Litter Decomposition in Wet Rubber and Fruit Agroforests: Below the Threshold for Tropical Peat Formation
Abstract
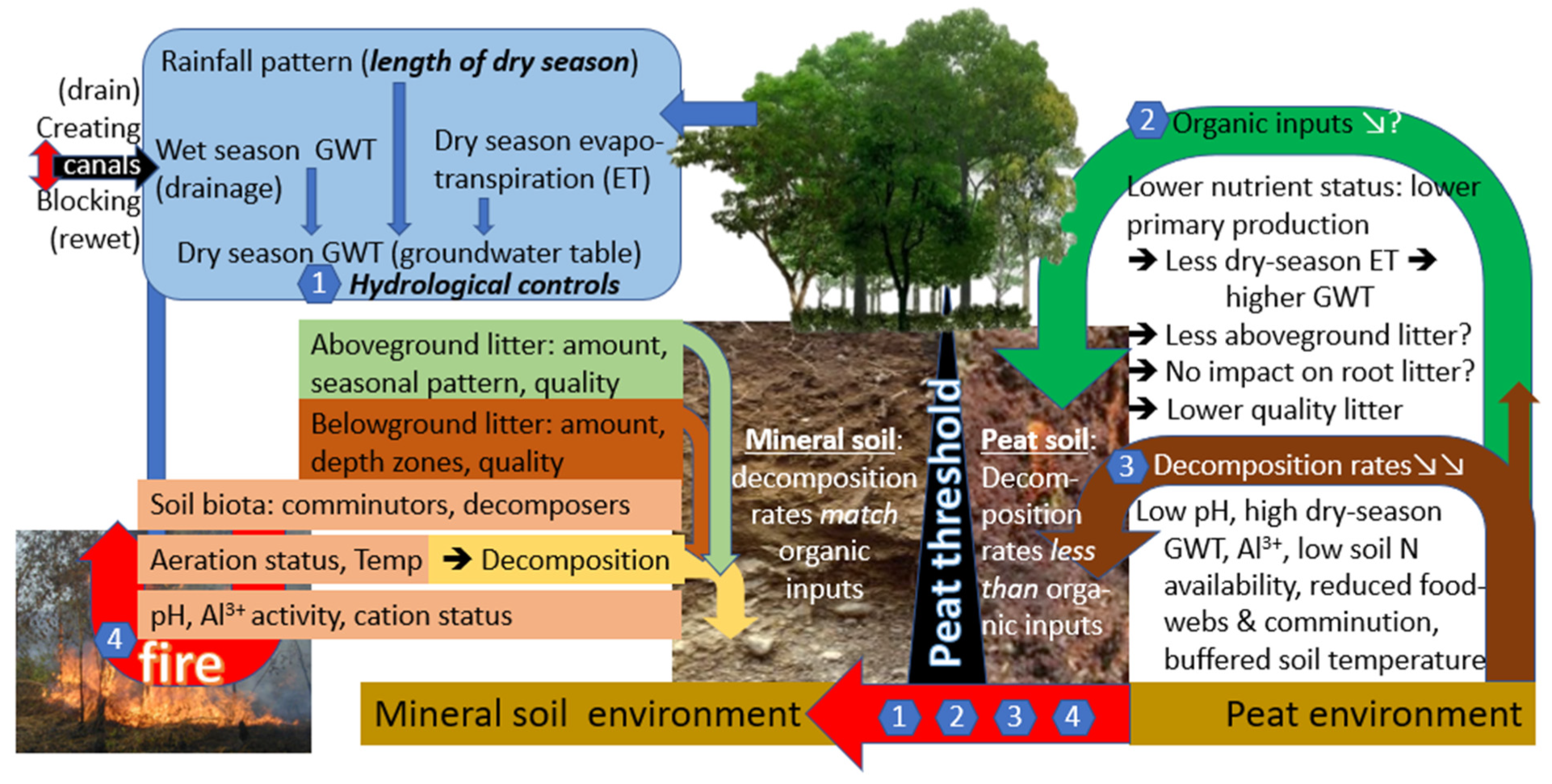
:1. Introduction
- How do rates of litter decomposition vary among agroforestry habitats (land cover types) on mineral soils but in peatland landscapes that differ in depth of drainage?
- How strong are home-field advantage (HFA) effects between the various habitat types when tested in a crossover experiment?
- Can HFA effects be related to soil and microclimatic conditions and/or to specific aspects of litter quality that may favor adapted decomposer communities?
2. Material and Methods
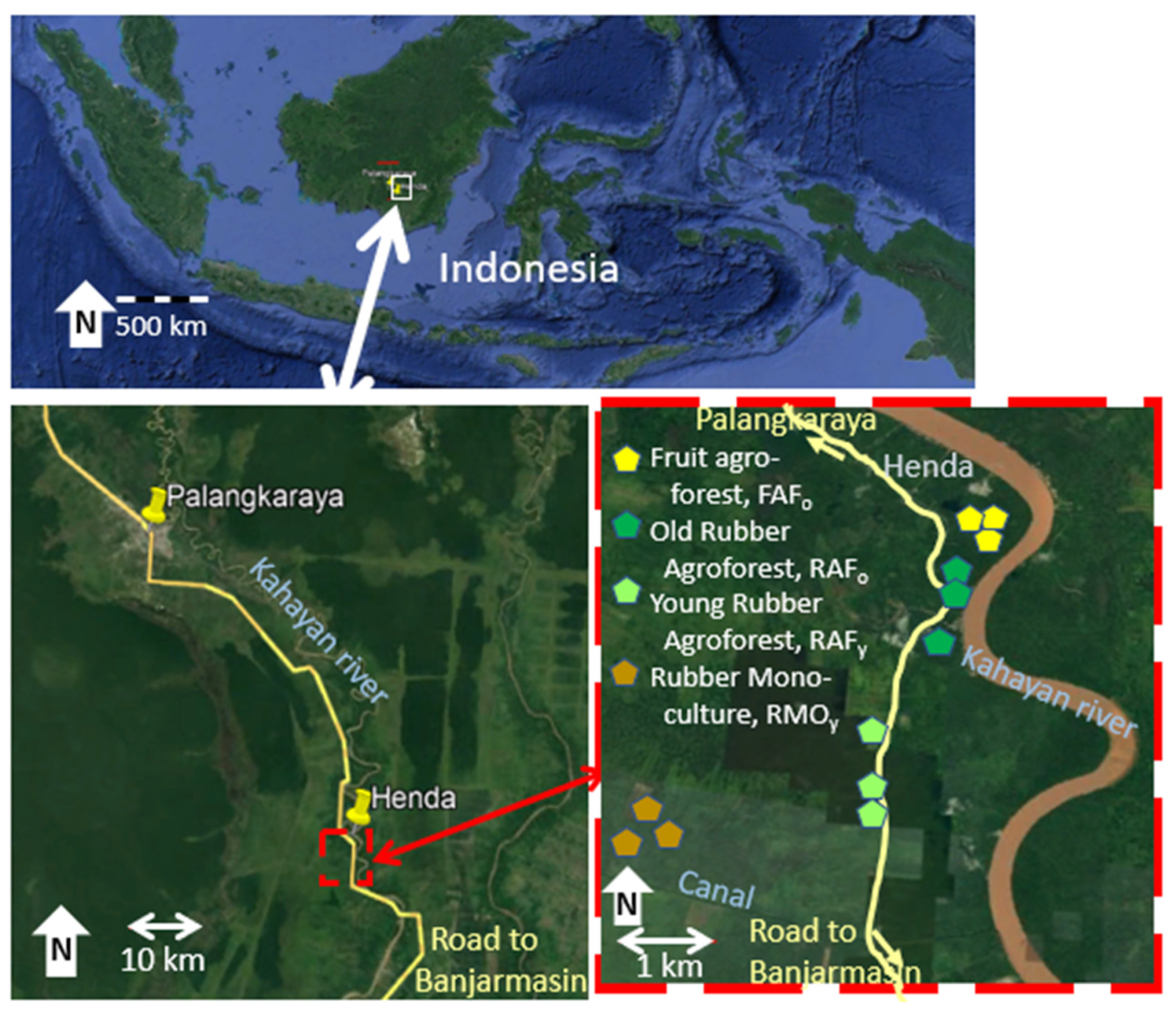
2.1. Research Location
- Fruit agroforest (FAFo), closest to the village and probably an older location of the village that shifted slightly to the south. Some of the trees are, according to local sources, at least one hundred years old. Important local fruit trees include “cempedak” (Artocarpus integer), “manggis” (Garcinia sp.), “rambai” (Baccaurea motleyana), “langsat” (Lansium domesticum), and “paken” (Durio kutejensis). There are also rubber (Hevea brasiliensis) trees in the plots.
- Old rubber agroforest (RAFo), planted with rubber around 150 years ago and relying on natural regeneration of these trees within the plot, along with local fruit trees including cempedak, rambai, durian (Durio sp.), “binjai” (Mangifera caesia), and rambutan (Nephelium lappaceum).
- Young rubber agroforest, (RAFy), planted with rubber around 30 years ago (according to local informants) and similar in tree composition to the RAFo plots. Local trees include “hampalam” (Mangifera sp.), manggis, rambutan, and “sentol” (Sandoricum koetjape).
2.2. Soil, Microclimate, and Vegetation Characteristics
2.3. Litterbags
2.4. Reciprocal Litter Exchange Experiment
2.5. Decomposition Rates
2.6. Home-Field Advantage
3. Results
3.1. Soil and Vegetation Characteristics
3.2. Microclimate
3.3. Litter Quality
3.4. Decomposition Rates
3.5. Home-Field Advantage (HFA)
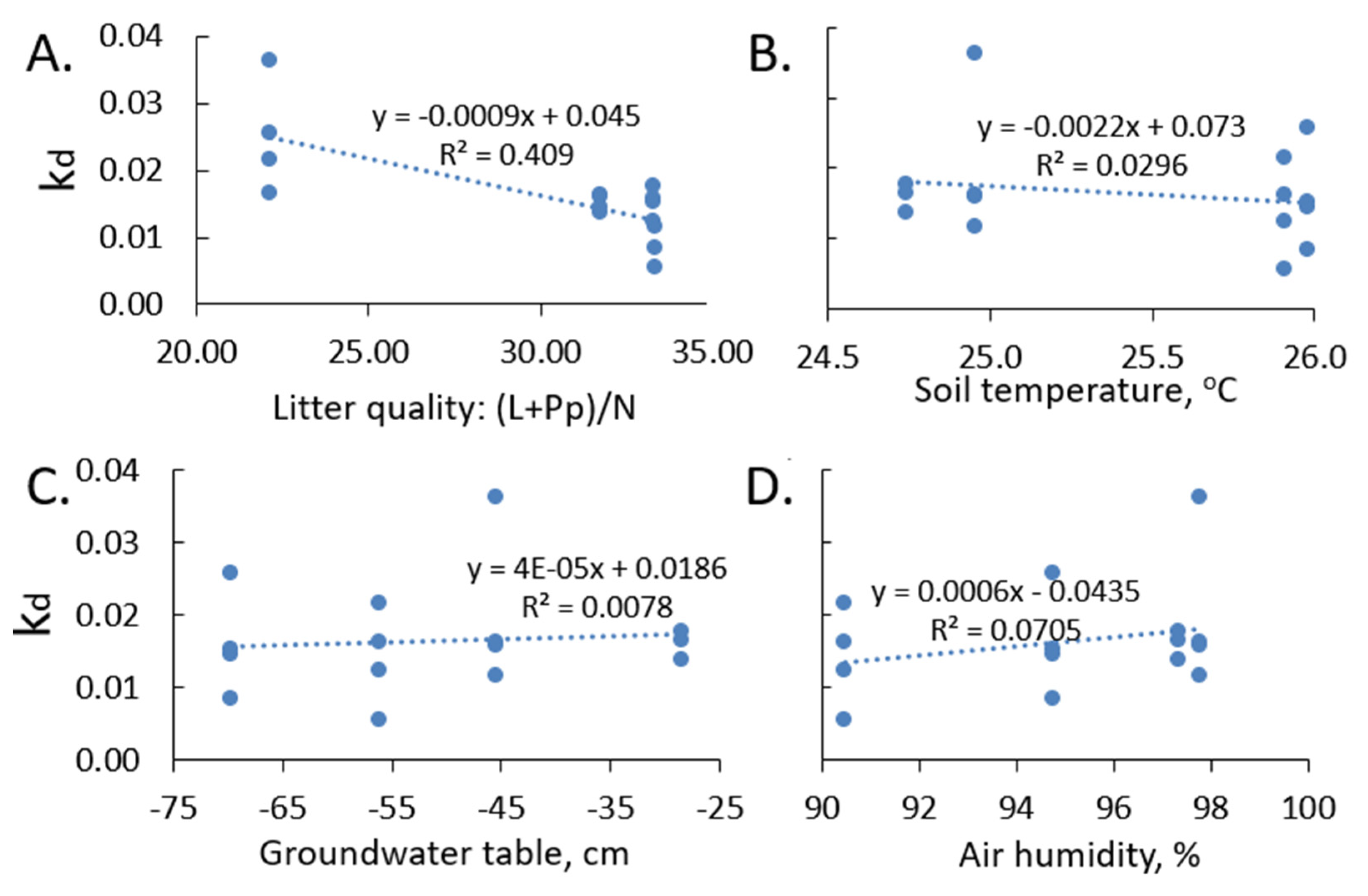
3.6. Litter Quality, Environmental Conditions, and HFA
4. Discussion
5. Conclusions
- The mean residence times for aboveground litter in wet agroforests were around 85 weeks, about 2 times longer than expected given litter quality and temperature, which was likely due to wet and acid soil conditions.
- Home-field advantage indices showing up to 80% faster decomposition in the home environment in specific habitat–litter combinations may reflect strong adaptation of decomposers to local litter sources.
- Overall, the ratio of lignin plus polyphenolics and nitrogen accounted for 40% of the variance in decomposition constant kd, indicating other more specific quality indicators can play a role.
- The measured litter residence times in wet agroforests support litter stocks larger than annual litterfall but are still far below the (as yet unknown) thresholds for peat formation.
- While the wet agroforests studied support landscape-level rewetting strategies and protect peat dome hinterlands from drying and fire, they do not contribute to in situ peat formation.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Mohr, E.C.J.; van Baren, F.A.; van Schuylenborgh, J. Tropical Soils, A Comprehensive Study of their Genesis, 3rd ed.; Mouton: Paris, France; Ichtiar Baru: Jakarta, Indonesia; Van Hoeve: The Hague, The Netherlands, 1972. [Google Scholar]
- Havinga, A.J.; Muller, J. In Memoriam Betje Polak (1901–1980), pioneer of peat research in The Netherlands and in the tropics. Acta Bot. Neerl. 1981, 30, 337–343. [Google Scholar] [CrossRef]
- Polak, B. Veen en veenontginning in Indonesia [Peat and peat exploitation in Indonesia]. Indones. J. Nat. Sci. 1952, 5, 146–160. [Google Scholar]
- Anda, M.; Ritung, S.; Suryani, E.; Hikmat, M.; Yatno, E.; Mulyani, A.; Subandiono, R.E. Revisiting tropical peatlands in Indonesia: Semi-detailed mapping, extent and depth distribution assessment. Geoderma 2021, 402, 115235. [Google Scholar] [CrossRef]
- Kiew, F.; Hirata, R.; Hirano, T.; Xhuan, W.G.; Aries, E.B.; Kemudang, K.; Wenceslaus, J.; San, L.K.; Melling, L. Carbon dioxide balance of an oil palm plantation established on tropical peat. Agric. For. Meteorol. 2020, 295, 108189. [Google Scholar] [CrossRef]
- Page, S.E.; Hooijer, A. In the line of fire: The peatlands of Southeast Asia. Philos. Trans. R. Soc. B Biol. Sci. 2016, 371, 20150176. [Google Scholar] [CrossRef] [Green Version]
- McCalmont, J.; Kho, L.K.; Teh, Y.A.; Lewis, K.; Chocholek, M.; Rumpang, E.; Hill, T. Short-and long-term carbon emissions from oil palm plantations converted from logged tropical peat swamp forest. Glob. Change Biol. 2021, 27, 2361–2376. [Google Scholar] [CrossRef]
- Dohong, A.; Aziz, A.A.; Dargusch, P. A review of the drivers of tropical peatland degradation in South-East Asia. Land Use Policy 2017, 69, 349–360. [Google Scholar] [CrossRef]
- Page, S.; Hosciło, A.; Wösten, H.; Jauhiainen, J.; Silvius, M.; Rieley, J.; Ritzema, H.; Tansey, K.; Graham, L.; Vasander, H. Restoration ecology of lowland tropical peatlands in Southeast Asia: Current knowledge and future research directions. Ecosystems 2009, 12, 888–905. [Google Scholar] [CrossRef]
- Swift, M.J.; Heal, O.W.; Anderson, J.M. Decomposition in Terrestrial Ecosystems; University of California Press: Berkeley, CA, USA, 1979. [Google Scholar]
- Handa, I.T.; Aerts, R.; Berendse, F.; Berg, M.P.; Bruder, A.; Butenschoen, O.; Chauvet, E.; Gessner, M.O.; Jabiol, J.; Makkonen, M.; et al. Consequences of biodiversity loss for litter decomposition across biomes. Nature 2014, 509, 218. [Google Scholar] [CrossRef] [Green Version]
- Bradford, M.A.; Berg, B.; Maynard, D.S.; Wieder, W.R.; Wood, S.A. Understanding the dominant controls on litter decomposition. J. Ecol. 2016, 104, 229–238. [Google Scholar] [CrossRef]
- Giweta, M. Role of litter production and its decomposition, and factors affecting the processes in a tropical forest ecosystem: A review. J. Ecol. Environ. 2020, 44, 1–9. [Google Scholar] [CrossRef]
- Anderson, J.M.; Ingram, J.S.I. Tropical Soil Biology and Fertility. A Handbook of Methods, 2nd ed.; CAB International: Wallingford, UK, 1993. [Google Scholar]
- Hairiah, K.; Utami, S.R.; Suprayogo, D.; Sunaryo, D.; Sitompul, S.M.; Lusiana, B.; Mulia, R.; van Noordwijk, M.; Cadisch, G. Pengelolaan Tanah Masam Secara Biologi. Refleksi Pengalaman dari Lampung Utara [Biological Management of Acid Soils: Reflections from Research in North Lampung]; International Centre for Research in Agroforestry: Bogor, Indonesia, 2000. [Google Scholar]
- Hairiah, K.; Sulistyani, H.; Suprayogo, D.; Purnomosidhi, P.; Widodo, R.H.; Van Noordwijk, M. Litter layer residence time in forest and coffee agroforestry systems in Sumberjaya, West Lampung. For. Ecol. Manag. 2006, 224, 45–57. [Google Scholar] [CrossRef]
- Hairiah, K.; Dewi, S.; Agus, F.; Velarde, S.; Ekadinata, A.; Rahayu, S.; van Noordwijk, M. Measuring Carbon Stocks: Across Land Use Systems: A Manual; World Agroforestry (ICRAF): Bogor, Indonesia, 2011. [Google Scholar]
- Palm, C.A.; Sanchez, P.A. Nitrogen release from the leaves of some tropical legumes as affected by their lignin and polyphenolic contents. Soil Biol. Biochem. 1991, 23, 83–88. [Google Scholar] [CrossRef]
- Zhang, D.; Hui, D.; Luo, Y.; Zhou, G. Rates of litter decomposition in terrestrial ecosystems: Global patterns and controlling factors. J. Plant Ecol. 2008, 1, 85–93. [Google Scholar] [CrossRef] [Green Version]
- Van Noordwijk, M.; Martikainen, P.; Bottner, P.; Cuevas, E.; Rouland, C.; Dhillion, S.S. Global change and root function. Glob. Change Biol. 1998, 4, 759–772. [Google Scholar] [CrossRef]
- Medrilzam, M.; Smith, C.; Aziz, A.A.; Herbohn, J.; Dargusch, P. Smallholder Farmers and the Dynamics of Degradation of Peatland Ecosystems in Central Kalimantan, Indonesia. Ecol. Econ. 2017, 136, 101–113. [Google Scholar] [CrossRef]
- Taufik, M.; Setiawan, B.I.; Van Lanen, H.A. Increased fire hazard in human-modified wetlands in Southeast Asia. Ambio 2019, 48, 363–373. [Google Scholar] [CrossRef]
- Jaenicke, J.; Wösten, H.; Budiman, A.; Siegert, F. Planning hydrological restoration of peatlands in Indonesia to mitigate carbon dioxide emissions. Mitig. Adapt. Strateg. Glob. Chang. 2010, 15, 223–239. [Google Scholar] [CrossRef] [Green Version]
- Jefferson, U.; Carmenta, R.; Daeli, W.; Phelps, J. Characterising policy responses to complex socio-ecological problems: 60 fire management interventions in Indonesian peatlands. Glob. Environ. Chang. 2020, 60, 102027. [Google Scholar] [CrossRef]
- Van Noordwijk, M.; Matthews, R.; Agus, F.; Farmer, J.; Verchot, L.; Hergoualc’h, K.; Dewi, S. Mud, muddle and models in the knowledge value-chain to action on tropical peatland conservation. Mitig. Adapt. Strateg. Glob. Change 2014, 19, 887–905. [Google Scholar] [CrossRef] [Green Version]
- Tata, H.L. Mixed farming systems on peatlands in Jambi and Central Kalimantan provinces, Indonesia: Should they be described as paludiculture? Mires Peat 2019, 25, 8. [Google Scholar]
- Widayati, A.; Tata, H.L.; van Noordwijk, M. Agroforestry on Peatlands: Combining Productive and Protective Functions as Part of Restoration; Policy Brief No. 70; Agroforestry Options for ASEAN Series No. 4; World Agroforestry Centre (ICRAF): Bogor, Indonesia, 2016. [Google Scholar]
- Wichtmann, W.; Schröder, C.; Joosten, H. Paludiculture-Productive Use of Wet Peatlands; Schweizerbart Science Publishers: Stuttgart, Germany, 2016. [Google Scholar]
- Ziegler, R. Paludiculture as a critical sustainability innovation mission. Res. Policy 2020, 49, 103979. [Google Scholar] [CrossRef]
- Froufe, L.C.M.; Schwiderke, D.K.; Castilhano, A.C.; Cezar, R.M.; Steenbock, W.; Seoane, C.E.S.; Bognnola, I.A.; Vezzani, F.M. Nutrient cycling from leaf litter in multistrata successional agroforestry systems and natural regeneration at Brazilian Atlantic Rainforest Biome. Agrofor. Syst. 2020, 94, 159–171. [Google Scholar] [CrossRef]
- Prescott, C.E. Do rates of litter decomposition tell us anything we really need to know? For. Ecol. Manag. 2005, 220, 66–74. [Google Scholar] [CrossRef]
- Silvianingsih, Y.A.; Hairiah, K.; Suprayogo, D.; van Noordwijk, M. Agroforests swiddening and livelihoods between restored peat domes and river: Effects of the 2015 fire ban in Central Kalimantan (Indonesia). Int. For. Rev. 2020, 22, 382–396. [Google Scholar] [CrossRef]
- Silvianingsih, Y.A.; Hairiah, K.; Suprayogo, D.; van Noordwijk, M. Kaleka agroforest in Central Kalimantan (Indonesia): Soil quality, hydrological protection of adjacent peatlands, and sustainability. Land 2021, 10, 856. [Google Scholar] [CrossRef]
- Hättenschwiler, S.; Tiunov, A.V.; Scheu, S. Biodiversity and litter decomposition in terrestrial ecosystems. Annu. Rev. Ecol. Evol. Syst. 2005, 36, 191–218. [Google Scholar] [CrossRef]
- Martinez-Yrizar, A.; Nuñez, S.; Burquez, A. Leaf litter decomposition in a southern Sonoran Desert ecosystem, northwestern Mexico: Effects of habitat and litter quality. Acta Oecol. 2007, 32, 291–300. [Google Scholar] [CrossRef]
- Gholz, H.L.; Wedin, D.A.; Smitherman, S.M.; Harmon, M.E.; Parton, W.J. Long-term dynamics of pine and hardwood litter in contrasting environments: Toward a global model of decomposition. Glob. Change Biol. 2000, 6, 751–765. [Google Scholar] [CrossRef]
- Delgado-Baquerizo, M.; García-Palacios, P.; Milla, R.; Gallardo, A.; Maestre, F.T. Soil characteristics determine soil carbon and nitrogen availability during leaf litter decomposition regardless of litter quality. Soil Biol. Biochem. 2015, 81, 134–142. [Google Scholar] [CrossRef]
- Tian, G.; Brussaard, L.; Kang, B.T. Biological effects of plant residues with contrasting chemical compositions under humid tropical conditions: Effects on soil fauna. Soil Biol. Biochem. 1993, 25, 731–737. [Google Scholar] [CrossRef] [Green Version]
- Krishna, M.P.; Mohan, M. Litter decomposition in forest ecosystems: A review. Energy Ecol. Environ. 2017, 2, 236–249. [Google Scholar] [CrossRef]
- Strickland, M.S.; Lauber, C.; Fierer, N.; Bradford, M.A. Testing the functional significance of microbial community composition. Ecology 2009, 90, 441–451. [Google Scholar] [CrossRef] [Green Version]
- Keiser, A.D.; Keiser, D.A.; Strickland, M.S.; Bradford, M.A. Disentangling the mechanisms underlying functional differences among decomposer communities. J. Ecol. 2014, 102, 603–609. [Google Scholar] [CrossRef]
- Cleveland, C.C.; Reed, S.C.; Keller, A.B.; Nemergut, D.R.; O’Neill, S.P.; Ostertag, R.; Vitousek, P.M. Litter quality versus soil microbial community controls over decomposition: A quantitative analysis. Oecologia 2014, 174, 283–294. [Google Scholar] [CrossRef] [PubMed]
- Hoorens, B.; Aerts, R.; Stroetenga, M. Does initial litter chemistry explain litter mixture effects on decomposition? Oecologia 2003, 137, 578–586. [Google Scholar] [CrossRef]
- Berg, B.; Steffen, K.T.; McClaugherty, C. Litter decomposition rate is dependent on litter Mn concentrations. Biogeochemistry 2007, 82, 29–39. [Google Scholar] [CrossRef]
- Di Lonardo, D.P.; Manrubia, M.; De Boer, W.; Zweers, H.; Veen, G.F.; Van der Wal, A. Relationship between home-field advantage of litter decomposition and priming of soil organic matter. Soil Biol. Biochem. 2018, 126, 49–56. [Google Scholar] [CrossRef]
- Ayres, E.; Steltzer, H.; Simmons, B.L.; Simpson, R.T.; Steinweg, J.M.; Wallenstein, M.D.; Mellor, N.; Parton, W.J.; Moore, J.C.; Wall, D.H. Home-field advantage accelerates leaf litter decomposition in forests. Soil Biol. Biochem. 2009, 41, 606–610. [Google Scholar] [CrossRef]
- Ayres, E.; Steltzer, H.; Berg, S.; Wall, D.H. Soil biota accelerate decomposition in high-elevation forests by specializing in the breakdown of litter produced by the plant species above them. J. Ecol. 2009, 97, 901–912. [Google Scholar] [CrossRef]
- Veen, G.F.; Sundqvist, M.K.; Wardle, D.A. Environmental factors and traits that drive plant litter decomposition do not determine home-field advantage effects. Funct. Ecol. 2015, 29, 981–991. [Google Scholar] [CrossRef] [Green Version]
- Wallenstein, M.D.; Haddix, M.L.; Ayres, E.; Steltzer, H.; Magrini-Bair, K.A.; Paul, E.A. Litter chemistry changes more rapidly when decomposed at home but converges during decomposition–transformation. Soil Biol. Biochem. 2013, 57, 311–319. [Google Scholar] [CrossRef]
- Kagata, H.; Ohgushi, T. Home-field advantage in decomposition of leaf litter and insect frass. Popul. Ecol. 2013, 55, 69–76. [Google Scholar] [CrossRef]
- Hairiah, K.; van Noordwijk, M.; Mardiani, M.O.; Alhusna, M.; Khurniawati, D.N.; Ayun, C.; Prayogo, C.; Rowe, R.L.; McNamara, N.P. Dietary diversity preference, home-field advantage, macrofauna and microclimate effects in aboveground litter decomposition in coffee and pine agroforestry. 2021. [Google Scholar]
- Bradford, M.A.; Veen, G.C.; Bonis, A.; Bradford, E.M.; Classen, A.T.; Cornelissen, J.H.C.; Crowther, T.W.; Jonathan, R.; Freschet, G.T.; Kardol, P.; et al. A test of the hierarchical model of litter decomposition. Nat. Ecol. Evol. 2017, 1, 1836. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; He, Z.; Hao, J.; Tian, K.; Jia, X.; Kong, X.; Akbar, S.; Bei, Z.; Tian, X. Effect of N addition on home-field advantage of litter decomposition in subtropical forests. For. Ecol. Manag. 2017, 398, 216–225. [Google Scholar] [CrossRef]
- Bridgham, S.D.; Richardson, C.J. Endogenous versus exogenous nutrient control over decomposition and mineralization in North Carolina peatlands. Biogeochemistry 2003, 65, 151–178. [Google Scholar] [CrossRef]
- Clarkson, B.R.; Moore, T.R.; Fitzgerald, N.B.; Thornburrow, D.; Watts, C.H.; Miller, S. Water table regime regulates litter decomposition in restiad peatlands, New Zealand. Ecosystems 2014, 17, 317–326. [Google Scholar] [CrossRef]
- Lopez-Sangil, L.; Hartley, I.P.; Rovira, P.; Casals, P.; Sayer, E.J. Drying and rewetting conditions differentially affect the mineralization of fresh plant litter and extant soil organic matter. Soil Biol. Biochem. 2018, 124, 81–89. [Google Scholar] [CrossRef] [Green Version]
- BMKG: Badan Meteorologi, Klimatologi dan Geofisika Palangka Raya. Kalimantan Tengah, 2019. Available online: https://www.bmkg.go.id/iklim (accessed on 4 September 2021).
- Badan Pusat Statistik (BPS). Kecamatan Jabiren Raya dalam Angka; National Bureau of Statistics: Jakarta, Indonesia, 2018.
- Hairiah, K.; van Noordwijk, M.; Sari, R.R.; Saputra, D.D.; Suprayogo, D.; Kurniawan, S.; Prayogo, C.; Gusli, S. Soil carbon stocks in Indonesian (agro) forest transitions: Compaction conceals lower carbon concentrations in standard accounting. Agric. Ecosyst. Environ. 2020, 294, 106879. [Google Scholar] [CrossRef]
- Schinner, F.; Öhlinger, R.; Kandeler, E.; Margesin, R. (Eds.) Methods in Soil Biology; Springer: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Walela, C.; Daniel, H.; Wilson, B.; Lockwood, P.; Cowie, A.; Harden, S. The initial lignin: Nitrogen ratio of litter from above and below ground sources strongly and negatively influenced decay rates of slowly decomposing litter carbon pools. Soil Biol. Biochem. 2014, 77, 268–275. [Google Scholar] [CrossRef]
- Vanlauwe, B.; Gachengo, C.; Shepherd, K.; Barrios, E.; Cadisch, G.; Palm, C.A. Laboratory validation of a resource quality-based conceptual framework for organic matter management. Soil Sci. Soc. Am. J. 2005, 69, 1135–1145. [Google Scholar] [CrossRef] [Green Version]
- Goering, H.U.; Van Soest, P.J. Forage Fiber Analyses (Apparatus, Reagents, Procedures, and Some Applications); Report No 379; US Agricultural Research Service: Washington, DC, USA, 1970.
- Van Soest, P.J.; McQueen, R.W. The chemistry and estimation of fibre. Proc. Nutr. Soc. 1973, 32, 123–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gessner, M.O. Proximate lignin and cellulose. In Methods to Study Litter Decomposition; Graça, M.A.S., Bärlocher, F., Gessner, M.O., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 115–120. [Google Scholar]
- Olson, J.S. Energy storage and the balance of producers and decomposers in ecological systems. Ecology 1963, 44, 322–331. [Google Scholar] [CrossRef] [Green Version]
- Fierer, N.; Craine, J.M.; McLauchlan, K.; Schimel, J.P. Litter quality and the temperature sensitivity of decomposition. Ecology 2005, 86, 320–326. [Google Scholar] [CrossRef] [Green Version]
- Austin, A.T.; Vivanco, L.; González-Arzac, A.; Pérez, L.I. There’s no place like home? An exploration of the mechanisms behind plant litter–decomposer affinity in terrestrial ecosystems. New Phytol. 2014, 204, 307–314. [Google Scholar] [CrossRef]
- Tjitrosemito, S.; Guhardja, E.; Qayim, I.; Leuschner, C. Litterfall production and leaf-litter decomposition at natural forest and cacao agroforestry in Central Sulawesi, Indonesia. Asian J. Biol. Sci. 2011, 4, 221–234. [Google Scholar]
- Nanda, F.; Muhammad, A.; Fatonah, S. Laju Produksi Serasah Dalam Dua Sistem Budidaya Karet Pada Lahan Gambut di Kawasan Rimbo Panjang, Riau. 2013. Available online: https://repository.unri.ac.id/xmlui/bitstream/handle/123456789/2399/Karya%20Ilmiah%20Fristya%20Nanda%2026%2001%202013.pdf?sequence=1&isAllowed=y (accessed on 4 September 2021).
- Sari, R.R.; Saputra, D.D.; Hairiah, K.; Rozendaal, D.; Roshetko, J.M.; van Noordwijk, M. Gendered species preferences link tree diversity and carbon stocks in cacao agroforest in Southeast Sulawesi, Indonesia. Land 2020, 9, 108. [Google Scholar] [CrossRef] [Green Version]
- Martius, C.; Höfer, H.; Garcia, M.V.; Römbke, J.; Förster, B.; Hanagarth, W. Microclimate in agroforestry systems in central Amazonia: Does canopy closure matter to soil organisms? Agrofor. Syst. 2004, 60, 291–304. [Google Scholar] [CrossRef]
- Ge, X.; Zeng, L.; Xiao, W.; Huang, Z.; Geng, X.; Tan, B. Effect of litter substrate quality and soil nutrients on forest litter decomposition: A review. Acta Ecol. Sin. 2013, 33, 102–108. [Google Scholar] [CrossRef]
- Isaac, S.R.; Nair, M.A. Biodegradation of leaf litter in the warm humid tropics of Kerala, India. Soil Biol. Biochem. 2005, 37, 1656–1664. [Google Scholar] [CrossRef]
- Mirmanto, E. Vegetation analyses of Sebangau peat swamp forest, Central Kalimantan. Biodivers. J. Biol. Divers. 2010, 11, 82–88. [Google Scholar] [CrossRef]
- Natalia, D.; Arisoesilaningsih, E.; Hairiah, K. Are high carbon stocks in agroforests and forest associated with high plant species diversity? Agrivita 2016, 39, 74–82. [Google Scholar] [CrossRef] [Green Version]
- Straková, P.; Penttilä, T.; Laine, J.; Laiho, R. Disentangling direct and indirect effects of water table drawdown on above-and belowground plant litter decomposition: Consequences for accumulation of organic matter in boreal peatlands. Glob. Change Biol. 2012, 18, 322–335. [Google Scholar] [CrossRef]
- Gießelmann, U.C.; Martins, K.G.; Brändle, M.; Schädler, M.; Marques, R.; Brandl, R. Lack of home-field advantage in the decomposition of leaf litter in the Atlantic Rainforest of Brazil. Appl. Soil Ecol. 2011, 49, 5–10. [Google Scholar] [CrossRef]
- John, M.G.S.; Orwin, K.H.; Dickie, I.A. No ‘home’ versus ‘away’ effects of decomposition found in a grassland–forest reciprocal litter transplant study. Soil Biol. Biochem. 2011, 43, 1482–1489. [Google Scholar] [CrossRef]
- Yule, C.M.; Gomez, L.N. Leaf litter decomposition in a tropical peat swamp forest in Peninsular Malaysia. Wetl. Ecol. Manag. 2009, 17, 231–241. [Google Scholar] [CrossRef]
- Chomel, M.; Guittonny-Larchevêque, M.; Fernandez, C.; Gallet, C.; DesRochers, A.; Paré, D.; Jackson, B.G.; Baldy, V. Plant secondary metabolites: A key driver of litter decomposition and soil nutrient cycling. J. Ecol. 2016, 104, 1527–1541. [Google Scholar] [CrossRef]
- Schmitt, L.; Perfecto, I. Coffee leaf litter decomposition: Short term home-field advantage in shaded coffee agro-ecosystems. Appl. Soil Ecol. 2021, 161, 103854. [Google Scholar] [CrossRef]
- Trinder, C.J.; Johnson, D.; Artz, R.R. Interactions among fungal community structure, litter decomposition and depth of water table in a cutover peatland. FEMS Microbiol. Ecol. 2008, 64, 433–448. [Google Scholar] [CrossRef] [Green Version]
- Bonanomi, G.; Incerti, G.; Giannino, F.; Mingo, A.; Lanzotti, V.; Mazzoleni, S. Litter quality assessed by solid state 13C NMR spectroscopy predicts decay rate better than C/N and Lignin/N ratios. Soil Biol. Biochem. 2013, 56, 40–48. [Google Scholar] [CrossRef]
- Górecki, K.; Rastogi, A.; Stróżecki, M.; Gąbka, M.; Lamentowicz, M.; Łuców, D.; Kayzer, D.; Juszczak, R. Water table depth, experimental warming, and reduced precipitation impact on litter decomposition in a temperate Sphagnum-peatland. Sci. Total Environ. 2021, 771, 145452. [Google Scholar] [CrossRef] [PubMed]
- Rejmánková, E.; Houdková, K. Wetland plant decomposition under different nutrient conditions: What is more important, litter quality or site quality? Biogeochemistry 2006, 80, 245–262. [Google Scholar] [CrossRef]
- Page, S.E.; Baird, A.J. Peatlands and global change: Response and resilience. Annu. Rev. Environ. Resour. 2016, 41, 35–57. [Google Scholar] [CrossRef]
- Chimner, R.A.; Ewel, K.C. A tropical freshwater wetland: II. Production, decomposition and peat formation. Wet. Ecol. Manag. 2005, 13, 671–684. [Google Scholar] [CrossRef]
- Brady, M.A. Organic Matter Dynamics of Coastal Peat Deposits in Sumatra, Indonesia. Ph.D. Thesis, Department of Forestry, University of British Columbia, Vancouver, BC, Canada, 1997. Available online: https://open.library.ubc.ca/cIRcle/collections/ubctheses/831/items/1.0075286 (accessed on 25 January 2022).
- Bacon, K.L.; Baird, A.J.; Blundell, A.; Bourgault, M.A.; Chapman, P.J.; Dargie, G.; Dooling, G.P.; Gee, C.; Holden, J.; Kelly, T.J.; et al. Questioning ten common assumptions about peatlands. Mires Peat 2017, 19, 1–23. Available online: https://eprints.whiterose.ac.uk/119944/1/map_19_12.pdf (accessed on 25 January 2022).
- Jauhiainen, J.; Takahashi, H.; Heikkinen, J.E.P.; Martikainen, P.J.; Vasander, H. Carbon fluxes from a tropical peat swamp forest floor. Glob. Change Biol. 2005, 11, 1788–1797. [Google Scholar] [CrossRef]
- Applegate, G.; Freeman, B.; Tular, B.; Sitadevi, L.; Jessup, T.C. Application of agroforestry business models to tropical peatland restoration. Ambio 2021, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Surahman, A.; Soni, P.; Shivakoti, G.P. Reducing CO2 emissions and supporting food security in Central Kalimantan, Indonesia, with improved peatland management. Land Use Policy 2018, 72, 325–332. [Google Scholar] [CrossRef]
- Budiman, I.; Sari, E.N.; Hadi, E.E.; Siahaan, H.; Januar, R.; Hapsari, R.D. Progress of paludiculture projects in supporting peatland ecosystem restoration in Indonesia. Glob. Ecol. Conserv. 2020, 23, e01084. [Google Scholar] [CrossRef]
- Van Noordwijk, M.; Ekadinata, A.; Leimona, B.; Catacutan, D.; Martini, E.; Tata, H.L.; Öborn, I.; Hairiah, K.; Prasit Wangpakapattanawong, M.R.; Dewi, S.; et al. Agroforestry options for degraded landscapes in Southeast Asia. In Agroforestry for Degraded Landscapes; Springer: Singapore, 2020; pp. 307–347. [Google Scholar]
- He, G.; Wang, K.; Zhong, Q.; Zhang, G.; van den Bosch, C.K.; Wang, J. Agroforestry reclamations decreased the CO2 budget of a coastal wetland in the Yangtze estuary. Agric. For. Meteorol. 2021, 296, 108212. [Google Scholar] [CrossRef]
- Giesen, W.; Sari, E.N.N. Tropical Peatland Restoration Report: The Indonesian Case; Berbak Green Prosperity Partnership—MCA: Jakarta, Indonesia, 2018; Available online: https://luk.staff.ugm.ac.id/rawa/GiesenNirmala2018TropicalPeatlandRestorationReportIndonesiaForBRG.pdf (accessed on 5 September 2021).






| Bulk Density, g cm−3 | Clay, % | Silt, % | Corg | pH i (H2O) | Earth-Worms, # m−2 | Soil Temp, °C | Rel. Humidity, Air, % | Mean Water Table, cm | |
|---|---|---|---|---|---|---|---|---|---|
| Fruit agroforest (FAFo) | 0.94 b | 76.7 a | 23.1 a | 2.04 a | 3.63 a | 75.1 bc | 24.7 a | 97.4 c | −28.5 a |
| Old rubber agroforest (RAFo) | 0.97 b | 68.6 a | 32.2 ab | 2.76 a | 3.64 a | 93.4 c | 25.0 a | 97.8 c | −45.4 b |
| Young rubber agroforest (RAFy) | 0.85 a | 60.9 ab | 39.0 b | 6.11 b | 3.71 b | 63.1 b | 25.9 b | 90.5 a | −56.1 b |
| Rubber monoculture (RMOy) | 0.83 a | 50.2 b | 49.6 c | 5.43 b | 3.66 a | 27.5 a | 26.0 b | 94.8 b | −69.6 c |
| s.e.d. | 0.04 | 5.07 | 4.9 | 1.06 | 0.03 | 19.7 | 0.46 | 2.37 | 12.3 |
| Habitat | Poles # | Medium Trees | Large Trees | Basal Area | Aboveground Tree Biomass | Litter Layer | Litter Layer |
|---|---|---|---|---|---|---|---|
| Individuals ha−1 | m2 ha−1 | Mg ha−1 | cm | Mg ha−1 | |||
| FAFo | 320 a | 142 a | 87 b | 26.8 ab | 299 b | 2.50 b | 10.32 b |
| RAFo | 347 a | 127 a | 102 b | 34.4 b | 281 b | 1.39 a | 6.45 a |
| RAFy | 527 b | 132 a | 28 a | 20.2 a | 144 a | 1.74 ab | 6.95 a |
| RMOy | 320 a | 217 b | 40 a | 20.8 a | 148 a | 2.35 b | 8.25 ab |
| s.e.d. | 76 | 27.75 | 19.08 | 3.71 | 42.7 | 0.38 | 1.22 |
| Polyphenol, Pp % | Lignin, L % | Ash, % | Cellulose, % | N, % | (L + Pp)N | |
|---|---|---|---|---|---|---|
| Fruit Agroforest (old) | 5.93 | 31.34 | 1.80 | 30.07 | 1.12 | 33.36 |
| SEM | 1.87 | 0.28 | 0.45 | 1.31 | 0.04 | 2.50 |
| Rubber agroforest (old) | 3.83 | 31.21 | 1.42 | 27.89 | 1.13 | 31.76 |
| SEM | 1.19 | 0.66 | 0.26 | 0.66 | 0.17 | 4.50 |
| Rubber agroforest (young) | 10.69 | 29.44 | 1.34 | 29.56 | 1.21 | 33.32 |
| SEM | 0.36 | 0.87 | 0.37 | 1.12 | 0.09 | 2.29 |
| Rubber monoculture (young) | 4.10 | 29.75 | 1.47 | 27.81 | 1.53 | 22.13 |
| SEM | 0.25 | 0.69 | 0.29 | 2.28 | 0.01 | 0.61 |
| Habitat | Litter Source | kd, Week−1 | Se of kd Estimate | R2, % | MRT, Week | T0.5, Week |
|---|---|---|---|---|---|---|
| FAFo | L-FAFo | 0.018347 | 0.00321 | 84.5 | 54.5 | 37.8 |
| L-RAFo | 0.013675 | 0.001738 | 91.2 | 73.1 | 50.7 | |
| L-RAFy | 0.017497 | 0.001615 | 95.1 | 57.2 | 39.6 | |
| L-RMOy | 0.016452 | 0.001485 | 95.3 | 60.8 | 42.1 | |
| RAFo | L-FAFo | 0.011497 | 0.003514 | 64.1 | 87.0 | 60.3 |
| L-RAFo | 0.016119 | 0.001461 | 95.3 | 62.0 | 43.0 | |
| L-RAFy | 0.015744 | 0.002460 | 87.2 | 63.5 | 44.0 | |
| L-RMOy | 0.036097 | 0.001998 | 98.2 | 27.7 | 19.2 | |
| RAFy | L-FAFo | 0.005481 | 0.002223 | 50.3 | 182.5 | 126.5 |
| L-RAFo | 0.016039 | 0.002165 | 90.1 | 62.3 | 43.2 | |
| L-RAFy | 0.012348 | 0.002612 | 78.8 | 81.0 | 56.1 | |
| L-RMOy | 0.021410 | 0.001019 | 98.7 | 46.7 | 32.4 | |
| RMOy | L-FAFo | 0.008407 | 0.001997 | 74.7 | 119.0 | 82.5 |
| L-RAFo | 0.014367 | 0.002651 | 83.0 | 69.6 | 48.2 | |
| L-RAFy | 0.015163 | 0.003427 | 76.5 | 66.0 | 45.7 | |
| L-RMOy | 0.025548 | 0.004177 | 86.2 | 39.1 | 27.1 | |
| Minimum | 0.005481 | 0.001019 | 50.3 | 27.7 | 19.2 | |
| Average | 0.016512 | 0.002359 | 84.3 | 72.0 | 49.9 | |
| Maximum | 0.036097 | 0.004177 | 98.7 | 182.5 | 126.5 | |
| Based on Decomposition Constant kd | Based on Mean Residence Time | |||||
|---|---|---|---|---|---|---|
| FAFo | RAFo | RAFy | FAFo | RAFo | RAFy | |
| RAFo | 37.1, 36.8 | −27.1, −26.9 | ||||
| RAFy | 51.4, 43.2 | −11.2, −11.1 | −33.9, −30.1 | 12.6, 12.5 | ||
| RMOy | 77.6, 82.2 | −9.7, −10.6 | −1.3, −9.7 | −43.7, −45.1 | 10.8, 11.9 | 1.3, 10.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silvianingsih, Y.A.; van Noordwijk, M.; Suprayogo, D.; Hairiah, K. Litter Decomposition in Wet Rubber and Fruit Agroforests: Below the Threshold for Tropical Peat Formation. Soil Syst. 2022, 6, 19. https://doi.org/10.3390/soilsystems6010019
Silvianingsih YA, van Noordwijk M, Suprayogo D, Hairiah K. Litter Decomposition in Wet Rubber and Fruit Agroforests: Below the Threshold for Tropical Peat Formation. Soil Systems. 2022; 6(1):19. https://doi.org/10.3390/soilsystems6010019
Chicago/Turabian StyleSilvianingsih, Yosefin Ari, Meine van Noordwijk, Didik Suprayogo, and Kurniatun Hairiah. 2022. "Litter Decomposition in Wet Rubber and Fruit Agroforests: Below the Threshold for Tropical Peat Formation" Soil Systems 6, no. 1: 19. https://doi.org/10.3390/soilsystems6010019
APA StyleSilvianingsih, Y. A., van Noordwijk, M., Suprayogo, D., & Hairiah, K. (2022). Litter Decomposition in Wet Rubber and Fruit Agroforests: Below the Threshold for Tropical Peat Formation. Soil Systems, 6(1), 19. https://doi.org/10.3390/soilsystems6010019








