Proteolytically Resistant Bioactive Peptide-Grafted Sr/Mg-Doped Hardystonite Foams: Comparison of Two Covalent Functionalization Strategies
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
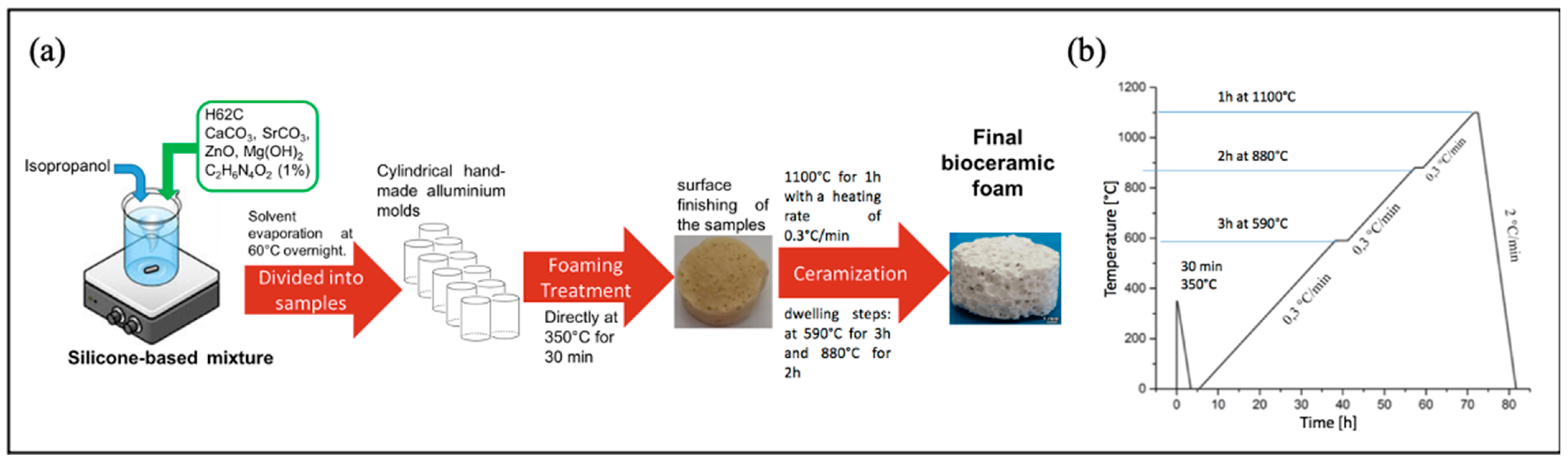
2.2. Sr/Mg-Doped HT Foams’ Synthesis and Characterization
2.3. Peptides’ Synthesis and Characterization
2.3.1. D2HVP
2.3.2. D2HVPF
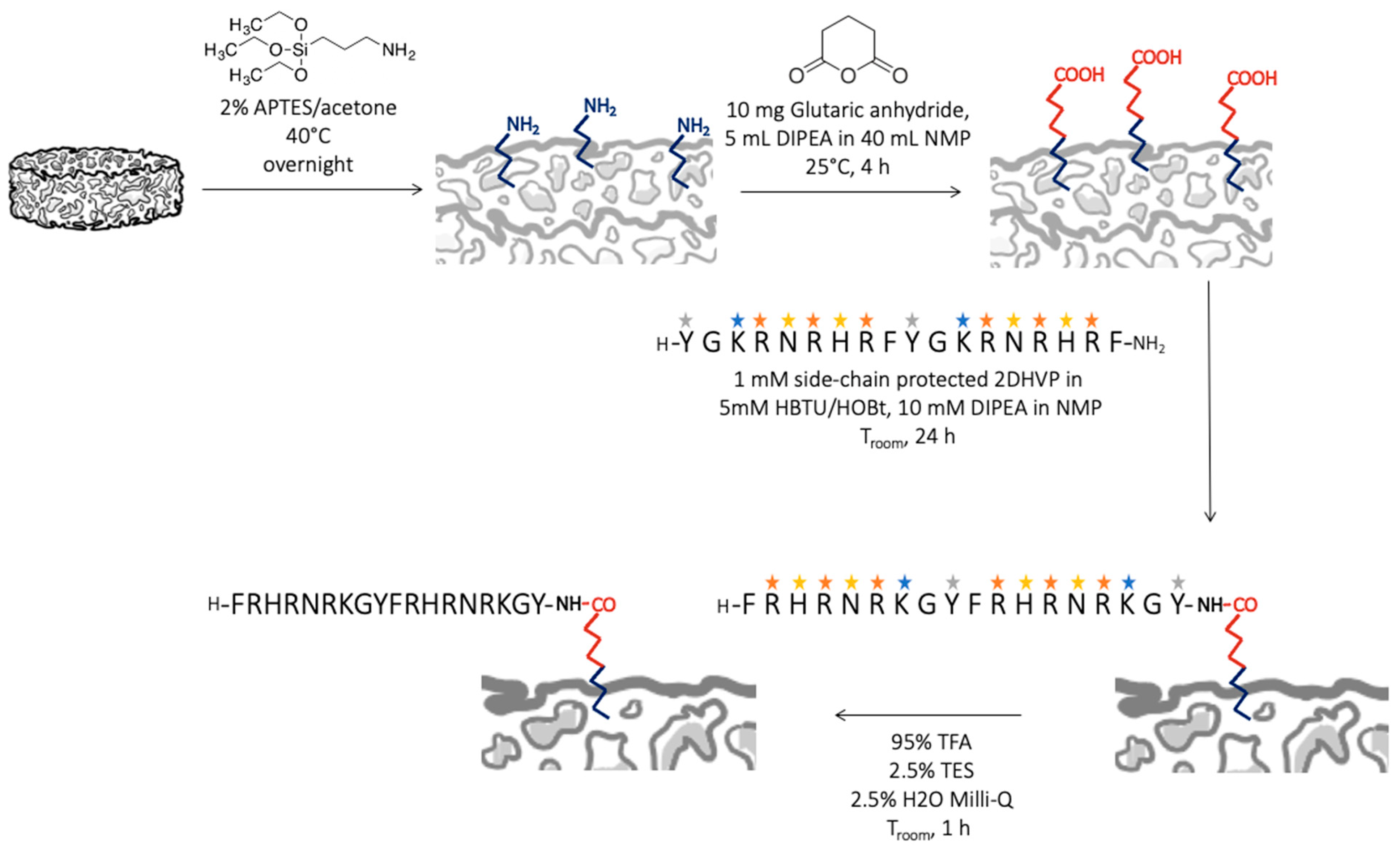
2.4. Bioceramic Functionalization and Characterization
2.4.1. Functionalization with D2HVP via a Side-Chain Protected Peptide
2.4.2. Functionalization with D2HVP via Aldehyde Group
2.5. Vibrational Raman and IR Spectroscopy
2.6. Inductively Coupled Plasma Mass Spectroscopy (ICP-MS)
2.7. Scanning Electron Microscopy (SEM)
2.8. Biological Assays
2.8.1. Cell Culture
2.8.2. Cell Staining
2.8.3. EDU Proliferation Assay
2.8.4. CFSE Proliferation Assay
2.8.5. Cytotoxicity Assay
2.8.6. Quantitative Real Time Polymerase Chain Reaction
2.8.7. Statistical Analysis
3. Results
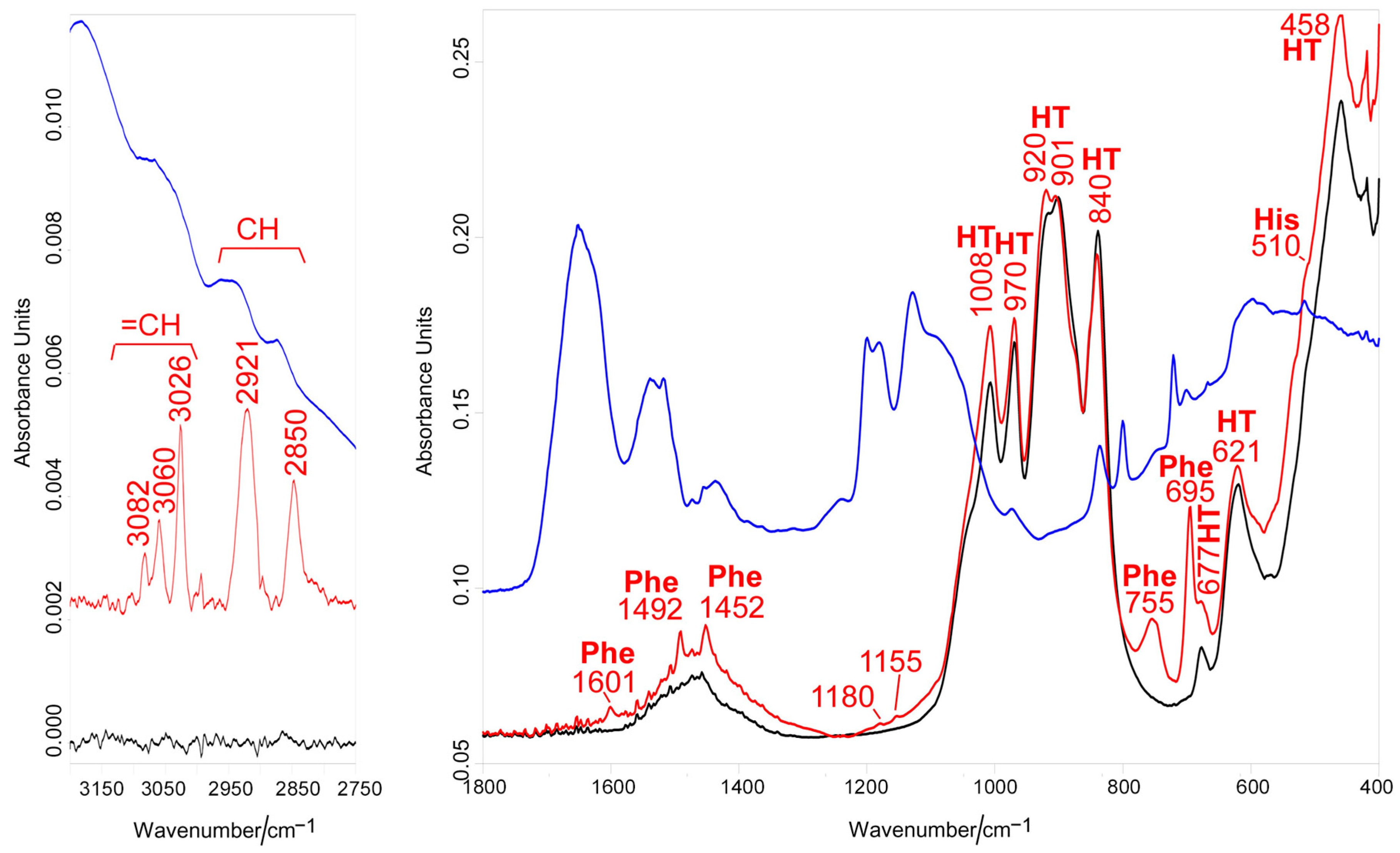
3.1. Vibrational Raman and IR Spectroscopies
3.2. Biological Assays
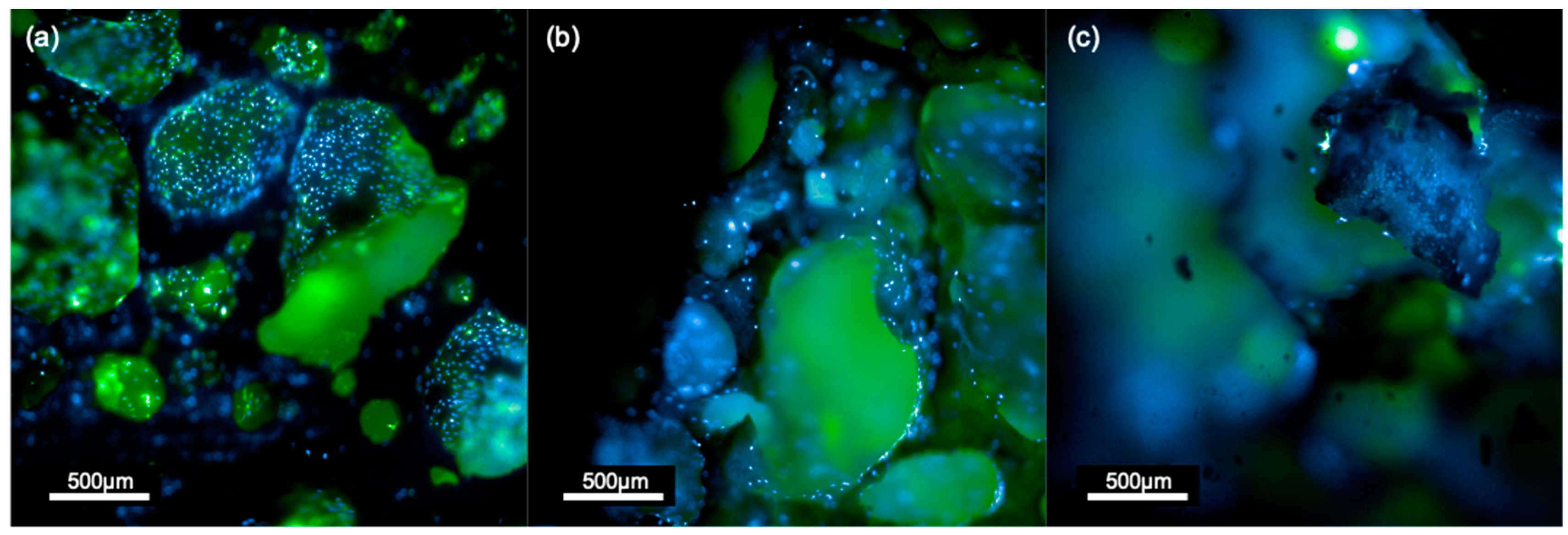
3.2.1. Sr/Mg-Doped HT Foams Functionalized via a Side-Chain Protected Peptide Showed No Osteoblasts Proliferation
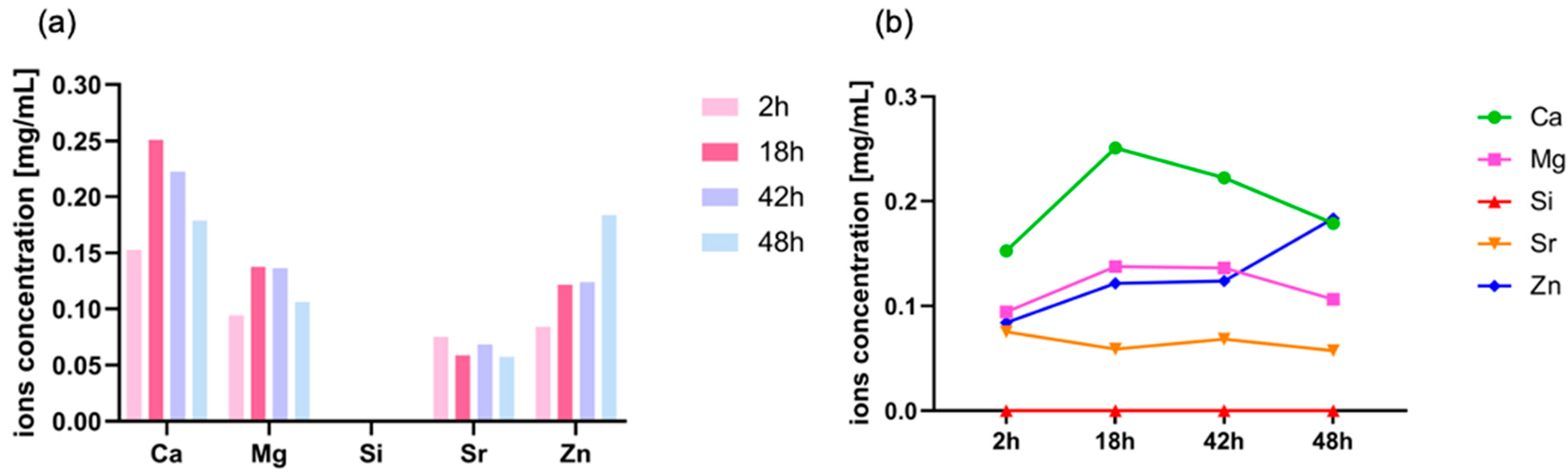
3.2.2. ICP-MS Analysis
3.2.3. SEM Analysis
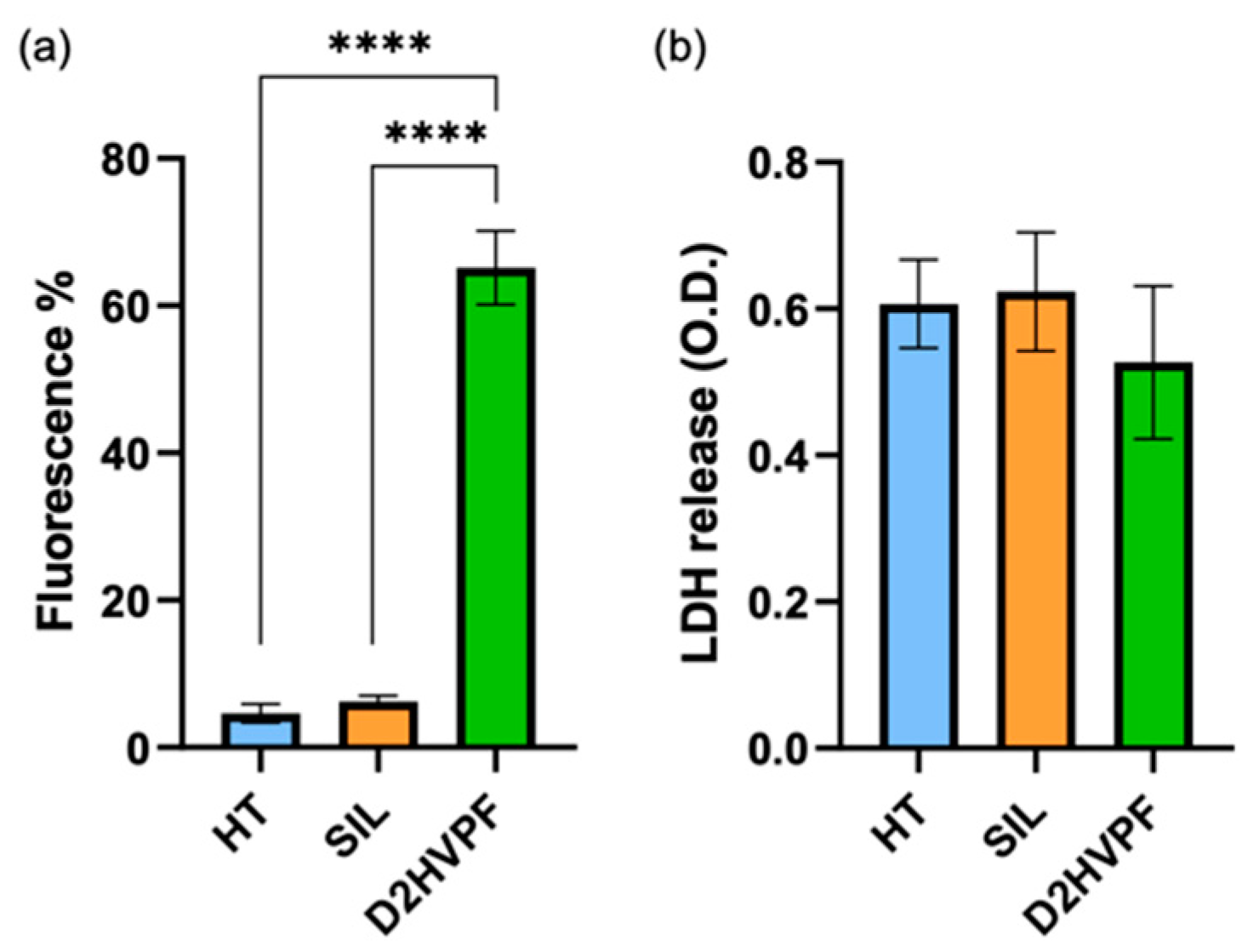
3.2.4. Functionalized Foams via Peptide-Aldehyde Enhanced Osteoblasts Proliferation without Inducing Cytotoxicity
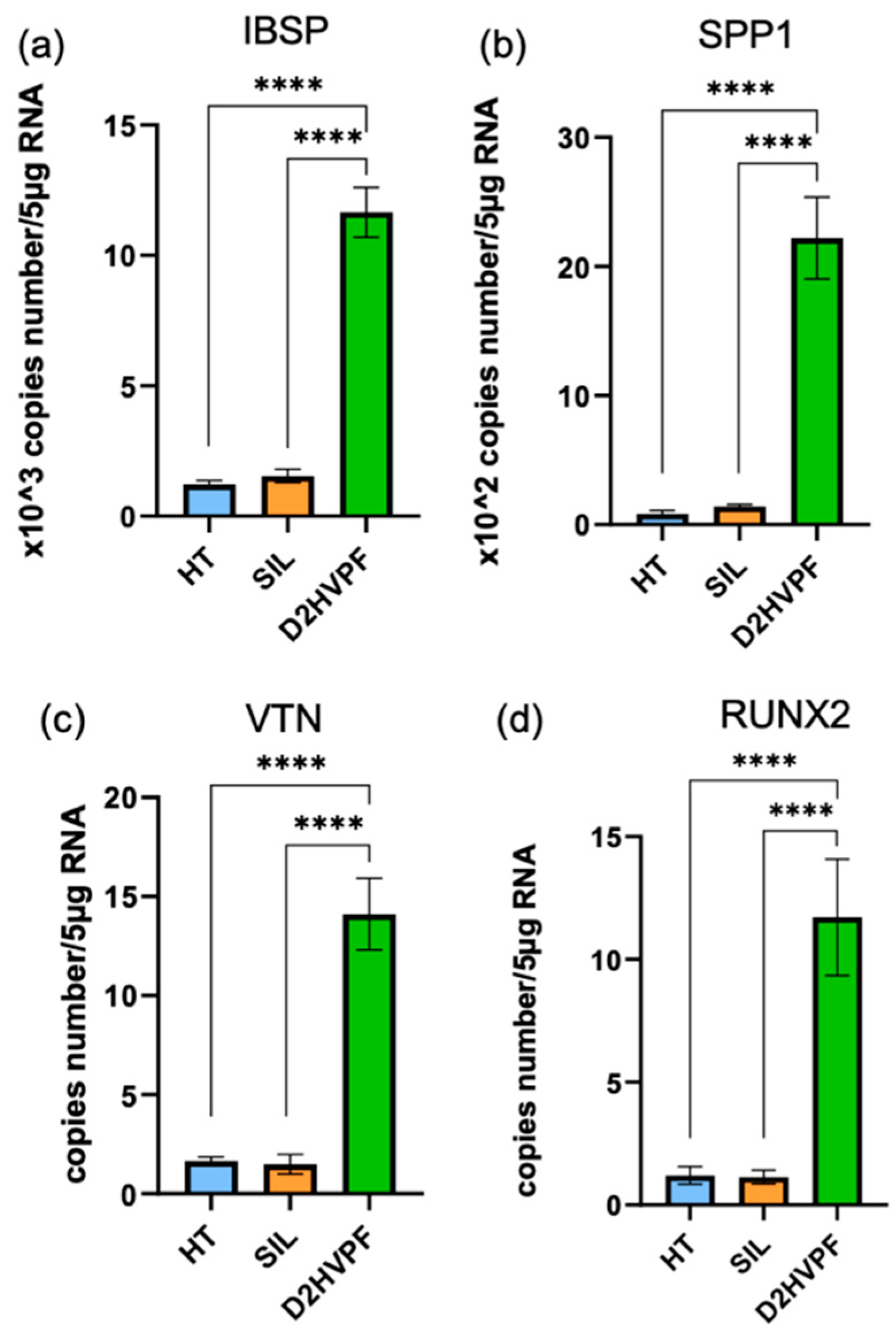
3.2.5. Functionalized Foams Induce Gene Expression of Human Osteoblasts
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Edgar, L.; McNamara, K.; Wong, T.; Tamburrini, R.; Katari, R.; Orlando, G. Heterogeneity of Scaffold Biomaterials in Tissue Engineering. Materials 2016, 9, 332. [Google Scholar] [CrossRef]
- Zander, N.E.; Orlicki, J.A.; Rawlett, A.M.; Beebe, T.P. Quantification of Protein Incorporated into Electrospun Polycaprolactone Tissue Engineering Scaffolds. ACS Appl. Mater. Interfaces 2012, 4, 2074–2081. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Lim, J.; Teoh, S.-H. Review: Development of Clinically Relevant Scaffolds for Vascularised Bone Tissue Engineering. Biotechnol. Adv. 2013, 31, 688–705. [Google Scholar] [CrossRef] [PubMed]
- Baino, F.; Novajra, G.; Vitale-Brovarone, C. Bioceramics and Scaffolds: A Winning Combination for Tissue Engineering. Front. Bioeng. Biotechnol. 2015, 3, 202. [Google Scholar] [CrossRef]
- Fiocco, L.; Bernardo, E.; Colombo, P.; Cacciotti, I.; Bianco, A.; Bellucci, D.; Sola, A.; Cannillo, V. Novel Processing of Bioglass Ceramics from Silicone Resins Containing Micro- and Nano-Sized Oxide Particle Fillers. J. Biomed. Mater. Res. Part A 2014, 102, 2502–2510. [Google Scholar] [CrossRef]
- Zamuner, A.; Brun, P.; Ciccimarra, R.; Ravanetti, F.; Veschini, L.; Elsayed, H.; Sivolella, S.; Iucci, G.; Porzionato, A.; Silvio, L.D.; et al. Biofunctionalization of Bioactive Ceramic Scaffolds to Increase the Cell Response for Bone Regeneration. Biomed. Mater. 2021, 16, 055007. [Google Scholar] [CrossRef]
- Fiocco, L.; Elsayed, H.; Ferroni, L.; Gardin, C.; Zavan, B.; Bernardo, E. Bioactive Wollastonite-Diopside Foams from Preceramic Polymers and Reactive Oxide Fillers. Materials 2015, 8, 2480–2494. [Google Scholar] [CrossRef]
- Bernardo, E.; Carlotti, J.-F.; Dias, P.M.; Fiocco, L.; Colombo, P.; Treccani, L.; Hess, U.; Rezwan, K. Novel Akermanite-Based Bioceramics from Preceramic Polymers and Oxide Fillers. Ceram. Int. 2014, 40, 1029–1035. [Google Scholar] [CrossRef]
- Elsayed, H.; Zocca, A.; Franchin, G.; Bernardo, E.; Colombo, P. Hardystonite Bioceramics from Preceramic Polymers. J. Eur. Ceram. Soc. 2016, 36, 829–835. [Google Scholar] [CrossRef]
- Elsayed, H.; Zocca, A.; Bernardo, E.; Gomes, C.M.; Günster, J.; Colombo, P. Development of Bioactive Silicate-Based Glass-Ceramics from Preceramic Polymer and Fillers. J. Eur. Ceram. Soc. 2015, 35, 731–739. [Google Scholar] [CrossRef]
- Mourino, V.; Cattalini, J.P.; Boccaccini, A.R. Metallic Ions as Therapeutic Agents in Tissue Engineering Scaffolds: An Overview of Their Biological Applications and Strategies for New Developments. J. R. Soc. Interface 2012, 9, 401–419. [Google Scholar] [CrossRef] [PubMed]
- Hoppe, A.; Güldal, N.S.; Boccaccini, A.R. A Review of the Biological Response to Ionic Dissolution Products from Bioactive Glasses and Glass-Ceramics. Biomaterials 2011, 32, 2757–2774. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Yu, Y.; Dai, K.; Ma, Z.; Liu, Y.; Wang, J.; Liu, C. Improved Osteogenesis and Angiogenesis of Magnesium-Doped Calcium Phosphate Cement via Macrophage Immunomodulation. Biomater. Sci. 2016, 4, 1574–1583. [Google Scholar] [CrossRef]
- Elsayed, H.; Gardin, C.; Ferroni, L.; Zavan, B.; Colombo, P.; Bernardo, E. Highly Porous Sr/Mg-Doped Hardystonite Bioceramics from Preceramic Polymers and Reactive Fillers: Direct Foaming and Direct Ink Writing. Adv. Eng. Mater. 2019, 21, 1800900. [Google Scholar] [CrossRef]
- Zamuner, A.; Brun, P.; Scorzeto, M.; Sica, G.; Castagliuolo, I.; Dettin, M. Smart Biomaterials: Surfaces Functionalized with Proteolytically Stable Osteoblast-Adhesive Peptides. Bioact. Mater. 2017, 2, 121–130. [Google Scholar] [CrossRef]
- Hughes, D.E.; Salter, D.M.; Dedhar, S.; Simpson, R. Integrin Expression in Human Bone. J. Bone Miner. Res. 1993, 8, 527–533. [Google Scholar] [CrossRef]
- Ramazanoglu, M.; Oshi, Y. Osseointegration and Bioscience of Implant Surfaces—Current Concepts at Bone-Implant Interface. In Implant Dentistry—A Rapidly Evolving Practice; InTech: London, UK, 2011. [Google Scholar]
- Bierbaum, S.; Hintze, V.; Scharnweber, D. Functionalization of Biomaterial Surfaces Using Artificial Extracellular Matrices. Biomatter 2012, 2, 132–141. [Google Scholar] [CrossRef]
- Hassert, R.; Beck-Sickinger, A.G. Tuning Peptide Affinity for Biofunctionalized Surfaces. Eur. J. Pharm. Biopharm. 2013, 85, 69–77. [Google Scholar] [CrossRef]
- Beutner, R.; Michael, J.; Schwenzer, B.; Scharnweber, D. Biological Nano-Functionalization of Titanium-Based Biomaterial Surfaces: A Flexible Toolbox. J. R. Soc. Interface 2010, 7 (Suppl. S1), S93–S105. [Google Scholar] [CrossRef]
- Dettin, M.; Zamuner, A.; Iucci, G.; Messina, G.M.L.; Battocchio, C.; Picariello, G.; Gallina, G.; Marletta, G.; Castagliuolo, I.; Brun, P. Driving H-Osteoblast Adhesion and Proliferation on Titania: Peptide Hydrogels Decorated with Growth Factors and Adhesive Conjugates. J. Pept. Sci. 2014, 20, 585–594. [Google Scholar] [CrossRef]
- Sapsford, K.E.; Algar, W.R.; Berti, L.; Gemmill, K.B.; Casey, B.J.; Oh, E.; Stewart, M.H.; Medintz, I.L. Functionalizing Nanoparticles with Biological Molecules: Developing Chemistries That Facilitate Nanotechnology. Chem. Rev. 2013, 113, 1904–2074. [Google Scholar] [CrossRef]
- Dettin, M.; Herath, T.; Gambaretto, R.; Iucci, G.; Battocchio, C.; Bagno, A.; Ghezzo, F.; Di Bello, C.; Polzonetti, G.; Di Silvio, L. Assessment of Novel Chemical Strategies for Covalent Attachment of Adhesive Peptides to Rough Titanium Surfaces: XPS Analysis and Biological Evaluation. J. Biomed. Mater. Res. Part A 2009, 91A, 463–479. [Google Scholar] [CrossRef]
- Bagno, A.; Piovan, A.; Dettin, M.; Chiarion, A.; Brun, P.; Gambaretto, R.; Fontana, G.; Di Bello, C.; Palù, G.; Castagliuolo, I. Human Osteoblast-like Cell Adhesion on Titanium Substrates Covalently Functionalized with Synthetic Peptides. Bone 2007, 40, 693–699. [Google Scholar] [CrossRef]
- Maheshwari, G.; Brown, G.; Lauffenburger, D.A.; Wells, A.; Griffith, L.G. Cell Adhesion and Motility Depend on Nanoscale RGD Clustering. J. Cell Sci. 2000, 113 Pt 1, 1677–1686. [Google Scholar] [CrossRef]
- Cacchioli, A.; Ravanetti, F.; Bagno, A.; Dettin, M.; Gabbi, C. Human Vitronectin–Derived Peptide Covalently Grafted onto Titanium Surface Improves Osteogenic Activity: A Pilot In Vivo Study on Rabbits. Tissue Eng. Part A 2009, 15, 2917–2926. [Google Scholar] [CrossRef]
- Conde, J.; Dias, J.T.; Grazú, V.; Moros, M.; Baptista, P.V.; de la Fuente, J.M. Revisiting 30 Years of Biofunctionalization and Surface Chemistry of Inorganic Nanoparticles for Nanomedicine. Front. Chem. 2014, 2, 48. [Google Scholar] [CrossRef]
- Tallawi, M.; Rosellini, E.; Barbani, N.; Cascone, M.G.; Rai, R.; Saint-Pierre, G.; Boccaccini, A.R. Strategies for the Chemical and Biological Functionalization of Scaffolds for Cardiac Tissue Engineering: A Review. J. R. Soc. Interface 2015, 12, 20150254. [Google Scholar] [CrossRef]
- Nanci, A.; Wuest, J.D.; Peru, L.; Brunet, P.; Sharma, V.; Zalzal, S.; McKee, M.D. Chemical Modification of Titanium Surfaces for Covalent Attachment of Biological Molecules. J. Biomed. Mater. Res. 1998, 40, 324–335. [Google Scholar] [CrossRef]
- Ruoslahti, E. RGD and other recognition sequences for integrins. Annu. Rev. Cell Dev. Biol. 1996, 12, 697–715. [Google Scholar] [CrossRef]
- Yang, M.; Zhang, Z.-C.; Liu, Y.; Chen, Y.-R.; Deng, R.-H.; Zhang, Z.-N.; Yu, J.-K.; Yuan, F.-Z. Function and Mechanism of RGD in Bone and Cartilage Tissue Engineering. Front. Bioeng. Biotechnol. 2021, 9, 773636. [Google Scholar] [CrossRef]
- Brun, P.; Scorzeto, M.; Vassanelli, S.; Castagliuolo, I.; Palù, G.; Ghezzo, F.; Messina, G.M.L.; Iucci, G.; Battaglia, V.; Sivolella, S.; et al. Mechanisms Underlying the Attachment and Spreading of Human Osteoblasts: From Transient Interactions to Focal Adhesions on Vitronectin-Grafted Bioactive Surfaces. Acta Biomater. 2013, 9, 6105–6115. [Google Scholar] [CrossRef]
- Zamuner, A.; Pasquato, A.; Castagliuolo, I.; Dettin, M.; Brun, P. Selective Grafting of Protease-Resistant Adhesive Peptides on Titanium Surfaces. Molecules 2022, 27, 8727. [Google Scholar] [CrossRef]
- Luna, L.G. Manual of Histologic Staining Methods of the Armed Forces Institute of Pathology; Blakiston Division, McGraw-Hill: New York, NY, USA, 1968. [Google Scholar]
- Chan, F.K.-M.; Moriwaki, K.; De Rosa, M.J. Detection of Necrosis by Release of Lactate Dehydrogenase Activity. In Immune Homeostasis: Methods and Protocols; Springer: Berlin/Heidelberg, Germany, 2013; pp. 65–70. [Google Scholar]
- Kaminskii, A.A.; Rhee, H.; Lux, O.; Eichler, H.J.; Bohatý, L.; Becker, P.; Liebertz, J.; Ueda, K.; Shirakawa, A.; Koltashev, V.V.; et al. Many-Phonon Stimulated Raman Scattering and Related Cascaded and Cross-Cascaded χ(3)-Nonlinear Optical Effects in Melilite-Type Crystal Ca2ZnSi2O7. Laser Phys. Lett. 2011, 8, 859–874. [Google Scholar] [CrossRef]
- Bistričić, L.; Volovšek, V.; Dananić, V. Conformational and Vibrational Analysis of Gamma-Aminopropyltriethoxysilane. J. Mol. Struct. 2007, 834–836, 355–363. [Google Scholar] [CrossRef]
- Zhu, G.; Zhu, X.; Fan, Q.; Wan, X. Raman Spectra of Amino Acids and Their Aqueous Solutions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2011, 78, 1187–1195. [Google Scholar] [CrossRef]
- Toyama, A.; Ono, K.; Hashimoto, S.; Takeuchi, H. Raman Spectra and Normal Coordinate Analysis of the N1−H and N3−H Tautomers of 4-Methylimidazole: Vibrational Modes of Histidine Tautomer Markers. J. Phys. Chem. A 2002, 106, 3403–3412. [Google Scholar] [CrossRef]
- Faria, J.L.B.; Almeida, F.M.; Pilla, O.; Rossi, F.; Sasaki, J.M.; Melo, F.E.A.; Mendes Filho, J.; Freire, P.T.C. Raman Spectra OfL-Histidine Hydrochloride Monohydrate Crystal. J. Raman Spectrosc. 2004, 35, 242–248. [Google Scholar] [CrossRef]
- Webster, T.J.; Schadler, L.S.; Siegel, R.W.; Bizios, R. Mechanisms of Enhanced Osteoblast Adhesion on Nanophase Alumina Involve Vitronectin. Tissue Eng. 2001, 7, 291–301. [Google Scholar] [CrossRef]
- Cassari, L.; Brun, P.; Di Foggia, M.; Taddei, P.; Zamuner, A.; Pasquato, A.; De Stefanis, A.; Valentini, V.; Saceleanu, V.M.; Rau, J.V.; et al. Mn-Containing Bioactive Glass-Ceramics: BMP-2-Mimetic Peptide Covalent Grafting Boosts Human-Osteoblast Proliferation and Mineral Deposition. Materials 2022, 15, 4647. [Google Scholar] [CrossRef]
- Chukanov, N.V.; Chervonnyi, A.D. Infrared Spectroscopy of Minerals and Related Compounds; Springer mineralogy; Springer: Cham, Germany, 2016; ISBN 978-3-319-25347-3. [Google Scholar]
- Socrates, G. Infrared Characteristic Group Frequencies: Tables and Charts, 2nd ed.; Wiley: Chichester, NY, USA, 1994; ISBN 978-0-471-94230-6. [Google Scholar]
- Boettcher, S.W.; Bartl, M.H.; Hu, J.G.; Stucky, G.D. Structural Analysis of Hybrid Titania-Based Mesostructured Composites. J. Am. Chem. Soc. 2005, 127, 9721–9730. [Google Scholar] [CrossRef]
- Delgado, J.M.; Blanco, R.; Orts, J.M.; Pérez, J.M.; Rodes, A. DFT and In-Situ Spectroelectrochemical Study of the Adsorption of Fluoroacetate Anions at Gold Electrodes. J. Phys. Chem. C 2009, 113, 989–1000. [Google Scholar] [CrossRef]
- Mokkarat, A.; Kruanetr, S.; Sakee, U. Facial Preparation of Trifluoroacetic Acid-Immobilized Amino-Functionalized Silica Magnetite Nano-Catalysts as a Highly Efficient and Reusable for Synthesis of β-Enaminones. J. Mol. Struct. 2022, 1259, 132745. [Google Scholar] [CrossRef]
- Ojima, J. Determining of Crystalline Silica in Respirable Dust Samples by Infrared Spectrophotometry in the Presence of Interferences. JRNL Occup Health 2003, 45, 94–103. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.M.; Molinelli, A.; Karlowatz, M.; Aleksandrov, A.; Orlando, T.; Mizaikoff, B. Infrared Attenuated Total Reflection Spectroscopy of Quartz and Silica Micro- and Nanoparticulate Films. J. Phys. Chem. C 2012, 116, 37–43. [Google Scholar] [CrossRef]
- Innocenzi, P. Infrared Spectroscopy of Sol–Gel Derived Silica-Based Films: A Spectra-Microstructure Overview. J. Non-Cryst. Solids 2003, 316, 309–319. [Google Scholar] [CrossRef]
- Terry, B. The Acid Decomposition of Silicate Minerals Part I. Reactivities and Modes of Dissolution of Silicates. Hydrometallurgy 1983, 10, 135–150. [Google Scholar] [CrossRef]
- Aina, V.; Perardi, A.; Bergandi, L.; Malavasi, G.; Menabue, L.; Morterra, C.; Ghigo, D. Cytotoxicity of Zinc-Containing Bioactive Glasses in Contact with Human Osteoblasts. Chem.-Biol. Interact. 2007, 167, 207–218. [Google Scholar] [CrossRef]
- Aina, V.; Malavasi, G.; Fiorio Pla, A.; Munaron, L.; Morterra, C. Zinc-Containing Bioactive Glasses: Surface Reactivity and Behaviour towards Endothelial Cells. Acta Biomater. 2009, 5, 1211–1222. [Google Scholar] [CrossRef]
- Fiocco, L.; Li, S.; Stevens, M.M.; Bernardo, E.; Jones, J.R. Biocompatibility and Bioactivity of Porous Polymer-Derived Ca-Mg Silicate Ceramics. Acta Biomater. 2017, 50, 56–67. [Google Scholar] [CrossRef]
- Oki, A.; Parveen, B.; Hossain, S.; Adeniji, S.; Donahue, H. Preparation Andin Vitro Bioactivity of Zinc Containing Sol-Gel-Derived Bioglass Materials. J. Biomed. Mater. Res. 2004, 69A, 216–221. [Google Scholar] [CrossRef]
- Brandao-Neto, J.V.; Stefan, V.; Mendonca, B.B.; Bloise, W.; Castro, A.V.B. The Essential Role of Zinc in Growth. Nutr. Res. 1995, 15, 335–358. [Google Scholar] [CrossRef]
- Tang, Z.-L.; Wasserloos, K.; St. Croix, C.M.; Pitt, B.R. Role of Zinc in Pulmonary Endothelial Cell Response to Oxidative Stress. Am. J. Physiol.-Lung Cell. Mol. Physiol. 2001, 281, L243–L249. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Yamaguchi, R. Action of Zinc on Bone Metabolism in Rats. Increases in Alkaline Phosphatase Activity and DNA Content. Biochem. Pharmacol. 1986, 35, 773–777. [Google Scholar] [CrossRef] [PubMed]
- Holloway, W.R.; Collier, F.M.; Herbst, R.E.; Hodge, J.M.; Nicholson, G.C. Osteoblast-Mediated Effects of Zinc on Isolated Rat Osteoclasts: Inhibition of Bone Resorption and Enhancement of Osteoclast Number. Bone 1996, 19, 137–142. [Google Scholar] [CrossRef] [PubMed]
- Lusvardi, G.; Malavasi, G.; Menabue, L.; Menziani, M.C. Synthesis, Characterization, and Molecular Dynamics Simulation of Na2O−CaO−SiO2−ZnO Glasses. J. Phys. Chem. B 2002, 106, 9753–9760. [Google Scholar] [CrossRef]
- Pors Nielsen, S. The Biological Role of Strontium. Bone 2004, 35, 583–588. [Google Scholar] [CrossRef]
- Guo, D.; Xu, K.; Zhao, X.; Han, Y. Development of a Strontium-Containing Hydroxyapatite Bone Cement. Biomaterials 2005, 26, 4073–4083. [Google Scholar] [CrossRef]
- Wong, C.T.; Lu, W.W.; Chan, W.K.; Cheung, K.M.C.; Luk, K.D.K.; Lu, D.S.; Rabie, A.B.M.; Deng, L.F.; Leong, J.C.Y. In Vivo Cancellous Bone Remodeling on a Strontium-Containing Hydroxyapatite (Sr-HA) Bioactive Cement. J. Biomed. Mater. Res. 2004, 68A, 513–521. [Google Scholar] [CrossRef]
- Wong, C.T.; Chen, Q.Z.; Lu, W.W.; Leong, J.C.Y.; Chan, W.K.; Cheung, K.M.C.; Luk, K.D.K. Ultrastructural Study of Mineralization of a Strontium-Containing Hydroxyapatite (Sr-HA) Cementin Vivo. J. Biomed. Mater. Res. 2004, 70A, 428–435. [Google Scholar] [CrossRef]
- Qiu, K.; Zhao, X.J.; Wan, C.X.; Zhao, C.S.; Chen, Y.W. Effect of Strontium Ions on the Growth of ROS17/2.8 Cells on Porous Calcium Polyphosphate Scaffolds. Biomaterials 2006, 27, 1277–1286. [Google Scholar] [CrossRef]
- Marie, P.J.; Ammann, P.; Boivin, G.; Rey, C. Mechanisms of Action and Therapeutic Potential of Strontium in Bone. Calcif. Tissue Int. 2001, 69, 121–129. [Google Scholar] [CrossRef] [PubMed]
- Verberckmoes, S.C.; De Broe, M.E.; D’Haese, P.C. Dose-Dependent Effects of Strontium on Osteoblast Function and Mineralization. Kidney Int. 2003, 64, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Morohashi, T.; Sano, T.; Yamada, S. Effects of Strontium on Calcium Metabolism in Rats. I. A Distinction between the Pharmacological and Toxic Doses. Jpn. J. Pharmacol. 1994, 64, 155–162. [Google Scholar] [CrossRef]
- Chen, T.-Y.; Chu, S.-Y.; Juang, Y.-D. Effects of Sintering Temperature on the Dielectric and Piezoelectric Properties of Sr Additive Sm-Modified PbTiO3 Ceramics. Sens. Actuators A: Phys. 2002, 102, 6–10. [Google Scholar] [CrossRef]
- Li, J.; Liao, H.; Hermansson, L. Sintering of Partially-Stabilized Zirconia and Partially-Stabilized Zirconia-Hydroxyapatite Composites by Hot Isostatic Pressing and Pressureless Sintering. Biomaterials 1996, 17, 1787–1790. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Ramaswamy, Y.; Kwik, D.; Zreiqat, H. The Effect of Strontium Incorporation into CaSiO3 Ceramics on Their Physical and Biological Properties. Biomaterials 2007, 28, 3171–3181. [Google Scholar] [CrossRef] [PubMed]
- Zreiqat, H.; Howlett, C.R.; Zannettino, A.; Evans, P.; Schulze-Tanzil, G.; Knabe, C.; Shakibaei, M. Mechanisms of Magnesium-Stimulated Adhesion of Osteoblastic Cells to Commonly Used Orthopaedic Implants. J. Biomed. Mater. Res. 2002, 62, 175–184. [Google Scholar] [CrossRef]
- Yamasaki, Y.; Yoshida, Y.; Okazaki, M.; Shimazu, A.; Uchida, T.; Kubo, T.; Akagawa, Y.; Hamada, Y.; Takahashi, J.; Matsuura, N. Synthesis of Functionally Graded MgCO3 Apatite Accelerating Osteoblast Adhesion. J. Biomed. Mater. Res. 2002, 62, 99–105. [Google Scholar] [CrossRef]
- Dietrich, E.; Oudadesse, H.; Lucas-Girot, A.; Mami, M. In Vitro Bioactivity of Melt-Derived Glass 46S6 Doped with Magnesium. J. Biomed. Mater. Res. Part A 2009, 88A, 1087–1096. [Google Scholar] [CrossRef]





| GENE [ACCESSION#] | SEQUENCE |
|---|---|
| GAPDH | a Fw: 5′-cgggaagcccatcacca-3′ |
| [NM_002046] | b Rv: 5′-ccggcctcaccccatt-3′ |
| IBSP | Fw: 5′-ttactaccaccaccagtgaagc-3′ |
| [NM_004967] | Rv: 5′-gatgcaaagccagaatggat-3′ |
| VTN | Fw: 5′-ggaggacatcttcgagcttct-3′ |
| [NM_000638] | Rv: 5′-gctaatgaactggggctgtc-3′ |
| SPP1 | Fw: 5′-aagtttcgcagacctgacatc-3′ |
| [NM_000582] | Rv: 5′-ggctgtcccaatcagaagg-3′ |
| RUNX2 | Fw: 5′-cagtgacaccatgtcagcaa-3′ |
| [NM_001024630] | Rv: 5′-gctcacgtcgctcattttg-3′ |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zamuner, A.; Zeni, E.; Elsayed, H.; Di Foggia, M.; Taddei, P.; Pasquato, A.; Di Silvio, L.; Bernardo, E.; Brun, P.; Dettin, M. Proteolytically Resistant Bioactive Peptide-Grafted Sr/Mg-Doped Hardystonite Foams: Comparison of Two Covalent Functionalization Strategies. Biomimetics 2023, 8, 185. https://doi.org/10.3390/biomimetics8020185
Zamuner A, Zeni E, Elsayed H, Di Foggia M, Taddei P, Pasquato A, Di Silvio L, Bernardo E, Brun P, Dettin M. Proteolytically Resistant Bioactive Peptide-Grafted Sr/Mg-Doped Hardystonite Foams: Comparison of Two Covalent Functionalization Strategies. Biomimetics. 2023; 8(2):185. https://doi.org/10.3390/biomimetics8020185
Chicago/Turabian StyleZamuner, Annj, Elena Zeni, Hamada Elsayed, Michele Di Foggia, Paola Taddei, Antonella Pasquato, Lucy Di Silvio, Enrico Bernardo, Paola Brun, and Monica Dettin. 2023. "Proteolytically Resistant Bioactive Peptide-Grafted Sr/Mg-Doped Hardystonite Foams: Comparison of Two Covalent Functionalization Strategies" Biomimetics 8, no. 2: 185. https://doi.org/10.3390/biomimetics8020185
APA StyleZamuner, A., Zeni, E., Elsayed, H., Di Foggia, M., Taddei, P., Pasquato, A., Di Silvio, L., Bernardo, E., Brun, P., & Dettin, M. (2023). Proteolytically Resistant Bioactive Peptide-Grafted Sr/Mg-Doped Hardystonite Foams: Comparison of Two Covalent Functionalization Strategies. Biomimetics, 8(2), 185. https://doi.org/10.3390/biomimetics8020185












