, D.; Chang, M.-J.; Hung, I.-M. The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries. Batteries 2023, 9, 515. https://doi.org/10.3390/batteries9100515
Mohanty D, Chang M-J, Hung I-M. The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries. Batteries. 2023; 9(10):515. https://doi.org/10.3390/batteries9100515
Chicago/Turabian StyleMohanty, Debabrata, Min-Jie Chang, and I-Ming Hung. 2023. "The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries" Batteries 9, no. 10: 515. https://doi.org/10.3390/batteries9100515
APA StyleMohanty, D., Chang, M.-J., & Hung, I.-M. (2023). The Effect of Different Amounts of Conductive Carbon Material on the Electrochemical Performance of the LiFePO4 Cathode in Li-Ion Batteries. Batteries, 9(10), 515. https://doi.org/10.3390/batteries9100515