Optical Switches for Lipid Membranes: Computed Molecular Projection Area as a Switch Selection Criterion
Abstract
:1. Introduction
2. Objects and Methodology
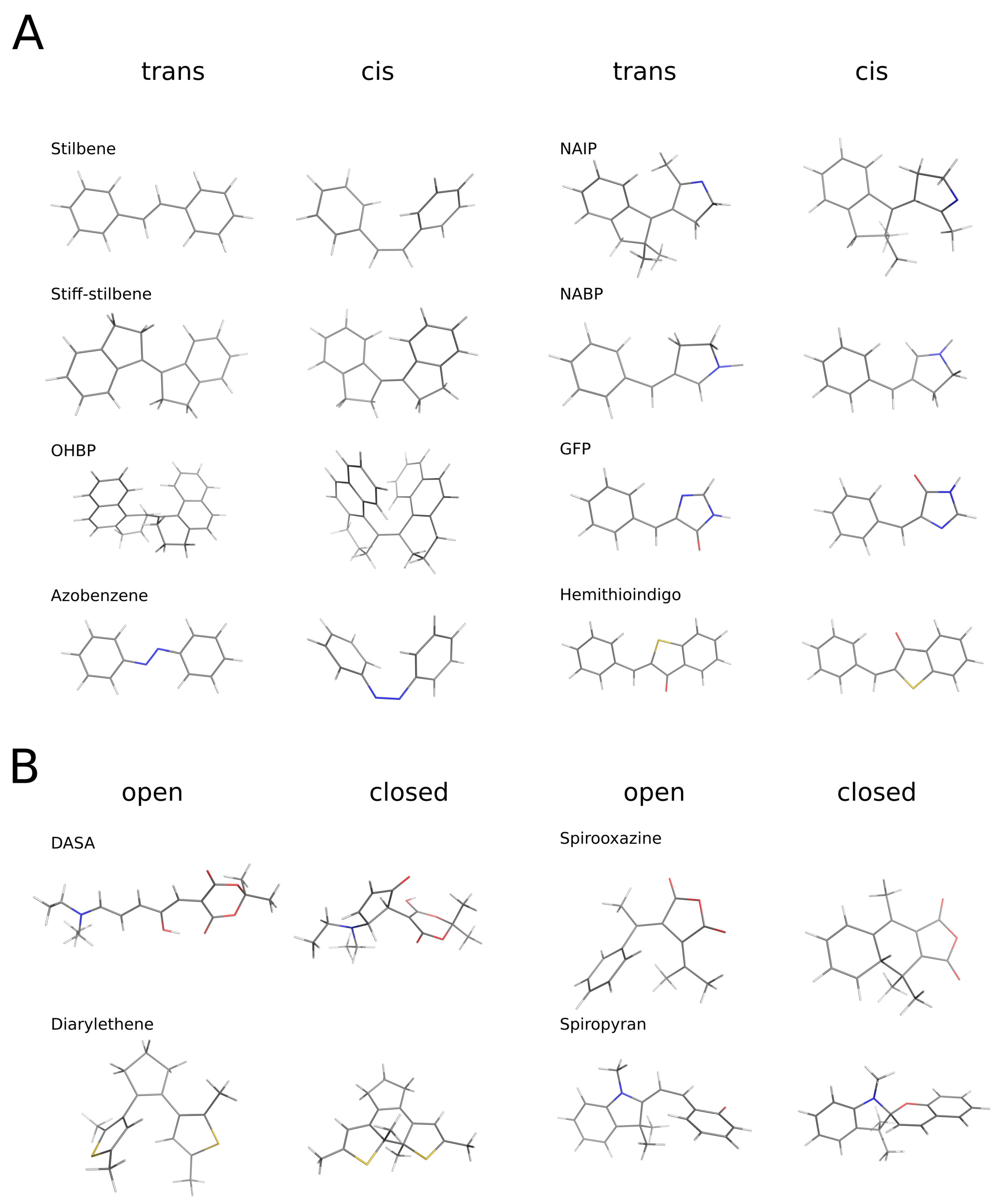
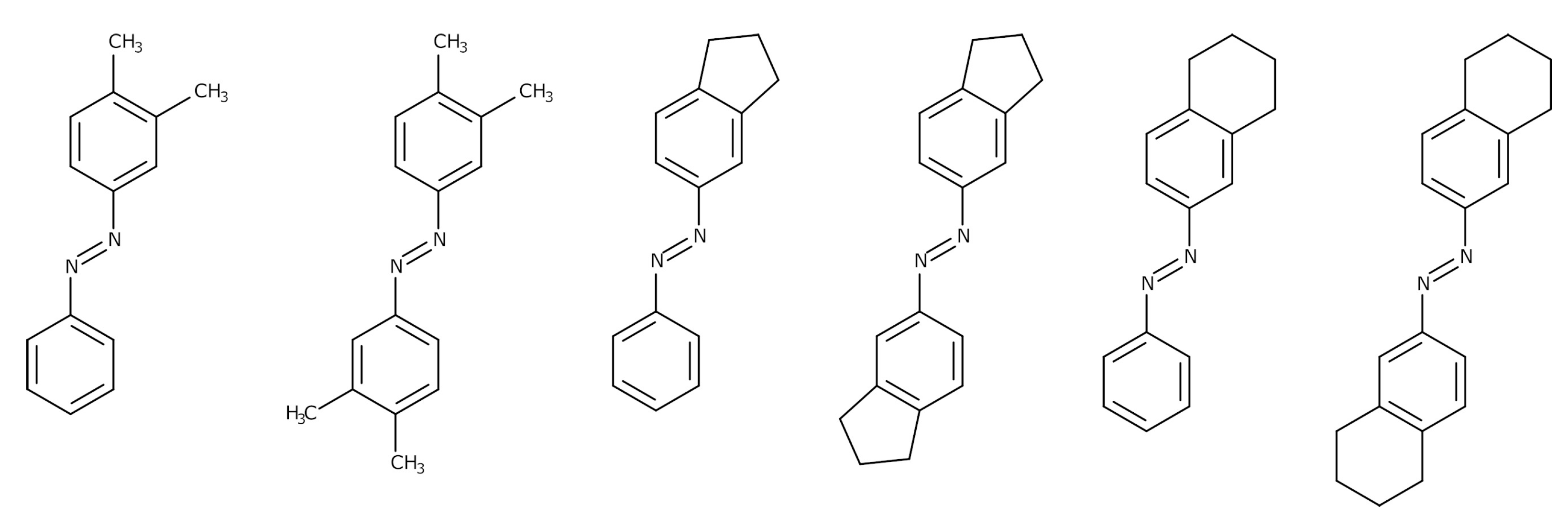
2.1. Objects
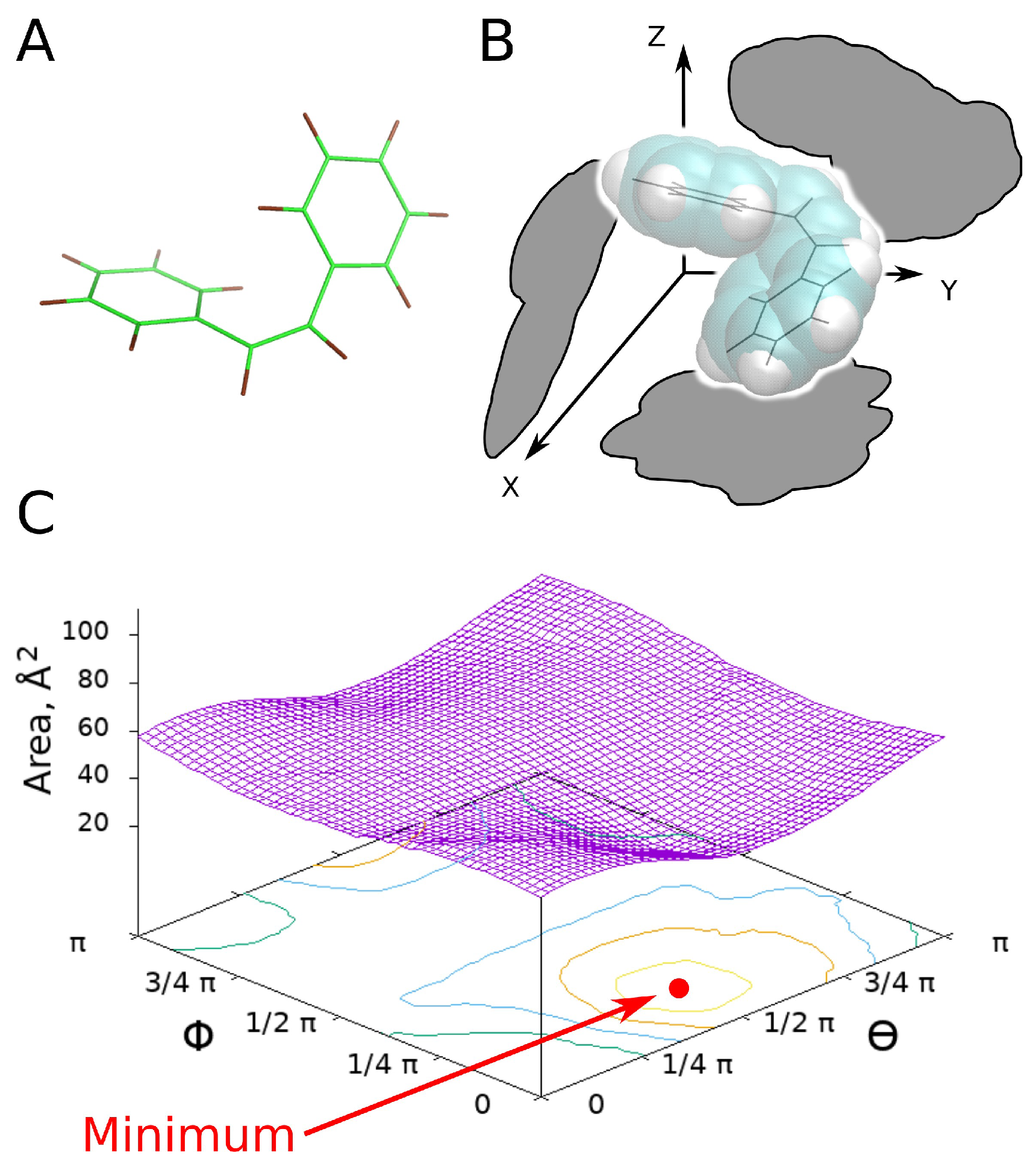
2.2. Physical Model
2.3. Calculation Algorithm
3. Results and Discussion
3.1. UV-Switch Geometry
3.2. Trans/cis (Open/Closed) Transition, and Change in the Projection Area of the Molecules
3.3. Structural Features of Promising Optical Switches
4. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Fujiwara, H.; Yonezawa, Y. Photoelectric response of a black lipid membrane containing an amphiphilic azobenzene derivative. Nature 1991, 351, 724–726. [Google Scholar] [CrossRef]
- Song, X.; Perlstein, J.; Whitten, D. Supramolecular aggregates of azobenzene phospholipids and related compounds in bilayer assemblies and other microheterogeneous media: Structure, properties, and photoreactivity. J. Am. Chem. Soc. 1997, 119, 9144–9159. [Google Scholar] [CrossRef]
- Hamada, T.; Sugimoto, R.; Nagasaki, T.; Takagi, M. Photochemical control of membrane raft organization. Soft Matter 2011, 7, 220–224. [Google Scholar] [CrossRef]
- Frank, J.; Franquelim, H.; Schwille, P.; Trauner, D. Optical Control of Lipid Rafts with Photoswitchable Ceramides. J. Am. Chem. Soc. 2016, 138, 12981–12986. [Google Scholar] [CrossRef]
- Saitov, A.; Akimov, S.; Galimzyanov, T.; Glasnov, T.; Pohl, P. Ordered Lipid Domains Assemble via Concerted Recruitment of Constituents from Both Membrane Leaflets. Phys. Rev. Lett. 2020, 124, 108102. [Google Scholar] [CrossRef] [Green Version]
- Frank, J.; Yushchenko, D.; Hodson, D.; Lipstein, N.; Nagpal, J.; Rutter, G.; Rhee, J.; Gottschalk, A.; Brose, N.; Schultz, C.; et al. Photoswitchable diacylglycerols enable optical control of protein kinase C. Nat. Chem. Biol. 2016, 12, 755–762. [Google Scholar] [CrossRef] [Green Version]
- Frank, J.; Moroni, M.; Moshourab, R.; Sumser, M.; Lewin, G.; Trauner, D. Photoswitchable fatty acids enable optical control of TRPV1. Nat. Commun. 2015, 6, 7118. [Google Scholar] [CrossRef] [Green Version]
- Pfeffermann, J.; Eicher, B.; Boytsov, D.; Hannesschlaeger, C.; Galimzyanov, T.; Glasnov, T.; Pabst, G.; Akimov, S.; Pohl, P. Photoswitching of model ion channels in lipid bilayers. J. Photochem. Photobiol. B 2021, 224, 112320. [Google Scholar] [CrossRef]
- Georgiev, V.; Grafmüller, A.; Bléger, D.; Hecht, S.; Kunstmann, S.; Barbirz, S.; Lipowsky, R.; Dimova, R. Area Increase and Budding in Giant Vesicles Triggered by Light: Behind the Scene. Adv. Sci. 2018, 5, 1800432. [Google Scholar] [CrossRef]
- García-Iriepa, C.; Marazzi, M.; Frutos, L.M.; Sampedro, D. E/Z Photochemical switches: Syntheses, properties and applications. RSC Adv. 2013, 3, 6241–6266. [Google Scholar] [CrossRef]
- Lerch, M.M.; Szymanski, W.; Feringa, B.L. The (photo)chemistry of Stenhouse photoswitches: Guiding principles and system design. Chem. Soc. Rev. 2018, 47, 1910. [Google Scholar] [CrossRef] [PubMed]
- Irie, M.; Fukaminato, T.; Matsuda, K.; Kobatake, S. Photochromism of diarylethene molecules and crystals: Memories, switches, and actuators. Chem. Rev. 2014, 114, 12174–12277. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; He, C.; Li, P.; Zhu, Y.-P. Recent progress in all-visible-light-triggered diarylethenes. Dye. Pigment. 2020, 182, 108623. [Google Scholar] [CrossRef]
- Bercovic, G.; Krongauz, V.; Weiss, V. Spiropyrans and spirooxazines for memories and switches. Chem. Rev. 2000, 100, 1741–1754. [Google Scholar] [CrossRef]
- Leith, G.A.; Martin, C.R.; Mathur, A.; Kittikhunnatham, P.; Park, K.C.; Shustova, N.R. Dynamically controlled electronic behavior of stimuli-responsive materials: Exploring dimensionality and connectivity. Adv. Energy Mater. 2022, 12, 2100441. [Google Scholar] [CrossRef]
- Ollila, O.H.S.; Risselada, H.J.; Louhivuori, M.; Lindahl, E.; Vattulainen, I.; Marrink, S.J. 3D Pressure field in lipid membranes and membrane-protein complexes. Phys. Rev. Lett. 2009, 102, 10–11. [Google Scholar] [CrossRef] [Green Version]
- Mukhin, S.I.; Kheyfets, B.B. Analytical approach to thermodynamics of bolalipid membranes. Phys. Rev. E 2010, 82, 51901. [Google Scholar] [CrossRef] [Green Version]
- Drozdova, A.A.; Mukhin, S.I. Lateral pressure profile in lipid membranes with curvature: Analytical calculation. J. Exp. Theor. Phys. 2017, 125, 357–363. [Google Scholar] [CrossRef]
- Tretiakova, D.S.; Alekseeva, A.S.; Galimzyanov, T.R.; Boldyrev, A.M.; Chernyadyev, A.Y.; Ermakov, Y.A.; Batishchev, O.V.; Vodovozova, E.L.; Boldyrev, I.A. Lateral stress profile and fluorescent lipid probes. FRET pair of probes that introduces minimal distortions into lipid packing. Biochim. Biophys. Acta (BBA)-Biomembr. 2018, 1860, 2337–2347. [Google Scholar] [CrossRef]
- Shchegravina, E.S.; Tretiakova, D.S.; Alekseeva, A.S.; Galimzyanov, T.R.; Utkin, Y.N.; Ermakov, Y.N.; Svirshchevskaya, E.V.; Negrebetsky, V.V.; Karpechenko, N.; Chernikov, V.P.; et al. Phospholipidic Colchicinoids as Promising Prodrugs Incorporated into Enzyme-Responsive Liposomes: Chemical, Biophysical, and Enzymological Aspects. Bioconjug. Chem. 2019, 30, 1098–1113. [Google Scholar] [CrossRef]
- Neese, F. The ORCA program system. WIREs Comput. Mol. Sci. 2012, 2, 73–78. [Google Scholar] [CrossRef]
- Weigend, F.; Ahlrichs, R. Balanced basis sets of split valence, triple zeta valence and quadruple zeta valence quality for H to Rn: Design and assessment of accuracy. Phys. Chem. Chem. Phys. 2005, 7, 3297–3305. [Google Scholar] [CrossRef] [PubMed]
- Alvarez, S. A cartography of the van der Waals territories. Dalt. Trans. 2013, 42, 8617–8636. [Google Scholar] [CrossRef] [PubMed] [Green Version]



| Isomer | Parameter | Stilbene | Stiff-Stilbene | OHBP | Azobenzene | NAIP | NABP | GFP | Hemithioindigo |
|---|---|---|---|---|---|---|---|---|---|
| Trans | (Å) | 78.04 | 93.44 | 107.3 | 76.28 | 77.78 | 67.72 | 69.02 | 90.23 |
| - | (Å) | 21.21 | 30.87 | 61.73 | 21.27 | 46.79 | 28.22 | 22.61 | 24.89 |
| - | 3.68 | 3.03 | 1.74 | 3.59 | 1.66 | 2.4 | 3.05 | 3.63 | |
| Cis | (Å) | 68.65 | 84.95 | 96.05 | 59.6 | 78.01 | 78.03 | 68.1 | 86.49 |
| - | (Å) | 35.30 | 40.38 | 65.06 | 36.55 | 43.18 | 28.71 | 22.28 | 24.63 |
| - | 1.94 | 2.1 | 1.48 | 1.63 | 1.81 | 2.54 | 3.06 | 3.51 |
| Isomer | Parameter | DASA | Diarylethene | Spirooxazine | Spiropyran |
|---|---|---|---|---|---|
| Open | (Å) | 97.22 | 82.29 | 74.53 | 85.90 |
| - | (Å) | 39.10 | 62.18 | 46.65 | 46.75 |
| - | 2.49 | 1.32 | 1.60 | 1.84 | |
| Close | (Å) | 94.75 | 85.75 | 78.19 | 82.18 |
| - | (Å) | 49.90 | 50.41 | 41.89 | 44.88 |
| - | 1.90 | 1.70 | 1.87 | 1.83 |
| Stilbene | Stiff-Stilbene | OHBP | Azobenzene | NAIP | NABP | GFP | Hemithioindigo | |
|---|---|---|---|---|---|---|---|---|
| S | 1.66 | 1.31 | 1.05 | 1.72 | 0.92 | 1.02 | 0.99 | 0.99 |
| (Å) | 12.09 | 9.51 | 3.33 | 15.28 | 3.61 | 0.49 | 5.94 | 0.26 |
| DASA | Diarylethene | Spirooxazine | Spiropyran | |
|---|---|---|---|---|
| S | 1.28 | 0.81 | 0.90 | 0.96 |
| (Å) | 10.0 | 11.77 | 4.76 | 1.87 |
| Isomer | Parameter | Stilbene | Stiff-Stilbene | OHBP | Azobenzene | NAIP | NABP | GFP | Hemithioindigo |
|---|---|---|---|---|---|---|---|---|---|
| Trans | Dihedral angle (1-2-3-4) | 180 | 179.8 | 155 | 180 | 167.1 | 178.6 | 180.0 | 180.0 |
| - | Angle (1-2-3) | 127.1 | 125.1 | 123.4 | 116.6 | 126.4 | 128.9 | 130.3 | 119.5 |
| - | Angle (2-3-4) | 127.1 | 125.1 | 123.4 | 116.6 | 130.5 | 126.4 | 122.3 | 132.6 |
| Cis | Dihedral angle (1-2-3-4) | 4.5 | 9.9 | 16.9 | 5.4 | 10.3 | 2.0 | 0.1 | 0 |
| - | Angle (1-2-3) | 129.7 | 123.2 | 123.6 | 124.4 | 131.4 | 129.0 | 134.9 | 131.8 |
| - | Angle (1-2-3) | 129.7 | 123.2 | 123.6 | 124.4 | 133.1 | 132.2 | 132.6 | 136.2 |
| Isomer | Parameter | Dimethyl mono | Dimethyl bis | Cyclopentyl mono | Cyclopentyl bis | Cyclohexyl mono | Cyclohexyl bis |
|---|---|---|---|---|---|---|---|
| Trans | (Å) | 85.71 | 99.56 | 87.03 | 98.65 | 92.66 | 108.41 |
| - | (Å) | 26.83 | 28.92 | 28.32 | 32.73 | 30.20 | 33.73 |
| - | 3.19 | 3.44 | 3.07 | 3.01 | 3.07 | 3.21 | |
| Cis | (Å) | 70.27 | 79.44 | 73.09 | 80.94 | 78.22 | 93.04 |
| - | (Å) | 42.47 | 50.1 | 42.24 | 52.71 | 44.12 | 51.21 |
| - | 1.65 | 1.59 | 1.73 | 1.54 | 1.77 | 1.82 | |
| - | S | 1.58 | 1.73 | 1.49 | 1.61 | 1.46 | 1.52 |
| - | (Å) | 15.44 | 21.18 | 13.92 | 19.98 | 13.92 | 17.48 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Boldyrev, I. Optical Switches for Lipid Membranes: Computed Molecular Projection Area as a Switch Selection Criterion. Colloids Interfaces 2022, 6, 30. https://doi.org/10.3390/colloids6020030
Boldyrev I. Optical Switches for Lipid Membranes: Computed Molecular Projection Area as a Switch Selection Criterion. Colloids and Interfaces. 2022; 6(2):30. https://doi.org/10.3390/colloids6020030
Chicago/Turabian StyleBoldyrev, Ivan. 2022. "Optical Switches for Lipid Membranes: Computed Molecular Projection Area as a Switch Selection Criterion" Colloids and Interfaces 6, no. 2: 30. https://doi.org/10.3390/colloids6020030
APA StyleBoldyrev, I. (2022). Optical Switches for Lipid Membranes: Computed Molecular Projection Area as a Switch Selection Criterion. Colloids and Interfaces, 6(2), 30. https://doi.org/10.3390/colloids6020030






