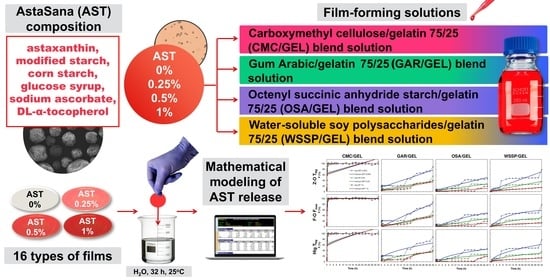
Mathematical Modeling of Water-Soluble Astaxanthin Release from Binary Polysaccharide/Gelatin Blend Matrices
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Film Preparation
2.3. Film Thickness
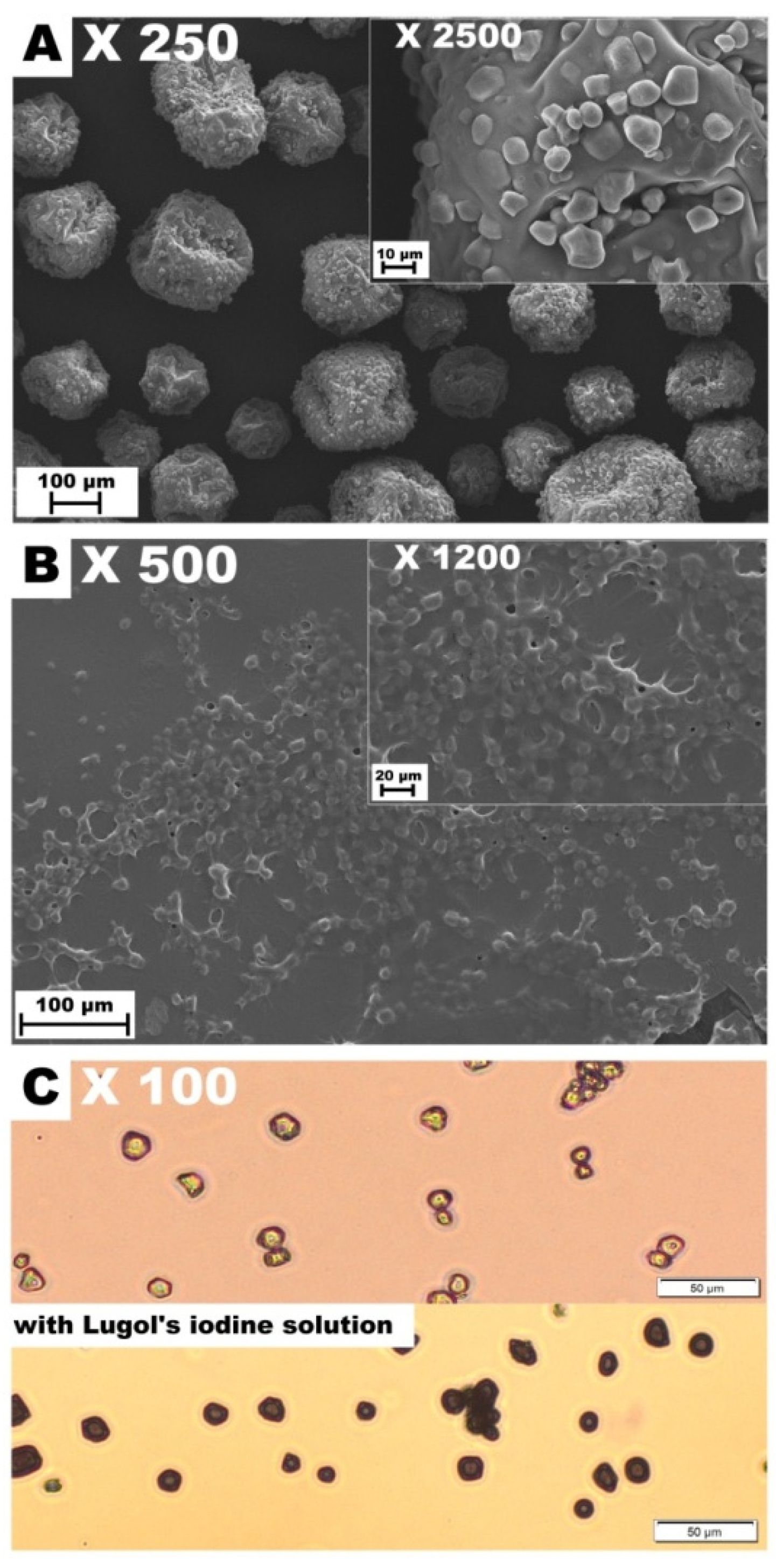
2.4. Scanning Electron Microscopy (SEM) and Light Microscopy
2.5. AST Release Study
2.6. Mathematical Modeling
3. Results
3.1. Microstructure and Film Thickness
3.2. Release of AST
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Nyamweya, N.N. Applications of polymer blends in drug delivery. Future J. Pharm. Sci. 2021, 7, 18. [Google Scholar] [CrossRef]
- Cutter, C.N. Microbial control by packaging: A review. Crit. Rev. Food Sci. Nutr. 2002, 42, 151–161. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, D. Biopolymer/candelilla wax emulsion films as carriers of ascorbic acid—A comparative study. Food Hydrocoll. 2016, 52, 543–553. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Biendl, M. Physicochemical and antioxidant properties of biopolymer/candelilla wax emulsion films containing hop extract—A comparative study. Food Hydrocoll. 2016, 60, 384–392. [Google Scholar] [CrossRef]
- Young, S.; Wong, M.; Tabata, Y.; Mikos, A.G. Gelatin as a delivery vehicle for the controlled release of bioactive molecules. J. Control. Release 2005, 109, 256–274. [Google Scholar] [CrossRef] [PubMed]
- Uz, M.; Altinkaya, S.A. Development of mono and multilayer antimicrobial food packaging materials for controlled release of potassium sorbate. LWT—Food Sci. Technol. 2011, 44, 2302–2309. [Google Scholar] [CrossRef] [Green Version]
- Łupina, K.; Kowalczyk, D.; Zięba, E.; Kazimierczak, W.; Mężyńska, M.; Basiura-Cembala, M.; Wiącek, A.E. Edible films made from blends of gelatin and polysaccharide-based emulsifiers—A comparative study. Food Hydrocoll. 2019, 96, 555–567. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Pytka, M.; Szymanowska, U.; Skrzypek, T.; Łupina, K.; Biendl, M. Release kinetics and antibacterial activity of potassium salts of iso-α-acids loaded into the films based on gelatin, carboxymethyl cellulose and their blends. Food Hydrocoll. 2020, 109, 106104. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Kordowska-Wiater, M.; Karaś, M.; Zięba, E.; Mężyńska, M.; Wiącek, A.E. Release kinetics and antimicrobial properties of the potassium sorbate-loaded edible films made from pullulan, gelatin and their blends. Food Hydrocoll. 2020, 101, 105539. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Baraniak, B. Effect of candelilla wax on functional properties of biopolymer emulsion films—A comparative study. Food Hydrocoll. 2014, 41, 195–209. [Google Scholar] [CrossRef]
- Chiwele, I.; Jones, B.E.; Podczeck, F. The shell dissolution of various empty hard capsules. Chem. Pharm. Bull. 2000, 48, 951–956. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Huang, J.; Zheng, X.; Liu, S.; Lu, K.; Tang, K.; Liu, J. Heat sealable soluble soybean polysaccharide/gelatin blend edible films for food packaging applications. Food Packag. Shelf Life 2020, 24, 100485. [Google Scholar] [CrossRef]
- Jridi, M.; Abdelhedi, O.; Salem, A.; Kechaou, H.; Nasri, M.; Menchari, Y. Physicochemical, antioxidant and antibacterial properties of fish gelatin-based edible films enriched with orange peel pectin: Wrapping application. Food Hydrocoll. 2020, 103, 105688. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Szymanowska, U.; Skrzypek, T.; Basiura-Cembala, M.; Łupina, K.; Biendl, M. Edible films based on gelatin, carboxymethyl cellulose, and their blends as carriers of potassium salts of iso-α-acids: Structural, physicochemical and antioxidant properties. Food Hydrocoll. 2021, 115. [Google Scholar] [CrossRef]
- Kowalczyk, D.; Skrzypek, T.; Basiura-Cembala, M.; Łupina, K.; Mężyńska, M. The effect of potassium sorbate on the physicochemical properties of edible films based on pullulan, gelatin and their blends. Food Hydrocoll. 2020, 105, 105837. [Google Scholar] [CrossRef]
- Łupina, K.; Kowalczyk, D.; Drozłowska, E. Polysaccharide/gelatin blend films as carriers of ascorbyl palmitate—A comparative study. Food Chem. 2020, 333, 127465. [Google Scholar] [CrossRef]
- Łupina, K.; Kowalczyk, D.; Kazimierczak, W. Gum Arabic/gelatin and water-soluble soy polysaccharides/gelatin blend films as carriers of astaxanthin—A comparative study of the kinetics of release and antioxidant properties. Polymers 2021, 13, 1062. [Google Scholar] [CrossRef]
- Fakhri, S.; Abbaszadeh, F.; Dargahi, L.; Jorjani, M. Astaxanthin: A mechanistic review on its biological activities and health benefits. Pharmacol. Res. 2018, 136, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, E. Astaxanthin as a Medical Food. Funct. Foods Health Dis. 2013, 3, 254–258. [Google Scholar] [CrossRef]
- Landon, R.; Gueguen, V.; Petite, H.; Letourneur, D.; Pavon-Djavid, G.; Anagnostou, F. Impact of Astaxanthin on Diabetes Pathogenesis and Chronic Complications. Mar. Drugs 2020, 18, 357. [Google Scholar] [CrossRef] [PubMed]
- Donoso, A.; González-Durán, J.; Muñoz, A.A.; González, P.A.; Agurto-Muñoz, C. Therapeutic uses of natural astaxanthin: An evidence-based review focused on human clinical trials. Pharmacol. Res. 2021, 166. [Google Scholar] [CrossRef]
- Faraone, I.; Sinisgalli, C.; Ostuni, A.; Armentano, M.F.; Carmosino, M.; Milella, L.; Russo, D.; Labanca, F.; Khan, H. Astaxanthin anticancer effects are mediated through multiple molecular mechanisms: A systematic review. Pharmacol. Res. 2020, 155, 104689. [Google Scholar] [CrossRef]
- Brendler, T.; Williamson, E.M. Astaxanthin: How much is too much? A safety review. Phyther. Res. 2019, 33, 3090–3111. [Google Scholar] [CrossRef] [PubMed]
- Capelli, B.; Bagchi, D.; Cysewski, G.R. Synthetic astaxanthin is significantly inferior to algal-based astaxanthin as an antioxidant and may not be suitable as a human nutraceutical supplement. Nutrafoods 2013, 12, 145–152. [Google Scholar] [CrossRef]
- Koller, M.; Muhr, A.; Braunegg, G. Microalgae as versatile cellular factories for valued products. Algal Res. 2014, 6, 52–63. [Google Scholar] [CrossRef]
- Colín-Chávez, C.; Soto-Valdez, H.; Peralta, E.; Lizardi-Mendoza, J.; Balandrán-Quintana, R. Diffusion of natural astaxanthin from polyethylene active packaging films into a fatty food simulant. Food Res. Int. 2013, 54, 873–880. [Google Scholar] [CrossRef]
- Zhang, Y.; Huo, M.; Zhou, J.; Zou, A.; Li, W.; Yao, C.; Xie, S. DDSolver: An add-in program for modeling and comparison of drug dissolution profiles. AAPS J. 2010, 12, 263–271. [Google Scholar] [CrossRef] [Green Version]
- Singh, J.; Colussi, R.; McCarthy, O.J.; Kaur, L. Potato Starch and Its Modification. In Advances in Potato Chemistry and Technology, 2nd ed.; Singh, J., Kaur, L., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2016; pp. 195–247. ISBN 9780128000021. Available online: https://www.sciencedirect.com/science/article/pii/B978012800002100008X (accessed on 1 February 2021).
- Ikeda, S.; Funami, T.; Zhang, G. Visualizing surface active hydrocolloids by atomic force microscopy. Carbohydr. Polym. 2005, 62, 192–196. [Google Scholar] [CrossRef]
- Costa, P.; Lobo, J.M.S. Modeling and comparison of dissolution profiles. Eur. J. Pharm. Sci. 2001, 13, 123–133. [Google Scholar] [CrossRef]
- Olejnik, A.; Kapuscinska, A.; Schroeder, G.; Nowak, I. Physico-chemical characterization of formulations containing endomorphin-2 derivatives. Amino Acids 2017, 49, 1719–1731. [Google Scholar] [CrossRef] [Green Version]
- Permanadewi, I.; Kumoro, A.C.; Wardhani, D.H.; Aryanti, N. Modelling of controlled drug release in gastrointestinal tract simulation. J. Phys. Conf. Ser. 2019, 1295, 012063. [Google Scholar] [CrossRef]
- Bruschi, M. Mathematical models of drug release. In Strategies to Modify the Drug Release from Pharmaceutical Systems; Bruschi, M., Ed.; Woodhead Publishing: Cambridge, UK, 2015; pp. 63–86. ISBN 9780081000922. [Google Scholar]
- Shah, S.N.H.; Asghar, S.; Choudhry, M.A.; Akash, M.S.H.; Rehman, N.U.; Baksh, S. Formulation and evaluation of natural gum-based sustained release matrix tablets of flurbiprofen using response surface methodology. Drug Dev. Ind. Pharm. 2009, 35, 1470–1478. [Google Scholar] [CrossRef]
- Batra, V.; Bhowmick, A.; Behera, B.K.; Ray, A.R. Sustained release of ferrous sulfate from polymer-coated gum arabica pellets. J. Pharm. Sci. 1994, 83, 632–635. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Khalid, N.; Shu, G.; Neves, M.A.; Kobayashi, I.; Nakajima, M. Complex coacervates from gelatin and octenyl succinic anhydride modified kudzu starch: Insights of formulation and characterization. Food Hydrocoll. 2019, 86, 70–77. [Google Scholar] [CrossRef]
- Builders, P.F.; Arhewoh, M.I. Pharmaceutical applications of native starch in conventional drug delivery. Starch/Staerke 2016, 68. [Google Scholar] [CrossRef]




| Model | Equation | Parameters |
|---|---|---|
| Zero-order with Tlag (Z-O Tlag) | k0, Tlag | |
| First-order with Fmax (F-O Fmax) | k1, Fmax | |
| Higuchi with Tlag (Hig Tlag) | kH, Tlag | |
| Hopfenberg with Tlag (Hop Tlag) | kHB, n, Tlag | |
| Korsmeyer–Peppas with Tlag (K-P Tlag) | kKP, n, Tlag | |
| Logistic 2 (Log 2) | α, β, Fmax | |
| Makoid–Banakar with Tlag (M-BTlag ) | kMB, n, k, Tlag | |
| Weibull with Fmax (Wb Fmax) | α, β, Ti, Fmax |
| Film | AST (%) | Thickness (µm) | t25% (h) |
|---|---|---|---|
| CMC/GEL | 0 | 91.32 ± 3.96 ab | - |
| 0.25 | 91.01 ± 3.23 ab | 0.09 (Wb Fmax) | |
| 0.5 | 95.88 ± 3.71 b | 0.15 (Wb Fmax) | |
| 1 | 94.17 ± 2.19 b | 0.09 (Wb Fmax) | |
| GAR/GEL | 0 | 88.90 ± 2.40 a | - |
| 0.25 | 86.74 ± 3.21 a | 7.20 (M-B Tlag) | |
| 0.5 | 87.26 ± 3.87 a | 22.27 (M-B Tlag) | |
| 1 | 88.88 ± 2.33 a | 24.00 (M-B Tlag) | |
| OSA/GEL | 0 | 88.33 ± 1.86 a | - |
| 0.25 | 86.39 ± 3.68 a | 13.34 (M-B Tlag) | |
| 0.5 | 87.72 ± 2.55 a | 27.26 (K-P Tlag) | |
| 1 | 86.67 ± 3.61 a | n.c. (M-B Tlag) | |
| WSSP/GEL | 0 | 88.88 ± 2.71 a | - |
| 0.25 | 88.21 ± 2.02 a | 6.16 (Log 2) | |
| 0.5 | 87.57 ± 3.73 a | 17.46 (Log 2) | |
| 1 | 86.75 ± 2.10 a | 50.95 (M-B Tlag) |
| Film | AST (%) | Z-O Tlag | F-O Fmax | Hig Tlag | Hop Tlag | K-P Tlag | Log 2 | M-B Tlag | Wb Fmax |
|---|---|---|---|---|---|---|---|---|---|
| CMC/GEL | 0.25 | 0.2105 | 0.9503 | 0.2426 | 0.9575 | 0.9494 | 0.9606 | 0.7610 | 0.9610 |
| 0.5 | 0.2194 | 0.9481 | 0.2567 | 0.9869 | 0.9410 | 0.9872 | 0.7517 | 0.9890 | |
| 1 | 0.2642 | 0.9824 | 0.3099 | 0.9824 | 0.9457 | 0.9821 | 0.9621 | 0.9829 | |
| GAR/GEL | 0.25 | 0.8899 | 0.5867 | 0.7899 | 0.9238 | 0.9304 | 0.6389 | 0.9380 | 0.9180 |
| 0.5 | 0.7780 | 0.5315 | 0.3689 | 0.9220 | 0.8642 | 0.5142 | 0.9341 | 0.7953 | |
| 1 | 0.8699 | 0.8289 | 0.7864 | 0.9231 | 0.8397 | 0.8397 | 0.9356 | 0.9106 | |
| OSA/GEL | 0.25 | 0.9335 | 0.8729 | 0.8139 | 0.9314 | 0.9313 | 0.8736 | 0.9461 | 0.9287 |
| 0.5 | 0.8852 | 0.7970 | 0.7472 | 0.8881 | 0.8906 | 0.8093 | 0.8861 | 0.7963 | |
| 1 | 0.5954 | 0.5911 | 0.8326 | 0.8446 | 0.8411 | 0.8404 | 0.8980 | 0.7422 | |
| WSSP/GEL | 0.25 | 0.8523 | 0.9248 | 0.9192 | 0.8956 | 0.9162 | 0.9275 | 0.9187 | 0.9226 |
| 0.5 | 0.8871 | 0.9412 | 0.9297 | 0.9037 | 0.9302 | 0.9439 | 0.9389 | 0.9425 | |
| 1 | 0.9281 | 0.9627 | 0.9832 | 0.9315 | 0.9827 | 0.9750 | 0.9967 | 0.9817 | |
| Average R2adjusted | 0.6928 | 0.8265 | 0.6650 | 0.9242 * | 0.9135 | 0.8577 | 0.9056 | 0.9059 | |
| Film | AST (%) | Z-O Tlag | F-O Fmax | Hig Tlag | |||
|---|---|---|---|---|---|---|---|
| k0 | Tlag | k1 | Fmax | kH | Tlag | ||
| CMC/GEL | 0.25 | 1.603 | −41.703 | 4.399 | 101.594 | 17.865 | −12.630 |
| 0.5 | 1.740 | −36.049 | 3.478 | 100.957 | 18.494 | −9.997 | |
| 1 | 1.790 | −34.293 | 3.230 | 99.919 | 18.721 | −9.220 | |
| GAR/GEL | 0.25 | 1.920 | −8.124 | 0.008 | 355.699 | 11.542 | −1.825 |
| 0.5 | 0.959 | −7.786 | 0.00009 | 15,279.747 | 6.265 | 1.758 | |
| 1 | 0.987 | −3.007 | 0.00008 | 14,031.338 | 8.906 | 13.589 | |
| OSA/GEL | 0.25 | 1.414 | −4.041 | 0.027 | 85.433 | 8.259 | 1.882 |
| 0.5 | 0.770 | −4.465 | 0.013 | 85.507 | 4.555 | 1.783 | |
| 1 | 0.354 | −1.155 | 0.00003 | 13,204.829 | 6.941 | 23.867 | |
| WSSP/GEL | 0.25 | 2.077 | −2.043 | 0.084 | 62.251 | 11.100 | 0.840 |
| 0.5 | 1.077 | −1.650 | 0.066 | 34.939 | 5.613 | 0.840 | |
| 1 | 0.443 | −2.518 | 0.070 | 14.438 | 2.392 | 0.670 | |
| Film | AST (%) | Hop Tlag | K-P Tlag | Log 2 | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| kHB | n | Tlag | kKP | n | Tlag | α | β | Fmax | ||
| CMC/GEL | 0.25 | 2.224 | 1.433 | −0.003 | 97.541 | 0.017 | 0.170 | 4.947 | 6.096 | 101.023 |
| 0.5 | 2.805 | 0.421 | −0.025 | 98.100 | 0.007 | 0.330 | 8.472 | 11.740 | 99.556 | |
| 1 | 0.009 | 371.689 | 0.008 | 86.585 | 0.055 | 0.140 | 2.552 | 3.563 | 100.352 | |
| GAR/GEL | 0.25 | 0.021 | 0.525 | −14.133 | 0.000 | 2.904 | −45.281 | −7.465 | 1.016 | 25,582.192 |
| 0.5 | 0.017 | 0.160 | −26.110 | 0.000 | 3.321 | −44.884 | −9.942 | 2.382 | 24,495.904 | |
| 1 | 0.023 | 0.222 | −7.182 | 0.372 | 1.348 | 0.006 | −10.486 | 3.112 | 13,213.903 | |
| OSA/GEL | 0.25 | 0.012 | 1.243 | −3.730 | 1.117 | 1.062 | −4.771 | −8.514 | 1.693 | 19,250.461 |
| 0.5 | 0.017 | 0.336 | −6.449 | 0.047 | 1.686 | −13.899 | −8.760 | 1.609 | 15,235.496 | |
| 1 | 0.026 | 0.051 | −6.165 | 0.000 | 3.982 | −0.068 | −17.133 | 10.354 | 118.171 | |
| WSSP/GEL | 0.25 | 0.000 | 212.634 | −0.439 | 11.051 | 0.501 | 0.840 | −3.559 | 4.060 | 60.547 |
| 0.5 | 0.000 | 396.451 | −0.901 | 4.454 | 0.577 | 0.696 | −4.110 | 4.293 | 32.359 | |
| 1 | 0.000 | 122.841 | −2.150 | 2.484 | 0.487 | 0.670 | −3.876 | 1.489 | 83.440 | |
| Film | AST (%) | M-B Tlag | Wb Fmax | ||||||
|---|---|---|---|---|---|---|---|---|---|
| kMB | n | k | Tlag | α | β | Ti | Fmax | ||
| CMC/GEL | 0.25 | 82.488 | 0.234 | 0.023 | 0.008 | 0.057 | 2.112 | −0.053 | 100.652 |
| 0.5 | 78.744 | 0.261 | 0.025 | 0.008 | 3.748 | 12.760 | −0.858 | 99.641 | |
| 1 | 89.040 | 0.097 | 0.008 | 0.140 | 0.321 | 0.858 | 0.027 | 99.944 | |
| GAR/GEL | 0.25 | 18.160 | −0.058 | −0.055 | −0.775 | 17,018.112 | 2.012 | −23.973 | 468.274 |
| 0.5 | 12.085 | −0.442 | −0.089 | −1.822 | 1309.071 | 1.554 | −14.722 | 153.488 | |
| 1 | 4.794 | 0.009 | −0.068 | 0.003 | 3059.906 | 1.849 | −10.398 | 132.949 | |
| OSA/GEL | 0.25 | 0.001 | 3.757 | 0.066 | −15.353 | 6993.898 | 1.065 | −4.780 | 7756.062 |
| 0.5 | 0.128 | 1.393 | −0.005 | −11.203 | 110.319 | 0.722 | 0.027 | 255.772 | |
| 1 | 0.600 | −0.333 | −0.147 | −0.014 | 25,028.532 | 2.473 | −4.127 | 63.672 | |
| WSSP/GEL | 0.25 | 6.969 | 0.819 | 0.023 | 0.622 | 21.504 | 1.409 | 0.001 | 54.429 |
| 0.5 | 1.575 | 1.197 | 0.038 | 0.013 | 38.243 | 1.561 | −0.067 | 28.572 | |
| 1 | 3.825 | 0.162 | −0.025 | 0.840 | 98.199 | 0.495 | 0.670 | 243.555 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Łupina, K.; Kowalczyk, D.; Skrzypek, T.; Baraniak, B. Mathematical Modeling of Water-Soluble Astaxanthin Release from Binary Polysaccharide/Gelatin Blend Matrices. Colloids Interfaces 2021, 5, 41. https://doi.org/10.3390/colloids5030041
Łupina K, Kowalczyk D, Skrzypek T, Baraniak B. Mathematical Modeling of Water-Soluble Astaxanthin Release from Binary Polysaccharide/Gelatin Blend Matrices. Colloids and Interfaces. 2021; 5(3):41. https://doi.org/10.3390/colloids5030041
Chicago/Turabian StyleŁupina, Katarzyna, Dariusz Kowalczyk, Tomasz Skrzypek, and Barbara Baraniak. 2021. "Mathematical Modeling of Water-Soluble Astaxanthin Release from Binary Polysaccharide/Gelatin Blend Matrices" Colloids and Interfaces 5, no. 3: 41. https://doi.org/10.3390/colloids5030041
APA StyleŁupina, K., Kowalczyk, D., Skrzypek, T., & Baraniak, B. (2021). Mathematical Modeling of Water-Soluble Astaxanthin Release from Binary Polysaccharide/Gelatin Blend Matrices. Colloids and Interfaces, 5(3), 41. https://doi.org/10.3390/colloids5030041