Challenges and the Way forward in Diagnosis and Treatment of Tuberculosis Infection
Abstract
:1. Introduction
2. Screening Tests for TBI
2.1. Skin Tests
2.2. Interferon-Gamma Release Assays (IGRAs)
2.2.1. QuantiFERON®-TB Gold-In Tube (QFT®-GIT), QuantiFERON®-TB Gold Plus (QFT®-Plus) and QIAreachTM QuantiFERON®-TB (QIAreachTM)
2.2.2. T-SPOT®.TB and T-Cell SelectTM
2.2.3. Beijing Wantai’s TB-IGRA (Wantai)
2.2.4. TS-SPOT
2.2.5. LIOFeron®TB/LTBI
2.3. Comparison of TST, QFT®, and T-SPOT®.TB in Screening of TBI among Different Populations
2.3.1. Children
2.3.2. Elderly
2.3.3. Immunocompromised Individuals
2.3.4. BCG-Vaccinated
2.3.5. High-Endemic TB Countries
2.3.6. Healthcare Workers
3. Evaluation of Host-Derived Biomarkers
3.1. Cytokines/Chemokines
3.1.1. Interferon-γ (IFN-γ)
3.1.2. Interleukin-2 (IL-2)
3.1.3. IFN-γ-Inducible Protein 10 kDa (IP-10)
3.1.4. Tumor Necrosis Factor-α (TNF-α)
3.1.5. Interleukin-10 (IL-10)
3.1.6. Vascular Endothelial Growth Factors (VEGF)
3.2. mRNAs and microRNAs
3.3. T-Cell Subsets
3.4. Gene Polymorphisms
3.5. Host Circulating Proteins and Metabolites
4. Evaluation of Mtb-Derived Biomarkers
4.1. Mtb Latency Antigens
4.2. Mtb Antigens Used for Serodiagnostic
4.3. Detection of Mtb DNA in TBI
4.4. Detection of Mtb Antigens in TBI
5. Clinical and Epidemiological Scoring
6. Treatment for TBI
6.1. Standard TBI Therapy
6.2. TBI Treatment in MDR Strains
7. Conclusions and Perspectives
- Unstimulated and stimulated multiplexed cytokine analysis instead of standalone marker-based on IFN-γ.
- Addition of Mtb latency Ags as stimulating Ags, and optimization of Ag stimulating time (from 24 to 72 h).
- Screening on TBI serum for Mtb Ags and specific Abs to Mtb secreted and latency Ags for the development of rapid diagnostic kits.
- Use of flow cytometry for simultaneous detection of T cell subsets and their signature cytokines.
- Study on mRNAs and microRNAs as diagnostics and therapeutics candidates for TB.
- Identification of markers not only for diagnostic purposes, but also able to assess the TB progression or reactivation risk.
- A non-invasive approach using urine for the detection of Mtb or host-related biomarkers.
- Use of Mtb-specific Ags or epitopes for development of skin test reagents.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- WHO. Global Tuberculosis Report 2021: Incidence of Tuberculosis (per 100,000 people). The World Bank. 2021. Available online: https://data.worldbank.org/indicator/SH.TBS.INCD?most_recent_value_desc=true (accessed on 10 May 2022).
- Floyd, K.; Glaziou, P.; Zumla, A.; Raviglione, M. The global tuberculosis epidemic and progress in care, prevention, and research: An overview in year 3 of the End TB era. Lancet Respir. Med. 2018, 6, 299–314. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.; Ippolito, G.; Mfinanga, S.; Ntoumi, F.; Yeboah-Manu, D.; Vilaplana, C.; Zumla, A.; Maeurer, M. Improving treatment outcomes for MDR-TB–Novel host-directed therapies and personalised medicine of the future. Int. J. Infect. Dis. 2019, 80, S62–S67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. Fact Sheets: Tuberculosis; World Health Organization: Geneva, Switzerland, 2020; Available online: http://www.who.int/en/news-room/fact-sheets/detail/tuberculosis (accessed on 10 June 2022).
- Silva, S.; Arinaminpathy, N.; Atun, R.; Goosby, E.; Reid, M. Economic impact of tuberculosis mortality in 120 countries and the cost of not achieving the Sustainable Development Goals tuberculosis targets: A full-income analysis. Lancet Glob. Health 2021, 9, e1372–e1379. [Google Scholar] [CrossRef]
- Alliance, T. Drug resistance: A response to Antimicrobial Resistance Includes Tackling TB. Available online: https://www.tballiance.org/why-new-tb-drugs/antimicrobial-resistance (accessed on 10 June 2022).
- WHO. WHO Consolidated Guidelines on Tuberculosis: Module 3: Diagnosis: Tests for TB Infection; World Health Organization: Geneva, Switzerland, 2022; Available online: https://www.who.int/publications/i/item/9789240056084 (accessed on 20 January 2023).
- Cohen, A.; Mathiasen, V.D.; Schön, T.; Wejse, C. The global prevalence of latent tuberculosis: A systematic review and meta-analysis. Eur. Respir. J. 2019, 54, 1900655. [Google Scholar] [CrossRef] [PubMed]
- Boom, W.H.; Schaible, U.E.; Achkar, J.M. The knowns and unknowns of latent Mycobacterium tuberculosis infection. J. Clin. Investig. 2021, 131, e136222. [Google Scholar] [CrossRef] [PubMed]
- Ai, J.-W.; Ruan, Q.-L.; Liu, Q.-H.; Zhang, W.-H. Updates on the risk factors for latent tuberculosis reactivation and their managements. Emerg. Microbes Infect. 2016, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Houben, R.M.G.J.; Dodd, P.J. The global burden of latent tuberculosis infection: A re-estimation using mathematical modelling. PLoS Med. 2016, 13, e1002152. [Google Scholar] [CrossRef] [Green Version]
- Knight, G.M.; McQuaid, C.F.; Dodd, P.J.; Houben, R.M.G.J. Global burden of latent multidrug-resistant tuberculosis: Trends and estimates based on mathematical modelling. Lancet Infect. Dis. 2019, 19, 903–912. [Google Scholar] [CrossRef] [Green Version]
- Reichler, M.R.; Khan, A.; Yuan, Y.; Chen, B.; McAuley, J.; Mangura, B.; Sterling, T.R.; Team, T.E.S.C.T.O. Duration of exposure among close contacts of patients with infectious tuberculosis and risk of latent tuberculosis infection. Clin. Infect. Dis. 2020, 71, 1627–1634. [Google Scholar] [CrossRef]
- WHO. Latent Tuberculosis Infection: Updated and Consolidated Guidelines for Programmatic Management; World Health Organization: Geneva, Switzerland, 2018; Available online: https://apps.who.int/iris/handle/10665/260233 (accessed on 20 June 2022).
- Hamada, Y.; Cirillo, D.M.; Matteelli, A.; Penn-Nicholson, A.; Rangaka, M.X.; Ruhwald, M. Tests for tuberculosis infection: Landscape analysis. Eur. Respir. J. 2021, 58, 2100167. [Google Scholar] [CrossRef]
- Force, U.P.S.T. Screening for latent tuberculosis infection in adults: US Preventive Services Task Force Recommendation Statement. JAMA 2016, 316, 962–969. [Google Scholar] [CrossRef]
- Stein, C.M.; Nsereko, M.; Malone, L.L.; Okware, B.; Kisingo, H.; Nalukwago, S.; Chervenak, K.; Mayanja-Kizza, H.; Hawn, T.R.; Boom, W.H. Long-term stability of resistance to latent Mycobacterium tuberculosis infection in highly exposed tuberculosis household contacts in Kampala, Uganda. Clin. Infect. Dis. 2019, 68, 1705–1712. [Google Scholar] [CrossRef] [PubMed]
- Verrall, A.J.; Alisjahbana, B.; Apriani, L.; Novianty, N.; Nurani, A.C.; van Laarhoven, A.; Ussher, J.E.; Indrati, A.; Ruslami, R.; Netea, M.G.; et al. Early clearance of Mycobacterium tuberculosis: The INFECT case contact cohort study in Indonesia. J. Infect. Dis. 2020, 221, 1351–1360. [Google Scholar] [CrossRef] [PubMed]
- WHO. Rapid Communication: TB Antigen-Based Skin Tests for the Diagnosis of TB Infection; World Health Organization: Geneva, Switzerland, 2022; Available online: https://www.who.int/publications/i/item/WHO-UCN-TB-2022.1 (accessed on 20 January 2023).
- WHO. Use of Alternative Interferon-Gamma Release Assays for the Diagnosis of TB Infection: WHO Policy Statement; World Health Organization: Geneva, Switzerland, 2022; Available online: https://www.who.int/publications/i/item/9789240042346 (accessed on 20 January 2023).
- WHO. Implementing Tuberculosis Diagnostics: Policy Framework; World Health Organization: Geneva, Switzerland, 2015; Available online: https://apps.who.int/iris/handle/10665/16271 (accessed on 20 January 2023).
- WHO. WHO Consolidated Guidelines on Tuberculosis: Module 3: Diagnosis: Rapid Diagnostics for Tuberculosis Detection, 2021 Update; World Health Organization: Geneva, Switzerland, 2021; Available online: https://www.who.int/publications/i/item/9789240029415 (accessed on 20 January 2023).
- CDC. Fact Sheets: Tuberculin Skin Testing; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2020. Available online: https://www.cdc.gov/tb/publications/factsheets/testing/skintesting.htm (accessed on 30 June 2022).
- Nayak, S.; Acharjya, B. Mantoux test and its interpretation. Indian Dermatol. Online J. 2012, 3, 2–6. [Google Scholar] [CrossRef] [PubMed]
- van Pinxteren Laurens, A.H.; Ravn, P.; Agger Else, M.; Pollock, J.; Andersen, P. Diagnosis of tuberculosis based on the two specific antigens ESAT-6 and CFP10. Clin. Diagn. Lab. Immunol. 2000, 7, 155–160. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruhwald, M.; Aggerbeck, H.; Gallardo, R.V.; Hoff, S.T.; Villate, J.I.; Borregaard, B.; Martinez, J.A.; Kromann, I.; Penas, A.; Anibarro, L.L.; et al. Safety and efficacy of the C-Tb skin test to diagnose Mycobacterium tuberculosis infection, compared with an interferon γ release assay and the tuberculin skin test: A phase 3, double-blind, randomised, controlled trial. Lancet Respir. Med. 2017, 5, 259–268. [Google Scholar] [CrossRef]
- Weldingh, K.; Andersen, P. ESAT-6/CFP10 skin test predicts disease in M. tuberculosis-infected guinea pigs. PLoS ONE 2008, 3, e1978. [Google Scholar] [CrossRef] [Green Version]
- Arend, S.M.; Franken, W.P.J.; Aggerbeck, H.; Prins, C.; van Dissel, J.T.; Thierry-Carstensen, B.; Tingskov, P.N.; Weldingh, K.; Andersen, P. Double-blind randomized Phase I study comparing rdESAT-6 to tuberculin as skin test reagent in the diagnosis of tuberculosis infection. Tuberculosis 2008, 88, 249–261. [Google Scholar] [CrossRef]
- Bergstedt, W.; Tingskov, P.N.; Thierry-Carstensen, B.; Hoff, S.T.; Aggerbeck, H.; Thomsen, V.O.; Andersen, P.; Andersen, A.B. First-in-man open clinical trial of a combined rdESAT-6 and rCFP-10 tuberculosis specific skin test reagent. PLoS ONE 2010, 5, e11277. [Google Scholar] [CrossRef]
- Slogotskaya, L.; Litvinov, V.; Kochetkov, Y.; Ovsyankina, E.; Kudlay, D.; Seltsovsky, P.; Nikolenko, N.; Ivanova, D. New skin test with recombinant protein CFP10-ESAT6 in patients (children and adults) with tuberculosis, non-tuberculosis disease and latent TB infection. Eur. Respir. J. 2012, 40, P416. [Google Scholar]
- Slogotskaya, L.; Bogorodskaya, E.; Ivanova, D.; Makarova, M.; Guntupova, L.; Litvinov, V.; Seltsovsky, P.; Kudlay, D.; Nikolenko, N. Sensitivity and specificity of new skin test with recombinant protein CFP10-ESAT6 in patients with tuberculosis and individuals with non- tuberculosis diseases. Eur. Respir. J. 2013, 42, 1995. [Google Scholar]
- Slogotskaya, L.; Bogorodskaya, E.; Ivanova, D.; Sevostyanova, T. Comparative sensitivity of the test with tuberculosis recombinant allergen, containing ESAT6-CFP10 protein, and Mantoux test with 2 TU PPD-L in newly diagnosed tuberculosis children and adolescents in Moscow. PLoS ONE 2018, 13, e0208705. [Google Scholar] [CrossRef] [PubMed]
- Hoff, S.T.; Peter, J.G.; Theron, G.; Pascoe, M.; Tingskov, P.N.; Aggerbeck, H.; Kolbus, D.; Ruhwald, M.; Andersen, P.; Dheda, K. Sensitivity of C-Tb: A novel RD-1-specific skin test for the diagnosis of tuberculosis infection. Eur. Respir. J. 2016, 47, 919–928. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hanif, S.; R, A.-A.; AS, M. Species-specific antigenic Mycobacterium tuberculosis proteins tested by delayed-type hypersensitivity response. Int. J. Tuberc. Lung Dis. 2010, 14, 489–494. [Google Scholar] [PubMed]
- Kalra, M.; Khuller, G.K.; Sheikh, J.A.; Verma, I. Evaluation of Mycobacterium tuberculosis specific RD antigens for delayed type hypersensitivity responses in guinea pig. Indian J. Exp. Biol. 2010, 48, 117–123. [Google Scholar] [PubMed]
- Lyashchenko, K.; Manca, C.; Colangeli, R.; Heijbel, A.; Williams, A.; Gennaro Maria, L. Use of Mycobacterium tuberculosis complex-specific antigen cocktails for a skin test specific for tuberculosis. Infect. Immun. 1998, 66, 3606–3610. [Google Scholar] [CrossRef] [Green Version]
- Luo, W.; Qu, Z.-L.; Xie, Y.; Xiang, J.; Zhang, X.-L. Identification of a novel immunodominant antigen Rv2645 from RD13 with potential as a cell-mediated immunity-based TB diagnostic agent. J. Infect. 2015, 71, 534–543. [Google Scholar] [CrossRef]
- Aagaard, C.; Brock, I.; Olsen, A.; Ottenhoff, T.H.M.; Weldingh, K.; Andersen, P. Mapping immune reactivity toward Rv2653 and Rv2654: Two novel low-molecular-mass antigens found specifically in the Mycobacterium tuberculosis complex. J. Infect. Dis. 2004, 189, 812–819. [Google Scholar] [CrossRef] [Green Version]
- Mori, T.; Sakatani, M.; Yamagishi, F.; Takashima, T.; Kawabe, Y.; Nagao, K.; Shigeto, E.; Harada, N.; Mitarai, S.; Okada, M.; et al. Specific detection of tuberculosis infection. Am. J. Respir. Crit. Care Med. 2004, 170, 59–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- CDC. Interferon Gamma Release Assay Testing for Latent Tuberculosis Infection: Physician Guidelines; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2021. Available online: http://www.bccdc.ca/resource-gallery/Documents/Communicable-Disease-Manual/Chapter%204%20-%20TB/TB_manual_IGRA_guidelines.pdf (accessed on 5 July 2022).
- CDC. Interferon-Gamma Release Assays (IGRAs)—Blood Tests for TB Infection; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2016. Available online: https://www.cdc.gov/tb/publications/factsheets/testing/igra.htm (accessed on 5 July 2022).
- Ronge, L.; Sloot, R.; Du Preez, K.; Kay, A.W.; Kirchner, H.L.; Grewal, H.M.S.; Mandalakas, A.M.; Hesseling, A.C. The magnitude of interferon gamma release assay responses in children with household tuberculosis contact is associated with tuberculosis exposure and disease status. Pediatr. Infect. Dis. J. 2021, 40, 763–770. [Google Scholar] [CrossRef]
- Buonsenso, D.; Delogu, G.; Perricone, C.; Grossi, R.; Careddu, A.; De Maio, F.; Palucci, I.; Sanguinetti, M.; Valentini, P.; Sali, M. Accuracy of QuantiFERON-TB Gold Plus test for diagnosis of Mycobacterium tuberculosis infection in children. J. Clin. Microbiol. 2020, 58, e00272-20. [Google Scholar] [CrossRef] [PubMed]
- Surve, S.; Bhor, V.; Naukariya, K.; Begum, S.; Munne, K.; Tipre, P.; Sutar, N.; Jaiswal, A.; Bhonde, G.; Chauhan, S.; et al. Discordance between TST and QFT-TBGold Plus for latent tuberculosis screening among under-five children: An interim analysis. J. Trop. Pediatr. 2021, 67, fmab103. [Google Scholar] [CrossRef] [PubMed]
- Carranza, C.; Pedraza-Sanchez, S.; de Oyarzabal-Mendez, E.; Torres, M. Diagnosis for latent tuberculosis infection: New alternatives. Front. Immunol. 2020, 11, 2006. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Teeter, L.D.; Graves, J.; Graviss, E.A. Characteristics associated with negative interferon-γ release assay results in culture-confirmed tuberculosis patients, Texas, USA, 2013–2015. Emerg. Inf. Dis. 2018, 24, 534–540. [Google Scholar] [CrossRef] [Green Version]
- Qiagen. QuantiFERON-TB Gold Plus (QFT-Plus) ELISA Package Insert. 2016. Available online: http://www.quantiferon.com/wp-content/uploads/2017/04/English_QFTPlus_ELISA_R04_022016.pdf (accessed on 10 July 2022).
- Pérez-Recio, S.; Pallarès, N.; Grijota-Camino Maria, D.; Sánchez-Montalvá, A.; Barcia, L.; Campos-Gutiérrez, S.; Pomar, V.; Rabuñal-Rey, R.; Balcells María, E.; Gazel, D.; et al. Identification of recent tuberculosis exposure using QuantiFERON-TB Gold Plus, a multicenter study. Microbiol. Spectr. 2021, 9, e00972–00921. [Google Scholar] [CrossRef]
- Kellar, K.L.; Gehrke, J.; Weis, S.E.; Mahmutovic-Mayhew, A.; Davila, B.; Zajdowicz, M.J.; Scarborough, R.; LoBue, P.A.; Lardizabal, A.A.; Daley, C.L.; et al. Multiple cytokines are released when blood from patients with tuberculosis is stimulated with Mycobacterium tuberculosis antigens. PLoS ONE 2011, 6, e26545. [Google Scholar] [CrossRef]
- Hoffmann, H.; Avsar, K.; Göres, R.; Mavi, S.C.; Hofmann-Thiel, S. Equal sensitivity of the new generation QuantiFERON-TB Gold plus in direct comparison with the previous test version QuantiFERON-TB Gold IT. Clin. Microbiol. Infect. 2016, 22, 701–703. [Google Scholar] [CrossRef] [Green Version]
- Winglee, K.; Hill, A.N.; Belknap, R.; Stout, J.E.; Ayers, T.L. Variability of interferon-γ release assays in people at high risk of tuberculosis infection or progression to tuberculosis disease living in the United States. Clin. Microbiol. Infect. 2022, 28, 1023.e1021–1023.e1027. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.W.; Gaur, R.L.; Tien, S.S.; Spangler, M.; Pai, M.; Banaei, N. Evaluation of QuantiFERON-TB Gold-Plus in health care workers in a low-incidence setting. J. Clin. Microbiol. 2017, 55, 1650–1657. [Google Scholar] [CrossRef] [Green Version]
- Nemes, E.; Rozot, V.; Geldenhuys, H.; Bilek, N.; Mabwe, S.; Abrahams, D.; Makhethe, L.; Erasmus, M.; Keyser, A.; Toefy, A.; et al. Optimization and interpretation of serial quantiferon testing to measure acquisition of Mycobacterium tuberculosis infection. Am. J. Respir. Crit. Care Med. 2017, 196, 638–648. [Google Scholar] [CrossRef]
- Nienhaus, A.; Ringshausen, F.C.; Costa, J.T.; Schablon, A.; Tripodi, D. IFN-γ release assay versus tuberculin skin test for monitoring TB infection in healthcare workers. Expert Rev. Anti. Infect. Ther. 2013, 11, 37–48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Brown, J.; Kumar, K.; Reading, J.; Harvey, J.; Murthy, S.; Capocci, S.; Hopkins, S.; Seneviratne, S.; Cropley, I.; Lipman, M. Frequency and significance of indeterminate and borderline Quantiferon Gold TB IGRA results. Eur. Respir. J. 2017, 50, 1701267. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, J.; Westman, A.; Bruchfeld, J.; Sturegård, E.; Gaines, H.; Schön, T. A borderline range for Quantiferon Gold In-Tube results. PLoS ONE 2017, 12, e0187313. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anibarro, L.; Trigo, M.; Villaverde, C.; Pena, A.; Cortizo, S.; Sande, D.; Pazos, R.; Gonzalez-Fernandez, A. Interferon-gamma release assays in tuberculosis contacts: Is there a window period? Eur. Respir. J. 2011, 37, 215–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fox, G.J.; Barry, S.E.; Britton, W.J.; Marks, G.B. Contact investigation for tuberculosis: A systematic review and meta-analysis. Eur. Respir. J. 2013, 41, 140–156. [Google Scholar] [CrossRef] [PubMed]
- Stieber, F.; Howard, J.; Manissero, D.; Boyle, J.; Ndunda, N.; Love, J.; Yang, M.; Schumacher, A.; Uchiyama, R.; Parsons, S.; et al. Evaluation of a lateral-flow nanoparticle fluorescence assay for TB infection diagnosis. Int. J. Tuberc. Lung Dis. 2021, 25, 917–922. [Google Scholar] [CrossRef] [PubMed]
- Fukushima, K.; Akagi, K.; Kondo, A.; Kubo, T.; Sakamoto, N.; Mukae, H. First clinical evaluation of the QIAreachTM QuantiFERON-TB for tuberculosis infection and active pulmonary disease. Pulmonology 2022, 28, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Saluzzo, F.; Mantegani, P.; Poletti de Chaurand, V.; Cirillo, D.M. QIAreach QuantiFERON-TB for the diagnosis of Mycobacterium tuberculosis infection. Eur. Respir. J. 2022, 59, 2102563. [Google Scholar] [CrossRef] [PubMed]
- Kaaba, C.; Ruperez, M.; Kosloff, B.; Ndunda, N.; Shanaube, K.; Ayles, H. Assessing usability of QIAreach QuantiFERON-TB platform in a high tuberculosis prevalence, low-resource setting. ERJ Open Res. 2021, 7, 00511–02021. [Google Scholar] [CrossRef]
- Immunotec, O. T-SPOT®.TB: Package Insert. 2019. Available online: https://www.tspot.com/wp-content/uploads/2020/01/The-T-SPOT.TB-test-package-insert.pdf (accessed on 10 July 2022).
- Janssens, J.-P.; Roux-Lombard, P.; Perneger, T.; Metzger, M.; Vivien, R.; Rochat, T. Quantitative scoring of an interferon-γ assay for differentiating active from latent tuberculosis. Eur. Respir. J. 2007, 30, 722–728. [Google Scholar] [CrossRef]
- Rego, K.; Pereira, K.; MacDougall, J.; Cruikshank, W. Utility of the T-SPOT. Tuberculosis 2018, 108, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Immunotec, O. T-Cell Select™. 2022. Available online: https://www.oxfordimmunotec.com/international/products-services/t-cell-select/ (accessed on 10 October 2022).
- Wantai. WANTAI TB-IGRA: Diagnostic Kit for T Cell Infected with Mycobacterium Tuberculosis. Available online: http://www.ystwt.cn/wp-content/uploads/2018/04/Wantai-TB-IGRA.pdf (accessed on 5 October 2022).
- Li, G.; Li, F.; Zhao, H.-M.; Wen, H.-L.; Li, H.-C.; Li, C.-L.; Ji, P.; Xu, P.; Wu, K.; Hu, Z.-D.; et al. Evaluation of a new IFN-γ release assay for rapid diagnosis of active tuberculosis in a high-incidence setting. Front. Cell. Infect. Microbiol. 2017, 7, 117. [Google Scholar] [CrossRef] [PubMed]
- Della Bella, C.; Spinicci, M.; Alnwaisri, H.F.M.; Bartalesi, F.; Tapinassi, S.; Mencarini, J.; Benagiano, M.; Grassi, A.; D’Elios, S.; Troilo, A.; et al. LIOFeron®TB/LTBI: A novel and reliable test for LTBI and tuberculosis. Int. J. Infect. Dis. 2020, 91, 177–181. [Google Scholar] [CrossRef] [Green Version]
- Lewinsohn, D.M.; Leonard, M.K.; LoBue, P.A.; Cohn, D.L.; Daley, C.L.; Desmond, E.; Keane, J.; Lewinsohn, D.A.; Loeffler, A.M.; Mazurek, G.H.; et al. Official American Thoracic Society/Infectious Diseases Society of America/Centers for Disease Control and Prevention Clinical Practice Guidelines: Diagnosis of Tuberculosis in Adults and Children. Clin. Infect. Dis. 2017, 64, 111–115. [Google Scholar] [CrossRef]
- McKenna, L.; Sari, A.H.; Mane, S.; Scardigli, A.; Brigden, G.; Rouzier, V.; Becerra, M.C.; Hesseling, A.C.; Amanullah, F. Pediatric tuberculosis research and development: Progress, priorities and funding opportunities. Pathogens 2022, 11, 128. [Google Scholar] [CrossRef]
- Mazurek, G.H.; Jereb, J.; Vernon, A.; LoBue, P.; Goldberg, S.; Castro, K. Updated guidelines for using Interferon Gamma Release Assays to detect Mycobacterium tuberculosis infection—United States, 2010. MMWR Recomm. Rep. 2010, 59, 1–25. [Google Scholar] [PubMed]
- Soler-Garcia, A.; Gamell, A.; Pérez-Porcuna, T.; Soriano-Arandes, A.; Santiago, B.; Tórtola, T.; Ruiz-Serrano, M.J.; Korta Murua, J.J.; Bustillo-Alonso, M.; Garrote-Llanos, M.I.; et al. Performance of QuantiFERON-TB Gold Plus assays in children and adolescents at risk of tuberculosis: A cross-sectional multicentre study. Thorax 2021, 77, 1193–1201. [Google Scholar] [CrossRef]
- Yun, K.W.; Kim, Y.K.; Kim, H.R.; Lee, M.K.; Lim, I.S. Usefulness of interferon-γ release assay for the diagnosis of latent tuberculosis infection in young children. Korean J. Pediatr. 2016, 59, 256–261. [Google Scholar] [CrossRef] [Green Version]
- Kay, A.W.; Islam, S.M.; Wendorf, K.; Westenhouse, J.; Barry, P.M. Interferon-γ release assay performance for tuberculosis in childhood. Pediatrics 2018, 141, e20173918. [Google Scholar] [CrossRef] [Green Version]
- Gaensbauer, J.; Young, J.; Harasaki, C.; Aiona, K.; Belknap, R.; Haas, M.K. Interferon-gamma release assay testing in children younger than 2 years in a US-based health system. Pediatr. Infect. Dis. J. 2020, 39, 803–807. [Google Scholar] [CrossRef]
- Ahmed, A.; Feng, P.-J.I.; Gaensbauer, J.T.; Reves, R.R.; Khurana, R.; Salcedo, K.; Punnoose, R.; Katz, D.J.; CONSORTIUM, f.t.T.E.S. Interferon-γ release assays in children <15 years of age. Pediatrics 2020, 145, e20191930. [Google Scholar] [CrossRef] [PubMed]
- Wendorf, K.A.; Lowenthal, P.; Feraud, J.; Cabanting, N.; Murto, C. Interferon-γ release assays for tuberculosis infection diagnosis in refugees < 5 years old. Pediatrics 2020, 146, e20200715. [Google Scholar] [CrossRef] [PubMed]
- Connell, T.G.; Ritz, N.; Paxton, G.A.; Buttery, J.P.; Curtis, N.; Ranganathan, S.C. A three-way comparison of tuberculin skin testing, QuantiFERON-TB Gold and T-SPOT.TB in children. PLoS ONE 2008, 3, e2624. [Google Scholar] [CrossRef] [Green Version]
- Bae, W.; Park, K.U.; Song, E.Y.; Kim, S.J.; Lee, Y.J.; Park, J.S.; Cho, Y.-J.; Yoon, H.I.; Yim, J.-J.; Lee, C.-T.; et al. Comparison of the sensitivity of QuantiFERON-TB Gold In-Tube and T-SPOT.TB according to patient age. PLoS ONE 2016, 11, e0156917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scordo, J.M.; Aguillón-Durán, G.P.; Ayala, D.; Quirino-Cerrillo, A.P.; Rodríguez-Reyna, E.; Joya-Ayala, M.; Mora-Guzmán, F.; Ledezma-Campos, E.; Villafañez, A.; Schlesinger, L.S.; et al. Interferon gamma release assays for detection of latent Mycobacterium tuberculosis in older Hispanic people. Int. J. Infect. Dis. 2021, 111, 85–91. [Google Scholar] [CrossRef]
- Dheda, K.; Barry, C.E.; Maartens, G. Tuberculosis. Lancet 2016, 387, 1211–1226. [Google Scholar] [CrossRef] [PubMed]
- Richeldi, L.; Losi, M.; D’Amico, R.; Luppi, M.; Ferrari, A.; Mussini, C.; Codeluppi, M.; Cocchi, S.; Prati, F.; Paci, V.; et al. Performance of tests for latent tuberculosis in different groups of immunocompromised patients. Chest 2009, 136, 198–204. [Google Scholar] [CrossRef] [PubMed]
- Auguste, P.; Tsertsvadze, A.; Pink, J.; Court, R.; McCarthy, N.; Sutcliffe, P.; Clarke, A. Comparing interferon-gamma release assays with tuberculin skin test for identifying latent tuberculosis infection that progresses to active tuberculosis: Systematic review and meta-analysis. BMC Infect. Dis. 2017, 17, 200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernández-Blázquez, A.; Argüelles Menéndez, P.; Sabater-Cabrera, C.; García-García, J.M.; Asensi Álvarez, V.; Palacios Gutiérrez, J.J. Diagnosis of tuberculous infection in immunosuppressed patients and/or candidates for biologics using a combination of 2 IGRA tests: T-SPOT.TB/QuantiFERON TB Gold In-Tube vs. T-SPOT.TB/QuantiFERON TB Gold Plus. Arch. Bronconeumol. 2020, 10, 30128–30129. [Google Scholar]
- Park, C.H.; Park, J.H.; Jung, Y.S. Impact of immunosuppressive therapy on the performance of latent tuberculosis screening tests in patients with inflammatory bowel disease: A systematic review and meta-analysis. J. Pers. Med. 2022, 12, 507. [Google Scholar] [CrossRef]
- Wong, S.H.; Gao, Q.; Tsoi, K.K.; Wu, W.K.; Tam, L.S.; Lee, N.; Chan, F.K.; Wu, J.C.; Sung, J.J.; Ng, S.C. Effect of immunosuppressive therapy on interferon γ release assay for latent tuberculosis screening in patients with autoimmune diseases: A systematic review and meta-analysis. Thorax 2016, 71, 64–72. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fritschi, N.; Curtis, N.; Ritz, N. Bacille Calmette Guérin (BCG) and new TB vaccines: Specific, cross-mycobacterial and off-target effects. Paediatr. Respir. Rev. 2020, 36, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Lange, C.; Aaby, P.; Behr, M.A.; Donald, P.R.; Kaufmann, S.H.E.; Netea, M.G.; Mandalakas, A.M. 100 years of Mycobacterium bovis bacille Calmette-Guérin. Lancet Infect. Dis. 2022, 22, e2–e12. [Google Scholar] [CrossRef] [PubMed]
- Iglesias, M.J.; Martin, C. Editorial Commentary: Nonspecific beneficial effects of BCG vaccination in high-income countries, should we extend recommendation of BCG vaccination? Clin. Infect. Dis. 2015, 60, 1620–1621. [Google Scholar] [CrossRef] [Green Version]
- de Castro, M.J.; Pardo-Seco, J.; Martinón-Torres, F. Nonspecific (Heterologous) Protection of neonatal BCG vaccination against hospitalization due to respiratory infection and sepsis. Clin. Infect. Dis. 2015, 60, 1611–1619. [Google Scholar] [CrossRef] [Green Version]
- Broset, E.; Pardo-Seco, J.; Kanno, A.I.; Aguilo, N.; Dacosta, A.I.; Rivero-Calle, I.; Gonzalo-Asensio, J.; Locht, C.; Leite, L.C.C.; Martin, C.; et al. BCG vaccination improves DTaP immune responses in mice and is associated with lower pertussis incidence in ecological epidemiological studies. EBioMedicine 2021, 65, 103254. [Google Scholar] [CrossRef]
- Domínguez, J.; Ruiz-Manzano, J.; Souza-Galvão, M.D.; Latorre, I.; Milà, C.; Blanco, S.; Jiménez, M.Á.; Prat, C.; Lacoma, A.; Altet, N.; et al. Comparison of two commercially available gamma interferon blood tests for immunodiagnosis of tuberculosis. Clin. Vaccine Immunol. 2008, 15, 168–171. [Google Scholar] [CrossRef] [Green Version]
- Pai, M.; Zwerling, A.; Menzies, D. Systematic review: T-cell–based assays for the diagnosis of latent tuberculosis infection: An update. Ann. Intern. Med. 2008, 149, 177–184. [Google Scholar] [CrossRef]
- Kurtz, T.; Feil, A.C.; Nascimento, L.S.; de Oliveira Abreu, P.; Scotta, M.C.; Pinto, L.A. Effect of neonatal bacille Calmette-Guérin on the tuberculin skin test reaction in the first 2 years of life. Int. J. Tuberc. Lung Dis. 2019, 23, 344–348. [Google Scholar] [CrossRef]
- Anibarro, L.; Trigo, M.; Villaverde, C.; Pena, A.; González-Fernández, A. Tuberculin skin test and interferon-γ release assay show better correlation after the tuberculin ‘window period’ in tuberculosis contacts. Scand. J. Infect. Dis. 2011, 43, 424–429. [Google Scholar] [CrossRef]
- Dowdy, D.W.; Behr, M.A. Are we underestimating the annual risk of infection with Mycobacterium tuberculosis in high-burden settings? Lancet Infect. Dis. 2022, 22, e271–e278. [Google Scholar] [CrossRef] [PubMed]
- Faust, L.; Ruhwald, M.; Schumacher, S.; Pai, M. How are high burden countries implementing policies and tools for latent tuberculosis infection? A survey of current practices and barriers. Health Sci. Rep. 2020, 3, e158. [Google Scholar] [CrossRef] [PubMed]
- Alyaquobi, F.; AlMaqbali, A.A.; Al-Jardani, A.; Ndunda, N.; Al Rawahi, B.; Alabri, B.; AlSadi, A.M.; AlBaloshi, J.A.; Al-Baloshi, F.S.; Al-Essai, N.A.; et al. Screening migrants from tuberculosis high-endemic countries for latent tuberculosis in Oman: A cross sectional cohort analysis. Travel Med. Infect. Dis. 2020, 37, 101734. [Google Scholar] [CrossRef] [PubMed]
- Campbell, J.R.; Krot, J.; Elwood, K.; Cook, V.; Marra, F. A systematic review on TST and IGRA tests used for diagnosis of LTBI in immigrants. Mol. Diagn. Ther. 2015, 19, 9–24. [Google Scholar] [CrossRef] [PubMed]
- Elfrink, F.; van den Hoek, A.; Mensen, M.E.; Sonder, G.J.B. Screening travellers to high-endemic countries for infection with Mycobacterium tuberculosis using interferon gamma release assay; a prospective study. BMC Infect. Dis. 2014, 14, 515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spruijt, I.; Joren, C.; Schimmel, H.; Procee, F.; Alam, Y.; van den Hof, S.; Erkens, C. The identification of prevalent tuberculosis disease through infection screening among high-risk migrants in the Netherlands. Eur. Respir. J. 2022, 59, 2103018. [Google Scholar] [CrossRef] [PubMed]
- Apriani, L.; McAllister, S.; Sharples, K.; Alisjahbana, B.; Ruslami, R.; Hill, P.C.; Menzies, D. Latent tuberculosis infection in healthcare workers in low- and middle-income countries: An updated systematic review. Eur. Respir. J. 2019, 53, 1801789. [Google Scholar] [CrossRef]
- Swaminathan, N.; Perloff, S.R.; Zuckerman, J.M. Prevention of Mycobacterium tuberculosis transmission in health care settings. Infect. Dis. Clin. N. Am. 2021, 35, 1013–1025. [Google Scholar] [CrossRef]
- Fox, G.J.; Redwood, L.; Chang, V.; Ho, J. The effectiveness of individual and environmental infection control measures in reducing the transmission of Mycobacterium tuberculosis: A systematic review. Clin. Infect. Dis. 2021, 72, 15–26. [Google Scholar] [CrossRef]
- Mok, J.H. Diagnosis and treatment of latent tuberculosis infection in healthcare workers. Tuberc. Respir. Dis. 2016, 79, 127–133. [Google Scholar] [CrossRef] [Green Version]
- Park, Y.; Kim, S.Y.; Kim, J.W.; Park, M.S.; Kim, Y.S.; Chang, J.; Kang, Y.A. Serial testing of healthcare workers for latent tuberculosis infection and long-term follow up for development of active tuberculosis. PLoS ONE 2018, 13, e0204035. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dobler, C.C.; Farah, W.H.; Alsawas, M.; Mohammed, K.; Breeher, L.E.; Murad, M.H.; Molella, R.G. Tuberculin Skin test conversions and occupational exposure risk in US healthcare workers. Clin. Infect. Dis. 2018, 66, 706–711. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sosa, L.E.; Njie, G.J.; Lobato, M.N.; Bamrah Morris, S.; Buchta, W.; Casey, M.L.; Goswami, N.D.; Gruden, M.; Hurst, B.J.; Khan, A.R.; et al. Tuberculosis screening, testing, and treatment of U.S. health care personnel: Recommendations from the National Tuberculosis Controllers Association and CDC, 2019. MMWR Morb. Mortal. Wkly. Rep. 2019, 68, 439–443. [Google Scholar] [CrossRef] [PubMed]
- Machingaidze, S.; Verver, S.; Mulenga, H.; Abrahams, D.-A.; Hatherill, M.; Hanekom, W.; Hussey, G.D.; Mahomed, H. Predictive value of recent QuantiFERON Conversion for tuberculosis disease in adolescents. Am. J. Respir. Crit Care Med. 2012, 186, 1051–1056. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Kim, N.; Park, H.; Kim, T.S.; Park, S.W.; Roh, E.Y.; Yoon, J.H.; Shin, S. The use of a borderline zone for the interpretation of interferon-gamma release assay results for serial screening of healthcare workers. PLoS ONE 2020, 15, e0235254. [Google Scholar] [CrossRef]
- Leung, W.L.; Law, K.L.; Leung, V.S.S.; Yip, C.W.; Leung, C.C.; Tam, C.M.; Kam, K.M. Comparison of intracellular cytokine flow cytometry and an enzyme immunoassay for evaluation of cellular immune response to active tuberculosis. Clin. Vaccine Immunol. 2009, 16, 344–351. [Google Scholar] [CrossRef] [Green Version]
- El-Sheikh, N.; Mousa, N.O.; Osman, A.; Tawfeik, A.M.; Taha, B.A.; Mahran, H.; Saleh, A.M.; El-shiekh, I.; Amin, W.; Elrefaei, M. Assessment of interferon gamma-induced protein 10 mRNA release assay for detection of latent tuberculosis infection in egyptian pediatric household contacts. Int. J. Infect. Dis. 2021, 109, 223–229. [Google Scholar] [CrossRef]
- Kumar, N.P.; Moideen, K.; Banurekha, V.V.; Nair, D.; Babu, S. Plasma proinflammatory cytokines are markers of disease severity and bacterial burden in pulmonary tuberculosis. Open Forum Infect. Dis. 2019, 6, ofz257. [Google Scholar] [CrossRef] [Green Version]
- Soeroto, A.Y.; Dahlan, Z.; Kartasasmita, C.B.; Parwati, I. Serum cytokines level can differentiate active pulmonary tuberculosis from latent TB. Eur. Respir. J. 2013, 42, P2842. [Google Scholar]
- Sheffee, N.S.; Rubio-Reyes, P.; Mirabal, M.; Calero, R.; Carrillo-Calvet, H.; Chen, S.; Chin, K.L.; Shakimi, N.A.S.; Anis, F.Z.; Suraiya, S.; et al. Engineered Mycobacterium tuberculosis antigen assembly into core-shell nanobeads for diagnosis of tuberculosis. Nanomed. Nanotechnol. Biol. Med. 2021, 34, 102374. [Google Scholar] [CrossRef]
- Robison, H.M.; Chapman, C.A.; Zhou, H.; Erskine, C.L.; Theel, E.; Peikert, T.; Lindestam Arlehamn, C.S.; Sette, A.; Bushell, C.; Welge, M.; et al. Risk assessment of latent tuberculosis infection through a multiplexed cytokine biosensor assay and machine learning feature selection. Sci. Rep. 2021, 11, 20544. [Google Scholar] [CrossRef] [PubMed]
- Hur, Y.-G.; Kang, Y.A.; Jang, S.-H.; Hong, J.Y.; Kim, A.; Lee, S.A.; Kim, Y.; Cho, S.-N. Adjunctive biomarkers for improving diagnosis of tuberculosis and monitoring therapeutic effects. J. Infect. 2015, 70, 346–355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, J.; Wang, S.; Lu, C.; Shao, L.; Gao, Y.; Zhou, Z.; Huang, H.; Zhang, Y.; Zhang, W. Multiple cytokine responses in discriminating between active tuberculosis and latent tuberculosis infection. Tuberculosis 2017, 102, 68–75. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Li, Y.; Shen, Y.; Wu, J.; Gao, Y.; Zhang, S.; Shao, L.; Jin, J.; Zhang, Y.; Zhang, W. Screening and identification of a six-cytokine biosignature for detecting TB infection and discriminating active from latent TB. J. Transl. Med. 2018, 16, 206. [Google Scholar] [CrossRef] [PubMed]
- Clifford, V.; Tebruegge, M.; Zufferey, C.; Germano, S.; Forbes, B.; Cosentino, L.; Matchett, E.; McBryde, E.; Eisen, D.; Robins-Browne, R.; et al. Cytokine biomarkers for the diagnosis of tuberculosis infection and disease in adults in a low prevalence setting. Tuberculosis 2019, 114, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Tebruegge, M.; Dutta, B.; Donath, S.; Ritz, N.; Forbes, B.; Camacho-Badilla, K.; Clifford, V.; Zufferey, C.; Robins-Browne, R.; Hanekom, W.; et al. Mycobacteria-specific cytokine responses detect tuberculosis infection and distinguish latent from active tuberculosis. Am. J. Respir. Crit. Care Med. 2015, 192, 485–499. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Suzukawa, M.; Akashi, S.; Nagai, H.; Nagase, H.; Nakamura, H.; Matsui, H.; Hebisawa, A.; Ohta, K. Combined analysis of IFN-γ, IL-2, IL-5, IL-10, IL-1RA and MCP-1 in QFT supernatant is useful for distinguishing active tuberculosis from latent infection. PLoS ONE 2016, 11, e0152483. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Won, E.-J.; Choi, J.-H.; Cho, Y.-N.; Jin, H.-M.; Kee, H.J.; Park, Y.-W.; Kwon, Y.-S.; Kee, S.-J. Biomarkers for discrimination between latent tuberculosis infection and active tuberculosis disease. J. Infect. 2017, 74, 281–293. [Google Scholar] [CrossRef] [PubMed]
- Chegou, N.N.; Black, G.F.; Kidd, M.; van Helden, P.D.; Walzl, G. Host markers in Quantiferon supernatants differentiate active TB from latent TB infection: Preliminary report. BMC Pulm. Med. 2009, 9, 21. [Google Scholar] [CrossRef] [PubMed]
- La Manna, M.P.; Orlando, V.; Li Donni, P.; Sireci, G.; Di Carlo, P.; Cascio, A.; Dieli, F.; Caccamo, N. Identification of plasma biomarkers for discrimination between tuberculosis infection/disease and pulmonary non tuberculosis disease. PLoS ONE 2018, 13, e0192664. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Essone, P.N.; Leboueny, M.; Maloupazoa Siawaya, A.C.; Alame-Emane, A.K.; Aboumegone Biyogo, O.C.; Dapnet Tadatsin, P.H.; Mveang Nzoghe, A.; Essamazokou, D.U.; Mvoundza Ndjindji, O.; Padzys, G.-S.; et al. M. tuberculosis infection and antigen specific cytokine response in healthcare workers frequently exposed to tuberculosis. Sci. Rep. 2019, 9, 8201. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kak, G.; Raza, M.; Tiwari, B.K. Interferon-gamma (IFN-γ): Exploring its implications in infectious diseases. Biomol. Concepts 2018, 9, 64–79. [Google Scholar] [CrossRef] [PubMed]
- Green, A.M.; DiFazio, R.; Flynn, J.L. IFN-γ from CD4 T cells is essential for host survival and enhances CD8 T cell function during Mycobacterium tuberculosis infection. J. Immunol. 2013, 190, 270–277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, J.A.; Duarte, R.; Nunes, C. Host factors associated to false negative and indeterminate results in an interferon-γ release assay in patients with active tuberculosis. Pulmonology 2020, 26, 353–362. [Google Scholar] [CrossRef]
- Pan, L.; Jia, H.; Liu, F.; Sun, H.; Gao, M.; Du, F.; Xing, A.; Du, B.; Sun, Q.; Wei, R.; et al. Risk factors for false-negative T-SPOT.TB assay results in patients with pulmonary and extra-pulmonary TB. J. Infect. 2015, 70, 367–380. [Google Scholar] [CrossRef] [PubMed]
- Hang, N.T.L.; Lien, L.T.; Kobayashi, N.; Shimbo, T.; Sakurada, S.; Thuong, P.H.; Hong, L.T.; Tam, D.B.; Hijikata, M.; Matsushita, I.; et al. Analysis of factors lowering sensitivity of interferon-γ release assay for tuberculosis. PLoS ONE 2011, 6, e23806. [Google Scholar] [CrossRef]
- Guo, J.; Li, Q.; Zhang, X.; Yao, C.; Liu, R.; Pang, Y.; Gao, M. Increased expression of IL-10 in peripheral blood mononuclear cells correlates with negative interferon-γ release assay results in culture-confirmed tuberculosis patients. Infect. Drug Resist. 2021, 41, 3135–3143. [Google Scholar] [CrossRef]
- Yamasue, M.; Komiya, K.; Usagawa, Y.; Umeki, K.; Nureki, S.I.; Ando, M.; Hiramatsu, K.; Nagai, H.; Kadota, J.I. Factors associated with false negative interferon-γ release assay results in patients with tuberculosis: A systematic review with meta-analysis. Sci. Rep. 2020, 10, 1607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, K.H.; Jeong, N.; Lim, J.U.; Lee, H.Y.; Lee, J.; Kim, S.C.; Kang, J.Y. Clinical relevance of false-negative interferon-gamma release assays in patients with tuberculous pleurisy in an intermediate tuberculosis burden country. J. Thorac. Dis. 2022, 14, 1009–1019. [Google Scholar] [CrossRef]
- Qiu, B.; Liu, Q.; Li, Z.; Song, H.; Xu, D.; Ji, Y.; Jiang, Y.; Tian, D.; Wang, J. Evaluation of cytokines as a biomarker to distinguish active tuberculosis from latent tuberculosis infection: A diagnostic meta-analysis. BMJ Open 2020, 10, e039501. [Google Scholar] [CrossRef]
- Liao, W.; Lin, J.-X.; Leonard, W.J. Interleukin-2 at the crossroads of effector responses, tolerance, and immunotherapy. Immunity 2013, 38, 13–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jafrasteh, A.; Karimi, A.; Hoseinialfatemi, S.M.; Azimi, L.; Tabarsi, P.; Nasehi, M.; Naseri, M.; Panahi Mishkar, A.; Sheikhi, M.; Mansour Ghanaie, R. Evaluation of Interleukin-2 to detect active and latent tuberculosis among household contacts of pulmonary tuberculosis cases. Arch. Pediatr. Infect. Dis. 2021, 9, e109398. [Google Scholar] [CrossRef]
- Tan, Y.; Tan, Y.; Li, J.; Hu, P.; Guan, P.; Kuang, H.; Liang, Q.; Yu, Y.; Chen, Z.; Wang, Q.; et al. Combined IFN-γ and IL-2 release assay for detect active pulmonary tuberculosis: A prospective multicentre diagnostic study in China. J. Transl. Med. 2021, 19, 289. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, F.; Shi, H.; Wu, P. Host biomarkers other than interferon gamma in QFT-TB supernatants for identifying active tuberculosis. Tuberculosis 2022, 136, 102256. [Google Scholar] [CrossRef] [PubMed]
- Santin, M.; Morandeira-Rego, F.; Alcaide, F.; Rabunal, R.; Anibarro, L.; Aguero-Balbin, R.; Casas-Garcia, X.; Perez-Escolano, E.; Navarro, M.D.; Sanchez, F.; et al. Detection of interleukin-2 is not useful for distinguishing between latent and active tuberculosis in clinical practice: A prospective cohort study. Clin. Microbiol. Infect. 2016, 22, 1007.e1001–1007.e1005. [Google Scholar] [CrossRef]
- Biselli, R.; Mariotti, S.; Sargentini, V.; Sauzullo, I.; Lastilla, M.; Mengoni, F.; Vanini, V.; Girardi, E.; Goletti, D.; D’ Amelio, R.; et al. Detection of interleukin-2 in addition to interferon-γ discriminates active tuberculosis patients, latently infected individuals, and controls. Clin. Microbiol. Infect. 2010, 16, 1282–1284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, Q.; Wei, W.; Sha, W. Potential Role for Mycobacterium tuberculosis specific IL-2 and IFN-γ responses in discriminating between latent infection and active disease after long-term stimulation. PLoS ONE 2016, 11, e0166501. [Google Scholar] [CrossRef] [Green Version]
- Sargentini, V.; Mariotti, S.; Carrara, S.; Gagliardi, M.C.; Teloni, R.; Goletti, D.; Nisini, R. Cytometric detection of antigen-specific IFN-γ/IL-2 secreting cells in the diagnosis of tuberculosis. BMC Infect. Dis. 2009, 9, 99. [Google Scholar] [CrossRef] [Green Version]
- Gourgouillon, N.; de Lauzanne, A.; Cottart, C.-H.; Curis, E.; Debord, C.; Guérin-El Khourouj, V.; Pédron, B.; Faye, A.; Sterkers, G. TNF-α/IL-2 ratio discriminates latent from active tuberculosis in immunocompetent children: A pilot study. Pediatr. Res. 2012, 72, 370–374. [Google Scholar] [CrossRef] [Green Version]
- Wei, Z.; Li, Y.; Wei, C.; Li, Y.; Xu, H.; Wu, Y.; Jia, Y.; Guo, R.; Jia, J.; Qi, X.; et al. The meta-analysis for ideal cytokines to distinguish the latent and active TB infection. BMC Pulm. Med. 2020, 20, 248. [Google Scholar] [CrossRef]
- Qiu, X.; Wang, H.; Tang, Y.; Su, X.; Ge, L.; Qu, Y.; Mu, D. Is interleukin-2 an optimal marker for diagnosing tuberculosis infection? A systematic review and meta-analysis. Ann. Med. 2020, 52, 376–385. [Google Scholar] [CrossRef] [PubMed]
- Liu, M.; Guo, S.; Hibbert, J.M.; Jain, V.; Singh, N.; Wilson, N.O.; Stiles, J.K. CXCL10/IP-10 in infectious diseases pathogenesis and potential therapeutic implications. Cytokine Growth Factor. Rev. 2011, 22, 121–130. [Google Scholar] [CrossRef] [PubMed]
- Whittaker, E.; Gordon, A.; Kampmann, B. Is IP-10 a better biomarker for active and latent tuberculosis in children than IFNγ? PLoS ONE 2008, 3, e3901. [Google Scholar] [CrossRef] [Green Version]
- Hong, J.Y.; Jung, G.S.; Kim, H.; Kim, Y.M.; Lee, H.J.; Cho, S.-N.; Kim, S.K.; Chang, J.; Kang, Y.A. Efficacy of inducible protein 10 as a biomarker for the diagnosis of tuberculosis. Int. J. Infect. Dis. 2012, 16, e855–e859. [Google Scholar] [CrossRef] [PubMed]
- Petrone, L.; Vanini, V.; Chiacchio, T.; Petruccioli, E.; Cuzzi, G.; Schininà, V.; Palmieri, F.; Ippolito, G.; Goletti, D. Evaluation of IP-10 in Quantiferon-Plus as biomarker for the diagnosis of latent tuberculosis infection. Tuberculosis 2018, 111, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Estévez, O.; Anibarro, L.; Garet, E.; Pallares, Á.; Pena, A.; Villaverde, C.; Del Campo, V.; González-Fernández, Á. Identification of candidate host serum and saliva biomarkers for a better diagnosis of active and latent tuberculosis infection. PLoS ONE 2020, 15, e0235859. [Google Scholar] [CrossRef]
- Lighter, J.; Rigaud, M.; Huie, M.; Peng, C.H.; Pollack, H. Chemokine IP-10: An adjunct marker for latent tuberculosis infection in children. Int. J. Tuberc. Lung Dis. 2009, 13, 731–736. [Google Scholar]
- Blauenfeldt, T.; Villar-Hernández, R.; García-García, E.; Latorre, I.; Holm Line, L.; Muriel-Moreno, B.; De Souza-Galvão Maria, L.; Millet Joan, P.; Sabriá, F.; Sánchez-Montalva, A.; et al. Diagnostic accuracy of interferon gamma-induced protein 10 mRNA release assay for tuberculosis. J. Clin. Microbiol. 2020, 58, e00848–00820. [Google Scholar] [CrossRef]
- Wergeland, I.; Assmus, J.; Dyrhol-Riise, A.M. Cytokine patterns in tuberculosis infection; IL-1ra, IL-2 and IP-10 differentiate borderline QuantiFERON-TB samples from uninfected controls. PLoS ONE 2016, 11, e0163848. [Google Scholar] [CrossRef]
- Uzorka, J.W.; Bakker, J.A.; van Meijgaarden, K.E.; Leyten, E.M.S.; Delfos, N.M.; Hetem, D.J.; Kerremans, J.; Zwarts, M.; Cozijn, S.; Ottenhoff, T.H.M.; et al. Biomarkers to identify Mycobacterium tuberculosis infection among borderline QuantiFERON results. Eur. Respir. J. 2022, 60, 2102665. [Google Scholar] [CrossRef]
- Qiu, X.; Tang, Y.; Zou, R.; Zeng, Y.; Yue, Y.; Li, W.; Qu, Y.; Mu, D. Diagnostic accuracy of interferon-gamma-induced protein 10 for differentiating active tuberculosis from latent tuberculosis: A meta-analysis. Sci. Rep. 2019, 9, 11408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Qiu, X.; Xiong, T.; Su, X.; Qu, Y.; Ge, L.; Yue, Y.; Zeng, Y.; Li, W.; Hu, P.; Mu, D. Accumulate evidence for IP-10 in diagnosing pulmonary tuberculosis. BMC Infect. Dis. 2019, 19, 924. [Google Scholar] [CrossRef] [Green Version]
- Qiu, X.; Tang, Y.; Yue, Y.; Zeng, Y.; Li, W.; Qu, Y.; Mu, D. Accuracy of interferon-γ-induced protein 10 for diagnosing latent tuberculosis infection: A systematic review and meta-analysis. Clin. Microbiol. Infect. 2019, 25, 667–672. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, A.J.; Lainati, F.; Aguilera Vasquez, N.; Chedid, C.; McGrath, S.; Benedetti, A.; MacLean, E.; Ruhwald, M.; Denkinger, C.M.; Kohli, M. Biomarkers that correlate with active pulmonary tuberculosis treatment response: A systematic review and meta-analysis. J. Clin. Microbiol. 2022, 60, e0185921. [Google Scholar] [CrossRef] [PubMed]
- Parameswaran, N.; Patial, S. Tumor necrosis factor-α signaling in macrophages. Crit. Rev. Eukaryot Gene Expr. 2010, 20, 87–103. [Google Scholar] [CrossRef]
- Wang, F.; Hou, H.; Xu, L.; Jane, M.; Peng, J.; Lu, Y.; Zhu, Y.; Sun, Z. Mycobacterium tuberculosis-specific TNF-α is a potential biomarker for the rapid diagnosis of active tuberculosis disease in Chinese population. PLoS ONE 2013, 8, e79431. [Google Scholar] [CrossRef]
- Harari, A.; Rozot, V.; Bellutti Enders, F.; Perreau, M.; Stalder, J.M.; Nicod, L.P.; Cavassini, M.; Calandra, T.; Blanchet, C.L.; Jaton, K.; et al. Dominant TNF-α+ Mycobacterium tuberculosis-specific CD4+ T cell responses discriminate between latent infection and active disease. Nat. Med. 2011, 17, 372–376. [Google Scholar] [CrossRef]
- Zhang, L.; Wan, S.; Zhou, Z.; Zhang, Y.; Liu, X. Utility of interferon gamma/tumor necrosis factor alpha FluoroSpot assay in differentiation between active tuberculosis and latent tuberculosis infection: A pilot study. BMC Infect. Dis. 2021, 21, 651. [Google Scholar] [CrossRef]
- Kim, J.Y.; Kang, Y.A.; Park, J.H.; Cha, H.H.; Jeon, N.Y.; Lee, S.W.; Lee, S.O.; Choi, S.H.; Kim, Y.S.; Woo, J.H.; et al. An IFN-γ and TNF-α dual release fluorospot assay for diagnosing active tuberculosis. Clin. Microbiol. Infect. 2020, 26, 928–934. [Google Scholar] [CrossRef]
- Kim, S.; Lee, H.; Kim, H.; Kim, Y.; Cho, J.-E.; Jin, H.; Kim, D.Y.; Ha, S.-J.; Kang, Y.A.; Cho, S.-N.; et al. Diagnostic performance of a cytokine and IFN-γ induced chemokine mRNA assay after Mycobacterium tuberculosis specific antigen stimulation in whole blood from infected individuals. J. Mol. Diagn. 2015, 17, 90–99. [Google Scholar] [CrossRef]
- Prabhavathi, M.; Pathakumari, B.; Raja, A. IFN-γ/TNF-α ratio in response to immuno proteomically identified human T-cell antigens of Mycobacterium tuberculosis—The most suitable surrogate biomarker for latent TB infection. J. Infect. 2015, 71, 238–249. [Google Scholar] [CrossRef] [PubMed]
- Couper, K.N.; Blount, D.G.; Riley, E.M. IL-10: The master regulator of immunity to infection. J. Immunol. 2008, 180, 5771–5777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bapat, P.R.; Husain, A.A.; Daginawala, H.F.; Agrawal, N.P.; Panchbhai, M.S.; Satav, A.R.; Taori, G.M.; Kashyap, R.S. The assessment of cytokines in Quantiferon supernatants for the diagnosis of latent TB infection in a tribal population of Melghat, India. J. Infect. Public Health 2015, 8, 329–340. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lohela, M.; Bry, M.; Tammela, T.; Alitalo, K. VEGFs and receptors involved in angiogenesis versus lymphangiogenesis. Curr. Opin. Cell Biol. 2009, 21, 154–165. [Google Scholar] [CrossRef] [PubMed]
- Delemarre, E.M.; van Hoorn, L.; Bossink, A.W.J.; Drylewicz, J.; Joosten, S.A.; Ottenhoff, T.H.M.; Akkerman, O.W.; Goletti, D.; Petruccioli, E.; Navarra, A.; et al. Serum biomarker profile including CCL1, CXCL10, VEGF, and adenosine deaminase activity distinguishes active from remotely acquired latent tuberculosis. Front. Immunol. 2021, 12. [Google Scholar] [CrossRef]
- Kumar, N.P.; Banurekha, V.V.; Nair, D.; Babu, S. Circulating angiogenic factors as biomarkers of disease severity and bacterial burden in pulmonary tuberculosis. PLoS ONE 2016, 11, e0146318. [Google Scholar] [CrossRef]
- Rensburg, I.C.v.; Loxton, A.G. Transcriptomics: The key to biomarker discovery during tuberculosis? Biomark. Med. 2015, 9, 483–495. [Google Scholar] [CrossRef]
- Petrilli, J.D.; Araújo, L.E.; da Silva, L.S.; Laus, A.C.; Müller, I.; Reis, R.M.; Netto, E.M.; Riley, L.W.; Arruda, S.; Queiroz, A. Whole blood mRNA expression-based targets to discriminate active tuberculosis from latent infection and other pulmonary diseases. Sci. Rep. 2020, 10, 22072. [Google Scholar] [CrossRef]
- de Araujo, L.S.; Vaas, L.A.I.; Ribeiro-Alves, M.; Geffers, R.; Mello, F.C.Q.; de Almeida, A.S.; Moreira, A.d.S.R.; Kritski, A.L.; Lapa E Silva, J.R.; Moraes, M.O.; et al. Transcriptomic biomarkers for tuberculosis: Evaluation of DOCK9. EPHA4, and NPC2 mRNA expression in peripheral blood. Front. Microbiol. 2016, 7, 1586. [Google Scholar] [CrossRef]
- Hashimoto, S.; Zhao, H.; Hayakawa, M.; Nakajima, K.; Taguchi, Y.h.; Murakami, Y. Developing a diagnostic method for latent tuberculosis infection using circulating miRNA. Transl. Med. Commun. 2020, 5, 25. [Google Scholar] [CrossRef]
- Wang, C.; Yang, S.; Sun, G.; Tang, X.; Lu, S.; Neyrolles, O.; Gao, Q. Comparative miRNA expression profiles in individuals with latent and active tuberculosis. PLoS ONE 2011, 6, e25832. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.S.-H.; Lee, S.-W.; Huang, K.-Y.; Lee, T.-Y.; Hsu, P.W.-C.; Weng, J.T.-Y. Systematic expression profiling analysis identifies specific microRNA-gene interactions that may differentiate between active and latent tuberculosis infection. BioMed Res. Int. 2014, 2014, 895179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hamada, Y.; Penn-Nicholson, A.; Krishnan, S.; Cirillo, D.M.; Matteelli, A.; Wyss, R.; Denkinger, C.M.; Rangaka, M.X.; Ruhwald, M.; Schumacher, S.G. Are mRNA based transcriptomic signatures ready for diagnosing tuberculosis in the clinic?—A review of evidence and the technological landscape. EBioMedicine 2022, 82, 104174. [Google Scholar] [CrossRef] [PubMed]
- Araujo, L.S.d.; Ribeiro-Alves, M.; Leal-Calvo, T.; Leung, J.; Durán, V.; Samir, M.; Talbot, S.; Tallam, A.; Mello, F.C.d.Q.; Geffers, R.; et al. Reprogramming of small noncoding RNA populations in peripheral blood reveals host biomarkers for latent and active Mycobacterium tuberculosis infection. mBio 2019, 10, e01037-19. [Google Scholar] [CrossRef] [Green Version]
- Zak, D.E.; Penn-Nicholson, A.; Scriba, T.J.; Thompson, E.; Suliman, S.; Amon, L.M.; Mahomed, H.; Erasmus, M.; Whatney, W.; Hussey, G.D.; et al. A blood RNA signature for tuberculosis disease risk: A prospective cohort study. Lancet 2016, 387, 2312–2322. [Google Scholar] [CrossRef]
- Darboe, F.; Mbandi, S.K.; Thompson, E.G.; Fisher, M.; Rodo, M.; van Rooyen, M.; Filander, E.; Bilek, N.; Mabwe, S.; Hatherill, M.; et al. Diagnostic performance of an optimized transcriptomic signature of risk of tuberculosis in cryopreserved peripheral blood mononuclear cells. Tuberculosis 2018, 108, 124–126. [Google Scholar] [CrossRef]
- Scriba, T.J.; Fiore-Gartland, A.; Penn-Nicholson, A.; Mulenga, H.; Kimbung Mbandi, S.; Borate, B.; Mendelsohn, S.C.; Hadley, K.; Hikuam, C.; Kaskar, M.; et al. Biomarker-guided tuberculosis preventive therapy (CORTIS): A randomised controlled trial. Lancet Infect. Dis. 2021, 21, 354–365. [Google Scholar] [CrossRef]
- Mulenga, H.; Fiore-Gartland, A.; Mendelsohn, S.C.; Penn-Nicholson, A.; Mbandi, S.K.; Nemes, E.; Borate, B.; Musvosvi, M.; Tameris, M.; Walzl, G.; et al. Evaluation of a transcriptomic signature of tuberculosis risk in combination with an interferon gamma release assay: A diagnostic test accuracy study. eClinicalMedicine 2022, 47, 101396. [Google Scholar] [CrossRef]
- Estévez, O.; Anibarro, L.; Garet, E.; Pallares, Á.; Barcia, L.; Calviño, L.; Maueia, C.; Mussá, T.; Fdez-Riverola, F.; Glez-Peña, D.; et al. An RNA-seq based machine learning approach identifies latent tuberculosis patients with an active tuberculosis profile. Front. Immunol. 2020, 11, 1470. [Google Scholar] [CrossRef]
- Phetsouphanh, C.; Zaunders, J.J.; Kelleher, A.D. Detecting antigen-specific T cell responses: From bulk populations to single cells. Int. J. Mol. Sci. 2015, 16, 18878–18893. [Google Scholar] [CrossRef]
- Pollock, K.M.; Whitworth, H.S.; Montamat-Sicotte, D.J.; Grass, L.; Cooke, G.S.; Kapembwa, M.S.; Kon, O.M.; Sampson, R.D.; Taylor, G.P.; Lalvani, A. T-cell immunophenotyping distinguishes active from latent tuberculosis. J. Infect. Dis. 2013, 208, 952–968. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silveira-Mattos, P.S.; Barreto-Duarte, B.; Vasconcelos, B.; Fukutani, K.F.; Vinhaes, C.L.; Oliveira-De-Souza, D.; Ibegbu, C.C.; Figueiredo, M.C.; Sterling, T.R.; Rengarajan, J.; et al. Differential expression of activation markers by Mycobacterium tuberculosis-specific CD4+ T cell distinguishes extrapulmonary from pulmonary tuberculosis and latent infection. Clin. Infect. Dis. 2020, 71, 1905–1911. [Google Scholar] [CrossRef] [PubMed]
- Mpande, C.A.M.; Rozot, V.; Mosito, B.; Musvosvi, M.; Dintwe, O.B.; Bilek, N.; Hatherill, M.; Scriba, T.J.; Nemes, E. Immune profiling of Mycobacterium tuberculosis-specific T cells in recent and remote infection. EBioMedicine 2021, 64, 103233. [Google Scholar] [CrossRef]
- Ubolyam, S.; Iampornsin, T.; Sophonphan, J.; Avihingsanon, A.; Suwanpimolkul, G.; Kawkitinarong, K.; Manosuthi, W.; Gatechompol, S.; Ananworanich, J.; Ruxrungtham, K.; et al. Performance of a simple flow cytometric assay in diagnosing active tuberculosis. Tuberculosis 2021, 126, 102017. [Google Scholar] [CrossRef]
- Escalante, P.; Peikert, T.; Van Keulen, V.P.; Erskine, C.L.; Bornhorst, C.L.; Andrist, B.R.; McCoy, K.; Pease, L.R.; Abraham, R.S.; Knutson, K.L.; et al. Combinatorial immunoprofiling in latent tuberculosis infection. toward better risk stratification. Am. J. Respir. Crit. Care Med. 2015, 192, 605–617. [Google Scholar] [CrossRef] [PubMed]
- Estevez, O.; Anibarro, L.; Garet, E.; Martínez, A.; Pena, A.; Barcia, L.; Peleteiro, M.; Fernández, Á. Multi-parameter flow cytometry immunophenotyping distinguishes different stages of tuberculosis infection. J. Infect. 2020, 81, 57–71. [Google Scholar] [CrossRef] [PubMed]
- Duncan, C.; Jamieson, F.; Mehaffy, C. Preliminary evaluation of exome sequencing to identify genetic markers of susceptibility to tuberculosis disease. BMC Res. Notes 2015, 8, 750. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, L.; Hu, Y.; Li, D.; Jiang, W.; Xu, B. Screening toll-like receptor markers to predict latent tuberculosis infection and subsequent tuberculosis disease in a Chinese population. BMC Med. Genet. 2015, 16, 19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, S.; Liu, X.; Wang, Y.; Zhang, M.; Wang, M.; He, J.-Q. Genetic polymorphisms of IFNG and IFNGR1 with latent tuberculosis infection. Dis. Markers 2019, 2019, 8410290. [Google Scholar] [CrossRef] [Green Version]
- Hu, Y.; Wu, L.; Li, D.; Zhao, Q.; Jiang, W.; Xu, B. Association between cytokine gene polymorphisms and tuberculosis in a Chinese population in Shanghai: A case–control study. BMC Immunol. 2015, 16, 8. [Google Scholar] [CrossRef] [Green Version]
- Chang, S.-Y.; Chen, M.-L.; Lee, M.-R.; Liang, Y.-C.; Lu, T.-P.; Wang, J.-Y.; Yan, B.-S. SP110 polymorphisms are genetic markers for vulnerability to latent and active tuberculosis infection in Taiwan. Dis. Markers 2018, 2018, 4687380. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cazarez-Navarro, G.; Palomares-Marín, J.; Rodríguez-Preciado, S.Y.; Pereira-Suárez, A.L.; Martínez-López, E.; Bacilio-Medrano, E.A.; Huerta-OlveraIván, S.; Hernández-Cañaveral, I. Association of TAP1 1177A>G and 2090A>G gene polymorphisms with latent tuberculosis infections in sheltered populations, in the metropolitan area of Guadalajara, Mexico: A pilot study. Rev. Inst. Med. Trop São Paulo 2021, 63, e55. [Google Scholar] [CrossRef] [PubMed]
- Cubillos-Angulo, J.M.; Arriaga, M.B.; Melo, M.G.M.; Silva, E.C.; Alvarado-Arnez, L.E.; de Almeida, A.S.; Moraes, M.O.; Moreira, A.S.R.; Lapa e Silva, J.R.; Fukutani, K.F.; et al. Polymorphisms in interferon pathway genes and risk of Mycobacterium tuberculosis infection in contacts of tuberculosis cases in Brazil. Int. J. Infect. Dis. 2020, 92, 21–28. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, S.; Li, G.; Bi, J.; Guo, Q.; Fu, X.; Wang, W.; Liu, S.; Xiao, G.; Ou, M.; Zhang, J.; et al. Association between functional nucleotide polymorphisms up-regulating transforming growth factor β1 expression and increased tuberculosis susceptibility. J. Infect. Dis. 2022, 225, 825–835. [Google Scholar] [CrossRef]
- Teklu, T.; Wondale, B.; Taye, B.; Hailemariam, M.; Bekele, S.; Tamirat, M.; Zewude, A.; Mohamed, T.; Medhin, G.; Legesse, M.; et al. Differences in plasma proteomes for active tuberculosis, latent tuberculosis and non-tuberculosis mycobacterial lung disease patients with and without ESAT-6/CFP10 stimulation. Proteome Sci. 2020, 18, 10. [Google Scholar] [CrossRef]
- Liu, L.; Deng, J.; Yang, Q.; Wei, C.; Liu, B.; Zhang, H.; Xin, H.; Pan, S.; Liu, Z.; Wang, D.; et al. Urinary proteomic analysis to identify a potential protein biomarker panel for the diagnosis of tuberculosis. IUBMB Life 2021, 73, 1073–1083. [Google Scholar] [CrossRef]
- Deng, J.; Liu, L.; Yang, Q.; Wei, C.; Zhang, H.; Xin, H.; Pan, S.; Liu, Z.; Wang, D.; Liu, B.; et al. Urinary metabolomic analysis to identify potential markers for the diagnosis of tuberculosis and latent tuberculosis. Arch. Biochem. Biophys. 2021, 704, 108876. [Google Scholar] [CrossRef]
- Mateos, J.; Estévez, O.; González-Fernández, Á.; Anibarro, L.; Pallarés, Á.; Reljic, R.; Gallardo, J.M.; Medina, I.; Carrera, M. High-resolution quantitative proteomics applied to the study of the specific protein signature in the sputum and saliva of active tuberculosis patients and their infected and uninfected contacts. J. Proteomics 2019, 195, 41–52. [Google Scholar] [CrossRef]
- Mateos, J.; Estévez, O.; González-Fernández, Á.; Anibarro, L.; Pallarés, Á.; Reljic, R.; Mussá, T.; Gomes-Maueia, C.; Nguilichane, A.; Gallardo, J.M.; et al. Serum proteomics of active tuberculosis patients and contacts reveals unique processes activated during Mycobacterium tuberculosis infection. Sci. Rep. 2020, 10, 3844. [Google Scholar] [CrossRef] [Green Version]
- Serra-Vidal, M.M.; Latorre, I.; Franken, K.L.C.M.; Díaz, J.; de Souza-Galvão, M.L.; Casas, I.; Maldonado, J.; Milà, C.; Solsona, J.; Jimenez-Fuentes, M.Á.; et al. Immunogenicity of 60 novel latency-related antigens of Mycobacterium tuberculosis. Front. Microbiol. 2014, 5, 517. [Google Scholar] [CrossRef]
- Leyten, E.M.S.; Lin, M.Y.; Franken, K.L.M.C.; Friggen, A.H.; Prins, C.; van Meijgaarden, K.E.; Voskuil, M.I.; Weldingh, K.; Andersen, P.; Schoolnik, G.K.; et al. Human T-cell responses to 25 novel antigens encoded by genes of the dormancy regulon of Mycobacterium tuberculosis. Microbes Infect. 2006, 8, 2052–2060. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Ma, H.; Wan, S.; Zhang, Y.; Gao, M.; Liu, X. Mycobacterium tuberculosis latency-associated antigen Rv1733c SLP improves the accuracy of differential diagnosis of active tuberculosis and latent tuberculosis infection. Chin. Med. J. 2022, 135, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Arroyo, L.; Marín, D.; Franken, K.L.M.C.; Ottenhoff, T.H.M.; Barrera, L.F. Potential of DosR and Rpf antigens from Mycobacterium tuberculosis to discriminate between latent and active tuberculosis in a tuberculosis endemic population of Medellin Colombia. BMC Infect. Dis. 2018, 18, 26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Goletti, D.; Butera, O.; Vanini, V.; Lauria, F.N.; Lange, C.; Franken, K.L.M.C.; Angeletti, C.; Ottenhoff, T.H.M.; Girardi, E. Response to Rv2628 latency antigen associates with cured tuberculosis and remote infection. Eur. Respir. J. 2010, 36, 135–142. [Google Scholar] [CrossRef] [PubMed]
- Adankwah, E.; Nausch, N.; Minadzi, D.; Abass, M.K.; Franken, K.L.M.C.; Ottenhoff, T.H.M.; Mayatepek, E.; Phillips, R.O.; Jacobsen, M. Interleukin-6 and Mycobacterium tuberculosis dormancy antigens improve diagnosis of tuberculosis. J. Infect. 2021, 82, 245–252. [Google Scholar] [CrossRef] [PubMed]
- Peña, D.; Rovetta, A.I.; Hernández Del Pino, R.E.; Amiano, N.O.; Pasquinelli, V.; Pellegrini, J.M.; Tateosian, N.L.; Rolandelli, A.; Gutierrez, M.; Musella, R.M.; et al. A Mycobacterium tuberculosis dormancy antigen differentiates latently infected Bacillus Calmette–Guérin-vaccinated individuals. EBioMedicine 2015, 2, 884–890. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amiano, N.O.; Morelli, M.P.; Pellegrini, J.M.; Tateosian, N.L.; Rolandelli, A.; Seery, V.; Castello, F.A.; Gallego, C.; Armitano, R.; Stupka, J.; et al. IFN-γ and IgG responses to Mycobacterium tuberculosis latency antigen Rv2626c differentiate remote from recent tuberculosis infection. Sci. Rep. 2020, 10, 7472. [Google Scholar] [CrossRef] [PubMed]
- Jee, B.; Singh, Y.; Yadav, R.; Lang, F. Small heat shock protein16.3 of Mycobacterium tuberculosis: After two decades of functional characterization. Cell Physiol. Biochem. 2018, 49, 368–380. [Google Scholar] [CrossRef]
- Belay, M.; Legesse, M.; Mihret, A.; Bekele, Y.; Ottenhoff, T.H.M.; Franken, K.L.M.C.; Bjune, G.; Abebe, F. Pro- and anti-inflammatory cytokines against Rv2031 are elevated during latent tuberculosis: A study in cohorts of tuberculosis patients, household contacts and community controls in an endemic setting. PLoS ONE 2015, 10, e0124134. [Google Scholar] [CrossRef]
- Demissie, A.; Leyten, E.M.S.; Abebe, M.; Wassie, L.; Aseffa, A.; Abate, G.; Fletcher, H.; Owiafe, P.; Hill, P.C.; Brookes, R.; et al. Recognition of stage-specific mycobacterial antigens differentiates between acute and latent infections with Mycobacterium tuberculosis. Clin. Vaccine Immunol. 2006, 13, 179–186. [Google Scholar] [CrossRef] [Green Version]
- Doddam, S.N.; Peddireddy, V.; Ahmed, N. Mycobacterium tuberculosis DosR regulon gene Rv2004c encodes a novel antigen with pro-inflammatory functions and potential diagnostic application for detection of latent tuberculosis. Front. Immunol. 2017, 8, 712. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, W.; Qi, Y.; Ren, C.; Wen, H.; Franken, K.L.M.C.; Ottenhoff, T.H.M.; Shen, J. Interferon-γ responses to Mycobacterium tuberculosis Rpf proteins in contact investigation. Tuberculosis 2013, 93, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wu, J.; Chen, J.; Gao, Y.; Zhang, S.; Zhou, Z.; Huang, H.; Shao, L.; Jin, J.; Zhang, Y.; et al. Evaluation of Mycobacterium tuberculosis-specific antibody responses for the discrimination of active and latent tuberculosis infection. Int. J. Infect. Dis. 2018, 70, 1–9. [Google Scholar] [CrossRef] [Green Version]
- WHO. WHO Warns against the Use of Inaccurate Blood Tests for Active Tuberculosis; World Health Organization: Geneva, Switzerland, 2011; Available online: https://www.who.int/news/item/20-07-2011-who-warns-against-the-use-of-inaccurate-blood-tests-for-active-tuberculosis (accessed on 10 August 2022).
- Li, Z.; Hu, J.; Liu, P.; Cui, D.; Di, H.; Wu, S. Microarray-based selection of a serum biomarker panel that can discriminate between latent and active pulmonary TB. Sci. Rep. 2021, 11, 14516. [Google Scholar] [CrossRef] [PubMed]
- Kumar, S.K.; Arya, S.; Aggarwal, A.; Kapoor, P.; Nath, A.; Misra, R.; Sinha, S. Immune responses to Mycobacterium tuberculosis membrane-associated antigens including alpha crystallin can potentially discriminate between latent infection and active tuberculosis disease. PLoS ONE 2020, 15, e0228359. [Google Scholar] [CrossRef] [PubMed]
- Castro-Garza, J.; García-Jacobo, P.; Rivera-Morales, L.G.; Quinn, F.D.; Barber, J.; Karls, R.; Haas, D.; Helms, S.; Gupta, T.; Blumberg, H.; et al. Detection of anti-HspX antibodies and HspX protein in patient sera for the identification of recent latent infection by Mycobacterium tuberculosis. PLoS ONE 2017, 12, e0181714. [Google Scholar] [CrossRef] [Green Version]
- Rajpal, S.K.; Snehal, S.W.; Miling, S.P.; Hemant, J.P.; Girdhar, M.T.; Hatim, F.D. Mycobacterium tuberculosis heat shock protein 16 as a potential marker for latent TB: A preliminary findings. J. Clin. Cell Immunol. 2011, 2, 1000115. [Google Scholar] [CrossRef]
- Coppola, M.; Arroyo, L.; van Meijgaarden, K.E.; Franken, K.L.M.C.; Geluk, A.; Barrera, L.F.; Ottenhoff, T.H.M. Differences in IgG responses against infection phase related Mycobacterium tuberculosis (Mtb) specific antigens in individuals exposed or not to Mtb correlate with control of TB infection and progression. Tuberculosis 2017, 106, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Kasempimolporn, S.; Thaveekarn, W.; Promrungreang, K.; Khow, O.; Boonchang, S.; Sitprija, V. Improved serodiagnostic sensitivity of strip test for latent tuberculosis. J. Clin. Diagn. Res. 2017, 11, DC01–DC03. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Song, X.; Zhao, Y.; Zhang, H.; Zhao, S.; Mao, F.; Bai, B.; Wu, S.; Shi, C. Mycobacterium tuberculosis secreted proteins as potential biomarkers for the diagnosis of active tuberculosis and latent tuberculosis infection. J. Clin. Lab. Anal. 2015, 29, 375–382. [Google Scholar] [CrossRef]
- Lee, J.Y.; Kim, B.-J.; Koo, H.-K.; Kim, J.; Kim, J.-M.; Kook, Y.-H.; Kim, B.-J. Diagnostic potential of IgG and IgA responses to Mycobacterium tuberculosis antigens for discrimination among active tuberculosis, latent tuberculosis infection, and non-infected individuals. Microorganisms 2020, 8, 979. [Google Scholar] [CrossRef] [PubMed]
- Maekura, R.; Kitada, S.; Osada-Oka, M.; Tateishi, Y.; Ozeki, Y.; Fujicawa, T.; Miki, M.; Jyunnko, O.; Mori, M.; Matsumoto, S. Serum antibody profiles in individuals with latent Mycobacterium tuberculosis infection. Microbiol. Immunol. 2019, 63, 130–138. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, S.-W.; Su, W.-J.; Chan, Y.-J.; Chuang, F.-Y.; Feng, J.-Y.; Chen, Y.-M. Mycobacterium tuberculosis–derived circulating cell-free DNA in patients with pulmonary tuberculosis and persons with latent tuberculosis infection. PLoS ONE 2021, 16, e0253879. [Google Scholar] [CrossRef]
- Bajgai, P.; Sharma, K.; Bansal, R.; Gupta, N.; Sharma, A.; Gupta, A. Detection of Mycobacterium tuberculosis genome in subretinal fluid of patients with latent tuberculosis infection. Ocul. Immunol. Inflamm. 2016, 24, 615–620. [Google Scholar] [CrossRef]
- Das, B.; Kashino, S.S.; Pulu, I.; Kalita, D.; Swami, V.; Yeger, H.; Felsher, D.W.; Campos-Neto, A. CD271(+) bone marrow mesenchymal stem cells may provide a niche for dormant Mycobacterium tuberculosis. Sci. Transl. Med. 2013, 5, 170ra113. [Google Scholar] [CrossRef] [PubMed]
- Belay, M.; Tulu, B.; Younis, S.; Jolliffe, D.A.; Tayachew, D.; Manwandu, H.; Abozen, T.; Tirfie, E.A.; Tegegn, M.; Zewude, A.; et al. Detection of <em>Mycobacterium tuberculosis</em> complex DNA in CD34-positive peripheral blood mononuclear cells of asymptomatic tuberculosis contacts: An observational study. Lancet Microbe 2021, 2, e267–e275. [Google Scholar] [CrossRef]
- Young, B.L.; Mlamla, Z.; Gqamana, P.P.; Smit, S.; Roberts, T.; Peter, J.; Theron, G.; Govender, U.; Dheda, K.; Blackburn, J. The identification of tuberculosis biomarkers in human urine samples. Eur. Respir. J. 2014, 43, 1719–1729. [Google Scholar] [CrossRef] [Green Version]
- Phan, L.M.T.; Kim, E.B.; Cheon, S.A.; Shim, T.S.; Kim, H.-J.; Park, T.J. Reliable naked-eye detection of Mycobacterium tuberculosis antigen 85B using gold and copper nanoshell-enhanced immunoblotting techniques. Sens. Actuators B Chem. 2020, 317, 128220. [Google Scholar] [CrossRef]
- Napolitano, D.R.; Pollock, N.; Kashino, S.S.; Rodrigues, V.; Campos-Neto, A. Identification of Mycobacterium tuberculosis ornithine carboamyltransferase in urine as a possible molecular marker of active pulmonary tuberculosis. Clin. Vaccine Immunol. 2008, 15, 638–643. [Google Scholar] [CrossRef] [Green Version]
- Mehaffy, C.; Dobos, K.M.; Nahid, P.; Kruh-Garcia, N.A. Second generation multiple reaction monitoring assays for enhanced detection of ultra-low abundance Mycobacterium tuberculosis peptides in human serum. Clin. Proteom. 2017, 14, 21. [Google Scholar] [CrossRef] [Green Version]
- Mehaffy, C.; Kruh-Garcia Nicole, A.; Graham, B.; Jarlsberg Leah, G.; Willyerd Charis, E.; Borisov, A.; Sterling Timothy, R.; Nahid, P.; Dobos Karen, M.; Land Geoffrey, A. Identification of Mycobacterium tuberculosis peptides in serum extracellular vesicles from persons with latent tuberculosis infection. J. Clin. Microbiol. 2020, 58, e00393–00320. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- WHO. WHO Consolidated Guidelines on Tuberculosis: Tuberculosis Preventive Treatment: Module 1: Prevention; World Health Organization: Geneva, Switzerland, 2020; Available online: https://www.who.int/publications/i/item/9789240001503 (accessed on 15 August 2022).
- Scolarici, M.; Dekitani, K.; Chen, L.; Sokol-Anderson, M.; Hoft, D.F.; Chatterjee, S. A scoring strategy for progression risk and rates of treatment completion in subjects with latent tuberculosis. PLoS ONE 2018, 13, e0207582. [Google Scholar] [CrossRef] [Green Version]
- Hesseling, A.C.; Mandalakas, A.M.; Kirchner, H.L.; Chegou, N.N.; Marais, B.J.; Stanley, K.; Zhu, X.; Black, G.; Beyers, N.; Walzl, G. Highly discordant T cell responses in individuals with recent exposure to household tuberculosis. Thorax 2009, 64, 840–846. [Google Scholar] [CrossRef] [Green Version]
- Li, R.; Nordio, F.; Huang, C.C.; Contreras, C.; Calderon, R.; Yataco, R.; Galea, J.T.; Zhang, Z.; Becerra, M.C.; Lecca, L.; et al. Two clinical prediction tools to improve tuberculosis contact investigation. Clin. Infect. Dis. 2020, 71, e338–e350. [Google Scholar] [CrossRef] [Green Version]
- Aksornchindarat, W.; Yodpinij, N.; Phetsuksiri, B.; Srisungngam, S.; Rudeeaneksin, J.; Bunchoo, S.; Klayut, W.; Sangkitporn, S.; Khawcharoenporn, T. T-SPOT®.TB test and clinical risk scoring for diagnosis of latent tuberculosis infection among Thai healthcare workers. J. Microbiol. Immunol. Infect. 2021, 54, 305–311. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines on the Management of Latent Tuberculosis Infection; World Health Organization: Geneva, Switzerland, 2015; Available online: https://www.who.int/publications/i/item/9789241548908 (accessed on 10 August 2022).
- Dobler, C.C.; Martin, A.; Marks, G.B. Benefit of treatment of latent tuberculosis infection in individual patients. Eur. Respir. J. 2015, 46, 1397–1406. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- CDC. Treatment Regimens for Latent TB Infection (LTBI); Centers for Disease Control and Prevention: Atlanta, GA, USA, 2020. Available online: https://www.cdc.gov/tb/topic/treatment/ltbi.htm (accessed on 15 August 2022).
- Swindells, S.; Ramchandani, R.; Gupta, A.; Benson, C.A.; Leon-Cruz, J.; Mwelase, N.; Jean Juste, M.A.; Lama, J.R.; Valencia, J.; Omoz-Oarhe, A.; et al. One month of rifapentine plus isoniazid to prevent HIV-related tuberculosis. N. Engl. J. Med. 2019, 380, 1001–1011. [Google Scholar] [CrossRef]
- CDC. Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV; Centers for Disease Control and Prevention: Atlanta, GA, USA, 2022. Available online: https://clinicalinfo.hiv.gov/en/guidelines/hiv-clinical-guidelines-adult-and-adolescent-opportunistic-infections/mycobacterium (accessed on 14 October 2022).
- Pradipta, I.S.; Houtsma, D.; van Boven, J.F.M.; Alffenaar, J.-W.C.; Hak, E. Interventions to improve medication adherence in tuberculosis patients: A systematic review of randomized controlled studies. NPJ Prim. Care Respir. Med. 2020, 30, 21. [Google Scholar] [CrossRef]
- Anibarro, L.; Casas, S.; Paz-Esquete, J.; Gonzalez, L.; Pena, A.; Guerra, M.R.; Sande, D.; Calviño, L.; Santin, M.; SEIMC. Treatment completion in latent tuberculosis infection at specialist tuberculosis units in Spain. Int. J. Tuberc. Lung Dis. 2010, 14, 701–707. [Google Scholar]
- Oren, E.; Bell, M.L.; Garcia, F.; Perez-Velez, C.; Gerald, L.B. Promoting adherence to treatment for latent TB infection through mobile phone text messaging: Study protocol for a pilot randomized controlled trial. Pilot Feasibility Stud. 2017, 3, 15. [Google Scholar] [CrossRef] [Green Version]
- Sterling, T.R.; Villarino, M.E.; Borisov, A.S.; Shang, N.; Gordin, F.; Bliven-Sizemore, E.; Hackman, J.; Hamilton, C.D.; Menzies, D.; Kerrigan, A.; et al. Three months of rifapentine and isoniazid for latent tuberculosis infection. N. Engl. J. Med. 2011, 365, 2155–2166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, S.J.; Lee, H.; Koo, G.W.; Min, J.-H.; Yeo, Y.; Park, D.W.; Park, T.S.; Moon, J.-Y.; Kim, S.-H.; Kim, T.H.; et al. Adherence to nine-month isoniazid for latent tuberculosis infection in healthcare workers: A prospective study in a tertiary hospital. Sci. Rep. 2020, 10, 6462. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, S.-Y.; Chiu, Y.-W.; Lu, P.-L.; Hwang, S.-J.; Chen, T.-C.; Hsieh, M.-H.; Chen, Y.-H. Three months of rifapentine and isoniazid for latent tuberculosis infection in hemodialysis patients: High rates of adverse events. J. Microbiol. Immunol. Infect. 2019, 52, 158–162. [Google Scholar] [CrossRef] [PubMed]
- Ilaiwy, G.; Dowdy, D.W. Cost effectiveness of three months of rifapentine and isoniazid for latent tuberculosis in Syrian refugees. J. Clin. Tuberc. Other Mycobact Dis. 2021, 24, 100262. [Google Scholar] [CrossRef] [PubMed]
- Borisov, A.S.; Bamrah Morris, S.; Njie, G.J.; Winston, C.A.; Burton, D.; Goldberg, S.; Yelk Woodruff, R.; Allen, L.; LoBue, P.; Vernon, A. Update of recommendations for use of once-weekly isoniazid-rifapentine regimen to treat latent mycobacterium tuberculosis infection. MMWR Morb. Mortal. Wkly. Rep. 2018, 67, 723–726. [Google Scholar] [CrossRef] [Green Version]
- Malik, A.A.; Gandhi, N.R.; Lash, T.L.; Cranmer, L.M.; Omer, S.B.; Ahmed, J.F.; Siddiqui, S.; Amanullah, F.; Khan, A.J.; Keshavjee, S.; et al. Effectiveness of preventive therapy for persons exposed at home to drug-resistant tuberculosis, Karachi, Pakistan. Emerg. Infect. Dis. 2021, 27, 805–812. [Google Scholar] [CrossRef]
- Nahid, P.; Mase, S.R.; Migliori, G.B.; Sotgiu, G.; Bothamley, G.H.; Brozek, J.L.; Cattamanchi, A.; Cegielski, J.P.; Chen, L.; Daley, C.L.; et al. Treatment of drug-resistant tuberculosis. An official ATS/CDC/ERS/IDSA clinical practice guideline. Am. J. Respir. Crit. Care Med. 2019, 200, e93–e142. [Google Scholar] [CrossRef]
- Ferguson, O.; Jo, Y.; Pennington, J.; Johnson, K.; Chaisson, R.E.; Churchyard, G.; Dowdy, D. Cost-effectiveness of one month of daily isoniazid and rifapentine versus three months of weekly isoniazid and rifapentine for prevention of tuberculosis among people receiving antiretroviral therapy in Uganda. J. Int. AIDS Soc. 2020, 23, e25623. [Google Scholar] [CrossRef]
- Dobler, C.C.; Batbayar, O.; Wright, C.M. Practical challenges and solutions to TB control in a lower-middle-income country: Experiences from Mongolia. Breathe 2018, 14, 180–183. [Google Scholar] [CrossRef] [Green Version]
- Horn, D.L.; Hewlett, D.; Alfalla, C.; Peterson, S.; Opal, S.M. Limited tolerance of ofloxacin and pyrazinamide prophylaxis against tuberculosis. N. Engl. J. Med. 1994, 330, 1241. [Google Scholar] [CrossRef]
- Younossian, A.B.; Rochat, T.; Ketterer, J.-P.; Wacker, J.; Janssens, J.-P. High hepatotoxicity of pyrazinamide and ethambutol for treatment of latent tuberculosis. Eur. Respir. J. 2005, 26, 462–464. [Google Scholar] [CrossRef] [PubMed]
- Bamrah, S.; Brostrom, R.; Dorina, F.; Setik, L.; Song, R.; Kawamura, L.M.; Heetderks, A.; Mase, S. Treatment for LTBI in contacts of MDR-TB patients, Federated States of Micronesia, 2009–2012. Int. J. Tuberc. Lung Dis. 2014, 18, 912–918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fox, G.J.; Nguyen, C.B.; Nguyen, T.A.; Tran, P.T.; Marais, B.J.; Graham, S.M.; Nguyen, B.H.; Velen, K.; Dowdy, D.W.; Mason, P.; et al. Levofloxacin versus placebo for the treatment of latent tuberculosis among contacts of patients with multidrug-resistant tuberculosis (the VQUIN MDR trial): A protocol for a randomised controlled trial. BMJ Open 2020, 10, e033945. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seddon, J.A.; Garcia-Prats, A.J.; Purchase, S.E.; Osman, M.; Demers, A.-M.; Hoddinott, G.; Crook, A.M.; Owen-Powell, E.; Thomason, M.J.; Turkova, A.; et al. Levofloxacin versus placebo for the prevention of tuberculosis disease in child contacts of multidrug-resistant tuberculosis: Study protocol for a phase III cluster randomised controlled trial (TB-CHAMP). Trials 2018, 19, 693. [Google Scholar] [CrossRef]
- NIH. Protecting Households on Exposure to Newly Diagnosed Index Multidrug-Resistant Tuberculosis Patients (PHOENIx MDR-TB). 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT03568383 (accessed on 10 August 2022).
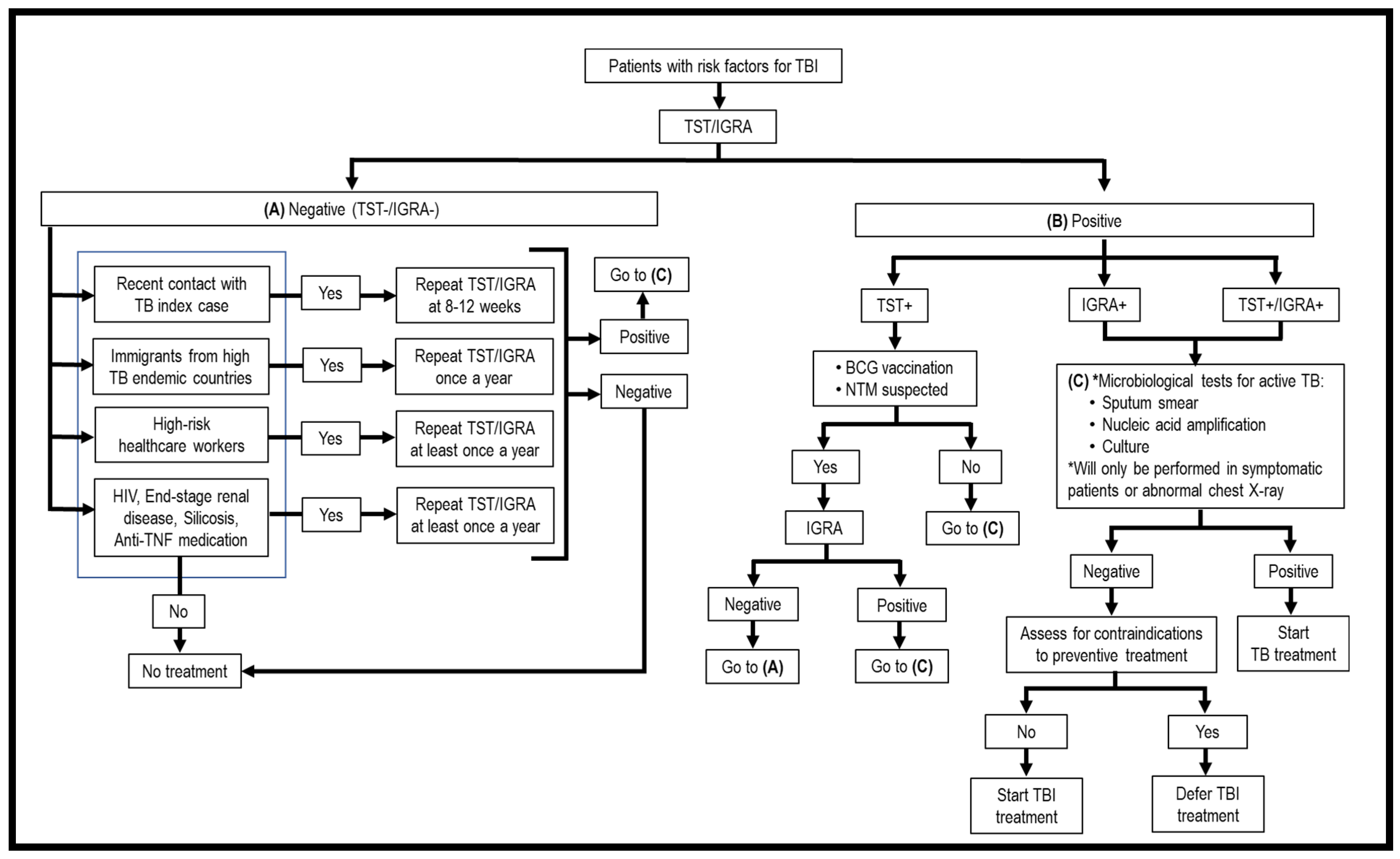

| Tuberculosis Infection (TBI) | Active Tuberculosis (ATB) |
|---|---|
|
|
| Criteria | TST | QFT® | T-SPOT®.TB |
|---|---|---|---|
| Sample | Skin test | Whole blood (Processed within 16 h) | PBMCs (Processed within 8 to 32 h) |
| Responses | Delayed-type hypersensitivity | T cells immune responses | |
| Frequency of patient visit | Two times visit | One time visit | |
| Sample processing step before the test | None | None | Isolation of PBMCs from whole blood and cell counting |
| Antigens | PPD | QFT®-GIT: ESAT-6, CFP-10, and TB7.7 QFT®-Plus:
|
|
| Protocol | 0.1 mL (5 IU) tuberculin injection at forearm | 1 mL of blood for individual tubes | 250,000 ± 50,000 cells/well |
| Platform | Induration | Enzyme-linked immunosorbent assay (ELISA) | Enzyme-linked immunospot assay (ELISPOT) |
| Principle | Measure size of induration after intradermal PPD inoculation | Quantify amount of INF-γ released by CD4 and CD8 T cells | Count number of cells that release IFN-γ |
| Maximum number of samples per run | Individual | QFT®-GIT: 28 samples; QFT®-Plus: 22 samples in a 96-well microtiter plate | 24 samples in a 96-well microtiter plate |
| Equipment | Ruler |
|
|
| Results interpretation |
|
|
|
| Indeterminate/Invalid | - |
|
|
| False-positive | BCG vaccination and NTM infections | Results not affected by BCG vaccination and most NTM infections with exception of M. kansasii, M. szulgai, and M. marinum | |
| Turnaround time | 48 to 72 h | 24 h | |
| Interpretation | Subjective | Not affected by bias | Subjective |
| Antigen | Participants | Sample | Stimulating Time | Assay | Cytokines/Chemokines Associated to TB Infection | Cytokines/Chemokines to Distinguish TBI and Active TB | Ref. |
|---|---|---|---|---|---|---|---|
| PPD (10 μg/mL) | 5–85 years old | PBMCs | 24 h | Bio-Plex Pro Human Cytokine 27-plex Assay (Bio-Rad, Hercules, CA, USA) | Stimulated:
| Stimulated:
| [117] |
| ESAT-6+CFP-10 (10 μg/mL) | 15–74 years old | Whole blood | 20–24 h | Bio-Plex Pro Human Cytokine 27-plex Assay (Bio-Rad, Hercules, CA, USA) | Unstimulated:
| Unstimulated:
| [118] |
| ESAT-6 (10 μg/mL), CFP-10 (10 μg/mL), or PPD (20 μg/mL) | 24–58 years old | Whole blood | 19 h | 8-plex human cytokine assay (Bio-Rad, Hercules, CA, USA) | Stimulated:
| Stimulated:
| [119] |
| ESAT-6 (10 μg/mL), CFP-10 (10 μg/mL), or PPD (20 μg/mL) | <18 years old | Whole blood | 20–24 h | 17-plex, Milliplex human cytokine/chemokine kits (Millipore Corp., Billerica, MA, USA) | Stimulated:
| Stimulated:
| [120] |
| ESAT6, CFP10, and MTB7.7-coated polyester beads | PTB and HCW (TST+ and TST−), Adults | Whole blood | Overnight | Human XL Cytokine Discovery Premixed 10-plex kit (R&D Systems, Minneapolis, MN, USA) | Stimulated:
| Stimulated:
| [114] |
| QFT®-GIT | 21–55 years old | Whole blood | 16–24 h | Bio-Plex Pro Human Cytokine 27-plex Assay (Bio-Rad, Hercules, CA, USA) | - | Unstimulated:
| [121] |
| QFT®-GIT | 15–89 years old | Whole blood | 16–24 h | 29-plex, Milliplex human cytokine/chemokine kits (Millipore Corp., Billerica, MA, USA) | Stimulated:
| Unstimulated:
| [122] |
| QFT®-GIT | 10–60 years old | Whole blood | Overnight | 29-plex LINCO-plex® kits (Millipore, St. Charles, MO, USA) | - | Unstimulated:
| [123] |
| QFT®-GIT | 17–84 years old | Whole blood | 16–24 h | 48-plex, Bio-Plex platform (Bio-Rad) | Unstimulated:
| Seven biomarkers:
| [124] |
| QFT®-GIT | Healthcare workers (HCW) and community control (CC), >18 years old | Whole blood | Overnight | Procartaplex11-plex (Life Technologies, Thermo Fisher Scientific, Waltham, MA, USA) | - | Stimulated:
| [125] |
| Participants | Sample | Assay | Results | Ref. |
|---|---|---|---|---|
| Adults | Whole blood | Microarray |
| [172] |
| Adults | Whole blood | RNA-Seq |
| [173] |
| Adults | Serum | RNA-Seq |
| [174] |
| Adults | PBMCs | Microarray |
| [175] |
| Adults | PBMCs | Microarray |
| [176] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chin, K.L.; Anibarro, L.; Sarmiento, M.E.; Acosta, A. Challenges and the Way forward in Diagnosis and Treatment of Tuberculosis Infection. Trop. Med. Infect. Dis. 2023, 8, 89. https://doi.org/10.3390/tropicalmed8020089
Chin KL, Anibarro L, Sarmiento ME, Acosta A. Challenges and the Way forward in Diagnosis and Treatment of Tuberculosis Infection. Tropical Medicine and Infectious Disease. 2023; 8(2):89. https://doi.org/10.3390/tropicalmed8020089
Chicago/Turabian StyleChin, Kai Ling, Luis Anibarro, Maria E. Sarmiento, and Armando Acosta. 2023. "Challenges and the Way forward in Diagnosis and Treatment of Tuberculosis Infection" Tropical Medicine and Infectious Disease 8, no. 2: 89. https://doi.org/10.3390/tropicalmed8020089
APA StyleChin, K. L., Anibarro, L., Sarmiento, M. E., & Acosta, A. (2023). Challenges and the Way forward in Diagnosis and Treatment of Tuberculosis Infection. Tropical Medicine and Infectious Disease, 8(2), 89. https://doi.org/10.3390/tropicalmed8020089






