Mosquito Fauna and Spatial Distribution in an Atlantic Forest Area in Rio de Janeiro State, Brazil, Reveal a High Risk of Transmission of Yellow Fever and Other Arboviruses
Abstract
1. Introduction
2. Material and Methods
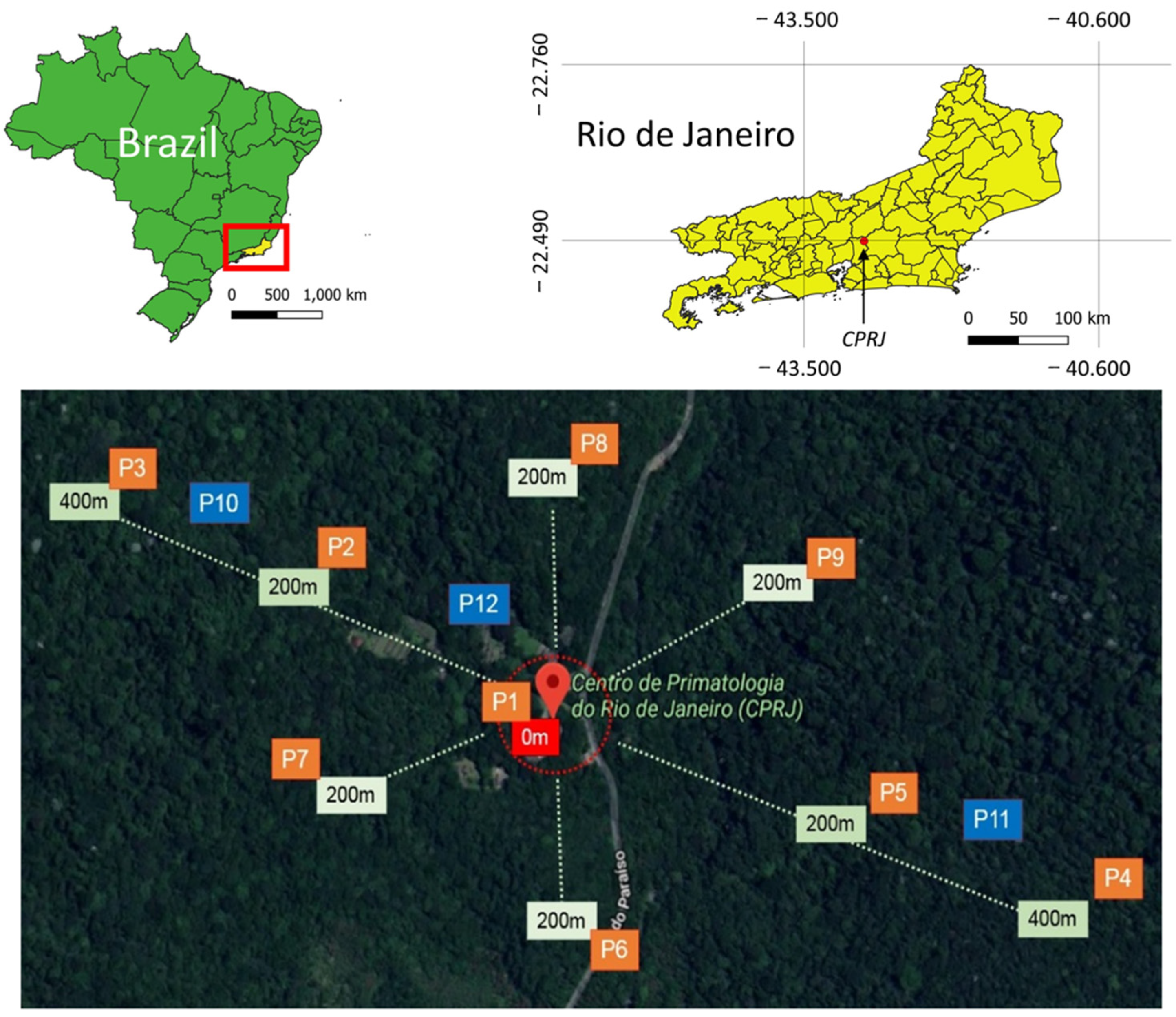
2.1. Study Area
2.2. Screening for YFV Natural Infections
2.3. Statistical Analysis
3. Results
| Genus/Species | Adults | Ovitrap | Total | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protected Human Attraction | BG-Sentinel | Total | Paddle | Water | Total | |||||||||
| Aedes aegypti (Linnaeus) | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 14 | 0.3% | 0 | 0.0% | 14 | 0.2% | 14 | 0.1% |
| Aedes albopictus (Skuse) | 8 | 1.7% | 2 | 0.1% | 10 | 0.3% | 11 | 0.2% | 2 | 0.2% | 13 | 0.2% | 23 | 0.2% |
| Aedes scapularis (Rondani) | 59 | 12.9% | 63 | 1.9% | 122 | 3.3% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 122 | 1.3% |
| Aedes terrens (Walker) | 4 | 0.9% | 8 | 0.2% | 12 | 0.3% | 788 | 16.5% | 0 | 0.0% | 788 | 14.0% | 800 | 8.6% |
| Anopheles cruzii (Dyar & Knab) | 1 | 0.2% | 0 | 0.0% | 1 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 1 | 0.0% |
| Culex urichii (Coquillett) | 0 | 0.0% | 1 | 0.0% | 1 | 0.0% | 0 | 0.0% | 444 | 52.7% | 444 | 7.9% | 445 | 4.8% |
| Culex (Mcx.) sp. | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 7 | 0.8% | 7 | 0.1% | 7 | 0.1% |
| Culex (Culex) spp. | 41 | 9.0% | 3030 | 92.5% | 3071 | 82.3% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 3071 | 32.8% |
| Haemagogus janthinomys Dyar/Hg. capricornii Lutz | 54 | 11.8% | 3 | 0.1% | 57 | 1.5% | 509 | 10.7% | 0 | 0.0% | 509 | 9.1% | 566 | 6.1% |
| Haemagogus leucocelaenus (Dyar & Shannon) | 99 | 21.6% | 17 | 0.5% | 116 | 3.1% | 3452 | 72.3% | 13 | 1.5% | 3465 | 61.7% | 3581 | 38.3% |
| Limatus durhamii Theobald | 11 | 2.4% | 2 | 0.1% | 13 | 0.3% | 0 | 0.0% | 60 | 7.1% | 60 | 1.1% | 73 | 0.8% |
| Limatus pseudomethysticus (Bonne-Wepster & Bonne) | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 311 | 36.9% | 311 | 5.5% | 311 | 3.3% |
| Mansonia titillans (Walker) | 0 | 0.0% | 6 | 0.2% | 6 | 0.2% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 6 | 0.1% |
| Mansonia sp. | 0 | 0.0% | 1 | 0.0% | 1 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 1 | 0.0% |
| Psorophora ferox (Humboldt) | 1 | 0.2% | 0 | 0.0% | 1 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 1 | 0.0% |
| Runchomyia cerqueirai (Stone) | 2 | 0.4% | 0 | 0.0% | 2 | 0.1% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 2 | 0.0% |
| Runchomyia frontosa (Theobald) | 24 | 5.2% | 27 | 0.8% | 51 | 1.4% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 51 | 0.5% |
| Runchomyia humboldti (Lane & Cerqueira) | 17 | 3.7% | 18 | 0.5% | 35 | 0.9% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 35 | 0.4% |
| Runchomyia reversa (Lane & Cerqueira) | 17 | 3.7% | 15 | 0.5% | 32 | 0.9% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 32 | 0.3% |
| Runchomyia sp. | 20 | 4.4% | 19 | 0.6% | 39 | 1.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 39 | 0.4% |
| Sabethes albiprivus Theobald | 3 | 0.7% | 0 | 0.0% | 3 | 0.1% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 3 | 0.0% |
| Sabethes chloropterus (Humboldt) | 21 | 4.6% | 0 | 0.0% | 21 | 0.6% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 21 | 0.2% |
| Toxorhynchites sp. | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 5 | 0.6% | 5 | 0.1% | 5 | 0.1% |
| Tricoprosopon sp. | 0 | 0.0% | 1 | 0.0% | 1 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 1 | 0.0% |
| Wyeomyia incaudata (Root) | 2 | 0.4% | 0 | 0.0% | 2 | 0.1% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 2 | 0.0% |
| Wyeomyia theobaldi) (Lane & Cerqueira) | 26 | 5.7% | 0 | 0.0% | 26 | 0.7% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 26 | 0.3% |
| Wyeomyia confusa (Lutz) | 0 | 0.0% | 1 | 0.0% | 1 | 0.0% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 1 | 0.0% |
| Wyeomyia spp. | 48 | 10.5% | 61 | 1.9% | 109 | 2.9% | 0 | 0.0% | 0 | 0.0% | 0 | 0.0% | 109 | 1.2% |
| Total | 458 | 100.0% | 3275 | 100.0% | 3733 | 100.0% | 4774 | 100.0% | 842 | 100.0% | 5616 | 100.0% | 9349 | 100.0% |
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Barrett, A.D.; Monath, T.P. Epidemiology and ecology of yellow fever virus. Adv. Virus Res. 2003, 61, 291–315. [Google Scholar] [CrossRef] [PubMed]
- Possas, C.; Lourenço-De-oliveira, R.; Tauil, P.L.; de Paula Pinheiro, F.; Pissinatti, A.; da Cunha, R.V.; Freire, M.; Martins, R.M.; Homma, A. Yellow fever outbreak in Brazil: The puzzle of rapid viral spread and challenges for immunisation. Memórias Do Inst. Oswaldo Cruz 2018, 113, e180278. [Google Scholar] [CrossRef]
- Staples, J.E.; Monath, T.P. Yellow Fever: 100 Years of Discovery. JAMA 2008, 27, 300–960. [Google Scholar] [CrossRef] [PubMed]
- Secretaria de Vigilância em Saúde. Manual de Vigilância de Epizootias em Primatas Não-Humanos, 2nd ed.; Ministério da Saúde: Brasília, Brazil, 2014. Available online: https://bvsms.saude.gov.br/bvs/publicacoes/guia_vigilancia_epizootias_primatas_entomologia.pdf (accessed on 21 May 2020).
- de Abreu, F.V.S.; Ferreira-de-Brito, A.; de Souza Azevedo, A.; Linhares, J.H.R.; de Oliveira Santos, V.; Hime Miranda, E.; Neves, M.S.A.S.; Yousfi, L.; Ribeiro, I.P.; dos Santos, A.A.C.; et al. Survey on Non-Human Primates and Mosquitoes Does not Provide Evidences of Spillover/Spillback between the Urban and Sylvatic Cycles of Yellow Fever and Zika Viruses Following Severe Outbreaks in Southeast Brazil. Viruses 2020, 12, 364. [Google Scholar] [CrossRef]
- Arnell, J.H. Mosquito studies (Diptera, Culicidae). XXXII. A revision of the genus Haemagogus. Contrib. Am. Entomol. Inst. 1973, 10, 1–174. [Google Scholar]
- Consoli, R.A.G.B.; de Oliveira, R.L. Principais Mosquitos de Importância Sanitária no Brasil; Fiocruz: Rio de Janeiro, Brazil, 1994; ISBN 85-85676-03-5. [Google Scholar]
- de Abreu, F.V.S.; Ribeiro, I.P.; Ferreira-de-Brito, A.; dos Santos, A.A.C.; de Miranda, R.M.; de Souza Bonelly, I.; Neves, M.S.A.S.; Bersot, M.I.; dos Santos, T.P.; Gomes, M.Q.; et al. Haemagogus leucocelaenus and Haemagogus janthinomys are the primary vectors in the major yellow fever outbreak in Brazil, 2016–2018. Emerg. Microbes Infect. 2019, 8, 218–231. [Google Scholar] [CrossRef] [PubMed]
- Delatorre, E.; de Abreu, F.V.S.; Ribeiro, I.P.; Gomez, M.M.; dos Santos, A.A.C.; Ferreira-de-Brito, A.; Neves, M.S.A.S.; Bonelly, I.; de Miranda, R.M.; Furtado, N.D.; et al. Distinct YFV lineages co-circulated in the Central-Western and Southeastern regions of Brazil from 2015 to 2018. Front. Microbiol. 2019, 10, 1079. [Google Scholar] [CrossRef]
- de Castro Gomes, A.; Torres, M.A.N.; de Paula, M.B.; Fernandes, A.; Marassá, A.M.; Consales, C.A.; Fonseca, D.F. Ecologia de Haemagogus e Sabethes (Diptera: Culicidae) em áreas epizoóticas do vírus da febre amarela, Rio Grande do Sul, Brasil. Epidemiol. Serviços Saúde 2010, 19, 101–113. [Google Scholar] [CrossRef]
- MMA Ministério do Meio Ambiente. Biodiversidade. 2019. Available online: http://www.mma.gov.br/biodiversidade (accessed on 21 May 2020).
- Myers, N.; Mittermeier, R.A.; Mittermeier, C.G.; da Fonseca, G.A.B.; Kent, J. Biodiversity hotspots for conservation priorities. Nature 2000, 403, 853–858. [Google Scholar] [CrossRef]
- Guedes, M.P. Culicidae (Diptera) No Brasil: Relações Entre Diversidade, Distribuição e Enfermidades. Oecol. Aust. 2012, 16, 283–296. [Google Scholar] [CrossRef]
- Scolforo, J.R.; Oliveira, A.D.; Filho, A.C.F.; Mello, J.M. Diversidade, equabilidade e similaridade no Domínio Atlântico. In Inventário Florestal de Minas Gerais: Floresta Estacional Semidecidual e Ombrófila; UFLA: Lavras, Brazil, 2008; pp. 337–390. [Google Scholar]
- Hamlet, A.; Ramos, D.G.; Gaythorpe, K.A.M.; Romano, A.P.M.; Garske, T.; Ferguson, N.M. Seasonality of agricultural exposure as an important predictor of seasonal yellow fever spillover in Brazil. Nat. Commun. 2021, 12, 3647. [Google Scholar] [CrossRef]
- Ilacqua, R.C.; Medeiros-Sousa, A.R.; Ramos, D.G.; Obara, M.T.; Ceretti-Junior, W.; Mucci, L.F.; Marrelli, M.T.; Laporta, G.Z. Reemergence of Yellow Fever in Brazil: The Role of Distinct Landscape Fragmentation Thresholds. J. Environ. Public Health 2021, 2021, 8230789. [Google Scholar] [CrossRef]
- de Abreu, F.V.S.; de Andreazzi, C.S.; Neves, M.S.A.S.; Meneguete, P.S.; Ribeiro, M.S.; Dias, C.M.G.; de Albuquerque Motta, M.; Barcellos, C.; Romão, A.R.; Magalhães, M.d.A.F.M.; et al. Ecological and environmental factors affecting transmission of sylvatic yellow fever in the 2017–2019 outbreak in the Atlantic Forest, Brazil. Parasites Vectors 2022, 15, 23. [Google Scholar] [CrossRef]
- da Silva Fernandes, A.T.; Moreira, S.B.; Gaspar, L.P.; Simões, M.; dos Reis Albuquerque Cajaraville, A.C.; Pereira, R.C.; de Barros Gomes, M.P.; Linhares, J.H.R.; de Oliveira Santos, V.; Santos, R.T.; et al. Safety and immunogenicity of 17DD attenuated yellow fever vaccine in howler monkeys (Alouatta spp.). J. Med. Primatol. 2021, 50, 36–45. [Google Scholar] [CrossRef]
- Fay, R.W.; Eliason, D.A. A perfered ovposition site as surveillance method for Aedes aegypti. Mosq. News 1966, 26, 531–534. [Google Scholar]
- Correa, R.R.; Ramalho, G.R. Revisão de Phoniomyia Theobald, 1903 (Diptera, Culicidae, Sabethini). Folia Clin. Biol. 1956, 25, 1–176. [Google Scholar]
- Forattini, O.P. Culicidologia Médica: Identificação, Biologia, Epidemiologia; Edusp—Editora da Universidade de São Paulo: São Paulo, Brazil, 2002. [Google Scholar]
- Marcondes, C.B.; Alencar, J. Revisão de mosquitos Haemagogus Williston (Diptera: Culicidae) do Brasil. Rev. Biomed. 2010, 21, 221–238. [Google Scholar]
- Domingo, C.; Patel, P.; Yillah, J.; Weidmann, M.; Méndez, J.A.; Nakouné, E.R.; Niedrig, M. Advanced Yellow Fever Virus Genome Detection in Point-of-Care Facilities and Reference Laboratories. J. Clin. Microbiol. 2012, 50, 4054–4060. [Google Scholar] [CrossRef] [PubMed]
- de Abreu, F.V.S.; Delatorre, E.; dos Santos, A.A.C.; Ferreira-de-Brito, A.; de Castro, M.G.; Ribeiro, I.P.; Furtado, N.D.; Vargas, W.P.; Ribeiro, M.S.; Meneguete, P.; et al. Combination of surveillance tools reveals that Yellow Fever virus can remain in the same Atlantic Forest area at least for three transmission seasons. Memórias Do Inst. Oswaldo Cruz 2019, 114, e190076. [Google Scholar] [CrossRef]
- Shannon, C.E. A Mathematical Theory of Communication. Bell Syst. Tech. J. 1948, 27, 379–423. [Google Scholar] [CrossRef]
- Hammer, Ø.; Harper, D.A.T.; Ryan, P.D. PAST: Paleontological Statistics Software Package for Education and Data Analysis PAST: Paleontological Statistics Software Package for Education and Data Analysis. Palaeontol. Electron. 2001, 4, 1–9. [Google Scholar]
- Montes, J. Fauna de Culicidae da Serra da Cantareira, São Paulo, Brasil. Rev. Saúde Pública 2005, 39, 578–584. [Google Scholar] [CrossRef] [PubMed]
- Guimarães, A.É.; Arlé, M. Mosquitos no Parque Nacional da Serra dos Órgãos, estado do Rio de Janeiro, Brasil: I-distribuição estacional. Memórias Do Inst. Oswaldo Cruz 1984, 79, 309–323. [Google Scholar] [CrossRef]
- dos Santos Silva, J.; Couri, M.S.; de Leão Giupponi, A.P.; Alencar, J. Mosquito fauna of the Guapiaçu Ecological Reserve, Cachoeiras de Macacu, Rio de Janeiro, Brazil, collected under the influence of different color CDC light traps. J. Vector Ecol. 2014, 39, 384–394. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Franco, O. História da Frebre Amarela no Brasil; Ministério da Saúde, Dep. Nac. Endem. Rurais: Rio e Janeiro, Brazil, 1969. [Google Scholar]
- Vasconcelos, P.F.; Costa, Z.G.; Da Rosa, E.S.T.; Luna, E.; Rodrigues, S.G.; Barros, V.L.; Dias, J.P.; Monteiro, H.A.; Oliva, O.F.; Vasconcelos, H.B.; et al. Epidemic of jungle yellow fever in Brazil, 2000: Implications of climatic alterations in disease spread. J. Med. Virol. 2001, 65, 598–604. [Google Scholar] [CrossRef]
- Davis, N.C.; Shannon, R.C. Studies on Yellow Fever in South America: V. Transmission Experiments With certain species of Culex and Aedes. J. Exp. Med. 1929, 50, 803–808. [Google Scholar] [CrossRef]
- Whitman, L.; Antunes, P.C.A. Studies on the Capacity of Various Brazilian Mosquitoes, Representing the Genera Psorophora, Aedes, Mansonia, and Culex, to Transmit Yellow Fever 1. Am. J. Trop. Med. Hyg. 1937, 17, 803–823. [Google Scholar] [CrossRef]
- da Cruz Cardoso, J.; de Almeida, M.A.B.; dos Santos, E.; da Fonseca, D.F.; Sallum, M.A.M.; Noll, C.A.; de Oliveira Monteiro, H.A.; Cruz, A.C.R.; Carvalho, V.L.; Pinto, E.V.; et al. Yellow fever virus in Haemagogus leucocelaenus and Aedes serratus mosquitoes, Southern Brazil, 2008. Emerg. Infect. Dis. 2010, 16, 1918–1924. [Google Scholar] [CrossRef]
- Mascheretti, M.; Tengan, C.H.; Sato, H.K.; Suzuki, A.; de Souza, R.P.; Maeda, M.; Brasil, R.; Pereira, M.; Tubaki, R.M.; Wanderley, D.M.V.; et al. Febre amarela silvestre: Reemergência de transmissão no estado de São Paulo, Brasil, 2009. Rev. Saúde Pública 2013, 47, 881–889. [Google Scholar] [CrossRef][Green Version]
- Pinheiro, G.G.; Rocha, M.N.; de Oliveira, M.A.; Moreira, L.A.; Filho, J.D.A. Detection of yellow fever virus in sylvatic mosquitoes during disease outbreaks of 2017–2018 in Minas Gerais State, Brazil. Insects 2019, 10, 136. [Google Scholar] [CrossRef]
- Soper, F.L. Yellow fever: The present situation (October 1938) with special reference to South America. Trans. R. Soc. Trop. Med. Hyg. 1938, 32, 297–322. [Google Scholar] [CrossRef]
- Kumm, H.W.; Cerqueira, N.L. The Role of Aëdes leucocelaenus in the Epidemiology of Jungle Yellow Fever in Brazil. Bull. Entomol. Res. 1951, 42, 195–199. [Google Scholar] [CrossRef]
- Vasconcelos, P.F.C.; Sperb, A.F.; Monteiro, H.A.O.; Torres, M.A.N.; Sousa, M.R.S.; Vasconcelos, H.B.; Mardini, L.B.L.F.; Rodrigues, S.G. Isolations of yellow fever virus from Haemagogus leucocelaenus in Rio Grande do Sul State, Brazil. Trans. R. Soc. Trop. Med. Hyg. 2003, 97, 60–62. [Google Scholar] [CrossRef] [PubMed]
- de Souza, R.P.; Petrella, S.; Coimbra, T.L.M.; Maeda, A.Y.; Rocco, I.M.; Bisordi, I.; Silveira, V.R.; Pereira, L.E.; Suzuki, A.; Dos Santos Silva, S.J.; et al. Isolation of yellow fever virus (YFV) from naturally infected Haemagogus (Conopostegus) leucocelaenus (diptera, culicidae) in São Paulo state, Brazil, 2009. Rev. Inst. Med. Trop. São Paulo 2011, 53, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Causey, O.R.; Laemmert, H.W.; Kumm, H.W. Dispersion of Forest Mosquitoes in Brazil: Further Studies 1. Am. J. Trop. Med. Hyg. 1950, 30, 301–312. [Google Scholar] [CrossRef] [PubMed]
- Hervé, J. Instituto Evandro Chagas: 50 Anos de Contribuição às Ciências Biológicas e à Medicina Tropical; Instituto Evandro Chagas: Belém, Brazil, 1986. [Google Scholar]
- Moreira, G.V.; Peixoto, C.; Ziccardi, M.; Lourenço-de-Oliveira, R.; Castro, M.G.; Dionisio, D.; Pissinati, A. Prevalence of Trypanosoma cruzi, Trypanosoma minasense and antibodies against arbovirus on non human primates (Callitrichidae) in captivity life. Ver. Bras. Med. Vet. 2000, 22, 252–254. [Google Scholar]
- de Miranda, R.M.; Fernandes, R.S.; da Silva-Fernandes, A.T.; Ferreira-de-Brito, A.; Moreira, S.B.; Pereira, R.C.; da Silva Mendes, Y.; de Lima, S.M.B.; Pissinatti, A.; Freire, M.d.S.; et al. Neotropical sylvatic mosquitoes and Aedes aegypti are not competent to transmit 17DD attenuated Yellow Fever Virus from vaccinated viremic New World non-human primates. Viruses 2022, 14, 2231. [Google Scholar] [CrossRef]
| Ecological Indices | Protected Human Attraction | BG-Sentinel | Ovitrap | |
|---|---|---|---|---|
| Paddle | Water | |||
| Richness (S) | 17 | 13 | 5 | 5 |
| Specimens | 369 | 182 | 4774 | 830 |
| Dominance (D) | 0.14 | 0.18 | 0.56 | 0.43 |
| Pielou equability (J) | 0.80 | 0.79 | 0.50 | 0.60 |
| Shannon’s diversity (H’) | 2.26 | 2.02 | 0.80 | 0.97 |
| Adults and Immatures | Immatures | Adults | Total | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 | P10 | P11 | P12 | |||
| Ovitrap | Water | 75 | 20 | 109 | 160 | 35 | 94 | 63 | 113 | 173 | - | - | - | 842 |
| 8.9% | 2.4% | 12.9% | 19.0% | 4.2% | 11.2% | 7.5% | 13.4% | 20.5% | - | - | - | 100.0% | ||
| Paddles | 534 | 573 | 1468 | 223 | 326 | 73 | 515 | 853 | 209 | - | - | - | 4774 | |
| 11.2% | 12.0% | 30.7% | 4.7% | 6.8% | 1.5% | 10.8% | 17.9% | 4.4% | - | - | - | 100.0% | ||
| Adult captures | BG-sentinel | 432 | 441 | 298 | 418 | 1686 | - | - | - | - | - | - | - | 3275 |
| 13.2% | 13.5% | 9.1% | 12.8% | 51.5% | - | - | - | - | - | - | - | 100.0% | ||
| Protected human attraction | - | - | - | - | - | - | - | - | - | 103 | 221 | 134 | 458 | |
| - | - | - | - | - | - | - | - | - | 22.5% | 48.3% | 29.3% | 100.0% | ||
| Species/Collection Point | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 | P10 | P11 | P12 | Total |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Aedes aegypti | 0 | 0 | 12 | 0 | 0 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 14 |
| Aedes albopictus | 0 | 0 | 5 | 1 | 0 | 0 | 4 | 0 | 5 | 2 | 0 | 6 | 23 |
| Aedes scapularis | 10 | 19 | 6 | 4 | 24 | 0 | 0 | 0 | 0 | 15 | 19 | 25 | 122 |
| Aedes terrens | 361 | 6 | 301 | 26 | 14 | 0 | 84 | 0 | 4 | 0 | 3 | 1 | 800 |
| Anopheles cruzii | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 1 |
| Culex urichi | 41 | 15 | 61 | 81 | 12 | 4 | 39 | 74 | 118 | 0 | 0 | 0 | 445 |
| Culex (Mcx.) sp. | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 6 | 0 | 0 | 0 | 7 |
| Culex (Culex) spp. | 403 | 397 | 263 | 373 | 1594 | 0 | 0 | 0 | 0 | 5 | 23 | 13 | 3071 |
| Haemagogus janthinomys/capricornii | 29 | 74 | 78 | 50 | 82 | 0 | 83 | 95 | 21 | 13 | 22 | 19 | 566 |
| Haemagogus leucocelaenus | 147 | 498 | 1096 | 154 | 233 | 73 | 344 | 756 | 181 | 35 | 44 | 20 | 3581 |
| Limatus durhamii | 19 | 0 | 17 | 15 | 1 | 10 | 0 | 0 | 0 | 4 | 2 | 5 | 73 |
| Limatus pseudomethysticus | 15 | 4 | 18 | 66 | 21 | 80 | 23 | 37 | 47 | 0 | 0 | 0 | 311 |
| Mansonia titillans | 1 | 1 | 0 | 2 | 2 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 6 |
| Mansonia sp. | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Psorophora ferox | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 0 | 0 | 1 |
| Runchomyia cerqueirai | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 0 | 2 |
| Runchomyia frontosa | 2 | 0 | 12 | 2 | 11 | 0 | 0 | 0 | 0 | 1 | 8 | 15 | 51 |
| Runchomyia humboldti | 0 | 2 | 5 | 0 | 11 | 0 | 0 | 0 | 0 | 7 | 4 | 6 | 35 |
| Runchomyia reversa | 2 | 2 | 0 | 1 | 10 | 0 | 0 | 0 | 0 | 0 | 10 | 7 | 32 |
| Runchomyia sp. | 4 | 6 | 0 | 7 | 2 | 0 | 0 | 0 | 0 | 7 | 7 | 6 | 39 |
| Sabethes albiprivus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 | 1 | 1 | 3 |
| Sabethes chloropterus | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 3 | 15 | 3 | 21 |
| Toxorhynchites sp. | 0 | 1 | 0 | 0 | 2 | 0 | 0 | 2 | 0 | 0 | 0 | 0 | 5 |
| Tricoprosopon sp. | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Wyeomyia (Pho.) incaudata | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 2 | 2 |
| Wyeomyia (Pho.) teoboldti | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 26 | 0 | 26 |
| Wyeomyia confusa | 0 | 0 | 0 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 1 |
| Wyeomyia sp. | 7 | 8 | 1 | 17 | 28 | 0 | 0 | 0 | 0 | 8 | 35 | 5 | 109 |
| Total | 1041 | 1034 | 1875 | 801 | 2047 | 167 | 578 | 966 | 382 | 103 | 221 | 134 | 9349 |
| Diversity indices | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 | P10 | P11 | P12 | |
| Richness (S) | 13 | 14 | 13 | 17 | 15 | 4 | 7 | 6 | 7 | 15 | 17 | 16 | |
| Specimens | 1041 | 1034 | 1875 | 801 | 2047 | 167 | 578 | 966 | 382 | 103 | 221 | 134 | |
| Shannon’s diversity (H’) | 1.48 | 1.21 | 1.33 | 1.69 | 0.87 | 0.97 | 1.22 | 0.77 | 1.3 | 2.16 | 2.43 | 2.4 | |
| Shannon’s equitability (J) | 0.57 | 0.46 | 0.52 | 0.60 | 0.32 | 0.70 | 0.63 | 0.43 | 0.67 | 0.80 | 0.86 | 0.87 |
| IS | P1 | P2 | P3 | P4 | P5 | P6 | P7 | P8 | P9 | P10 | P11 | P12 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P1 | * | 0.74 | 0.69 | 0.80 | 0.79 | 0.33 | 0.40 | 0.32 | 0.40 | 0.57 | 0.67 | 0.69 |
| P2 | * | 0.59 | 0.71 | 0.83 | 0.33 | 0.48 | 0.50 | 0.48 | 0.48 | 0.58 | 0.60 | |
| P3 | * | 0.67 | 0.64 | 0.47 | 0.60 | 0.53 | 0.60 | 0.57 | 0.53 | 0.62 | ||
| P4 | * | 0.81 | 0.38 | 0.50 | 0.35 | 0.50 | 0.63 | 0.65 | 0.73 | |||
| P5 | * | 0.42 | 0.45 | 0.48 | 0.45 | 0.60 | 0.69 | 0.71 | ||||
| P6 | * | 0.55 | 0.60 | 0.55 | 0.21 | 0.19 | 0.20 | |||||
| P7 | * | 0.62 | 0.86 | 0.36 | 0.25 | 0.35 | ||||||
| P8 | * | 0.62 | 0.19 | 0.17 | 0.18 | |||||||
| P9 | * | 0.27 | 0.25 | 0.35 | ||||||||
| P10 | * | 0.81 | 0.84 | |||||||||
| P11 | * | 0.85 | ||||||||||
| P12 | * |
| Mosquito Species | Number of Tested Specimens |
|---|---|
| Aedes albopictus | 4 |
| Aedes scapularis | 73 |
| Aedes terrens | 7 |
| Hg. janthinomys/capricornii | 57 |
| Hg. leucocelaenus | 116 |
| Culex sp. | 2 |
| Runchomyia sp. | 5 |
| Sabethes albiprivus | 1 |
| Sabethes chloropterus | 14 |
| Wyeomyia (Pho.) sp. | 8 |
| Total | 287 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Miranda, R.M.; Ferreira-de-Brito, A.; Silva, J.d.S.; Xavier, A.d.S.; Freitas Silva, S.O.; Alencar, J.; Lourenço-de-Oliveira, R. Mosquito Fauna and Spatial Distribution in an Atlantic Forest Area in Rio de Janeiro State, Brazil, Reveal a High Risk of Transmission of Yellow Fever and Other Arboviruses. Trop. Med. Infect. Dis. 2022, 7, 410. https://doi.org/10.3390/tropicalmed7120410
de Miranda RM, Ferreira-de-Brito A, Silva JdS, Xavier AdS, Freitas Silva SO, Alencar J, Lourenço-de-Oliveira R. Mosquito Fauna and Spatial Distribution in an Atlantic Forest Area in Rio de Janeiro State, Brazil, Reveal a High Risk of Transmission of Yellow Fever and Other Arboviruses. Tropical Medicine and Infectious Disease. 2022; 7(12):410. https://doi.org/10.3390/tropicalmed7120410
Chicago/Turabian Stylede Miranda, Rafaella Moraes, Anielly Ferreira-de-Brito, Júlia dos Santos Silva, Alexandre da Silva Xavier, Shayenne Olsson Freitas Silva, Jeronimo Alencar, and Ricardo Lourenço-de-Oliveira. 2022. "Mosquito Fauna and Spatial Distribution in an Atlantic Forest Area in Rio de Janeiro State, Brazil, Reveal a High Risk of Transmission of Yellow Fever and Other Arboviruses" Tropical Medicine and Infectious Disease 7, no. 12: 410. https://doi.org/10.3390/tropicalmed7120410
APA Stylede Miranda, R. M., Ferreira-de-Brito, A., Silva, J. d. S., Xavier, A. d. S., Freitas Silva, S. O., Alencar, J., & Lourenço-de-Oliveira, R. (2022). Mosquito Fauna and Spatial Distribution in an Atlantic Forest Area in Rio de Janeiro State, Brazil, Reveal a High Risk of Transmission of Yellow Fever and Other Arboviruses. Tropical Medicine and Infectious Disease, 7(12), 410. https://doi.org/10.3390/tropicalmed7120410







