The Impact of Green Physical Crosslinking Methods on the Development of Sericin-Based Biohydrogels for Wound Healing
Abstract
:1. Introduction
2. Experimental Section
2.1. Materials
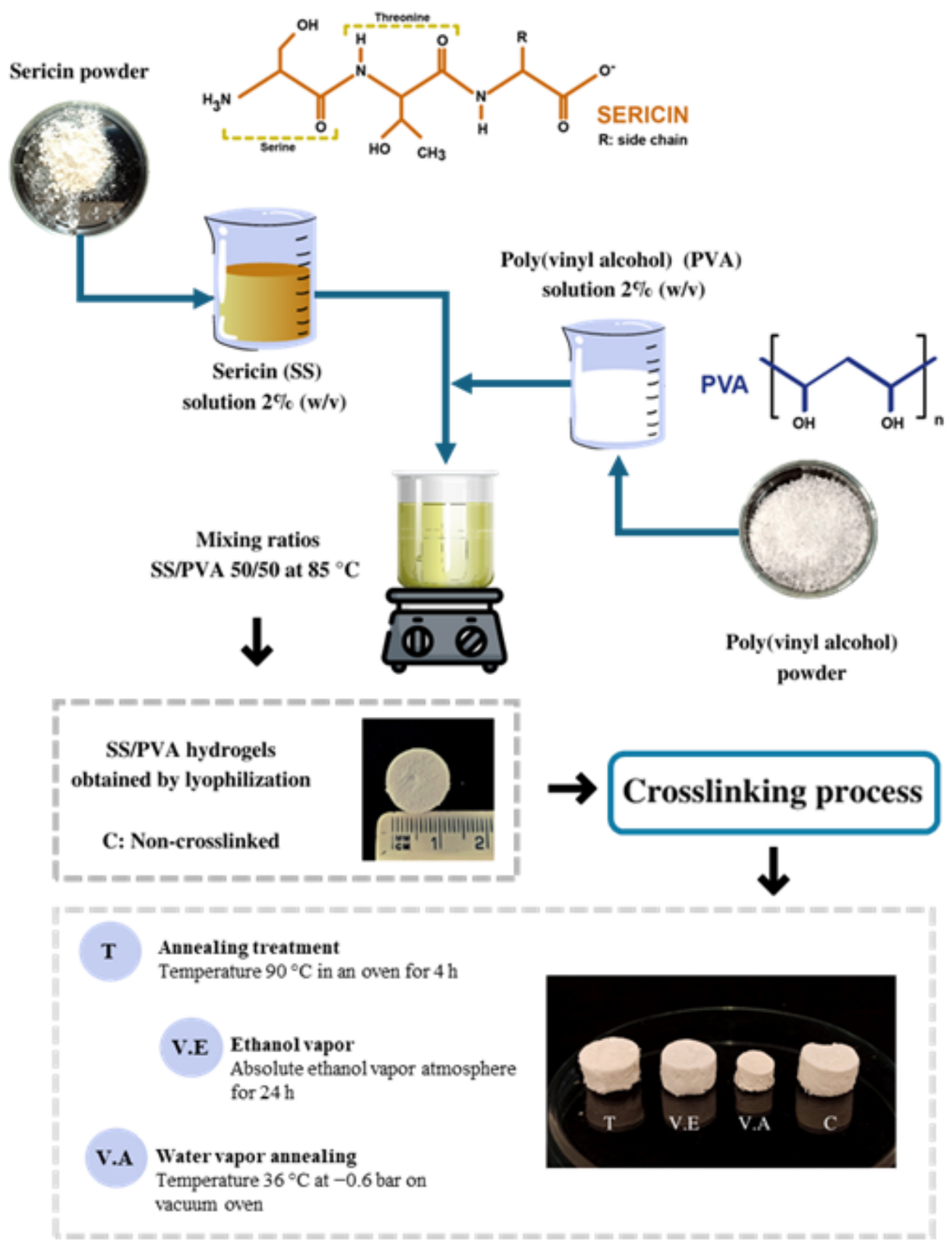
2.2. Preparation of Sericin Powder
2.3. Preparation of SS/PVA Hydrogels
2.4. Crosslinking of SS/PVA Hydrogels
2.5. Characterization Methods
2.5.1. X-ray Diffraction
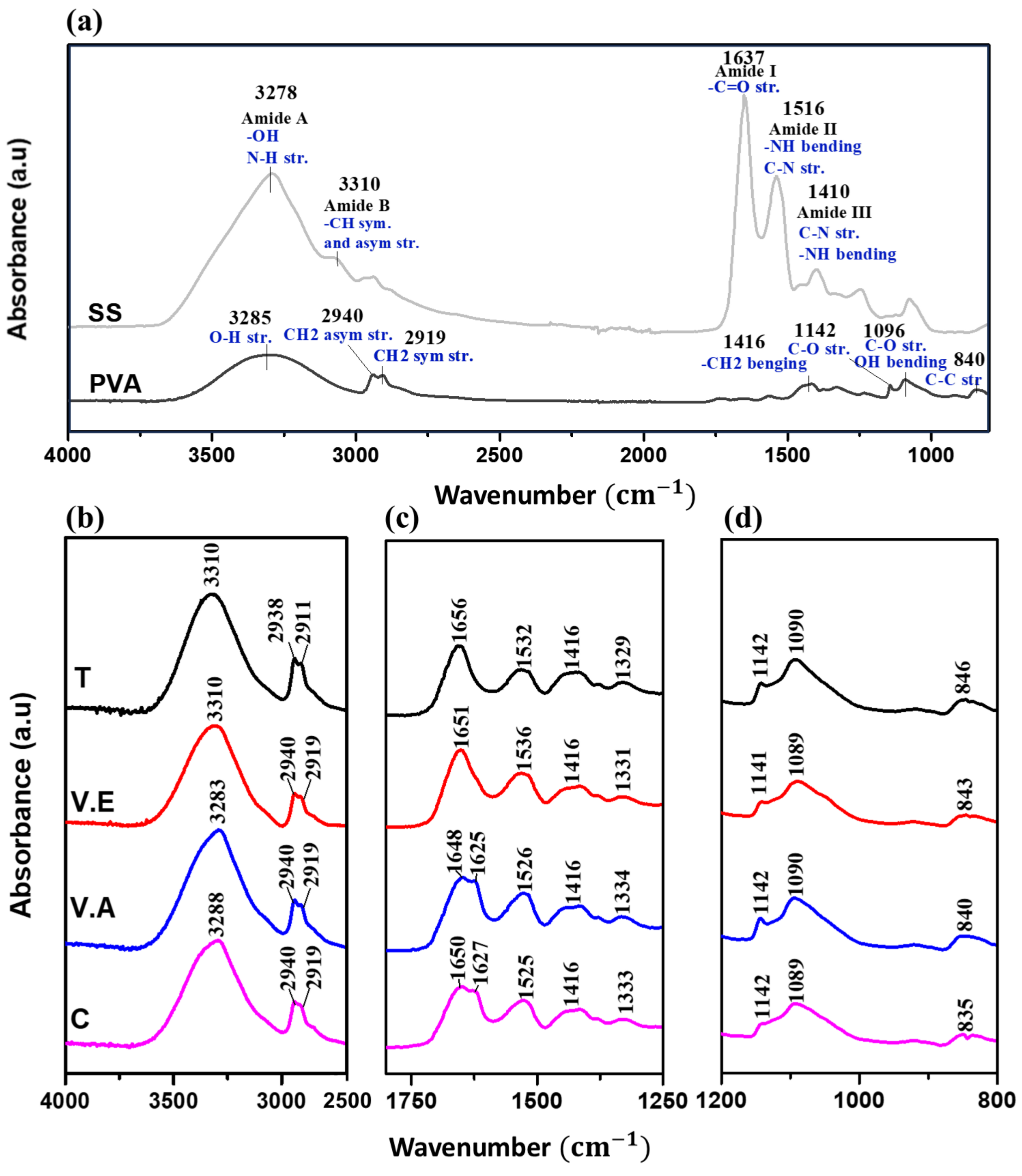
2.5.2. Fourier-Transform Infrared Spectroscopy (FTIR)
2.5.3. Scanning Electron Microscopy (SEM)
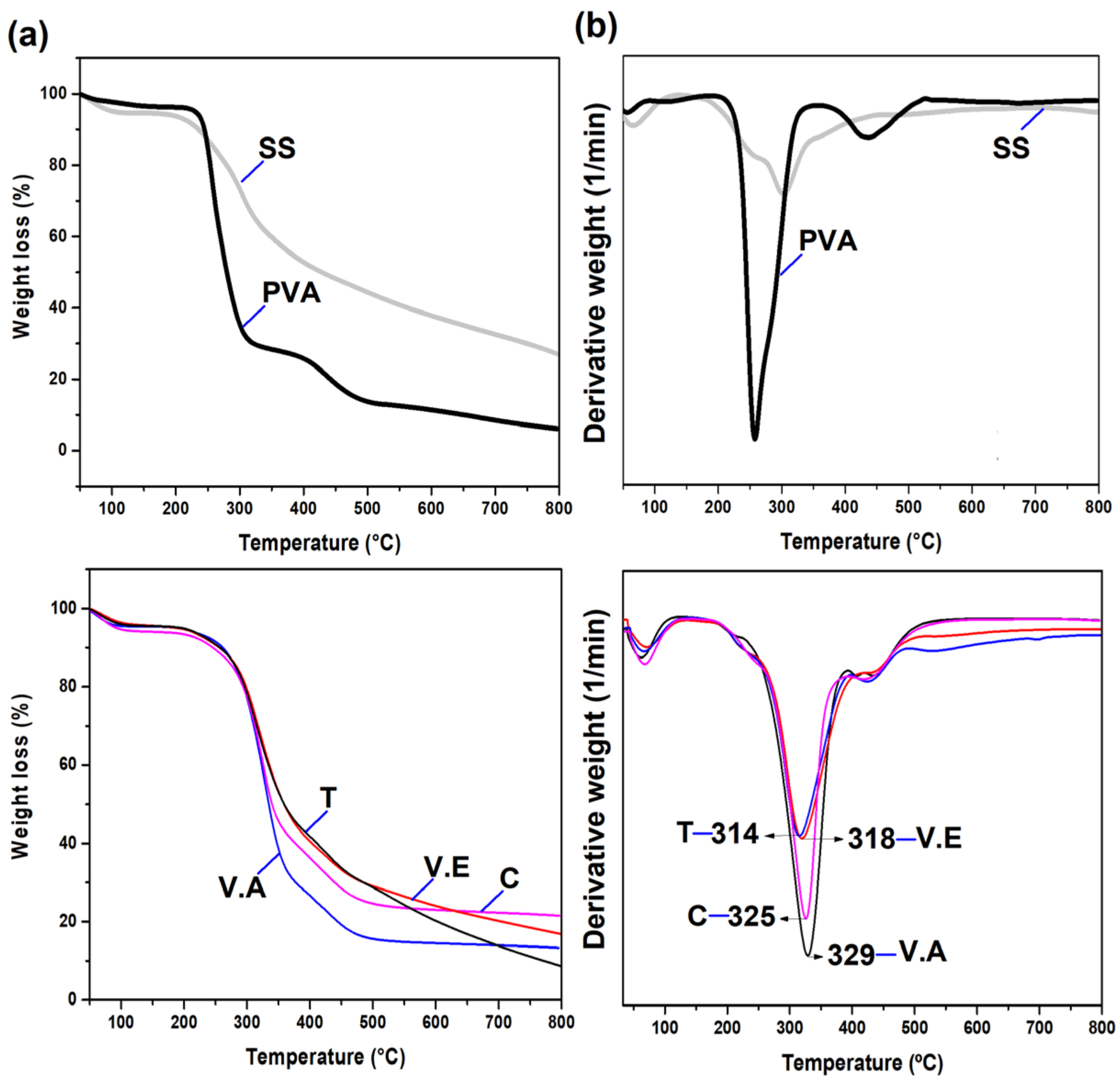
2.5.4. Thermogravimetric Analysis (TGA)
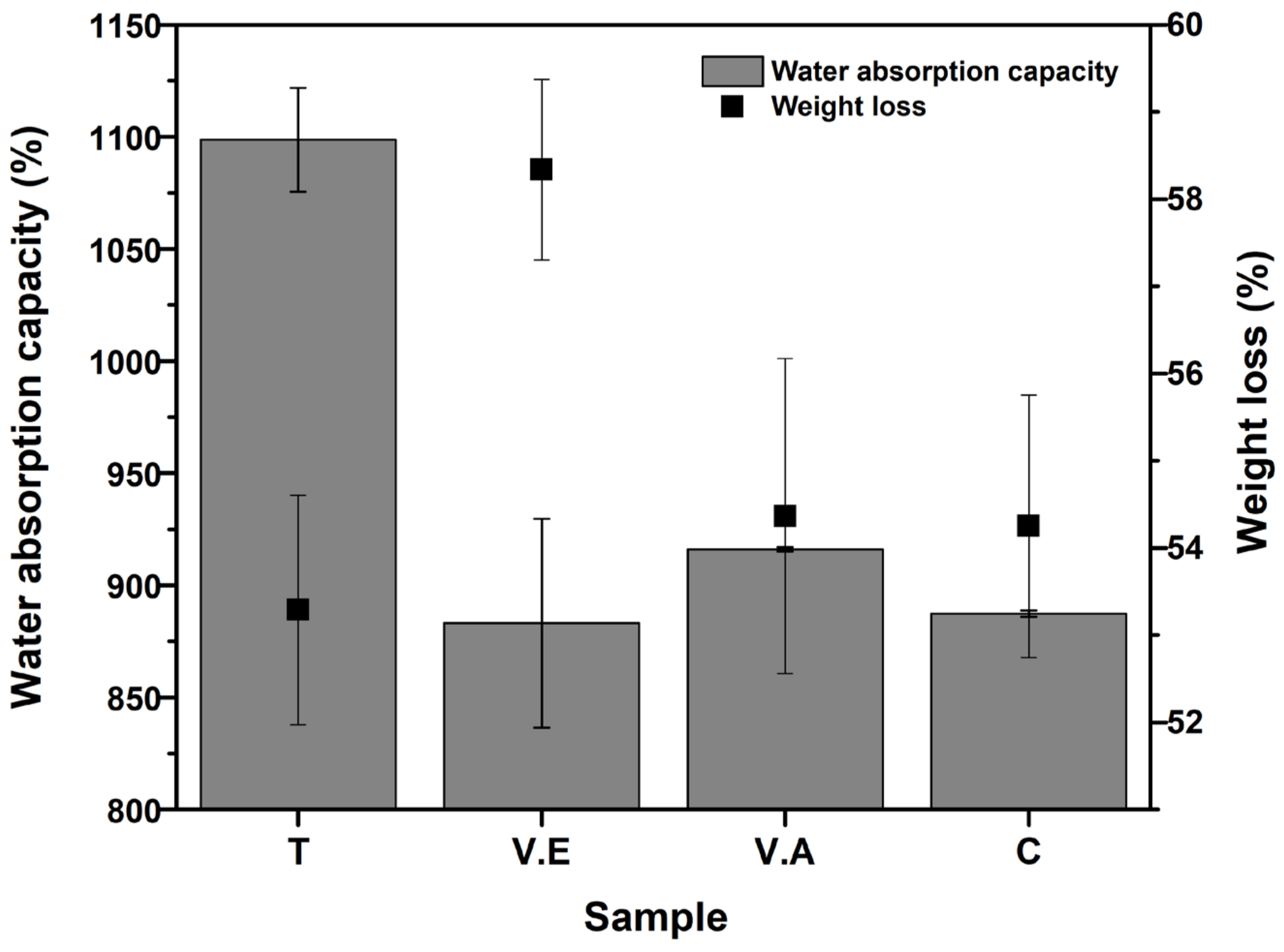
2.5.5. Water Absorption Capacity
2.5.6. Silk Sericin Release from the Hydrogels
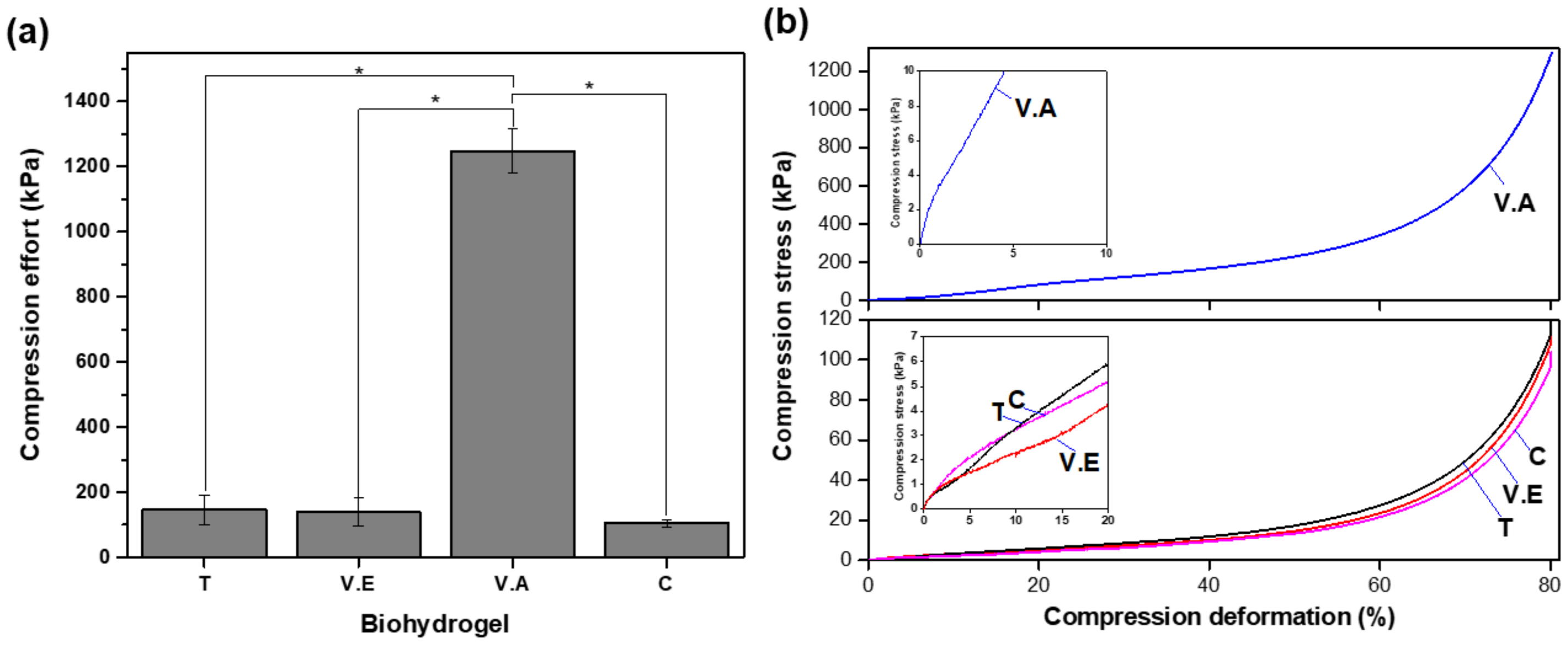
2.5.7. Compressive Strength
2.5.8. Statistical Analysis
3. Results and Discussion
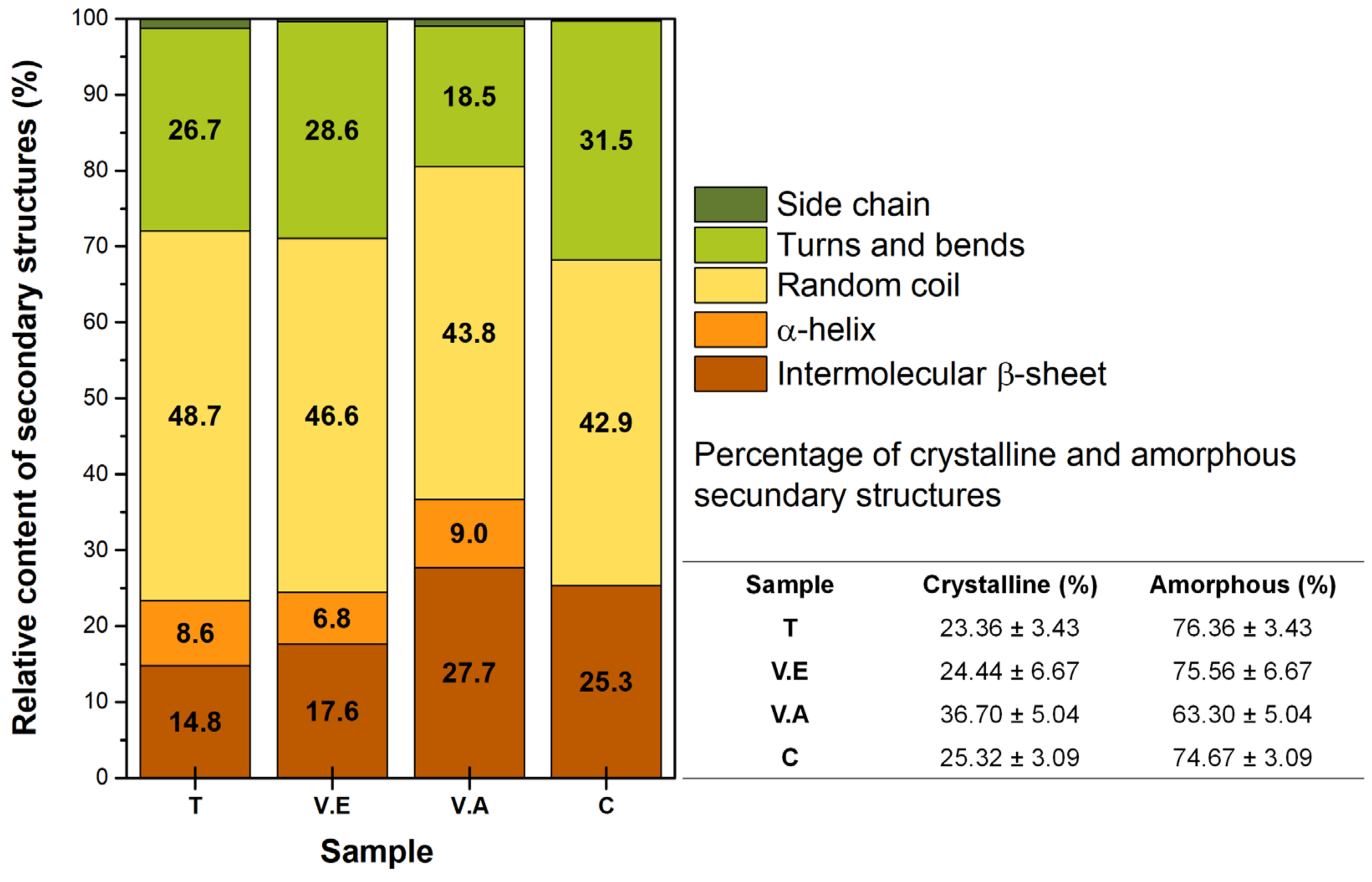
3.1. Impact of Physical Crosslinking on the Structure of Biohydrogels
3.2. Morphological Analysis
3.3. Performance Analysis
3.3.1. Compressive Strength
3.3.2. Water Absorption Capacity and Mass Loss
3.3.3. Silk Sericin Release from the Hydrogels
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Hoffman, A. Hydrogels for Biomedical Applications. Adv. Drug Deliv. Rev. 2012, 64, 18–23. [Google Scholar] [CrossRef]
- Napavichayanun, S.; Bonani, W.; Yang, Y.; Motta, A.; Aramwit, P. Fibroin and Polyvinyl Alcohol Hydrogel Wound Dressing Containing Silk Sericin Prepared Using High-Pressure Carbon Dioxide. Adv. Wound Care 2019, 8, 452–462. [Google Scholar] [CrossRef]
- Ullah, F.; Othman, M.B.H.; Javed, F.; Ahmad, Z.; Akil, H.M. Classification, Processing and Application of Hydrogels: A Review. Mater. Sci. Eng. C 2015, 57, 414–433. [Google Scholar] [CrossRef] [PubMed]
- Kamoun, E.A.; Chen, X.; Mohy Eldin, M.S.; Kenawy, E.-R.S. Crosslinked Poly(Vinyl Alcohol) Hydrogels for Wound Dressing Applications: A Review of Remarkably Blended Polymers. Arab. J. Chem. 2015, 8, 1–14. [Google Scholar] [CrossRef]
- Zhou, X.; Hao, Y.; Yuan, L.; Pradhan, S.; Shrestha, K.; Pradhan, O.; Liu, H.; Li, W. Nano-Formulations for Transdermal Drug Delivery: A Review. Chin. Chem. Lett. 2018, 29, 1713–1724. [Google Scholar] [CrossRef]
- Gonzalez, A.C.D.O.; Andrade, Z.D.A.; Costa, T.F.; Medrado, A.R.A.P. Wound Healing—A Literature Review. An. Bras. Dermatol. 2016, 91, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Chen, C.; Zhang, X.; Wen, X.; Xiao, Y.; Li, L.; Xu, Q.; Fu, F.; Diao, H.; Liu, X. Layer-by-Layer Coating of Carboxymethyl Chitosan-Gelatin-Alginate on Cotton Gauze for Hemostasis and Wound Healing. Surf. Coat. Technol. 2021, 406, 126644. [Google Scholar] [CrossRef]
- Dong, Z.; Wang, Q.; Du, Y. Alginate/Gelatin Blend Films and Their Properties for Drug Controlled Release. J. Memb Sci. 2006, 280, 37–44. [Google Scholar] [CrossRef]
- Pacheco, M.S.; Kano, G.E.; Paulo, L.d.A.; Lopes, P.S.; de Moraes, M.A. Silk Fibroin/Chitosan/Alginate Multilayer Membranes as a System for Controlled Drug Release in Wound Healing. Int. J. Biol. Macromol. 2020, 152, 803–811. [Google Scholar] [CrossRef] [PubMed]
- Kumar, J.P.; Bhardwaj, N.; Mandal, B.B. Cross-Linked Silk Sericin-Gelatin 2D and 3D Matrices for Prospective Tissue Engineering Applications. RSC Adv. 2016, 6, 105125–105136. [Google Scholar] [CrossRef]
- Aramwit, P.; Ratanavaraporn, J.; Siritientong, T. Improvement of Physical and Wound Adhesion Properties of Silk Sericin and Polyvinyl Alcohol Dressing Using Glycerin. Adv. Skin. Wound Care 2015, 28, 358–367. [Google Scholar] [CrossRef]
- Wang, S.L.; Zhuo, J.J.; Fang, S.M.; Xu, W.; Yu, Q.Y. Silk Sericin and Its Composite Materials with Antibacterial Properties to Enhance Wound Healing: A Review. Biomolecules 2024, 14, 723. [Google Scholar] [CrossRef]
- Punyamoonwongsa, P.; Klayya, S.; Sajomsang, W.; Kunyanee, C.; Aueviriyavit, S. Silk Sericin Semi-Interpenetrating Network Hydrogels Based on PEG-Diacrylate for Wound Healing Treatment. Int. J. Polym. Sci. 2019, 2019, 4740765. [Google Scholar] [CrossRef]
- Gupta, S.; Alrabaiah, H.; Christophe, M.; Rahimi-Gorji, M.; Nadeem, S.; Bit, A. Evaluation of Silk-Based Bioink during Pre and Post 3D Bioprinting: A Review. J. Biomed. Mater. Res. B Appl. Biomater. 2021, 109, 279–293. [Google Scholar] [CrossRef]
- Padamwar, M.N.; Pawar, A.P. Silk Sericin and Its Applications: A Review. J. Sci. Y Indiustrial Res. 2004, 63, 323–329. [Google Scholar]
- Wang, Z.; Wang, J.; Yang, J.; Luo, Z.; Yang, W.; Xie, H.; Huang, K.; Wang, L. Article A Neuroprotective Sericin Hydrogel as an Effective Neuronal Cell Carrier for the Repair of Ischemic Stroke. ACS Appl. Mater. Interfaces 2015, 7, 24629–24640. [Google Scholar] [CrossRef] [PubMed]
- Jaramillo-Quiceno, N.; Rueda-Mira, S.; Marín, J.F.S.; Álvarez-López, C. Development of a Novel Silk Sericin-Based Hydrogel Film by Mixture Design. J. Polym. Res. 2023, 30, 120. [Google Scholar] [CrossRef]
- Sonjan, S.; Ross, G.M.; Mahasaranon, S.; Sinkangam, B.; Intanon, S.; Ross, S. Biodegradable Hydrophilic Film of Crosslinked PVA/Silk Sericin for Seed Coating: The Effect of Crosslinker Loading and Polymer Concentration. J. Polym. Environ. 2021, 29, 323–334. [Google Scholar] [CrossRef]
- Chiellini, E.; Corti, A.; Antone, S.D.; Solaro, R. Biodegradation of Poly (Vinyl Alcohol) Based Materials. Prog. Polym. Sci. 2003, 28, 963–1014. [Google Scholar] [CrossRef]
- Bakhsheshi-rad, H.; Fauzi, A.; Aziz, M.; Akbari, M.; Hadisi, Z.; Omidi, M.; Chen, X. International Journal of Biological Macromolecules Development of the PVA/CS Nano Fi Bers Containing Silk Protein Sericin as a Wound Dressing: In Vitro and in Vivo Assessment. Int. J. Biol. Macromol. 2020, 149, 513–521. [Google Scholar] [CrossRef]
- Yamdej, R.; Pangza, K. Superior Physicochemical and Biological Properties of Poly (Vinyl Alcohol)/Sericin Hydrogels Fabricated by a. J. Bioact. Compat. Polym. 2016, 32, 32–44. [Google Scholar] [CrossRef]
- He, H.; Cai, R.; Wang, Y.; Tao, G.; Guo, P.; Zuo, H.; Chen, L.; Liu, X.; Zhao, P.; Xia, Q. Preparation and Characterization of Silk Sericin/PVA Blend Film with Silver Nanoparticles for Potential Antimicrobial Application. Int. J. Biol. Macromol. 2017, 104, 457–464. [Google Scholar] [CrossRef]
- Wang, L.C.; Chen, X.G.; Zhong, D.Y.; Xu, Q.C. Study on Poly(Vinyl Alcohol)/Carboxymethyl-Chitosan Blend Film as Local Drug Delivery System. J. Mater. Sci. Mater. Med. 2007, 18, 1125–1133. [Google Scholar] [CrossRef]
- Chen, Y.; Li, J.; Lu, J.; Ding, M.; Chen, Y. Synthesis and Properties of Poly(Vinyl Alcohol) Hydrogels with High Strength and Toughness. Polym. Test 2022, 108, 107516. [Google Scholar] [CrossRef]
- Hennink, W.E.; van Nostrum, C.F. Novel Crosslinking Methods to Design Hydrogels. Adv. Drug Deliv. Rev. 2012, 64, 223–236. [Google Scholar] [CrossRef]
- Ampawong, S.; Aramwit, P. In Vivo Safety and Efficacy of Sericin/Poly(Vinyl Alcohol)/Glycerin Scaffolds Fabricated by Freeze-Drying and Salt-Leaching Techniques for Wound Dressing Applications. J. Bioact. Compat. Polym. 2017, 32, 582–595. [Google Scholar] [CrossRef]
- Lagos, S.I.Z.; Salas, J.M.; Zapata, M.E.V.; Hernández, J.H.M.; Tovar, C.D.G. Optimization by Central Composite Experimental Design of the Synthesis of Physically Crosslinked Chitosan Spheres. Biomimetics 2020, 5, 63. [Google Scholar] [CrossRef]
- Matos, R.J.R.; Silva, J.C.; Soares, P.I.P.; Borges, J.P. Polyvinylpyrrolidone Nanofibers Incorporating Mesoporous Bioactive Glass for Bone Tissue Engineering. Biomimetics 2023, 8, 206. [Google Scholar] [CrossRef] [PubMed]
- Punjataewakupt, A.; Reddy, N.; Aramwit, P. Enhancing Clinical Applications of PVA Hydrogel by Blending with Collagen Hydrolysate and Silk Sericin. J. Polym. Res. 2022, 29, 110. [Google Scholar] [CrossRef]
- Tao, G.; Wang, Y.; Cai, R.; Chang, H.; Song, K.; Zuo, H.; Zhao, P.; Xia, Q.; He, H. Design and Performance of Sericin/Poly(Vinyl Alcohol) Hydrogel as a Drug Delivery Carrier for Potential Wound Dressing Application. Mater. Sci. Eng. C 2019, 101, 341–351. [Google Scholar] [CrossRef]
- Hu, X.; Shmelev, K.; Sun, L.; Gil, E.-S.; Park, S.-H.; Cebe, P.; Kaplan, D.L. Regulation of Silk Material Structure by Temperature-Controlled Water Vapor Annealing. Biomacromolecules 2011, 12, 1686–1696. [Google Scholar] [CrossRef]
- Serna, M.E. Ciencia Transdisciplinar En La Nueva Era; Instituto Antioqueño de Investigación: Medellín, Colombia, 2022; Volume I, ISBN 9786289513530. [Google Scholar]
- Arango, M.C.; Osorio, Y.M.; Osorno, J.B.; Parra, S.B.; Alvarez-López, C. Effect of Ethanol Post-Treatments over Sericin Scaffolds for Tissue Engineering Applications. J. Polym. Environ. 2022, 31, 1800–1811. [Google Scholar] [CrossRef]
- Apte, G.; Repanas, A.; Willems, C.; Mujtaba, A.; Schmelzer, C.E.H.; Raichur, A.; Syrowatka, F.; Groth, T. Effect of Different Crosslinking Strategies on Physical Properties and Biocompatibility of Freestanding Multilayer Films Made of Alginate and Chitosan. Macromol. Biosci. 2019, 19, 1900181. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, J.S.; Alvarez, V.A. The Effect of the Annealing on the Poly(Vinyl Alcohol) Obtained by Freezing-Thawing. Thermochim. Acta 2011, 521, 184–190. [Google Scholar] [CrossRef]
- Chen, K.; Zong, T.; Chen, Q.; Liu, S.; Xu, L.; Zhang, D. Preparation and Characterization of Polyvinyl Alcohol/Sodium Alginate/Carboxymethyl Cellulose Composite Hydrogels with Oriented Structure. Soft Mater. 2022, 20, 99–108. [Google Scholar] [CrossRef]
- Fan, L.; Wang, H.; Zhang, K.; He, C. Regenerated Silk Fibroin Nanofibrous Matrices Treated with 75% Ethanol Vapor for Tissue-Engineering Applications Regenerated Silk Fibroin Nanofibrous Matrices Treated with 75% Ethanol Vapor for Tissue-Engineering Applications. J. Biomater. Sci. 2012, 5063, 497–508. [Google Scholar] [CrossRef]
- Zuo, B.; Liu, L.; Wu, Z. Effect on Properties of Regenerated Silk Fibroin Fiber Coagulated with Aqueous Methanol/Ethanol. J. Appl. Polym. Sci. 2007, 106, 53–59. [Google Scholar] [CrossRef]
- Karahaliloglu, Z.; Kilicay, E.; Denkbas, E.B. Antibacterial Chitosan/Silk Sericin 3D Porous Scaffolds as a Wound Dressing Material. Artif. Cells Nanomed. Biotechnol. 2017, 45, 1172–1185. [Google Scholar] [CrossRef]
- Wang, Z.; Zhang, Y.; Zhang, J.; Huang, L.; Liu, J.; Li, Y.; Zhang, G.; Kundu, S.C.; Wang, L. Exploring Natural Silk Protein Sericin for Regenerative Medicine: An Injectable, Photoluminescent, Cell-Adhesive 3D Hydrogel. Sci. Rep. 2015, 4, 7064. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.; Talukdar, S.; Kundu, S.C. Potential of 2D Crosslinked Sericin Membranes with Improved Biostability for Skin Tissue Engineering. Cell Tissue Res. 2012, 347, 783–794. [Google Scholar] [CrossRef]
- Yang, J.; Xu, F.; Han, C.R. Metal Ion Mediated Cellulose Nanofibrils Transient Network in Covalently Cross-Linked Hydrogels: Mechanistic Insight into Morphology and Dynamics. Biomacromolecules 2017, 18, 1019–1028. [Google Scholar] [CrossRef]
- Aziz, S.B.; Marf, A.S.; Dannoun, E.M.A.; Brza, M.A.; Abdullah, R.M. The Study of the Degree of Crystallinity, Electrical Equivalent Circuit, and Dielectric Properties of Polyvinyl Alcohol (PVA)-Based Biopolymer Electrolytes. Polymers 2020, 12, 2184. [Google Scholar] [CrossRef] [PubMed]
- Omar, A.; Wali, A.; Arken, A.; Gao, Y.; Aisa, H.A.; Yili, A. Covalent Binding of Flavonoids with Silk Sericin Hydrolysate: Anti-inflammatory, Antioxidant, and Physicochemical Properties of Flavonoid–Sericin Hydrolysate Conjugates. J. Food Biochem. 2022, 46, e14125. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.; Kong, L.X. A Thermal Degradation Mechanism of Polyvinyl Alcohol/Silica Nanocomposites. Polym. Degrad. Stab. 2007, 92, 1061–1071. [Google Scholar] [CrossRef]
- Dash, B.C.; Mandal, B.B.; Kundu, S.C. Silk Gland Sericin Protein Membranes: Fabrication and Characterization for Potential Biotechnological Applications. J. Biotechnol. 2009, 144, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Bakadia, B.M.; Zhong, A.; Li, X.; Boni, B.O.O.; Ahmed, A.A.Q.; Souho, T.; Zheng, R.; Shi, Z.; Shi, D.; Lamboni, L.; et al. Biodegradable and Injectable Poly(Vinyl Alcohol) Microspheres in Silk Sericin-Based Hydrogel for the Controlled Release of Antimicrobials: Application to Deep Full-Thickness Burn Wound Healing. Adv. Compos. Hybrid. Mater. 2022, 5, 2847–2872. [Google Scholar] [CrossRef]
- Teramoto, H.; Mitsushiro, M. Analysis of Structural Properties and Formation of Sericin Fiber by Infrared Spectroscopu. J. Insect Biotechnol. Sericology 2003, 6, 157–162. [Google Scholar]
- Jackson, M.; Mantsch, H.H. The Use and Misuse of FTIR Spectroscopy in the Determination of Protein Structure. Crit. Rev. Biochem. Mol. Biol. 1995, 30, 95–120. [Google Scholar] [CrossRef]
- Adochitei, A.; Drochioiu, G. Rapid Characterization of Peptide Secondary Structure by FT-IR Spectroscopy. Rev. Roum. de Chim. 2011, 56, 783–791. [Google Scholar]
- Barth, A. Infrared Spectroscopy of Proteins. Biochim. Biophys. Acta Bioenerg. 2007, 1767, 1073–1101. [Google Scholar] [CrossRef]
- Mandal, B.; Priya, A.S.; Kundu, S.C. Novel Silk Sericin/Gelatin 3-D Scaffolds and 2-D Films: Fabrication and Characterization for Potential Tissue Engineering Applications. Acta Biomater. 2009, 5, 3007–3020. [Google Scholar] [CrossRef]
- Aramwit, P.; Siritientong, T.; Kanokpanont, S.; Srichana, T. Formulation and Characterization of Silk Sericin-PVA Scaffold Crosslinked with Genipin. Int. J. Biol. Macromol. 2010, 47, 668–675. [Google Scholar] [CrossRef] [PubMed]
- Enniys, L. Spectrophotometric and Turbidimetric Methods for Measuring Proteins. Methods Enzym. 1957, 3, 447–454. [Google Scholar]
- Lungu, A.; Albu, M.G.; Stancu, I.C.; Florea, N.M.; Vasile, E.; Iovu, H. Superporous Collagen-Sericin Scaffolds. J. Appl. Polym. Sci. 2013, 127, 2269–2279. [Google Scholar] [CrossRef]
- Krimm, S.; Bandekar, J. Vibrational Spectroscopy and Conformation of Peptides, Polypeptides, and Proteins. Adv. Protein Chem. 1986, 38, 181–364. [Google Scholar] [CrossRef]
- Ai, L.; He, H.; Wang, P.; Cai, R.; Tao, G.; Yang, M.; Liu, L. Rational Design and Fabrication of ZnONPs Functionalized Sericin/PVA Antimicrobial Sponge. Int. J. Mol. Sci. 2019, 20, 4796. [Google Scholar] [CrossRef]
- Kumkun, P.; Tuancharoensri, N.; Ross, G.; Mahasaranon, S.; Jongjitwimol, J.; Topham, P.D.; Ross, S. Green Fabrication Route of Robust, Biodegradable Silk Sericin and Poly(Vinyl Alcohol) Nanofibrous Scaffolds. Polym. Int. 2019, 68, 1903–1913. [Google Scholar] [CrossRef]
- Jipa, I.M.; Stoica, A.; Stroescu, M.; Dobre, L.M.; Dobre, T.; Jinga, S.; Tardei, C. Potassium Sorbate Release from Poly(Vinyl Alcohol)-Bacterial Cellulose Films. Chem. Pap. 2012, 66, 138–143. [Google Scholar] [CrossRef]
- Jaramillo-Quiceno, N.; Callone, E.; Dirè, S.; Álvarez-López, C.; Motta, A. Boosting Sericin Extraction through Alternative Silk Sources. Polym. J. 2021, 53, 1425–1437. [Google Scholar] [CrossRef]
- Ou, K.; Dong, X.; Qin, C.; Ji, X.; He, J. Properties and Toughening Mechanisms of PVA/PAM Double-Network Hydrogels Prepared by Freeze-Thawing and Anneal-Swelling. Mater. Sci. Eng. C 2017, 77, 1017–1026. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Xu, J.; Zhong, F.; Rotello, V.M. Strategies for Fabricating Protein Films for Biomaterial Applications. Adv. Sustain. Syst. 2021, 5, 2000167. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.J.; Park, J.; Karageorgiou, V.; Kim, U.J.; Valluzzi, R.; Cebe, P.; Kaplan, D.L. Water-Stable Silk Films with Reduced β-Sheet Content. Adv. Funct. Mater. 2005, 15, 1241–1247. [Google Scholar] [CrossRef]
- Callone, E.; Dirè, S.; Hu, X.; Motta, A. Processing Influence on Molecular Assembling and Structural Conformations in Silk Fibroin: Elucidation by Solid-State NMR. ACS Biomater. Sci. Eng. 2016, 2, 758–767. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, B.D.; Omenetto, F.; Chui, K.; Kaplan, D.L. Processing Methods to Control Silk Fibroin Film Biomaterial Features. J. Mater. Sci. 2008, 43, 6967–6985. [Google Scholar] [CrossRef]
- Elzayat, A.M.; Adam-Cervera, I.; Albus, M.; Cháfer, A.; Badia, J.D.; Pérez-Pla, F.F.; Muñoz-Espí, R. Polysaccharide/Silica Microcapsules Prepared via Ionic Gelation Combined with Spray Drying: Application in the Release of Hydrophilic Substances and Catalysis. Polymers 2023, 15, 4116. [Google Scholar] [CrossRef] [PubMed]
- Begum, M.H.A.; Hossain, M.M.; Gafur, M.A.; Kabir, A.N.M.H.; Tanvir, N.I.; Molla, M.R. Preparation and Characterization of Polyvinyl Alcohol–Starch Composites Reinforced with Pulp. SN Appl. Sci. 2019, 1, 1091. [Google Scholar] [CrossRef]
- Wei, Z.; Dai, S.; Huang, J.; Hu, X.; Ge, C.; Zhang, X.; Yang, K.; Shao, P.; Sun, P.; Xiang, N. Soy Protein Amyloid Fibril Scaffold for Cultivated Meat Application. ACS Appl. Mater. Interfaces 2023, 15, 15108–15119. [Google Scholar] [CrossRef]
- Zhang, K.; Ye, Q.; Yan, Z. Influence of Post-Treatment with 75% (v/v) Ethanol Vapor on the Properties of SF/P (LLA-CL) Nanofibrous Scaffolds. Int. J. Mol. Sci. 2012, 13, 2036–2047. [Google Scholar] [CrossRef]
- Lamboni, L.; Gauthier, M.; Yang, G.; Wang, Q. Silk Sericin: A Versatile Material for Tissue Engineering and Drug Delivery. Biotechnol. Adv. 2015, 33, 1855–1867. [Google Scholar] [CrossRef]
- Sun, H.; Maji, S.; Chandrakasan, A.P.; Marelli, B. Integrating Biopolymer Design with Physical Unclonable Functions for Anticounterfeiting and Product Traceability in Agriculture. Sci. Adv. 2023, 9, eadf1978. [Google Scholar] [CrossRef]
- Kwak, H.W.; Lee, H.; Lee, M.E.; Jin, H. Facile and Green Fabrication of Silk Sericin Films Reinforced with Bamboo-Derived Cellulose Nano Fi Brils. J. Clean Prod. 2018, 200, 1034–1042. [Google Scholar] [CrossRef]
- Aramwit, P.; Siritienthong, T.; Srichana, T.; Ratanavaraporn, J. Accelerated Healing of Full-Thickness Wounds by Genipin-Crosslinked Silk Sericin/PVA Scaffolds. Cells Tissues Organs 2013, 197, 224–238. [Google Scholar] [CrossRef] [PubMed]
- Aramwit, P.; Yamdech, R.; Ampawong, S. Controlled Release of Chitosan and Sericin from the Microspheres-Embedded Wound Dressing for the Prolonged Anti-Microbial and Wound Healing Efficacy. AAPS J. 2016, 18, 647–658. [Google Scholar] [CrossRef] [PubMed]
- Azeredo, H.M.C.; Waldron, K.W. Crosslinking in Polysaccharide and Protein Films and Coatings for Food Contact—A Review. Trends Food Sci. Technol. 2016, 52, 109–122. [Google Scholar] [CrossRef]
- Siritientong, T.; Angspatt, A.; Ratanavaraporn, J.; Aramwit, P. Clinical Potential of a Silk Sericin-Releasing Bioactive Wound Dressing for the Treatment of Split-Thickness Skin Graft Donor Sites. Pharm. Res. 2014, 31, 104–116. [Google Scholar] [CrossRef]
- Das, G.; Shin, H.S.; Campos, E.V.R.; Fraceto, L.F.; del Pilar Rodriguez-Torres, M.; Mariano, K.C.F.; de Araujo, D.R.; Fernández-Luqueño, F.; Grillo, R.; Patra, J.K. Sericin Based Nanoformulations: A Comprehensive Review on Molecular Mechanisms of Interaction with Organisms to Biological Applications. J. Nanobiotechnol. 2021, 19, 30. [Google Scholar] [CrossRef]
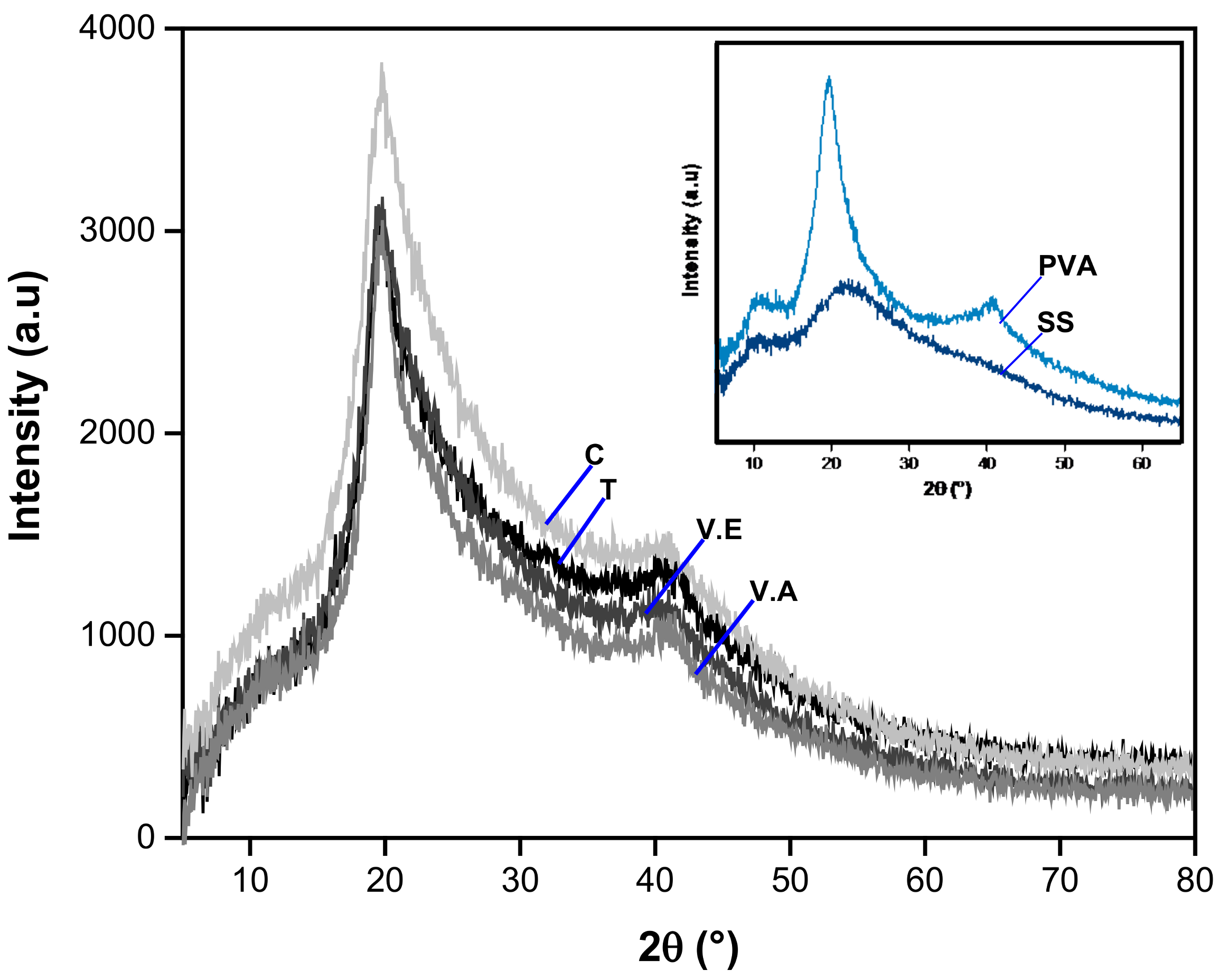
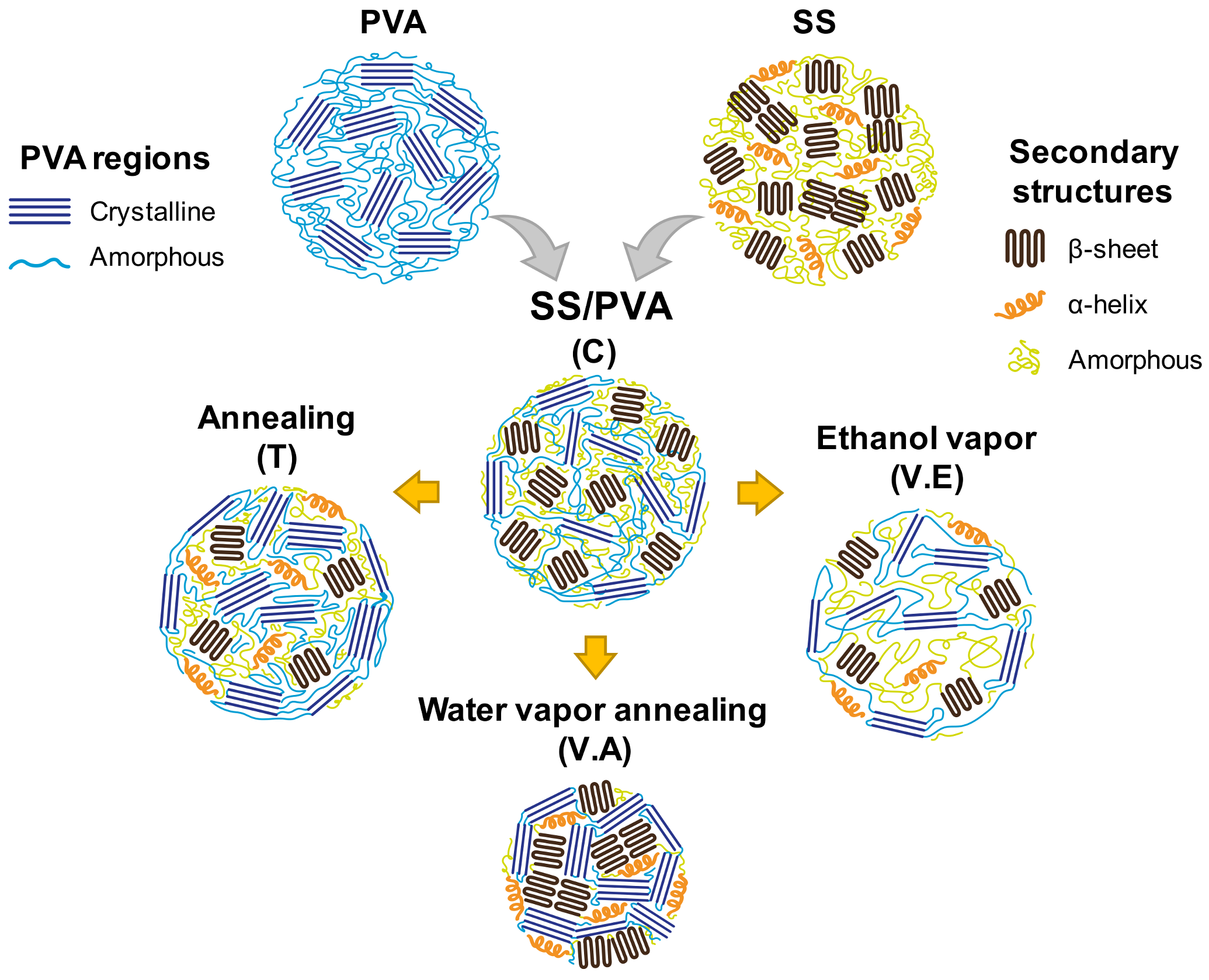



| Region | Diffraction Peak (2θ) | Polymer |
|---|---|---|
| Crystalline | 12.0 | SS and PVA (overlapping) |
| 19.7 | PVA | |
| 22.0 | SS and PVA (overlapping) | |
| 40.6 | PVA | |
| Amorphous | 31.0 | PVA |
| 44.0 | SS |
| Assignment | Wavenumber Ranges (cm−1) | Type of Structure |
|---|---|---|
| Side chain | 1590–1605 | Amorphous |
| Intermolecular β-sheet | 1610–1625 and 1697–1700 | Crystalline |
| Random coil | 1636–1645 | Amorphous |
| α-helix | 1658–1664 | Crystalline |
| Turns and bends | 1666–1695 | Amorphous |
| Type of Hydrogel | Average Pore Diameter (mm) | Morphology |
|---|---|---|
| T | Does not apply | Heterogeneous pores—interlayer voids |
| V.E | 11.03 ± 8.26 | Elongated pores—interlayer contraction |
| V.A | 10.08 ± 6.65 | Elongated pores—interlayer contraction |
| C | Does not apply | Heterogeneous pores—small cavities |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arango, M.C.; Jaramillo-Quiceno, N.; Badia, J.D.; Cháfer, A.; Cerisuelo, J.P.; Álvarez-López, C. The Impact of Green Physical Crosslinking Methods on the Development of Sericin-Based Biohydrogels for Wound Healing. Biomimetics 2024, 9, 497. https://doi.org/10.3390/biomimetics9080497
Arango MC, Jaramillo-Quiceno N, Badia JD, Cháfer A, Cerisuelo JP, Álvarez-López C. The Impact of Green Physical Crosslinking Methods on the Development of Sericin-Based Biohydrogels for Wound Healing. Biomimetics. 2024; 9(8):497. https://doi.org/10.3390/biomimetics9080497
Chicago/Turabian StyleArango, Maria C., Natalia Jaramillo-Quiceno, José David Badia, Amparo Cháfer, Josep Pasqual Cerisuelo, and Catalina Álvarez-López. 2024. "The Impact of Green Physical Crosslinking Methods on the Development of Sericin-Based Biohydrogels for Wound Healing" Biomimetics 9, no. 8: 497. https://doi.org/10.3390/biomimetics9080497








